Abstract
Introduction:
Dementia and hearing loss pose a substantial global health challenge, and understanding their association is crucial. This study aims to determine the prevalence of hearing loss in dementia patients and investigate the risk factors for dementia severity including hearing loss.
Methods:
This study was a cross-sectional study. Patients with dementia diagnosed by a psychiatrist or neurologist were invited to participate in this study. Audiometry and tympanometry were performed for hearing investigation. The severity of dementia was determined by the Thai Mental State Examination questionnaire, and the SD-SLP-01 aphasia screening questionnaire and associate factors were collected.
Results:
A total of 88 participants were included in the study. Two participants withdrew because they were unable to complete hearing tests. The prevalence of hearing loss in this study was 94.2%. The prevalence of aphasia was 25.6%. The most common type of hearing loss is sensorineural hearing loss. Twenty-five percent of participants had moderate to severe hearing loss. A statistically significant difference was observed in the mean Thai Mental State Examination scores across different degrees of hearing loss (p = 0.040). The factors that contributed to the severity of dementia included aphasia (OR: 14.40, 95% CI: 4.53–45.73, p < 0.001) and severe hearing loss (OR: 55.00, 95% CI: 0.83–3,650.97, p = 0.014).
Conclusions:
Our findings revealed an extremely high prevalence of hearing loss in the dementia population. Furthermore, a statistically significant association was observed between severe hearing loss and dementia.
Introduction
Dementia is a clinical syndrome characterized by a decline in cognitive function significant enough to interfere with daily activities. This decline encompasses impairments in memory, language, perception, attention, and problem-solving abilities. In addition, individuals may experience changes in emotional regulation and behavior. 1 Common types of dementia include Alzheimer’s disease, vascular dementia, mixed dementia, dementia with Lewy bodies, and frontotemporal dementia.2–5
Dementia is associated with a range of health complications that extend beyond cognitive decline, significantly impacting both physical and mental well-being. Individuals with dementia often experience neuropsychiatric symptoms such as depression, anxiety, and apathy, which can exacerbate the progression of the disease and diminish the quality of life. 6
An estimated 55 million people are living with dementia worldwide, and according to the World Health Organization survey, they expected that by 2050 the number of patients will increase to 152 million (71% of the world’s population). The cost of treating people with dementia was estimated at 1 trillion in 2018 and is projected to rise to 2 trillion by 2030. 7 Physical inactivity, smoking, depression, social isolation, hearing loss, hypertension, obesity, diabetes, and low educational attainment are well established, potentially modifiable risk factors for dementia.8–11
The prevalence of hearing loss in the normal population according to The Global Burden of Disease study, which incorporated mild and unilateral hearing loss, estimated that the population with hearing loss rose from 30.7 million (30.0%) in 2010 to 62.0 million (33.1%) in 2021.12,13
Hearing loss was associated with a higher chance of having dementia. One study conducted in older adults in the United States found that dementia prevalence among participants with moderate to severe hearing loss was higher than prevalence among participants with normal hearing (prevalence ratio = 1.61, 95% CI: 1.09–2.38). 14 Study in Germany found that dementia prevalence increased by approximately 0.23% when the prevalence of hearing impairment increased by 1 standard deviation. 15
Two-thirds of the elderly population experience hearing loss, yet only 20% seek treatment. 16 An average out-of-pocket hearing care cost of $2500 was unaffordable for over three-quarters of Americans with functional hearing loss. 17
An elevation of 10 dB in pure tone average (PTA) is associated with a 16% increased likelihood of dementia. 18 Hearing loss can be improved with hearing aids. Using a hearing aid may prevent cognitive decline. One study in Japan found that hearing aid use showed a significant suppressive effect on cognitive impairment in those with moderate hearing loss who always use a hearing aid (odds ratio = 0.54, 95% CI: 0.30–1.00, p = 0.049). 19
To our knowledge, this is the first study to objectively quantify the prevalence of hearing loss in dementia patients and investigate the risk factors for dementia severity including hearing loss.
Methods
Study design
This study was a cross-sectional study in a Psychiatric Clinic, Srinagarind Hospital, Thailand from April 2022 to March 2023.
Participants
We invited patients who were diagnosed with dementia by a neuropsychologist or neurologist and were able to undergo hearing tests to participate in this study. The exclusion criteria were ear infection, epilepsy, craniofacial anomalies, cerebellopontine angle tumor, vestibular schwannoma, acoustic neuroma, and syndrome or genetic disorder. We collected demographic data on modifiable risk factors for dementia (Table 1). 9
Demographic data.

Cognitive severity test and aphasia screening test
All patients were assessed cognitive tests using the Thai Mental State Examination (TMSE), a standard cognitive test in Thailand. 20 The severity of dementia, as classified by TMSE scores, is defined as follows: 0–12 (severe), 13–18 (moderate), and 19–30 (mild). The TMSE score of less than 19 was used as a cut point for moderate to severe dementia. 8 SD-SLP-01 aphasia screening test was assessed by a speech-language pathologist. 21
Hearing evaluation
All patients had their ears examined by an otolaryngologist and received a hearing test by an audiologist. Audiometry uses the Hughson–Westlake method 22 using air conduction pathways at a frequency range of 250–8000 Hz and bone conduction pathways at a frequency range of 500–4,000 Hz. The PTA was calculated using the hearing sensitivity at 500, 1000, and 2000 Hz. The tympanometry with 226 Hz. probe was also performed bilaterally.
Definition of hearing loss
American Speech-Language-Hearing Association defined the types of hearing loss as sensorineural hearing loss, conductive hearing loss, and mixed hearing loss and defined the degree of hearing loss from audiometry as follows: −10 to 25 dB (decibels) was normal hearing, 26–40 dB was mild hearing loss, 41–55 dB was moderate hearing loss, 56–70 dB was moderate-severe hearing loss, 71–90 dB was severe hearing loss, and 91+ dB was profound hearing loss. 23 High-frequency loss was identified as the pure tone threshold of audiometry at frequencies 2000, 4000, and 8000 Hz greater than 25 dB HL. 24 In this study, the PTA audiometry in the better ear was used to classify the hearing loss.
Statistical analysis
Statistical analysis was performed using SPSS software (IBM Company, USA). Mean, standard deviation (SD), percentage, and 95% confidence interval were used to analyze data on the population characteristics and prevalence of hearing loss in patients with dementia. Chi-square was used to analyze significant differences in population characteristics. Pearson’s correlation was used to analyze the correlation between TMSE score and degree of hearing loss. To evaluate the risk factors for severe dementia, the univariate logistic regression was used and presented as an odds ratio. A p-value < 0.05 was considered significant.
Using a sample size estimation equation for a single proportion, assuming that 35% of the dementia population has hearing loss, 25 a sample size of 88 would be required to achieve an absolute precision of 10% with 95% confidence.
Results
Out of 180 consecutive patients who were invited, 88 agreed to enroll in this study. Two participants withdrew because they were unable to complete the hearing test. So, the total number of 86 participants was included in the analysis.
There were 54 females (62.8%) and 32 males (37.2%). The mean age is 75.52 ± 7.26 years, ranging from 58 to 90 years. Alzheimer’s disease was the most common type of dementia (43.0%) followed by vascular dementia (38.4%). The most common underlying conditions among patients were hypertension (52.3%), diabetes (26.7%), and high cholesterol (22.1%). Regarding smoking status, 68.6% were non-smokers, while 31.4% were smokers. In terms of social integration, 82.6% of patients maintained communication with others, particularly family members. In addition, 43.0% of patients engaged in regular physical activity and exercise as part of their daily routine. The prevalence of aphasia in the study population was 25.6% (Table 1).
The prevalence of hearing loss in this study was 94.2% (95% CI: 0.86–0.98). Sensorineural hearing loss was present in 91.9% of patients. Regarding the severity, mild hearing loss was observed in 39.5% of patients, followed by high-frequency hearing loss in 29.1% of patients (Table 2).
Prevalence of hearing loss in dementia patients.

SNHL: sensorineural hearing loss.
Table 3 presents the mean TMSE scores across different degrees of hearing loss, showing a statistically significant difference (p = 0.040) based on a one-way ANOVA test. Individuals with normal hearing had the highest mean TMSE score (24.60 ± 3.05), while those with severe hearing loss had the lowest (13.50 ± 6.36). These findings suggest a potential association between greater hearing loss severity and lower cognitive function, as measured by TMSE (Figure 1).
The mean TMSE scores and the severity of hearing loss.

One-way ANOVA test.
Statistically significant.
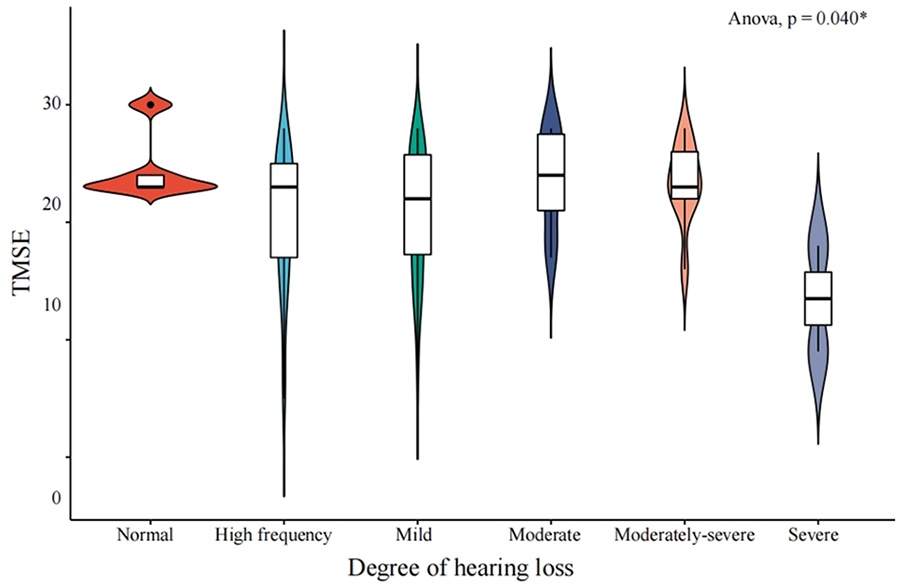
Violin plot of mean TMSE score and severity of hearing loss.
For the factors that can affect the severity of dementia, a TMSE score of less than 19 points was classified as moderate to severe dementia. 8 We found sex, age, underlying diseases, smoking status, social isolation status, and exercise were not a statistically significant factor for moderate–severe dementia. Aphasia (OR: 14.40, 95% CI: 4.53–45.73, p < 0.001) and severe hearing loss (OR: 55.00, 95% CI: 0.83–3,650.97, p = 0.014) were risk factors for moderate–severe dementia (Table 4).
Risk factors for moderate–severe dementia.

Odds ratio.
Chi-square test.
statistically significant difference.
Discussion
Dementia is a multifactorial condition. Hearing loss was one of the possible factors affecting the severity of dementia. The mechanism was proposed as an increase in cognitive load by the delay of auditory information decreases the working capacity of the brain (cognitive reserve).26,27
In this study, dementia was found more common in females (62.8%), the mean age was 75.52 years old which was comparable with the survey of dementia in Thailand which found dementia commonly in females (59.1%) and mean age of 67.96 years. 28 Many studies also found that the elderly were the primary age group to develop dementia.29,30
The most common type of dementia was Alzheimer’s disease (43.0%) followed by vascular dementia (38.4%). This finding was consistent with one study in 2021 that found Alzheimer’s disease (50%) and vascular dementia (24%) were the most common cause of dementia. 20 One review found the prevalence of Alzheimer’s disease ranged from 34% to 54% and vascular dementia ranged from 18% to 32% in dementia populations. 29
We found hypertension (52.3%), diabetes (26.7%), and cholesterol (22.1%) were the most common underlying diseases. These underlying diseases are risk factors for dementia related to blood vessels that carry blood to the brain. Diabetes affects insulin and the metabolic system, the brain must decrease insulin production, resulting in increased sugar in the bloodstream, causing impaired cognitive function. 9
In this study, we found a smoking prevalence of 31.4%, which is comparable to the 2017 Lancet Commissions report, where 27.4% of individuals over 65 years old were smokers, making it the third highest risk factor. 12 Smoking is known to increase the risk of vascular complications, including cardiovascular pathology, and exposure to cigarette smoke introduces neurotoxins that contribute to stroke, silent infarction, oxidative stress, atherosclerosis, and inflammation. These factors collectively heighten the risk of Alzheimer’s disease and all forms of dementia. 31
In this study, 43% of participants engaged in daily physical activity, while 31.4% were physically inactive due to mobility limitations or walking difficulties. A study on physical activity among older adults in rural Thailand reported a high prevalence of physical activity (58.7%), whereas 41.3% did not engage in regular exercise. 32 Another systematic review found that physical inactivity ranged from 28.8% to 33.8%. 33
A 6-month functional brain MRI study demonstrated that regular exercise improves cognitive network function, supporting the findings of this study. 34 Consistently, our results showed that participants who exercised regularly had higher TMSE scores (>19) in 29.07% of cases, compared to 19.0% in those who never exercised.
We found the prevalence of hearing loss in patients with dementia was 94.2% (95% CI: 0.86–0.98). The most common type of hearing loss is sensorineural hearing loss (91.9%).
We found a statistically significant difference in the mean TMSE scores across different degrees of hearing loss (p = 0.040), suggesting a potential association between greater hearing loss severity and lower cognitive function. Notably, the severe hearing loss group had the lowest mean TMSE score, although this group included only two patients.
The better ear is the ear that is used for hearing perception and listening in everyday life. When the dominant ear experiences difficulties, it leads to more challenging communication and increases cognitive load. 27 Severe hearing loss can significantly impair sound perception, such that patients cannot understand speech even when shouted at, unlike those with mild or moderate hearing loss who may miss certain consonant or vowel sounds. This effect results in a higher cognitive load for individuals with severe hearing loss compared to those with mild or moderate loss, which may contribute to dementia. 35
One study found no association between mild to moderate hearing loss and changes in social engagement. However, progressive hearing loss was linked to a gradual, imperceptible decline in social habits. 36 Another study also found that the severity of hearing loss was significantly correlated with hearing handicaps (p < 0.05). More often, participants revealed social and situational than emotional hearing handicaps (p < 0.05). 37 Severe hearing loss has been consistently shown to be a strong predictor of social isolation.38–40
This study found no significant correlation between social isolation and dementia. Thailand and South Korea share a cultural emphasis on extended families and strong community ties. This social fabric can provide significant support for the elderly, as demonstrated by South Korean research linking religion, family, and community involvement to reduced feelings of isolation and worthlessness among hearing-impaired seniors. 38
For risk factor analysis, we also found that severe hearing loss (OR: 55.00, 95% CI: 0.83–3650.97, p = 0.014) and aphasia (OR: 14.40, 95% CI: 4.53–45.73, p < 0.001) were a risk factor for moderate–severe dementia.
Aphasia is a disorder that results from damage to portions of the brain that are responsible for language. Deterioration of the anterior temporal lobe causes language impairment in both verbal and non-verbal communication. It is associated with dementia and is more common in patients with Alzheimer’s and other types of dementia. 41
The association between primary progressive aphasia and dementia was also found in previous studies. Both conditions had memory problems, and atrophy in the right prefrontal and insular cortex.42–44
To our knowledge, this is one of the first studies to objectively quantify the prevalence of hearing loss in dementia patients. Our findings reveal a surprisingly high prevalence of hearing loss in this population. However, a key limitation of this study is its small sample size, which restricts the statistical power and poses challenges for data analysis. In addition, the distribution of hearing loss severity across different dementia types results in low correlation values.
Healthcare professionals should recognize that there is a high prevalence of hearing loss and aphasia in dementia patients. Early hearing interventions such as using hearing aids and cochlear implants may prevent or reduce cognitive decline and dementia.45,46 The plan for prevention and multidisciplinary therapeutic strategies including a neurologist, psychiatrist, otolaryngologist, audiologist, and speech pathologist is needed. 47 Further research is warranted to investigate the effects of hearing aids on patients with dementia.
Conclusions
Our findings revealed an extremely high prevalence of hearing loss in the dementia population. Furthermore, a statistically significant correlation was observed between severe hearing loss and dementia.
Footnotes
Acknowledgements
The authors thank the participants for their cooperation and for providing valuable data.
Ethical considerations
This study was approved by the Center for Ethics in Human Research, Khon Kaen University (Approval ID: HE651020). This study was conducted in accordance with the principles embodied in the Declaration of Helsinki.
Consent to participate
All patients who could understand the information provided written informed consent by themselves, while legally authorized representatives of patients who were not capable of understanding the information provided the written informed consent on their behalf.
Author contributions
Conceptualization: Pradchaya Doungta and Patorn Piromchai; Data curation: Patorn Piromchai; Formal analysis: Pradchaya Doungta and Patorn Piromchai; Funding acquisition: Patorn Piromchai; Investigation: Pradchaya Doungta; Methodology: Pradchaya Doungta, Pongsatorn Paholpak, Panida Thanawirattananit, and Patorn Piromchai; Supervision: Patorn Piromchai; Validation: Patorn Piromchai; Writing – original draft: Pradchaya Doungta and Patorn Piromchai; Writing – review & editing: Pongsatorn Paholpak, Panida Thanawirattananit, Benjamas Prathanee, and Patorn Piromchai. All authors have read and agreed to the published version of the manuscript.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Faculty of Medicine, Khon Kaen University (Grant number IN65221).
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available upon request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
