Abstract
Objective:
Subarachnoid hemorrhage has high mortality and substantial long-term morbidity. Although SARS-CoV-2 infection is associated with hypercoagulopathy, patients may also present subarachnoid hemorrhage. We aimed to synthesize the clinical and paraclinical characteristics, treatment responses, and outcomes of this neurologic complication.
Methods:
The population included adults and children, and studies investigated patients with subarachnoid hemorrhagein the context of SARS-CoV-2 infection. We included prospective, retrospective, observational, or interventional studies. We systematically reviewed LitCOVID and the COVID-19 WHO database until February 2023, following a published protocol. Results are presented in tabular form to provide a descriptive summary of the findings.
Results:
We included 107 patients (30 aneurysmal SAH—aSAH, 52 non-aSAH confirmed by vascular imaging, and 25 without vascular imaging). Subarachnoid hemorrhage was concomitant or developed a few weeks after infection. Severity of COVID-19 ranged from asymptomatic (27.8% aSAH, 5.5% non-aSAH) to severe (50% aSAH, 75% non-aSAH). Patients presented inflammatory markers (25% aSAH, 81%non-aSAH), thrombocytopenia (12.5% aSAH, 28.6% non-aSAH), and impaired coagulation (0% aSAH, 28.6% non-aSAH). SARS-CoV-2 reverse transcription-polymerase chain reaction (RT-PCR) in the cerebrospinal fluid was negative in seven patients. Brain samples were positive for SARS-CoV-2 in one case.
Conclusions:
An inadequate infection response may change aneurysmal size and morphology, with rupture tendency. Renine–angiotensine–aldosteron system disruption with hypertension and cough increases pressure on aneurysmal walls. Furthermore, mycotic and dissecting aneurysms were reported. Non-aSAH accompanies infection-related complications such as cerebral venous sinus thrombosis, reversible cerebral vasoconstriction syndrome, posterior reversible encephalopathy syndrome, or intracerebral hemorrhage. Cytokine release syndrome, endothelial dysfunction, and SARS-CoV-2 affinity for angiotensin-converting enzyme 2 receptors may induce vasculitic processes. Cough precipitates arterial dissection. Hypoxic brain injury, immune thrombocytopenia, and some COVID-19 medications increase bleeding risks.
Introduction
Since the coronavirus (COVID-19) pandemic, neurologic complications of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection have been increasingly recognized. Up to one-third of patients with COVID-19 infection develop neurologic complications, including cerebral hemorrhage. While the association with acute ischemic stroke is well established,1,2 the association with intracranial hemorrhage, including SAH, is less clear. Furthermore, due to the novelty of SARS-CoV-2 infection, management of patients is based mainly on other infections.
The present systematic review aims to provide a thorough insight into existing literature by reporting data on SAH in patients with SARS-CoV-2 infection. We aim to investigate the clinical and paraclinical characteristics and the diagnostic and therapeutic challenges in this pathology, highlighting research gaps that require further attention.
Methods
We performed a systematic review following a previously published protocol. 3
Search strategy
We searched LitCOVID and WHO COVID-19 databases up to February 2023. These databases are curated for SARS-CoV-2 infection articles. Therefore, we used only the keyword “subarachnoid,” and we did not need to use search terms like “coronavirus,” “COVID-19,” or “SARS-CoV-2.” LitCOVID provides access to over 431.010 relevant articles in PubMed. The WHO database indexes articles from Embase, Web of Science, Scopus, ProQuest Central, gray literature, and several other databases. Also, we searched a preprint server (MedRxiv) using appropriate terms. Search filters were not used, as we aimed to generate a comprehensive research list. In addition, reference lists of the included articles were hand-searched to identify any further research. We used bibliographic software (EndNote 20, Clarivate Analytics, Philadelphia, PA, USA) to store, organize, and manage all retrieved references.
Study eligibility
The Population, Concept, and Context (PCC) mnemonics for the present scoping review were adults (over 18 years old) and children (P), with studies investigating patients with SAH (C) in the context of SARS-CoV-2 infection (C). We included prospective or retrospective observational or interventional studies. There were no restrictions on age, gender, or region.
We included conference abstracts only if the authors did not publish a full article on the study. Commentaries, correspondences, editorials, opinion articles, and narrative reviews were excluded.
Statistical analysis
We performed descriptive statistics, presenting patient demographics, clinical characteristics, ancillary investigations, treatments, and outcomes. Categorical variables are presented as numbers (percentages). In further analysis, differences for subgroups of aSAH and non-aSAH patients were tested for categorical variables using Fisher’s exact test (two-tailed testing for a significance level of p < 0.05).
This research did not require the acquisition of new patient data and, hence, was considered exempt from ethical approval.
Results
A PRISMA flow diagram displaying the case selection process is presented in Online Resource 1. Our search identified 564 articles through database searching, with 380 articles after de-duplicating records. In the title and screening phase, we removed 244 articles as they did not fulfill the eligibility criteria. Finally, we identified 58 articles for inclusion, reporting on 107 cases: 30 with a-SAH,4–19 52 with non-aSAH (confirmed by vascular imaging),7,19–49 and 25 patients with documented SAH but without vascular imaging.33,34,49–61 Table 1 presents the demographic characteristics of patients. A detailed presentation of the characteristics of included cases can be found in Online Resource 2. Among the included research, 37 articles were case reports and 21 case series.
Demographics of patients with SAH.

Immunological status and comorbidities
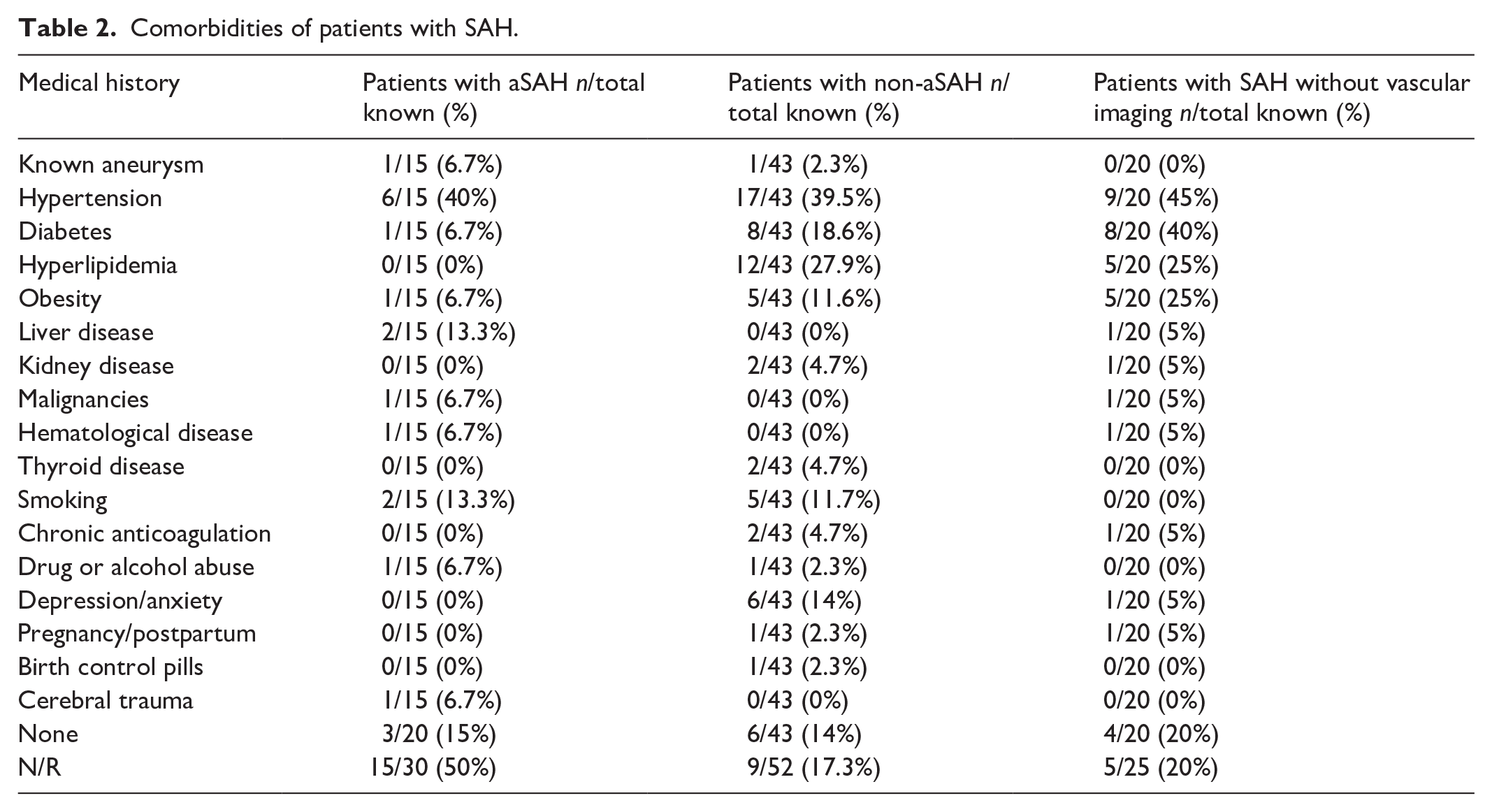
Only one patient with aSAH had previous vaccination with a one-dose COVID-19 mRNA vaccine. Nonetheless, vaccination status is not reported in 31 (29%) individuals. Unremarkable medical and drug history was noted in 3 (15%) patients with aSAH, 6 (14%) with non-aSAH, and 4 (20%) individuals with SAH without vascular imaging. The most important findings are presented in Table 2.
Comorbidities of patients with SAH.
Features of SARS-CoV-2 infection
Infection was confirmed by RT-PCR in 23 (76.7%) aSAH cases; one patient had a positive antigen test on admission, with negative RT-PCR on days 3 and 4. 17 Another patient had negative RT-PCR but positive serum SARS-CoV-2 IgG/IgA antibody. 19 Methods of diagnosing COVID-19 were unclear in 5 (16.7%) cases.
Among non-aSAH patients, infection was documented by positive RT-PCR in 38 (70.1%); the cycle threshold (Ct) was specified in one patient (Ct = 37). 44 One case was diagnosed based on a positive rapid antigen test on admission. 35 Another patient had a positive lateral flow test 10 days after neurological symptoms onset, with subsequent negative. 40 One individual was diagnosed based on a chest computed tomography (CT) scan; he had negative RT-PCR, IgG, and IgM tests. 34 Another patient had typical chest CT findings and recent history of cough and malaise; repeated RT-PCR was negative, but serological tests confirmed infection. 24 One case also had a negative RT-PCR but positive serum IgG/IgA antibodies. 19 In this subgroup, methods of diagnosing COVID-19 were unclear in 9 (17.3%) cases.
In patients with SAH but no information on vascular imaging, the diagnosis of SARS-CoV-2 infection was based on a positive RT-PCR test in 16 (64%) cases, but Ct is not specified. One patient with SAH 4 months after the initial COVID-19 episode had negative RT-PCR, but the serum COVID-19 rapid test detected IgM and IgG antibodies. 53 Another case had repeated negative RT-PCR tests, with positive antibody testing. 56 In one instance, the diagnosis was based solely on clinical and chest CT findings. 57 No data were provided on methods for diagnosing infection in 6 (24%) patients.
Severity of COVID-19 ranged from asymptomatic (27.8% aSAH, 5.5% non-aSAH) to severe (50% aSAH, 75% non-aSAH) (Table 3).
Clinical assessment of patients with SAH.
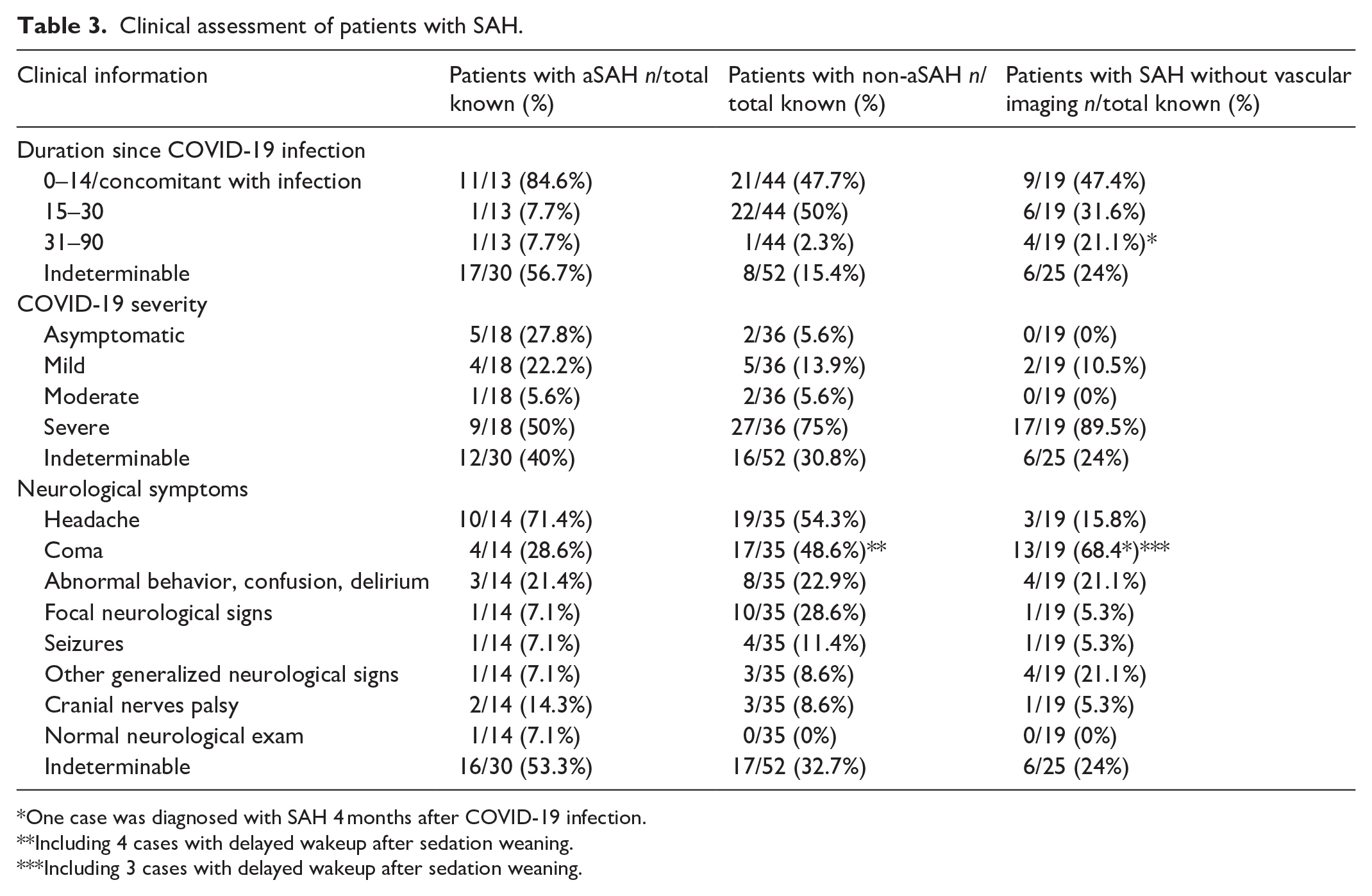
One case was diagnosed with SAH 4 months after COVID-19 infection.
Including 4 cases with delayed wakeup after sedation weaning.
Including 3 cases with delayed wakeup after sedation weaning.
Patients with aSAH received Remdesivir,16,18 Favipiravir,14,18 Tocilizumab, 18 Azithromycin, 12 Hydroxychloroquine,11,14 or corticosteroids.10,12,15–19 Of 10 patients with data on COVID-19 medication, 5 (50%) received anticoagulants.12,15–18 Patients with non-aSAH were treated with Lopinavir /Ritonavir,23,24,34,42,46 Remdesivir,30,31,35,38 Umifenovir, 42 Barticinib, 34 Anakirna, 38 Clazakizumab, 38 Tocilizumab, 38 Azithromycin,23–25,34,38,41 Hydroxychloroquine,23,24,34,36,38,46 corticosteroids,22,30,34,37,38,41,42,46 and intravenous immunoglobin.42,48 Some patients received Osetalmivir23,25,41 empirically. Among 35 (67.3%) patients with information on treatment, anticoagulants were prescribed to 20 (57.1%).31–34,36,38,39,41,45,46,48 Also, 2 (5.7%) did not receive any medication for infection.22,47 Details on the COVID-19 treatments are presented in Online Resource 3.
Clinical characteristics of SAH
As presented in Table 3, aSAH was concomitant with COVID-19 infection in 11 cases (84.6%). Only one patient (7.7%) with invasive mucormycosis rhinosinusitis and cavernous sinus thrombosis presented aneurysmal bleeding within 18 days of COVID-19 symptoms, 16 and one case with mycotic aneurysms (7.7%) within 42 days. 16
Among patients with non-aSAH, 21 (47.7%) presented with bleeding in the subarachnoid space within 2 weeks of infection, and 22 (50%) presented with SAH within 15–30 days. One patient with a few weeks of severe cough history had convexity SAH and vertebral artery dissection. 27
In aSAH group, the most common complaint was headache (71.4%), followed by coma (28.6%), and confusion or delirium (21.4%). Focal neurological signs were present in one case (7.1%). 11 One patient (7.1%) known right posterior communicating artery aneurysm had a normal neurological examination; he presented sudden headache and vomiting after physical activity. 17
In patients with non-aSAH, a headache was noted for 19 (54.3%); 17 (48.6%) were comatose, including 4 cases with delayed wakeup after sedation weaning.34,36 An altered mental status with abnormal behavior or confusion was present in 8 (22.9%), and focal neurological signs were reported for 10 (28.6%). All cases had an abnormal neurological examination.
Ancillary investigations
The detailed data on ancillary investigation results are presented in Table 4 and Online Resource 2. Among eight aSAH patients with available data, thrombocytopenia was reported for one (12.5%) case of invasive mucormycosis and cavernous sinus thrombosis. 16 Coagulation7,12,15,17,18 tests were within normal ranges in all cases. In the non-aSAH group, 12 (28.6%) had a decreased thrombocyte level, and 12 (28.6%) had impaired coagulation tests. Blood workup was normal in three patients (7.1%), including one with pituitary apoplexy and one with RCVS.21,35,45
Ancillary testing.

N/A: not applicable; PCR: polymerase chain reaction; ICH: intracerebral hemorrhage; IVH: intraventricular hemorrhage; CVST: cerebral venous sinus thrombosis; RCVS: reversible cerebral vasoconstriction syndrome; SDH: subdural hematoma; PRES: posterior reversible encephalopathy syndrome.
Cerebrospinal fluid (CSF) samples were obtained in four patients with aSAH and five with non-aSAH. Among aSAH patients, three had negative RT-PCR for SARS-CoV-2.5,9,19 However, one case had positive anti-SARS-CoV-2 IgG and IgA. 19 Two patients presented pleocytosis and a high red blood cell count.7,19 Among non-aSAH cases, none had increased white blood cells count in CSF. Two patients (40%) had proteinorachia,19,33 but three individuals (60%) had normal protein levels.36,46 The authors performed RT-PCR for SARS-COV-2 in four individuals, with negative results.19,33,36 The presence of anti-SARS-CoV-2 IgG was assessed in one patient, with negative findings. 36
An EEG was performed on three patients with non-aSAH29,33,36 and three with SAH without vascular imaging33,49,59 (Online Resource 2).
All cases underwent neuroimaging. In the aSAH subgroup, 8/30 (26.7%) cases underwent CT angiography, 2/30 (6.7%) had MR angiography, 2/30 (6.7%) were investigated with digital subtraction angiography, and 15/30 (50%) had angiography; in 3/30(10%) cases the imaging modality is not specified. The aSAH was detected by CT in 26/30 (86.7%) cases. The brain imaging modality is not specified in four (13.3%) patients. In the non-aSAH subgroup, the authors performed CT angiography (15/52 patients, 28.8%) MR angiography (8/52, 15.4%), digital subtraction angiography (6/52, 11.5%), angiography (5/52, 9.6%), and three-dimensional CT angiogram (1/52, 1.9%). Three cases (5.8%) underwent histopathological examination. Details on the imaging modality were not provided in 14/52 (26.9%) cases. In this subgroup, the preferred brain imaging modality was CT (45/52, 86.5%); in one patient (1.9%), the non-aSAH was detected by MRI, and the neuroimaging methods were not specified in 6/52 (11.5%) cases. In patients with SAH without vascular imaging, the most commonly used brain imaging method was CT (20/25, 80%).
Intraventricular hemorrhage (IVH) accompanied a-SAH in five patients (16.7%)5,6,11,15,18; among these, two had arterial dissection,5,18 and one also had ICH. 11 Intraparenchymal bleeding was present in another patient with SAH due to an aneurysm at the MCA origin and cerebellar ischemic stroke, possibly secondary to CVST. 13 CVST was also reported in a patient with mycotic aSAH in the context of invasive mucormycosis. 16 Cerebral infarction was present in 2 (6.7%) cases, one with mycotic aSAH 16 and one with possible CVST. 13
ICH accompanied non-aSAH in 19 (36.5%) patients.22,25,26,31,34–38,43,45,49. Among them, eight also had IVH, including one patient with CVST and cerebral infarct with hemorrhagic transformation, 49 six patients under sedation receiving therapeutic anticoagulation,36,38 and one comatose patient with basal ganglia hematoma, ventricular extension, and SAH. 34 IVH was also reported in a patient with perimesencephalic SAH following recovery from mild SARS-CoV-2 infection, 47 one with thrombocytopenia, receiving anticoagulants and mild multifocal SAH, 38 and one case of severe COVID-19, receiving extracorporeal membrane oxygenation (ECMO) and anticoagulants. 32 SAH appeared in the context of CVST in 7 (13.5%) cases,24,29,37,40–42,49 associated with cerebral infarction in four instances.24,29,41,49 Ischemic stroke was reported in six additional patients,20,22,28,33,36,46 including three with RCVS.20,22,46 In total, 7 (13.5%) cases were diagnosed with RCVS, including one with convexity SAH alone, 22 one with an associated subdural hematoma, 22 one with bilateral convexity SAH and vertebral artery dissection, 27 and one with convexity SAH and intracerebral hemorrhage (ICH) 45 ; the remaining three patients also had cerebral infarctions. Among non-aSAH, 4 (7.7%) individuals had a subdural hematoma. In one instance, SAH was associated with acute interhemispheric subdural hemorrhage. 7 A patient receiving aspirin and clopidogrel was administered therapeutic doses of enoxaparin; he presented a large ICH, an acute/hyperacute subdural hematoma, and SAH. 31 The third case presented ICH with associated small subdural hemorrhage and SAH. 43 PRES was reported in 2 (3.8%) patients. In one critically ill patient, the postmortem brain MRI revealed bilateral frontal and parietal SAH and signal abnormalities suggestive of PRES. 39 Another severe COVID-19 case had difficulty arousing after tracheal extubation; neuroimaging revealed cerebral hematoma, SAH, diffuse multiple microhemorrhages, and PRES. 34 Arterial dissection was detected in 3 (5.8%) patients in the context of RCSV20,27 or solely with SAH. 20 An unruptured aneurysm was found in 2 (3.8%) patients.32,34
An anatomopathological examination was performed in eight patients, all with non-aSAH: five autopsies,25,31,36,39,42 three biopsies,21,34 and one intraoperative examination. 44 In one severe COVID-19 patient, postmortem examination revealed acute pathology, scattered punctate SAH, and rare brainstem microhemorrhages. Neuropathological examinations were otherwise unremarkable. SARS-CoV-2 RNA was detected in the lung, trachea, subcarinal lymph node, kidney, large intestine, spleen, liver, heart, and blood. 25 Another patient receiving dual antiplatelet therapy and therapeutic doses of enoxaparin presented SAH, associated with ICH and subdural hematoma. 31 Autopsy demonstrated multiple small cerebral hemorrhages, pontine microbleeds, and basal ganglia microinfarcts in a severe COVID-19 case. Around the rostral surface of the cerebellum, there was an extensive SAH. Also, systemic endothelial damage and endotheliitis affected several organs. 36 Martin et al. 39 reported a patient with SAH and PRES who received mechanical ventilation and had four episodes of cardiorespiratory arrest; he underwent thrombolysis for pulmonary embolism on the day of death. Postmortem examination revealed reactive microglia, “red neurons,” Alzheimer’s type II glia, edema, congestion, perivascular hemorrhages, hemosiderin deposits, and axonal Wallerian degeneration. RT‑PCR was positive for SARS-CoV-2 infection of endothelial cells, as there was positive detection of the N-antigen in the cytoplasm. Immunohistochemistry was positive. 39 In a patient with Acinetobacter baumannii coinfection, the microscopic examination revealed fresh focal SAH and cortical venous thrombosis. Cerebral parenchyma showed scattered amyloid globules. RT-PCR for SARS-CoV-2 was negative in brain tissue. Pathological findings included multiple infarcts with thrombosis of the lung, spleen, and kidney. 42
Hernández-Fernández et al. 34 reported on the biopsy of a patient with SAH, ICH, and PRES with difficulty in arousing after tracheal extubation. 34 There was friable brain tissue with a dark aspect and fibrin microthrombi at different points. The hematoma originated from cortical arterioles, the examination being negative for arteriolosclerosis and cerebral amyloid angiopathy. 34 Also, a case of SAH and ICH had fibrin microthrombi in different brain areas, rare wall alterations of small arterioles, capillaries, and venules, with the disappearance of endothelial cells, capillaries degeneration due to edema or extravasation of macromolecules, and local inflammation. Fibrinoid necrosis was only found in one small vessel. In viable vessels, the reactive aspect of endothelial cells was striking, and sometimes, intranuclear and cytoplasmic clearance were seen. 34
A patient with episodes of thunderclap headache after COVID-19 presented a massive hemorrhage on CT. Surgery revealed not the suspected ICH but a massive space-occupying SAH. Histology demonstrated an old hematoma with additional fresh portions, but there was no evidence of vascular disease, malformation, or neoplasia. 44
Alyami et al. 21 presented a patient with SAH and pituitary apoplexy, with a histopathological examination used to diagnose the pituitary somatotroph adenoma. 21
Neurologic/neurosurgical treatment and patient outcomes
As shown in Online Resource 3, patients received neurological or neurosurgical treatment. Duration of follow-up varied, with death reported for 8 (40%) aSAH and 17 (38.6%) non-aSAH. Characteristics of deceased patients are presented in Table 5.
Characteristics of deceased patients.

Discussion
Since the first SARS-CoV-2 infection report, researchers noted the development of ischemic and hemorrhagic stroke, including SAH, following COVID-19. In this systematic review, we synthesize the evidence that aSAH and non-aSAH may occur in patients with COVID-19, highlighting this temporal association and comparing these entities.
SAH peaked in the fifth decade, but all patients in the aSAH group were less than 71 years old. In the non-aSAH group, 12.2% were older. In patients without COVID-19, it is unclear whether SAH peaks in the sixth decade or continues rising into old age.65,66 The aSAH was more frequent in men, but in the non-aSAH subgroup, the incidence was higher in women. Previous literature reports incidental unruptured intracranial aneurysms are higher in females, 67 but data are inconsistent. A systematic review found that sex does not significantly predict rupture risk. 68 However, another meta-analysis found that female sex was associated with greater odds of SAH. 69 A systematic review, including all types of SAH, reported that incidence in women was 1.24 times higher than in men; this gender difference started at age 55 and increased after that. 66 Our results align with studies from cohorts covering younger age groups which found an apparent excess in men in younger groups. In contrast, older patients show either equal sex incidence or a female predominance. 65
Medical history was unremarkable in 14%–15% of patients. Hypertension was the most frequent comorbidity in both aSAH (40%) and non-aSAH (39.5%), followed by liver disease in aSAH (13.3%) and hyperlipidemia in non-aSAH (27.9%, p = 0.0251). Previous research has demonstrated that cirrhosis was independently associated with an increased risk of aSAH, 70 consistent with studies reporting an increased risk of hemorrhagic stroke among individuals with cirrhosis. 71 Smoking history was present in 13.3% of patients with a ruptured aneurysm and 11.7% with non-aSAH. The percentage of hypertensives 72 and smokers 65 is similar to patients without COVID-19.
Aneurysmal bleeding occurred in most cases in the first 2 weeks of COVID-19 (p = 0.0251), while about half of patients with non-aSAH presented with bleeding within 15–30 days after infection (p = 0.0087). Half of aSAH patients had severe COVID-19, and 27.8% presented asymptomatic infection (p = 0.0344). In contrast, two-thirds of non-aSAH patients had severe infections, and only 5.6% were asymptomatic. These findings could be because non-aSAH occurred in the context of a COVID-19 complication, like CVST or RCVS.
Severe headache was more frequent in patients with aneurysmal rupture, but coma was less frequent. Non-aSAH was diagnosed in several patients with severe/critical COVID-19 when they had delayed wakeup after sedation weaning. Focal neurological signs (e.g., hemiparesis, aphasia) were more frequent in non-aSAH cases. In the aSAH subgroup, one patient with a headache presented a normal neurological examination, while in non-aSAH, additional neurological findings were always present.
In one-quarter of patients with aSAH, inflammatory markers were detected at onset. Lymphocytosis and lymphopenia were present in 12.5%, and thrombocytopenia was rare. In non-aSAH, inflammatory markers were present at onset in most cases, but the incidence of lymphocytosis and lymphopenia was largely similar to aSAH. A decreased level of thrombocytes and prolonged coagulation were more frequent in non-aSAH. D-dimer levels were increased in both subgroups in more than half of patients, but in non-aSAH, it may indicate the presence of COVID-19 complications, like CVST.
Anti-SARS-Cov2 IgG and IgA were positive in CSF in one aSAH patient, but negative in another non-aSAH case. RT-PCR was negative in both subgroups. The role of CSF SARS-CoV2 IgG and IgA antibodies and mechanisms of neuronal damage is under debate. It is plausible that neuroinflammatory processes in the CSF result from homeostatic responses of microglia and astroglia to systemic pathologies such as ischemia, hypoxia, or systemic critical illnesses rather than active inflammation or neurovirulence. 19
The absence of viral RNA, pleocytosis, and other inflammatory changes in the CSF favors the hypothesis that SARS-CoV-2 does not actively traffic into CNS, causing neuroinflammation. These findings do not support the presumed neurovirulence of SARS-CoV-2 in the pathogenesis of neurological complications. 19
In one case, brain samples were positive for SARS-CoV-2 by RT-PCR and immunohistochemistry. A recent review reports that only a few studies have found SARS-CoV-2 viral expression in neurons of the cerebral cortex, brainstem, and glial cells. 73 The most frequent neuropathological findings were hypoxic/ischemic and hemorrhagic lesions, with no specific neuropathological alterations. The authors concluded there is still no evidence of neurotropism for SARS-CoV-2, as the great diversity of autopsy approaches, sample preparation, anti-SARS-CoV-2 antibodies, and immunohistochemistry protocols, as well as the clinical history of each patient, may be one of the many causes for divergent results. 73 In aSAH, the cytokine storm and sympathetic activation may play a role in the aneurysm formation and rupture. Nonetheless, the blood–brain barrier is not disrupted in milder forms of infection, so SARS-CoV-2 cannot cross into CSF. 5
EEG was seldom performed and showed generalized or focal abnormalities. However, these findings are nonspecific and can be seen in many conditions, including metabolic and hypoxic encephalopathy, neurodegenerative disease, systemic infection, and encephalitis. 74
In SARS-CoV-2 infection, neuroimaging aids in detecting an underlying cause of SAH, like aneurysmal rupture, CVST, RCVS, PRES, arterial dissection, or extension of intraparenchymal bleeding. In aSAH, the most frequent additional finding was the intraventricular extension of SAH (16.7%), while ischemic stroke was rare (6.7%). In non-aSAH, ICH was the most frequent coexistent pathology (36.5%, p = 0.0033), followed by cerebral infarction (19.2%), CVST, and RCVS (both in 13.5%).
Death rates were similar to patients without COVID-19 (40% in aSAH, 38.6% in non-aSAH). In aSAH, the death rate was higher in males and smokers. Hypertension or diabetes did not increase the death rate. The percentage of deaths was higher in patients with severe SAH or COVID-19 and among those receiving anticoagulants, steroids, and antivirals, but not among cases treated with Hydroxychloroquine.
In the non-aSAH group, deaths were higher in females, individuals with hypertension, diabetes, and smokers. Severe SAH or COVID-19 increases the risk of death. Also, among the deceased cases, using anticoagulants, steroids, and Hydroxychloroquine increased the death rate, but not the administration of antivirals. Anticoagulants may increase bleeding risks, including the risk of SAH with poor outcomes. Steroids may decrease the anticoagulant effects but may impair vascular integrity, increasing the risk of bleeding when used together. Also, low Hydroxychloroquine blood levels have been associated with thrombotic events, 75 and among SAH patients, there were several with CVST. However, randomized placebo-controlled trials are needed to evaluate the effects of different medications in patients with SAH and COVID-19 to determine how they influence recovery time or outcomes.
The prognosis was better in the non-aSAH subgroup. However, full recovery was noted only in a minority of patients (5% aSAH, 18.2% non-aSAH). The duration of follow-up was short, and the presence of SAH following SARS-CoV-2 infection reflects a heterogeneity of mechanisms, including thrombosis (e.g., CVST), PRES, mucormycosis, pituitary apoplexy, and arterial dissection, and firm conclusions cannot be drawn.
The mechanisms underlying cerebral bleeding are not fully elucidated, with multiple factors being implicated: microthrombosis with secondary hemorrhage, dysregulated coagulation, vascular hyperpermeability in the context of SARS-CoV-2 infection, cytokine storm, endotheliitis, and vasculitis. 33 For example, the affinity of SARS-CoV-2 for angiotensin-converting enzyme 2 (ACE-2) receptors, expressed in endothelial and arterial smooth muscle cells in the brain, may trigger local inflammation that causes a vasculitic process. Therefore, the viral infection will damage the intracranial arteries, predisposing the vessel wall to rupture, a mechanism that could explain the pathogenesis of hemorrhagic stroke.
Some authors proposed that the hyperinflammatory state present in COVID-19, with hypercytokinemia and inflammation, could contribute to changes in size, morphology, and tendency to aneurysmal rupture to vascular degeneration, leading to aneurysm formation and size or morphology change, resulting in rupture and bleeding. 10 Early studies on the association between viral infections and SAH failed to support the hypotheses that intracranial aneurysms may develop because the initiating event of a viral infection produces any direct arterial damage, and aneurysm rupture may be temporally related to the infection. 76 The authors tested the following viruses: influenza A, influenza B, the respiratory syncytial virus (RSV), herpes simplex virus (HSV), and rubella. However, more recent research on patients with hepatitis B reported a significant association between HBsAg positive, anti-HBc positive, and cerebral aneurysm rupture. 77
Furthermore, in patients with SARS-CoV-2 infection, cytokine levels were frequently elevated in the CSF (often associated with blood-CSF barrier dysfunction) and serum, partly remaining positive at high levels for weeks/months. The CSF profile in COVID-19 with neurological symptoms was reported to be mainly characterized by blood-CSF barrier disruption in the absence of intrathecal inflammation, compatible with cerebrospinal endotheliopathy. Persistent blood-CSF barrier dysfunction and elevated cytokine levels may contribute to acute symptoms. 78 Also, disruption of the renine–angiotensine–aldosteron system may lead to uncontrolled hypertension, and severe cough increases the pressure on the aneurysmal wall. Furthermore, aSAH was reported in mycotic aneurysms and dissecting aneurysms.
In non-aSAH, the increased bleeding risk is multifactorial. A small percentage of cases may present an occult aneurysm; they are not diagnosed in initial angiographic studies but may be identified on repeat angiography. 79 Also, cerebral amyloid angiopathy can cause SAH in older adults. 80 Certain risk factors, including smoking, hypertension, and excessive alcohol intake, have statistically significant and consistent associations with an increased risk of SAH in case–control and longitudinal studies. 81
In patients with COVID-19, several additional risk factors may be present, including anticoagulation therapy. Also, extensive hemorrhaging, a result of the systemic anticoagulation required to reduce circuit clotting, is one of the most common complications during ECMO. 82 Nonetheless, the effects of other environmental factors like microplastics and nanoplastics83–85 or vaporized alcohol disinfectant 86 still need to be investigated.
In 36.5% of cases, SAH accompanied infection-related complications such as CVST, RCVS, PRES, or arterial dissection. Additionally, 36.5% had associated ICH. In the rest of the patients, SAH could be explained by a combination of cytokine release syndrome with endothelial dysfunction. Autopsy studies found systemic endothelial damage and endotheliitis, suggesting that the affinity of SARS-CoV-2 for ACE2 receptors in the arterial wall may trigger local inflammation, inducing a vasculitic process. Cough may also precipitate arterial dissection, and patients may present SAH in the context of hypoxic brain injury. Not negligible, immune thrombocytopenia and some medications used to treat COVID-19 increase the bleeding risks.
The SAH remains a rare occurrence following SARS-CoV-2 infection. Nonetheless, our findings emphasize the need for a high suspicion index, as SAH may be present in individuals with asymptomatic SARS-CoV-2 infection or ICU patients under sedation. SAH may indicate a potentially severe complication like CVST, RCVS, PRES, pituitary apoplexy, or arterial dissection. An international registry of such cases would aid in thoroughly examining SAH in the context of COVID-19, providing an opportunity to collect and report cases in a standardized manner across all ranges of severity in all settings.
The present work is the first systematic review with data on individual cases, comparing aSAH and non-aSAH as distinct subgroups. Several authors reviewed hemorrhagic stroke87,88 or cerebrovascular diseases in general 89 in SARS-CoV-2 infection but without a specific focus on SAH. A previous systematic review on SAH in COVID-19 patients, 90 up to September 2021, included 17 case reports, 11 case series, and two retrospective cohort studies, with 345 included cases. Nonetheless, focusing on case reports and case series that offer data on individual patients provides a more detailed examination of patient characteristics, treatment details, and outcomes, leading to a more nuanced understanding of the relationship between SARS-CoV-2 infection and SAH compared to previous reviews that included aggregated data from retrospective series. Despite methodological constraints, observing individual patients provides essential insights into etiology, pathogenesis, evolution, and possible treatments. 91 Case reports and case studies describing new events are the first-line evidence for further hypothesis testing with statistical approaches. 91 Furthermore, case reports and series can reduce the risk of publication and selection bias compared to those that only report summary statistics. Finally, our review may include more recent studies, providing a more up-to-date understanding of the topic.
Nonetheless, our findings are limited by the lack of standardized reporting of individual cases and the quality and extent of information in included reports, which were inconsistent. Also, truncated follow-up data impedes conclusions on outcomes and prognosis.
Conclusions
The development of SAH following COVID-19, although rare, requires a high suspicion index as it may indicate potentially severe complications. The presence of SAH reflects a heterogeneity of mechanisms, including thrombosis, PRES, mucormycosis, pituitary apoplexy, and arterial dissection. The increased bleeding risk in non-aSAH is multifactorial, with about one-third of cases accompanied by infection-related complications such as CVST, RCVS, or PRES and one-third having associated ICH. In the rest of the patients, SAH could be explained by a combination of cytokine release syndrome and endothelial dysfunction. Furthermore, autopsy studies found systemic endothelial damage and endotheliitis, suggesting that the affinity of SARS-CoV-2 for ACE2 receptors in the arterial wall may trigger local inflammation and a vasculitic process. Immune thrombocytopenia and some COVID-19 medications also increase bleeding risks. We emphasize the need for a high suspicion index and an international registry to examine SAH in COVID-19 contexts.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241299643 – Supplemental material for Subarachnoid hemorrhage in patients with SARS-CoV-2 infection: A scoping review
Supplemental material, sj-docx-1-smo-10.1177_20503121241299643 for Subarachnoid hemorrhage in patients with SARS-CoV-2 infection: A scoping review by Elena Cecilia Rosca, Almonzer Al-Qiami, Amalia Cornea and Mihaela Simu in SAGE Open Medicine
Footnotes
Acknowledgements
None.
Author contributions
AC, MS, and ECR conceptualized and designed the study. ECR drafted the manuscript. ECR, AC, and MS performed article searching. ECR, AA-Q, and AC extracted data. ECR and AC analyzed the data. AC, AA-Q, MS, and ECR reviewed and edited the final manuscript. All authors read, reviewed, and approved the final article.
Availability of data and materials
All relevant data are available within the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. The APC was funded by the “Victor Babes” University of Medicine and Pharmacy, Timisoara.
Human ethics and consent to participate declarations
Not applicable.
Institutional review board statement
Not applicable.
Informed consent
Not applicable.
Consent for publication
Not applicable.
Supplemental material
Supplemental material for this article is available online.
