Abstract
Introduction:
Severe COVID-19 can result in long-term sequelae known as “chronic COVID,” characterized by a wide range of persistent physical and mental symptoms. Chest imaging and pulmonary function test alterations have been observed in recovered patients. Most studies focus on up to a 3-month follow-up after symptom onset or hospital discharge, with few reports on long-term follow-up and limited evidence regarding disease progression in Latin America.
Methods:
This study aims to describe the clinical characteristics and changes in pulmonary function, imaging, and quality of life in severe and critical COVID-19 patients requiring ICU admission in a high-complexity hospital in Latin America. A prospective cohort of survivors underwent clinical, radiological, pulmonary function, and quality of life assessments 6 and 12 months post-discharge.
Results:
One hundred twelve patients were included, all of whom attended the 6-month follow-up, and 99 returned for the 12-month follow-up. Most subjects had no previous respiratory symptoms or significant medical history. At the end of the follow-up period, 74% of the patients showed interstitial infiltrates in chest tomography and a higher frequency of fibroatelectatic tracts and parenchymal bands. Pulmonary function tests returned to normal ranges, except for carbon monoxide diffusion, but no altered scores were reported in the questionnaires.
Conclusion:
Despite residual radiological findings, most parameters studied in severe and critical COVID-19 survivors improved over the 12-month follow-up period. Regardless of the imaging abnormalities, the improvement in variables such as symptomatic relief and normal pulmonary function suggests that these alterations are transient. Carbon monoxide diffusion did not normalize by the end of the follow-up, which is consistent with the abnormalities reported in multiple studies, indicating a potential disease-related pattern.
Introduction
In 2019, the SARS-CoV-2 virus was identified as the cause of pneumonia in Wuhan, China. Since then, it has led to the disease known as COVID-19, which was declared a pandemic in February 2020. According to the World Health Organization (WHO), there have been 767,364,883 reported cases and 6,938,353 deaths worldwide due to this virus. In Colombia, as of June 2023, there have been 6,367,600 confirmed cases and 142,748 deaths. 1
A high percentage of patients infected with this virus are asymptomatic,2,3 and the vast majority who develop the disease present mild symptoms. 4 Some individuals progress to severe forms of COVID-19, such as pneumonia, which can rapidly advance to acute respiratory distress syndrome (ARDS) or acute respiratory failure.5,6 Moreover, the virus can cause complications in other systems, such as the neurological and cardiovascular systems, and even affect coagulation, especially in critically ill patients. Intensive Care Unit (ICU) admission rates vary from 2% to 32%, with mortality rates ranging from 1% to 17% in different populations with COVID-19.7–9
The National Institutes of Health (NIH) has classified adults with SARS-CoV-2 infection into five groups, including asymptomatic or presymptomatic infection, mild illness, moderate illness, severe illness, and critical illness. 10 Special attention has been given to severe disease, defined as patients with oxygen saturation (SpO2) less than 94%, a ratio of arterial partial pressure of oxygen to fraction of inspired oxygen (PaO2/FiO2) less than 300 mm Hg, a respiratory rate greater than 30 breaths/min, or lung infiltrates greater than 50%, as well as critical illness, described as patients who have respiratory failure, septic shock, and/or multiple organ dysfunction. 10
Generally, individuals with severe disease require more time to recover compared to those with mild illness. However, the long-term consequences of severe COVID-19 are increasingly being recognized and are becoming more concerning. 11 Terms such as “post-COVID-19” or “chronic COVID” have been used to describe the associated signs and symptoms. According to the Centers for Disease Control and Prevention (CDC) and the WHO, the post-COVID condition presents a wide range of physical and mental symptoms that develop during or after the virus infection, persist for at least 2 months, impact the patient’s quality of life, and cannot be explained by any other diagnosis.12,13
In these cases, the persistence of symptoms such as fatigue, shortness of breath, joint pain, and chest pain has been observed after hospital discharge. 14 Other post-COVID-19 alterations described include neurological symptoms such as anosmia (loss of smell), headache, anxiety, and depression, as well as issues with memory and concentration.15,16 Persistent abnormalities in lung imaging studies and lung function impairment have also been observed.17,18
In chest computed tomography (CT) scans of COVID-19 patients performed 90 days after the onset of symptoms or hospital discharge, abnormalities have been observed in more than 50% of the patients, with ground-glass opacities and the presence of parenchymal bands or fibrous stripes being notable findings. Furthermore, lung function tests conducted on these patients 1 month after hospital discharge have shown abnormalities in at least 40% of cases, including a decrease in diffusion capacity and a predominantly restrictive pattern. 19
In the current literature, studies have been conducted on patients who have survived ARDS due to other causes. These patients have been followed up at 6, 12, and even 24 months after discharge from the ICU. These studies have revealed a significant negative impact on these patients’ quality of life, mental health, return to work, participation in social activities, and physical health.20–22
Although initially, most studies evaluating the sequelae of COVID-19 followed patients up for 1–3 months after symptom onset or hospital discharge, 18 there are reports of follow-ups ranging from 6 to 12 months in Asian and European populations.23–26 Currently, there is limited evidence available to support the follow-up of these patients for more than a year, as well as in other populations like Latin America.
The objective of this study is to describe the clinical characteristics and changes in pulmonary function, imaging, and quality of life in patients with severe and critical disease secondary to SARS-CoV-2 infection who required ICU admission after 6 and 12 months from discharge in a high-complexity hospital in Colombia.
Materials and methods
A prospective cohort study was conducted, which included adult survivors of COVID-19 diagnosed with severe or critical disease and admitted to the ICU at Fundación Valle del Lili (FVL), a high-complexity hospital located in Cali, Colombia. The diagnosis of COVID-19 was confirmed through a positive result in the RT-PCR test and/or the presence of positive antibodies and/or viral antigens specific to COVID-19. The severity of COVID-19 was classified based on the criteria set by the NIH. 10
Patient data were collected from the institutional COVID-19 registry of FVL, covering the period from March 2020 to March 2022. The selection of participants was based on the chronological order of their admission to the emergency department. Individuals with mild or moderate severity of COVID-19, those who did not undergo chest imaging during their hospitalization, or those who declined to participate in the study were not considered eligible candidates. Eligible candidates were invited to attend an in-person consultation at FVL, where the study’s objectives and activities were explained. Subsequently, the informed consent form was read, described, and signed by the participants.
Two visits were scheduled to complete the proposed activities, one 6 months and another 12 months after hospital discharge. During each visit, participants underwent several pulmonary function tests, including a 6-min walk test, spirometry, and lung diffusion tests. Diagnostic chest imaging was performed using the same modality during hospitalization: X-ray or CT scans. Participants also completed validated scales for the Colombian population, including the Beck Anxiety Inventory (BAI), 27 Beck Depression Inventory second edition (BDI-2), 28 and St. George’s Respiratory Questionnaire. 29 The English versions of these scales are provided in the Supplemental Material. The visits concluded with a medical consultation with a pulmonology specialist.
The Biomedical Research Ethics Committee of FVL approved the research protocol and the corresponding informed consent, registered under Protocol No. 01631 with Act No. 20 of 2020. Additionally, the study adhered to the recommendations of the Declaration of Helsinki and the Council for International Organizations of Medical Sciences. The protocol is registered on ClinicalTrials.gov with the ID NCT04559100.
Variables
Demographic characteristics and medical history information were collected. During the medical consultations conducted by a pulmonologist, data were obtained from a structured list of signs and symptoms. For the diagnostic imaging of the chest, predetermined variables related to pathological findings were considered. These images were evaluated by two experienced radiologists specializing in chest image interpretation. These radiologists conducted a blinded review of both X-rays and CT scans. The evaluation of chest images by the radiologists was not performed in a predefined order, thereby enabling each radiologist to review images of the same patient at varying follow-up intervals.
In the 6-min walk test, the distance covered in meters was recorded. In spirometry, the following measurements were taken: forced expiratory volume in 1 s (FEV1) in liters, % of predicted FEV1 post-bronchodilator, % of predicted forced vital capacity (FVC) post-bronchodilator, and the FEV1/FVC ratio. In the diffusion test, the measurements collected were CO2 in ml/min/Hg and % of predicted CO2 diffusion value. For the three applied questionnaires, the total score of each was considered.
For the variables related to diagnostic imaging, data were collected at three time points: during hospitalization for COVID-19, at the 6-month post-hospital discharge visit, and at the 12-month post-hospital discharge visit. Information was obtained for the other variables at both scheduled visits. If a participant showed average results in the pulmonary function tests and diagnostic imaging at the 6-month visit, these data were duplicated for the 12-month visit, assuming that the participant would continue to have expected results in these diagnostic tests. This approach was used to reduce patients’ exposure to radiation and minimize the need for additional imaging at the 12-month follow-up.
Statistical analysis
The sample size calculation for frequencies in a population was based on a study that indicated an 80% alteration in pulmonary function tests in survivors of ARDS. 30 Assuming a population size of 1000 people, with a hypothetical frequency of the outcome factor at 80% and a confidence limit of 7%, the total sample size required was 112 participants for a 95% Confidence Interval. The calculation was performed using the OpenEpi web application, available at: https://www.openepi.com/SampleSize/SSPropor.htm. Descriptive analysis was performed with measures of central tendency and dispersion for quantitative variables based on the normality of their distribution, which was evaluated using the Shapiro-Wilk test. For qualitative variables, we calculated absolute frequencies and percentages.
To test the difference in proportions in two dependent populations, the McNemar test with continuity correction is used for dichotomous variables, and the Stuart-Maxwell test is employed for polytomous variables. To assess differences in quantitative variables in two dependent populations, the Wilcoxon test with continuity correction is used for medians, and the paired t-test is employed. A significance level of 5% was employed for all tests. The statistical analysis was performed using the R statistical software version 4.2.1 (R Foundation for Statistical Computing) through RStudio 2022.12.0 (https://www.r-project.org).
Results
In the institutional COVID-19 registry of FVL, 12,283 unique patients who were admitted to the emergency department were included. Of these, 3164 patients were classified with a diagnosis of severe or critical COVID-19, and 2293 of them were hospitalized in the ICU. Patients were contacted in chronological order of their emergency department admission date until the proposed sample size was reached. A total of 120 patients were contacted, of which eight did not agree to participate. Ultimately, 112 subjects were included in the study.
These participants underwent the scheduled activities during the 6 and 12-month follow-up visits. All 112 subjects attended the 6-month follow-up visit, while only 99 returned for the 12-month follow-up. Most patients had chest CT scans during their hospitalization, and the same imaging modality was used for the 6- and 12-month follow-up visits. However, not all patients had CT scans during hospitalization due to pandemic regulations and the treating physician’s discretion. The activities performed in the study are detailed in Figure 1.

Patients participating in the study activities.
During the two follow-up visits, one patient was excluded from pulmonary function tests because of a history of neuromuscular pathology that prevented them from performing these tests. At the 6-month follow-up visit, the following missing data were found: for spirometry, one patient had no recorded data, and four patients needed to meet reproducibility criteria; for the pulmonary diffusion test, two patients had no recorded data.
At the 12-month follow-up visit, 13 patients did not attend the scheduled check-up. During the 6-min walk test, one patient was unable to complete it due to a hypertensive crisis. Two patients could not achieve proper coupling with the tracheostomy in the spirometry and diffusion test. Additionally, two patients had no recorded data for the diffusion test. In the surveys, some participants only completed part of the form for the scales, making it impossible to obtain a total score.
The median age of the participants was 60 years, with a higher proportion of men (68%). Most of the included patients had at least one comorbid condition (69%), with overweight being considered. Sixty-eight percent of the participants were overweight, followed by hypertension as the second most frequent comorbidity (43%). Only one patient had a history of asthma, two patients had a history of Chronic Obstructive Pulmonary Disease, and five patients had a history of cancer other than lung cancer. Please refer to Table 1 and Supplemental Table 1 for more details.
Demographic and clinical characteristics of the study population upon admission to the hospitalization (n = 112).

Median (Q1–Q3).
Body mass index (BMI) ⩾ 25.
Includes overweight.
During the evaluation with the pulmonology specialist, most patients did not report respiratory symptoms in the follow-up visits at 6 and 12 months. However, in the 12-month visit, patients mentioned feeling less fatigue, myalgia, and arthralgia than in the first 6-month visit. In most cases, no abnormal respiratory auscultation sounds were found during the physical examination in both visits. Three patients had a tracheostomy during the consultation visit and one had it closed before the 12-month follow-up. One patient who did not have a tracheostomy at the 6-month follow-up required its placement due to COVID-19 reinfection (the patient did not comply with the vaccination schedule). Please see Table 2 and Supplemental Table 2 for more information.
Symptoms and clinical signs at the first and second follow-up visits after hospital discharge.

McNemar’s Chi-squared test with continuity correction. The bolded information corresponds to p-Values < 0.05.
Forty patients had follow-ups with chest X-rays, of whom 4 did not attend the 12-month visit. One patient had no pathological imaging findings during hospitalization. The predominant infiltrates during hospitalization were ground-glass opacities followed by consolidation, with a peripheral and diffuse distribution and perihilar location. Most patients did not present pathological findings during the follow-up visits at 6 and 12 months. The most frequently found abnormality in the follow-up visits was interstitial infiltrates in approximately 25% of the patients.
Four patients did not receive follow-up imaging at 12 months because they did not attend this visit. In the three evaluation time points, most patients did not present pleural effusion or decreased lung volume; however, this latter finding improved in the two visits compared to hospitalization (Table 3 and Supplemental Table 3).
Radiological findings during hospitalization, first, and second follow-up visits after hospital discharge.

The tests performed with the Stuart-Maxwell test are presented in italics.
Comparison of hospitalization and first 6-month consultation.
Comparison hospitalization and second 12-month consultation.
Comparison of first 6-month consultation and second 12-month consultation.
McNemar’s Chi-squared test with continuity correction.
The bolded information corresponds to p-Values < 0.05.
A total of 72 patients underwent chest CT scans. However, retrieving the image information was impossible in two of them: one during hospitalization and the other at the 12-month visit. Additionally, 9 patients did not attend the 12-month follow-up. During hospitalization, approximately 62% of the patients had consolidations as the predominant pattern in the chest CT scan, followed by ground-glass opacities. The predominant profusion was multifocal, with a peribronchovascular and peripheral distribution and diffuse location. However, in the follow-up visits, there was a change in the predominant pattern of lung infiltrates. At 6 months, interstitial infiltrates and ground-glass opacities were the most frequent, both with a prevalence of 41%. At 12 months, it was found that 74% of the patients presented interstitial infiltrates.
In the chest CT scans obtained during the follow-up visits at 6 and 12 months, a decrease in the presence of the disordered cobblestone pattern and pleural effusion was observed compared to the images during hospitalization. At the 6-month visit, there was a more significant lung volume compromise than the 12-month consultation. At the 6-month and 12-month follow-up visits, fibroatelectatic tracts, bronchiectasis, parenchymal bands, and scarring emphysema were observed to a greater extent than in the CT scans during hospitalization. Additionally, subpleural reticulations increased progressively in the follow-ups, while no significant changes were observed in pleural thickening (Table 3 and Supplemental Table 3).
In the follow-up pulmonary function tests at 12 months, the distance covered in the 6-min walk test was observed, with a median that increased from 454 to 484 m. Additionally, an improvement in various spirometry parameters, including FEV1 and FVC, was observed. The diffusing capacity of the lungs for carbon monoxide (DLCO) also improved in the same examination. Patients who did not show pulmonary infiltrates in the chest imaging during the 6-month visit did not demonstrate improvement in lung function tests on the second visit compared to the first visit. On the other hand, those who had pulmonary infiltrates in the chest imaging during the 6-month visit showed improvement in all assessed parameters at 12 months (Supplemental Tables 4–6).
Patients without pulmonary infiltrates in chest imaging at 6 months obtained better results in pulmonary function tests than patients with pulmonary infiltrates in chest imaging during both follow-up visits (Figure 2—Supplemental Tables 7 and 8).

Results of pulmonary function tests stratified by radiographic findings in follow-up visits.
During the 6-month follow-up consultation, the St. George Respiratory Questionnaire score was 12.4 points, which decreased to 6.3 points at the 12-month follow-up. However, no significant changes were observed in the scores of the BAI and BDI-2 in both measurement periods (Table 4). Figures 3–5 show the radiological images of several representative cases, including chest scans from the acute phase and those from 6 or 12 months after the event.
Scores from respiratory symptoms, anxiety, and depression questionnaires at the first and second post-discharge follow-up visits.

Wilcoxon signed rank test with continuity correction.
The bolded information corresponds to p-Values < 0.05.
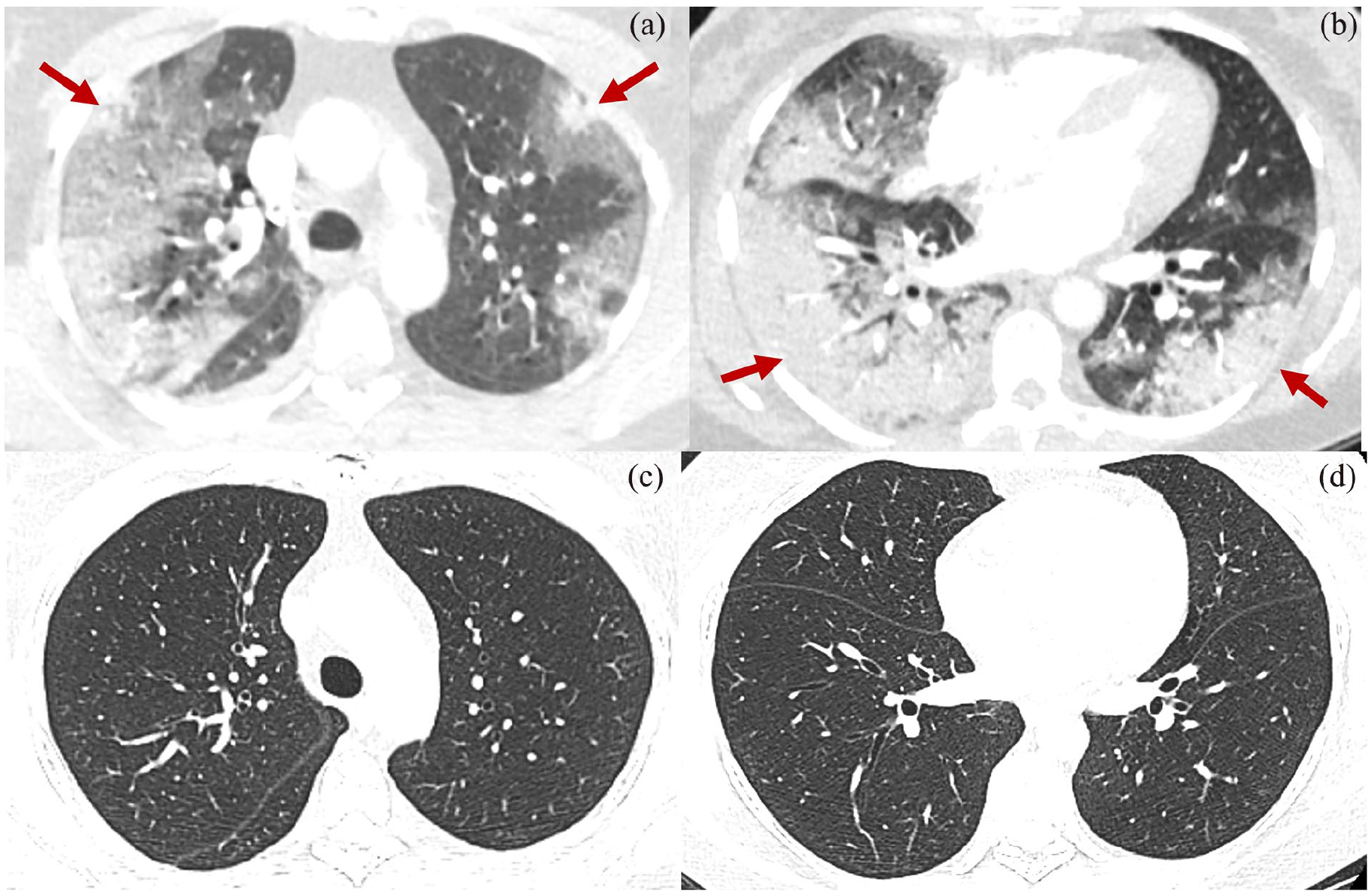
(a and b) Chest scan showing ground-glass opacities and areas of consolidation predominantly peripheral and peribronchovascular (red arrows). (c and d) Chest scan 6 months later showing complete resolution of the infiltrates.

Chest scan. (a) Axial section, (b) coronal section, showing areas of consolidation (red arrows) and ground-glass opacities (blue arrows). (c) Axial section, (d) coronal section. Chest scan taken 12 months after the acute COVID-19 event. Linear hyperdense tracts (orange arrows) and subpleural reticular opacities (black arrows) are observed.

Chest scan. (a) In the acute phase of COVID-19, bilateral basal consolidations (red arrows) and multifocal ground-glass infiltrates (blue arrows) are observed. (b) Chest scan 6 months later showing multifocal ground-glass areas (blue arrows), subpleural reticular opacities (black arrows), and subpleural parenchymal bands (orange arrow). (c) Chest scan 12 months after the acute event, multifocal ground-glass areas (blue arrows), subpleural parenchymal bands (orange arrow), and traction bronchiectasis (yellow arrow) are found.
Discussion
Scientific literature has shown that individuals with severe COVID-19 not only require more time to recover compared to mild forms of the disease but also experience long-term sequelae that are currently a cause of concern after surpassing the peaks of the pandemic. 11 Previous research has indicated that survivors of ARDS undergo significant respiratory symptoms, a decline in health-related quality of life, and enduring abnormalities in chest imaging, spirometry, and DLCO. These findings suggest that most ARDS patients do not fully recover their pre-illness health status due to underlying lung injuries and long-term pulmonary sequelae.20,30,31
There is currently a strong interest in investigating the long-term repercussions of COVID-19, especially in patients who have experienced the disease in its severe or critical form, akin to ARDS, and have necessitated ICU management. Only some studies have assessed these patients beyond 6 months after hospital discharge.23,24,32 The present study followed a cohort of severe and critical COVID-19 survivors for 12 months. Overall, no residual symptoms were observed at this time point, and there were no alterations in lung imaging, though remnants of the infection were present. Pulmonary function tests tended toward normalcy except for DLCO, and quality of life assessments showed normal results.
The persistence of clinical symptoms has been described as one of the sequelae of COVID-19, which has been evaluated in studies conducted in different parts of the world. The research conducted in Wuhan-China studied clinical sequelae in 538 COVID-19 survivors 3 months after hospital discharge. It was found that the majority of patients, especially women, continued to experience general symptoms (49%), primarily fatigue, sweating, myalgia, and arthralgia, and respiratory symptoms (39%), with a predominance of dyspnea following activity, discomfort, and chest pain. 33
In an Italian study, the persistence of symptoms was assessed in 143 COVID-19-recovered patients 1 month after hospital discharge. The findings showed that 32% of patients had 1 or 2 symptoms, while 55% had more than three. The most commonly reported symptoms were fatigue, dyspnea, arthralgia, and chest pain. 14 It is important to note that in this study, only 12% of the evaluated patients were admitted to the ICU, and the study by Xiong et al. 33 needs to describe the number of patients admitted to the ICU. Additionally, the follow-up conducted in both studies was at most 3 months after hospital discharge. 32
In contrast, although clinical symptoms related to the disease were evident in the cohort under study during the 6-month follow-up visit, most subjects did not report symptoms nor presented abnormalities during the physical examination. The main symptom reported was neuropathy, followed by fatigue, cough, myalgia, arthralgia, discomfort, and chest pain. However, in the 12-month assessment, it was found that symptoms such as fatigue, myalgia, and arthralgia showed significant improvement. Although other symptoms also decreased, no statistically significant difference was found between the two follow-up consultations.
Another 12-month follow-up study also observed improvement in symptom reporting between the 6-month and 12-month visits. 24 Additionally, although most patients had fewer abnormalities in lung sounds during the physical examination at the 12-month visit compared to the 6-month visit, this difference was not statistically significant. Our population’s lower frequency of clinical abnormalities could be related to the low frequency of pre-existing pulmonary medical conditions, representing a predominantly healthy population.
It has been observed that in patients with severe COVID-19, abnormalities in chest radiographs are common. 34 Post-mortem studies have revealed that these patients experience diffuse changes in lung tissue, such as alveolar damage, exudate, and fibrosis, which may explain the chronic respiratory symptoms in survivors, as well as the radiological alterations such as ground-glass opacities and reticulations suggestive of pulmonary fibrosis.17,35,36
In our study, we observed that 97.5% of patients with severe COVID-19 presented abnormalities in chest radiographs taken upon hospital admission; only one patient did not have pulmonary infiltrates, which may be related to the early phase of the disease. The findings mainly included ground-glass opacities, consolidation with peripheral and diffuse distribution, and parahilar and basal localization, consistent with the information reported in the literature. 37
According to the study by Rousan et al., 38 which examined 88 patients with positive results in the RT-PCR test for COVID-19 without specifying severity, approximately 33% of patients with radiological abnormalities in initial radiographs experienced a recovery phase in which radiological findings returned to normal after the first 18 days.
In our study, more than 70% of patients no longer presented abnormalities in chest radiographs at the 12-month follow-up visit, showing a significant improvement in the previously seen findings in CXRs taken during hospitalization, are similar to what has been described by other authors. 39 Additionally, some patients in the 12-month follow-up continued to present abnormalities in chest x-ray with a predominance of the interstitial pattern, suggestive of fibrotic changes, as observed in other studies.23,39
On the other hand, we found that most cases with decreased lung volumes and pleural effusion in the image taken during hospitalization showed significant improvement in these findings in the follow-up radiographs compared to the radiographs taken during hospitalization, which are less likely to be found as sequelae of severe COVID-19.
When evaluating chest CT images in our cohort of patients, we observed that all of them presented abnormalities in the CT scan taken during hospitalization, with consolidation being the most frequent finding, followed by ground-glass opacities. Analyzing the patients’ evolution, we found that the pattern of interstitial infiltrates was predominant in 74% of the cases in the 12-month follow-up CT.
Compared with a systematic review with an average follow-up of 3 months after hospital discharge, our findings showed a higher frequency of residual abnormalities in the chest CT. While the previous study observed a frequency of 57% of abnormalities in the CT scan, in our cohort, we found a higher proportion of alterations. 19
The specific findings in our cohort are consistent with those of the mentioned study, with ground-glass opacities being the most observed, while consolidation was less frequent. Additionally, we found similar frequencies in other findings, such as pleural thickening, pleural effusion, and bronchiectasis. 19 Another study has also reported changes in the predominance of abnormalities in the hospitalization CT scans, shifting from consolidation and ground-glass opacities to reticular opacities in the 12-month follow-up. 24
Our follow-up cohort observed a significant and progressive increase in findings considered remnants of COVID-19 infection. However, these findings do not correspond to the presence of pulmonary fibrosis. Fibroatelectatic bands, parenchymal bands, and subpleural reticulations showed statistically significant increases, which appeared from the 6-month visit and predominated at the 12-month visit. These findings were more frequent than those reported in a previous systematic review. 19
In most of our patients, the initial findings observed in the CT scan during hospitalization evolved into fibroatelectatic bands, parenchymal bands, subpleural reticulation, bronchiectasis, and scarred emphysema in the two follow-up visits. However, it is essential to note that these findings cannot be considered permanent as longer-term follow-up is required to determine their possible resolution.
During the 6-month follow-up, it was observed that, on average, the evaluated patients had normal FEV1 values (83.1%), a median of normal FVC (80.5%), and a normal FEV1/FVC ratio (0.85). These results differ from the findings of a meta-analysis that included studies with follow-up periods ranging from 1 to 6 months (with an average time of 90 days) after hospital discharge, which indicated that about half of the patients had abnormalities in pulmonary function tests, with 16% showing a restrictive pattern and 8% an obstructive pattern, 19 aligning with another systematic review. 18
However, a decrease in DLCO was observed in our population, with an average of 63.6%. These results may be related to the findings of other studies where approximately 35% 19 to 40% 18 of patients showed abnormalities in this test. These results highlight the importance of continuously evaluating lung function in patients with COVID-19, even after 6 months of hospital discharge, to identify possible sequelae and provide them with appropriate follow-up.
In the 12-month follow-up, patients had, on average, a normal FEV1 (83.1%), a median of normal FVC (87.5%), and a normal FEV1/FVC ratio (0.84). This indicates that, in most patients in this follow-up, spirometry did not show alterations related to disease sequelae, consistent with other longer-term studies, such as the one conducted in the UK with 240 days of follow-up, where 297 out of 1083 patients had a normal FVC. 17
The diffusion capacity in our population remained below the normal range at 12 months of follow-up (67.1%). However, it was higher than that observed at 6 months, consistent with the findings of the previously mentioned study, where 177 out of 510 patients had a decreased carbon monoxide (CO) diffusion capacity. 17 This pattern of normal spirometry results with persistent CO diffusion capacity alteration at 12 months of follow-up has also been described in survivors of ARDS due to causes other than COVID-19 28 and in patients who survived severe or critical COVID-19. 24
Regarding the comparison between the two follow-up visits in the entire study population, improvement was observed in all measured parameters in the second visit. An increase in the median of 30 m walked between the 6-month and 12-month visits corresponds to a clinically and statistically significant change. This improvement was also determined to a lesser extent in another study at 12 months. 32 The FEV1 and FVC showed a statistically significant average increase of 7%; the FEV1/FVC ratio did not show clinically relevant changes. As mentioned before, although CO diffusion was altered in both visits, it had an average increase of approximately 4% (Supplemental Table 4).
When stratifying the findings by abnormalities in the diagnostic images, it is observed that patients without abnormalities have pulmonary function test results within the normal range and do not show statistically significant changes between visits, except for the 6-min walk test, which had an average increase of 44 m (Supplemental Table 5). In contrast, the population with abnormalities in the diagnostic images had normal FEV1 at the 6-month visit, but the FVC was less than 80%, indicating a predominance of restrictive changes.
On the second visit, these findings improved, suggesting that the restrictive changes were transient. The meters in the 6-min walk test increased by a median of 33 m, and CO diffusion increased on average by 4%, both statistically significant changes (Supplemental Table 6).
When comparing patients with lung infiltrates and without lung infiltrates in the two visits, it is observed that in the 6-month visit, the population with abnormalities had worse results in all pulmonary function tests (Supplemental Table 7). However, when comparing these two populations at the 12-month visit, no relevant differences are observed in FEV1 and FVC. However, lower statistically significant values in the 6-min walk test and CO diffusion are appreciated in the population with abnormalities in diagnostic images (Supplemental Table 8).
The findings in our study are consistent with another study that compares patients with and without abnormalities in CT scans at 12 months of follow-up. 24 This reflects the recovery of pulmonary function, which tends to return to normal in the long term (12 months), regardless of abnormalities in diagnostic images, except for CO diffusion, which remains altered.
Regarding the results of the total score of the St. George Respiratory Questionnaire, our study observed low scores in both measurement visits, implying a lower impact of the disease on the participants’ health status. However, there was an improvement in the total score at the 12-month visit (median of 6.3 points) compared to the 6-month visit (median of 12.4 points), with an approximate 50% decrease, and this change was statistically significant.
Another study of a prospective cohort in Italy has evidenced the reduction in the score in post-COVID follow-up. At the time of diagnosis, most patients had a score greater than 30, and at the 2-month follow-up, 65% had a score of 0, although not all patients returned to their baseline before COVID-19. 40
It has been suggested that viral infections, such as COVID-19, can generate neuropsychiatric syndromes of multifactorial etiology,41,42 which can persist after the disease.33,43,44 A systematic review of the literature on psychiatric consequences in patients with COVID-19 observed that, in a follow-up after 60 days, depressive symptoms were present in 10.5% and anxiety symptoms in 12.3%, indicating a reduction compared to the acute phase of the disease, 32.6%, and 35.7%, respectively. 43
Other studies have described scores in self-report scales for affective symptoms within the range of normality or with a tendency to decrease at 1 year of follow-up.32,44 In line with the improvement of anxious and depressive symptoms exposed in the literature, the evaluated patients presented very low anxiety and minimal depression in the two post-hospitalization follow-up consultations, according to the cutoff points established in the applied scales. Based on this, the frequency of significant affective disorders is shallow in this population in the long term. However, this study did not directly evaluate differences between their pre-COVID state and during hospitalization.
The present prospective study is one of the few studies that, up to the present moment, have proposed the follow-up of patients who suffered from severe and critical COVID-19. It involves standardized clinical, radiological, and pulmonary function assessments and quality of life findings. Additionally, it includes two follow-up time points at 6 and 12 months and compares them. Furthermore, it provides a more extended follow-up period than other published studies, and the visits were conducted in person, allowing for more accurate and reliable first-hand information from the patients and their medical records, which is lacking in other approaches such as telephone follow-up or retrospective designs.
Unlike systematic reviews that include subjects with mixed COVID-19 severity, this study focused on patients who required admission to the ICU during the acute stage of their illness, regardless of whether they underwent endotracheal intubation. These particularities put them at a higher risk of developing pulmonary sequelae after hospital discharge, making them a group of particular clinical importance.
The selection of patients for this study was based on an institutional COVID-19 registry that undergoes a double data quality check, providing greater confidence in strict compliance with the inclusion criteria. Most of the measurements evaluated in this study are part of the routine clinical follow-up of post-COVID-19 patients. However, it is noted as a limitation that the sample size calculation did not consider power analysis or other statistical considerations beyond those already mentioned.
As with all studies addressing post-COVID sequelae, baseline measurements of patients before developing this disease were not available. However, having reference values for most outcomes studied allows for approximating normality and estimating patient clinical status changes. It is not appropriate to generalize the conclusions of this study to all severities of COVID-19, as patients with non-severe or non-critical classifications were excluded, given the expectation that those with mild to moderate COVID-19 would not present post-COVID sequelae. Similarly, in this study, it is not possible to establish whether the results presented are related to the administration of the COVID-19 vaccine, as this study began activities 1 year before the onset of mass vaccination in Colombia, and therefore, vaccination status data were not systematically and uniformly collected.
For all evaluated patients, chest CT scans were not possible during the acute phase of COVID-19 hospitalization, as transportation was complicated during the health emergency, and there were significant isolation restrictions. While there is no healthy control group, this limitation is compensated for by comparing the same patients at different post-COVID follow-up visits.
Finally, it is not possible to conclude the resolution of long-term alterations beyond the 12-month evaluation; therefore, studies with more extended follow-up periods are required to identify if specific residual alterations, such as decreased CO diffusion, return to their reference values or, on the contrary, become a permanent sequela. The loss of follow-up of some patients and the lack of information for a few measurements do not affect the presented statistical estimates and their results.
Conclusions
In conclusion, in the population of patients who survived severe and critical COVID-19, residual radiological findings of the infection are observed up to 12 months after hospital discharge—additionally, evidence of a transition from a predominance of ground-glass opacities and consolidation to interstitial infiltrates. No alterations were observed in spirometry in either of the two visits conducted, and improvement was observed in all parameters studied at 12 months, regardless of abnormalities in diagnostic imaging. However, patients with this compromise may require more recovery time. CO diffusion did not normalize at 12 months, which has been established in multiple studies in SARS survivors and post-COVID-19 patients, suggesting a possible pattern related to this disease.
The findings suggest that most of the observed sequelae are transient, and lung function tends to normalize in the long term. However, more extended follow-up studies are needed to assess the behavior of the CO diffusion test. In contrast to what is described in the literature, the findings from the medical history and physical examination at 6 and 12 months were insignificant in our population. No significant alterations were found in the applied scales, with low scores recorded in both follow-up visits. As described in the literature, an improvement in the scores is observed at the last 12-month follow-up visit.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241275369 – Supplemental material for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America
Supplemental material, sj-docx-1-smo-10.1177_20503121241275369 for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America by Liliana Fernández-Trujillo, Juan Sebastián Galindo-Sánchez, Angie Cediel, Carlos A García, Eliana I Morales, Jessica Largo and María A Amezquita-Dussan in SAGE Open Medicine
Supplemental Material
sj-docx-2-smo-10.1177_20503121241275369 – Supplemental material for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America
Supplemental material, sj-docx-2-smo-10.1177_20503121241275369 for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America by Liliana Fernández-Trujillo, Juan Sebastián Galindo-Sánchez, Angie Cediel, Carlos A García, Eliana I Morales, Jessica Largo and María A Amezquita-Dussan in SAGE Open Medicine
Supplemental Material
sj-docx-3-smo-10.1177_20503121241275369 – Supplemental material for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America
Supplemental material, sj-docx-3-smo-10.1177_20503121241275369 for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America by Liliana Fernández-Trujillo, Juan Sebastián Galindo-Sánchez, Angie Cediel, Carlos A García, Eliana I Morales, Jessica Largo and María A Amezquita-Dussan in SAGE Open Medicine
Supplemental Material
sj-docx-4-smo-10.1177_20503121241275369 – Supplemental material for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America
Supplemental material, sj-docx-4-smo-10.1177_20503121241275369 for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America by Liliana Fernández-Trujillo, Juan Sebastián Galindo-Sánchez, Angie Cediel, Carlos A García, Eliana I Morales, Jessica Largo and María A Amezquita-Dussan in SAGE Open Medicine
Supplemental Material
sj-docx-5-smo-10.1177_20503121241275369 – Supplemental material for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America
Supplemental material, sj-docx-5-smo-10.1177_20503121241275369 for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America by Liliana Fernández-Trujillo, Juan Sebastián Galindo-Sánchez, Angie Cediel, Carlos A García, Eliana I Morales, Jessica Largo and María A Amezquita-Dussan in SAGE Open Medicine
Supplemental Material
sj-docx-6-smo-10.1177_20503121241275369 – Supplemental material for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America
Supplemental material, sj-docx-6-smo-10.1177_20503121241275369 for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America by Liliana Fernández-Trujillo, Juan Sebastián Galindo-Sánchez, Angie Cediel, Carlos A García, Eliana I Morales, Jessica Largo and María A Amezquita-Dussan in SAGE Open Medicine
Supplemental Material
sj-docx-7-smo-10.1177_20503121241275369 – Supplemental material for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America
Supplemental material, sj-docx-7-smo-10.1177_20503121241275369 for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America by Liliana Fernández-Trujillo, Juan Sebastián Galindo-Sánchez, Angie Cediel, Carlos A García, Eliana I Morales, Jessica Largo and María A Amezquita-Dussan in SAGE Open Medicine
Supplemental Material
sj-docx-8-smo-10.1177_20503121241275369 – Supplemental material for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America
Supplemental material, sj-docx-8-smo-10.1177_20503121241275369 for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America by Liliana Fernández-Trujillo, Juan Sebastián Galindo-Sánchez, Angie Cediel, Carlos A García, Eliana I Morales, Jessica Largo and María A Amezquita-Dussan in SAGE Open Medicine
Supplemental Material
sj-docx-9-smo-10.1177_20503121241275369 – Supplemental material for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America
Supplemental material, sj-docx-9-smo-10.1177_20503121241275369 for Six and twelve-month respiratory outcomes in a cohort of severe and critical COVID-19 survivors: A prospective monocentric study in Latin America by Liliana Fernández-Trujillo, Juan Sebastián Galindo-Sánchez, Angie Cediel, Carlos A García, Eliana I Morales, Jessica Largo and María A Amezquita-Dussan in SAGE Open Medicine
Footnotes
Acknowledgements
Not applicable.
Author contributions
All authors have read and approved the manuscript and significantly contributed to this paper. LF-T: Conception and design, manuscript writing and correction, and final approval of the manuscript. JSG-S: Literature review, data collection, analytic calculations, manuscript writing, result interpretation, and final approval of the manuscript. AC: diagnostic chest imaging interpretation and final approval of the manuscript. CAG: diagnostic chest imaging interpretation and final approval of the manuscript. EIM: Conception and design, manuscript writing and correction, and final approval of the manuscript. JL: analytic calculations, result interpretation, and final approval of the manuscript. MAA-D: Literature review, manuscript writing and correction, final approval of the manuscript.
Availability of data and materials
Datasets used and/or analyzed during the current study are available from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval and consent to participate
This manuscript was written in compliance with the ethical standards of the institutional ethics committee and with the 1964 Helsinki Declaration. We have the approval of the Ethics Committee in Biomedical Research from Fundación Valle del Lili. This is supported in letter No. 20 of 2020, which is available with the Corresponding Author if needed.
Consent for publication
A copy of each consent is available from the corresponding author where necessary.
Prior presentation of abstract statement
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
