Abstract
Quality has been a concern of the World Health Organization since its inception and is defined as fitness for use. Since our ancestors began trading several millennia ago, Falsified Pharmaceutical Products has been a recurring problem and still threatens economic stability and public health. Its definition various from country to country and according to World Health Organization, 2017, it is ‘a product that is deliberately and fraudulently mislabelled with respect to identity and/or source’. The implementation of anti-falsified nanomaterial technologies is the prominent preventive measure to track and/or detect Falsified Pharmaceutical Products. Software and hardware companies had made encouraging progress towards implementing Radio Frequency Identification devices for ensuring the authenticity of pharmaceutical products. The purpose of the review was to critically appraise Radio Frequency Identification devices technology for the purpose of track and trace Falsified Pharmaceutical Products circulating in the market. Different search engines such as Google Scholar, Science Direct and PubMed were applied and mesh terms and keywords were searched. Different guides and related books were investigated in addition to the articles. Radio Frequency Identification devices technology is a compact electronic device that contains a small chip and reader with antenna that enables wireless transmission of identity of pharmaceutical products. The authenticated Radio Frequency Identification devices model is used for pharmaceutical products’ authentication from origin of pharmaceutical industry to the pharmacy at any point along the chain of the distribution. Popular pharmaceutical products, such as OxyContin and Sildenafil Citrate, which are particular targets of falsification have mandated the use of Radio Frequency Identification devices technology.
Introduction
During the last several decades, the quality of pharmaceutical products has not been a matter of much concern to prescribers, pharmacists or consumers. 1 Customers were simply taking it by relying on the health professionals and in turn, heath care practitioners serve their customers in the behave of priding various pharmaceutical plants. Although the recent rise in internet purchasing may fuel a resurgence of these problems, currently consumers expect and rely on a high level of quality from the drug products they purchase. The reason is that pharmaceutical product manufacturers were closely regulated industrial activities.1,2 The quality of pharmaceutical products was a concernment of the World Health Organization (WHO) since its inception. The setting of global standards was requested in WHO constitution; one of the responsibilities was to develop, establish and promote international standards with respect to pharmaceutical products. The government allocates a substantial budget to pharmaceutical products. The proportion higher in developing countries; it may exceed 40%. 3
Food and Drug Administration (FDA) began an initiative to address current Good Manufacturing Practices (CGMPs) for the 21st century around 20 years ago. This effort involved taking a new look at both the regulatory and industrial systems for ensuring drug quality. The initiative highlighted the need for major changes in both the regulators and the pharmaceutical industry’s approaches. One central, and not yet fully resolved, issue was the need for a consensus definition of pharmaceutical quality for regulatory purposes.1,4 Such a definition was much harder to achieve than it might seem at first glance. According to Woodcock, literatures do not offer a consensus definition of quality of any pharmaceutical products or services. Nevertheless, a concept common to most definitions involves meeting or exceeding customers’ needs. 1
The quality of pharmaceutical products was not an unfortunate; it must be the result of supersmart efforts of responsible bodies. The pharmaceutical products’ quality was essential to assure the maximum level of patient’s satisfaction. The criteria for their qualities of any pharmaceutical products in dosage form are its efficacy, potency, safety, stability, patient acceptability and regulatory compliance. 5 Cases where consumers are unable to sort out value for themselves are termed market failures by economists. For people taking medicines, however, more is at stake than just their money: their health or even their lives could be jeopardized by a drug quality problem. 1
In the pharmaceutical industry scenario, quality of pharmaceutical products has proposed ‘fitness for use’; the pharmaceutical product befitted its pre-specified quality attributes or regulatory specifications. 1 Pharmacopoeias provide standards, specifications and test methods that are expected to be used in the pharmaceutical industry to ensure the quality of pharmaceuticals. 5 For instance, US Pharmacopoeia (USP) monographs contain compendia assays for evaluating the quality of marketed pharmaceutical products. It had also been stated that a drug manufactured in compliance with CGMPs was a good-quality product.1,3 Furthermore, regulatory bodies were continually developing their requirements to ensure the safety, quality and efficacy for pharmaceutical development and manufacturing. 5
Now a days, there was an increment of globalization of pharmaceutical products’ manufacturing. The products were manufactured here and there at different regions and countries, and they move on a number of corridors of the international market 6 ; the pharmaceutical market hence had different standards of quality. Some National Medicine Regulatory Authorities (NMRA) were unable to assure the quality of pharmaceutical products circulating in their territory and the vulnerable customers might be exposed to the poor-quality products.7,8 Thus, the current pharmaceutical market situation had the potential to quickly infiltrate these poor-quality pharmaceutical products anywhere before appropriate detection were possible. 9 There are two main categories of poor-quality pharmaceutical products: substandard and falsified.6–9
Falsified pharmaceutical products
Pharmaceutical falsification had become a big problem for public health and economy. 10 It results in major financial losses, not only to manufacturers but also reduces the general welfare. 11 Since ancestors began trading several millennia ago, Falsified Pharmaceutical Products (FPPs) had a recurring problem, with history punctuated by crises on anti-malarials; fake cinchona bark in the 1600s and fake quinine in the 1800s. 9 FPPs is a substantial problem that is growing worldwide and affects most countries. 12
FPPs is defined differently in different countries. The definitions used in the various WHO member states show that the nature of the problem of these medicines varies from country to country. The first international meeting on FPPs was held by WHO in 1992 at Geneva, Switzerland. The purpose was to assist member states of WHO in efforts to prevent the infiltration of FPPs. 13 Hence, several countries came with their own protocols as to what constitutes a FPPs, and there was no consensus. 14 FPPs can be described as a medicine that is presented in a way to look like the original medicine, although it is not. In legal terms, it is trademark infringement.
According to Black’s law dictionary, FPPs used to describe а medicine made by someone other than the original manufacturer, by copying or imitating an original product without authority or right, with the intention to deceive or defraud and then marketing the copied, counterfeited or forged medicine as the original. 15 Furthermore, according to WHO, 2017, FPPs is defined as ‘a product that is deliberately and fraudulently mislabelled with respect to identity and/or source’. 16 They include ‘products without the active ingredient, with an insufficient quantity of the active ingredient, with the wrong active ingredient, toxins or with fake packaging’.6,9,16 The amount of active ingredient does not provide sufficient information to accurately determine if a medicine is falsified.
Many classes of drugs have been falsified, and several factors are associated with it: poor regulation, a higher number of intermediaries, lack of awareness, and many more. Pharmaceutical falsification will likely increase in the future as health care costs continue to rise globally. 17 The use of FPPs frequently results in treatment failure and in worse scenario lead to death.5,6,9–11,14,16 Fore instances, drugs having high stoke turn rates have high potential to deceive like sildenafil citrate18–20 and oxycontin.21–24
Anti-falsified pharmaceuticals’ technologies
The anti-falsifying measure includes strong regulatory control with analytical techniques like nanomaterials that allow the protection of the drug supply chain from FPPs.17,25 The security of the pharmaceutical supply chain can be strengthened by innovative identification and packaging technologies which were difficult to copy. 26 FPPs can lead to drug recalls and liability suits. In addition, brand loyalty is compromised as consumers perceive additional risks when using a company’s products. An effective anti-falsifying strategy avoids such issues in order to ensure patient safety. 27
Principle ways of combating pharmaceutical falsification includes legal actions on illicit drug smugglers, customer education/information, private investigations, cooperation with enforcement agencies and counter-measures using analytical technologies.6,10,14,17,27 The implementation of anti-falsifying technologies is the prominent preventive measure.10,17,27 In addition to providing authentication, they make the production of a convincing copy of a drug more difficult and costly. 26
The ideal anti-falsifying technology need to fulfill good security level (non-cloneable), higher product authentication ability, proven standards, difficult to remove and reapply, easy to check, have automatic authentication and legally compliant with the pharmaceutical manufacturer. 28 The FDA recommends the use of multiple, periodically changing, authentication measures on a product specific basis. 29 Software and hardware companies made positive strides in 2006 towards the deployment of Radio Frequency Identification (RFID) devices to guarantee the authenticity of pharmaceutical products provided to consumers via retailers.10,30 The purpose of the review was to critically appraise RFID technology for the purpose of track and trace FPPs circulating in legal market.
Method
This study is a review which aimed to determine the role of RFID technology in FPPs detection. In this regard, different search engines such as Google Scholar, Science Direct and PubMed were applied and mesh terms and keywords like ‘quality, falsified medicine, counterfeit medicine, radio frequency, Radio Frequency Identification and RFID technology’ were searched. Different guides and related books were investigated in addition to the articles. Descriptions, characteristics and applications of RFID technology were mentioned in most articles.
Radio frequency identification technology
Radio frequency method
Radio-analytical chemistry is about the use of radioactive nuclides and nuclear radiation for analytical purposes. It depends on type and energy of radiation emitted. 31 The field of Radio Frequency (RF) nano-electronics focuses on the fundamental study of devices that are enabled by nanotechnology and operate within a frequency range from about 100 MHz to 100 GHz. This range includes frequencies identified as radio frequencies, 32 but different sources specify different upper- and lower-bound ranges. 33 In the past few decades, the emergence and growth of nanotechnology have proceeded hand in hand with the discovery and investigation of new forms of matter as nano-scale material systems, spanning from atomically two-dimensional materials to nanowires to individual atoms. 32
RF refers to an oscillation rate of an alternating electric current/voltage or electromagnetic field. It usually refers to electrical rather than mechanical oscillations.31,33 The ultimate goal of RF nano-electronics is to leverage the new materials and new phenomena that have been revealed by scaling down to the nanoscale world in order to investigate new RF devices that will be of interest both for fundamental study and eventual commercial application. 32
Radio waves belong beyond infrared and/or microwaves. The analytical method-based radio wave is subject to a great deal of research and lead to more effective innovative technology. Aviation, broadcasting, mobile telephony, navigation, defense and other technologies were based on radio spectrum.32,34 RF electromagnetic energy is part of natural environment, emitted by sources like the Sun, Earth and the Ionosphere. Other sources of RF electromagnetic energy include microwave ovens, radar, specific industrial uses and various medical applications. There are continually emerging technologies that use RF electromagnetic energy particularly in telecommunications and there is some concern of potential health effects which is not fully alleviated by existing scientific data. 33 RFID device is hence an integral part of life that needs radio technology in order to communicate digital information between a stationary location and a movable product.10,35
Radio frequency identification synopsis
RFID refers to a small electronic device which contains a small chip and an antenna that enables wireless transmission of identity.10,36 It is a technology for tracing and tracking pharmaceutical products using a small radio transmitter and a reader. 37 Moreover, RFID refers to the application of tag using radio waves applied to pharmaceutical product for the purpose of pharmaceutical products’ identification. 36 Just the tag is dipped in the pharmaceutical product packaging and the products move on supply chain of the international market which contains information on label.
RFID become an automatic technology that utilizes RF waves to transmit data between reader and tag to identify, track and/or locate the pharmaceutical product anywhere. 38 This device provides a unique identification of the product and is a contactless smart card which does not require physical contact between the scanner and the tagged item. 39
The RFID tags had stored detail information about the pharmaceutical products that need to be tracked.30,37 Depending on the transmitter and reader, these tags can be utilized to scan and read up to a long distance.37,40 It can be either implanted or used as a wristband that stores crucial data.
History
Understanding in science advanced slowly until the 1600s. Meanwhile, explosion of observational knowledge of electricity, magnetism, and optics increased between the 1600s and 1800s. 35 The history of recognition via radio wave technology goes back to M. Faraday’s discovery that ‘radio wave is a form of energy’.35,41 Furthermore, Scottish physicist J.C. Maxwell revealed his electromagnetic theory in 1864. In addition to creating radio waves, German physicist H.R. Hertz confirmed Maxwell’s electromagnetic theory in 1887. Hertz is credited as the first to transmit and receive radio waves. 35
The birth of radar in the early 20th century was considered as significant technical development during World War II. Radar sends radio waves to locate and detect through the reflection of these waves. The position and speed can be ascertained by looking at this reflection. Since the military recognized the importance of radar, many of the early advances had shrouded in secrecy. 35 In 1946, L. Theremin invented a tool for the government of the Soviet Union that was able to transfer the radio waves caused by any events in the form of sound to the desired location. These sound waves would translate reflection of radio waves into an understandable language by mobilizing the diaphragm that was connected to a vibrator device. 41
Another technology that was very nearly similar to RFID was named Identification Friend or Foe (IFF). 41 IFF was invented in Britain in 1939 and was used as an efficient tool in World War II in order to make it easier to identify German aircrafts as enemy aircrafts (Figure 1).41,42 L. Theremin’s tool was considered as the first RFID-based device, but some of the sources believe that RFID technology was common among the experts since 1920 and has been completed in 1960s–19705.10,35,41

Identification friend or foe (IFF) device. 41
Basic components
RFID enhances item predictability for specific operations and aids in the recognition of contextual knowledge. However, research on RFID components is required before utilizing this technology in the health sector. 43 Different components of RFID (tag, antenna and reader), their working principles (Figure 2) and a brief description of each component is given as follows.

Working of different components of RFID. 41
Tag
RFID tag, or transponder, is programmed with unique information by electronic means, and it has to be read by an antenna.30,36,44 It is the fundamental component of the RFID system and is individually identifiable to its own unique identification number system. The internal memory of the tag stores the distinctive identification numbers.43,45 Every RFID tag (smart label) could contain 96 bits of information and a 40 bits serial number. 44
RFID tag in its most simplistic form comprised two parts: an antenna for transmitting and receiving signals and chip or integrated circuit (IC) which stores the tag’s ID and other information. RFID tags are affixed to items in order to track them using RFID reader and antenna.36,46 The main purpose of IC is to transmit the tag’s unique identifier. 47
RFID tags are of three types of – passive, semi-passive and active.43,44,48 However, depending on the modes of storage of data, the tags can be of three types: Read Write (RW), Read Only (RO) and Write Once Read Many (WORM). In RW type, the data can be added or overwritten. In RO tags, the data can be stored at the time of make and later the data cannot be added or overwritten. In the WORM type, the data can be added once and overwriting is not possible later.43,47,49
Active tags: They are more sophisticated. They are larger and costlier to produce; possess an internal battery that is used to run the microchip’s circuitry and to transmit a signal to the reader. Active tags have a stronger signal and are more reliable than passive tags as they can conduct a session with a RFID reader.47,50 They operate generally at higher frequencies. The signals are captured by the reader over a longer distance. 50 Because of onboard power supply, the active RFID tags transmit the signals at higher power levels than the passive tags due to which they are more effective in RF challenged conditions. 51 Practical ranges for the active RFID tags are hundreds of meters. They have an extended memory and have about 10 years of battery life. 52
Passive tags: Internal power supply is not included within the passive RFID tags and hence they depend only on the RFID reader to transmit data. They are also known as pure passive, reflective or beam powered tags. 47 The reader supplies the electromagnetic waves to produce a current in the antenna of the tags. Then, the tag reflects the RF signal and produces information by modulating the reflected signal to the reader. In most passive tags, a small electric current produced in the antenna by RF signal provides power to transmit data. They operate comparatively at lower frequencies with a lower capturing range.47,51 Passive tags communication signal is based on the power it gets from antenna as well. 43 Due to lack of onboard power supply, RFID passive tags are quite smaller 47 so that they can be embedded in a sticker or under the skin and are generally used for relatively short distances. The major advantage of passive tags is that they operate without a battery and hence, they are much less expensive and much smaller in size than active ones. But they have also drawbacks like short distance for reading the tag and requirement of a higher-powered reader.
Semi-passive/semi-active tags: Both types of tags contain power supplied on board. The main difference is how the battery is used. Batteries in semi-passive tags are only used to power the internal circuitry. They still need to be presented inside the near filed in order to absorb power for data transmission between readers and themselves. 47 Semi-active tags are identical to the active tags as they also have an inbuilt power supply; but semi-passive tags do not transmit a signal until the reader transmits the signal first. 53
Reader
The RFID readers (also called interrogators) are the system’s brain and is required for all systems to operate. They are devices that transmit and receive RF waves in order to communicate with tags (fixed or mobile).44,46,47
Antenna
RFID antennas are necessary elements in order to convert the RFID reader’s signal into RF waves that can be picked up by tags. Without some type of antenna, whether integrated or standalone, the reader cannot properly send and receive signals to tags.44,46 Unlike readers, antennas are dumb devices that receive their power directly from the reader. When the reader’s energy is transmitted to the antenna, the antenna generates RF field and subsequently, RF signal is transmitted to the tags in the vicinity. The antenna’s efficiency in generating waves in a specific direction is known as the antenna’s gain. To put it simply, the higher the gain, the more powerful and further reaching RF field an antenna will have.44,46,47
Antenna is attached to a microchip which emits the radio signals to activate the tag and to read & write the data. This assembly is usually enclosed within a protective layer which is determined by the type of application. A transceiver with decoder is used to retrieve the data stored on the RFID tag. This decoder decodes the data encoded in the IC of tags. These data are then passed to the host computer for processing. 44 The RFID antenna gives off RFID waves along a horizontal or vertical plane, which is described as the antenna’s polarity. If the RF field is a horizontal plane, it is described as horizontally linear and the same principle applies to an RFID antenna that creates a vertical plane.44,46,47
Principle of operation
Tag or transponder device contains an electronic circuit that is attached to an object which needs to have an identification code. 41 When the tag is placed near or within the reader area, the magnetic field generated by the reader activates the tag. Then, the tags continuously send data through the radio pulse. Finally, data received by the reader will be processed by a computer system or related software such as Enterprise Resource Planning (ERP) and Supply Chain Management Systems as shown from Figure 3.37,41 Identifying items through RFID technology is more accurate and involves less work than using barcode scanning and manual processes, as they do not need line of sight; the reader can access the information on the tag without touching it as long as it is within a range and can scan several items simultaneously. As well, unlike barcodes, the information held on an RFID tag can be changed or updated.37,45

RFID operation system.
In general, the RFID system consists of an antenna to scan information, a receiver with a decoder to interpret the information and a transmitter where the information is programmed. The antenna sends radio signals in a short range. Radio emissions establish a communication platform with the sender (tag) and send energy to the tag in order to communicate. When a tag or RFID card passes through working range of an antenna, it detects the activation signal sent by an antenna. This signal activates RFID internal ICs and as a result, the information on tag will be sent to reader. 41
As shown above, both figures explain RFID working process. Tag (three types) is attached to the object/medicine to be checked with identification code. When the tag is placed near or within the reader area, the antenna scans and the magnetic field activates the tag. Then, the tags continuously send data. Finally, data received by the reader is processed by a computer system.37,41
Applications on falsified pharmaceutical product detection
Nowadays, the world faces a growing public health problem with FPPs infiltrating its legitimate drug supply chain and being sold at the full price commanded by legitimate drugs in retail pharmacies and hospitals. 54 For instance, in United States and China, FPPs found in the regular market includes Lipitor®, Procrit®, Viagra® and Zyprexa® in which they usually produce high volume and retail at a high cost. 55 A system wide challenge faced by both pharmaceutical manufacturers and retailers involves determining the product custody at various levels along the supply chain. The complex pharmaceutical distribution infrastructure ensures difficultly in maintaining supply chain integrity as products move from the manufacturer to the point of sale. 56 The FDA has stepped up its efforts to improve the safety and security of the national drug supply by encouraging companies to use electronic technology to tag product packaging. Such technology may prevent shipment diversions and falsifying of pharmaceutical products because it can enable wholesalers and pharmacists to determine packaged drug products’ identities and dosage throughout the entire supply chain.29,57
There are other methods for identifying products as authentic. The good method has been RFID used to identify FPPs or potentially other information on a microchip attached. This data are then transmitted into a computer that can retrieve the data and match the item’s serial number. 58 The authenticated RFID model is expected to support item level authentication from origin to the pharmacy as point of sale and at any point along the chain of custody between the beginning and end points in the distribution chain. 56 RFID represents a form of wireless identification which provides an effective tool for tracking moving objects throughout the manufacturing and shipping process. The chips allow manufacturers and distributors more precise on tracking of their drug products throughout the supply chain. 58 RFID tags are so tiny that consumers cannot clearly see them.36,37,44,47,59 RFID’s capability to distinctively recognize each object and firmly capture information without line of sight has many benefits in the pharmaceutical industry including insight, visibility and efficiency, accountability and brand protection, product safety, recalls and regulatory requirements and reducing falsification. 60
In recent globalization, RFID technology has been advancing and widening its application fields by penetrating into the various industries with the benefits of efficiency, costs saving and fast processing.10,60 As a result, nowadays it is widely used in logistics, inventory, security, supply chain, manufacturing and retails industries. 60 According to the literatures used in this review, RFID technology has become the leading candidate for the tracking of the authenticity of drugs. A complete RFID serialization requires a costly infrastructure and some huge and rich pharmaceutical companies use this technology to track those drugs which are highly susceptible to falsifying. 10 For instance, Johnson & Johnson has emerged as a leader among pharmaceutical companies by establishing standards for RFID technology and participating in RFID pilot studies. The company employs RFID to comply with retailers’ mandates that certain products be shipped with RFID tags. Johnson & Johnson has performed tests using RFID tags to track promotional display shipping and recently employs the technology with surgical implements. 10 The other company using RFID for tracking drugs is Pfizer. 61 Pfizer began tagging bottles of Viagra® for US distribution with RFID prior to 2009. In actually by 2006, Pfizer had already spent $5 million to implement RFID with Viagra®.10,57,60–62
Pharmaceutical products are the main target of falsifiers. RFID is being considered as an important tool to fight against FPPs as shown in the in Figure 4. 63 The drug’s electronic Pedigree (ePedigree) within the chain of custody from the point of manufacture to the point of sale and the integrity of the manufacture and distribution of the pharmaceutical product are maintained.63,64 A drug pedigree is an electronic file that records the details of distribution of a prescription drug from its manufacture through wholesale transactions, until it is received by the dispenser.17,64 RFID is known for its wonderful ability to uniquely identify each item. Sales of FPPs are predicted to explode if it is not dealt with effectively and rapidly. RFIDs have ability to document the sale and transfer of a drug through every step of the supply chain, from the manufacturer to the retail outlet. Popular drug products such as OxyContin and Sildenafil citrate which are particular targets of falsifiers have mandated the use of RFID technology.10,63 In Pfizer’s RFID enactment, the tags are placed on drug packages. The authenticity of the product can be verified by scanning the RFID serial number of a particular package and confirming its authenticity with Pfizer directly, using their personal authentication.61,63
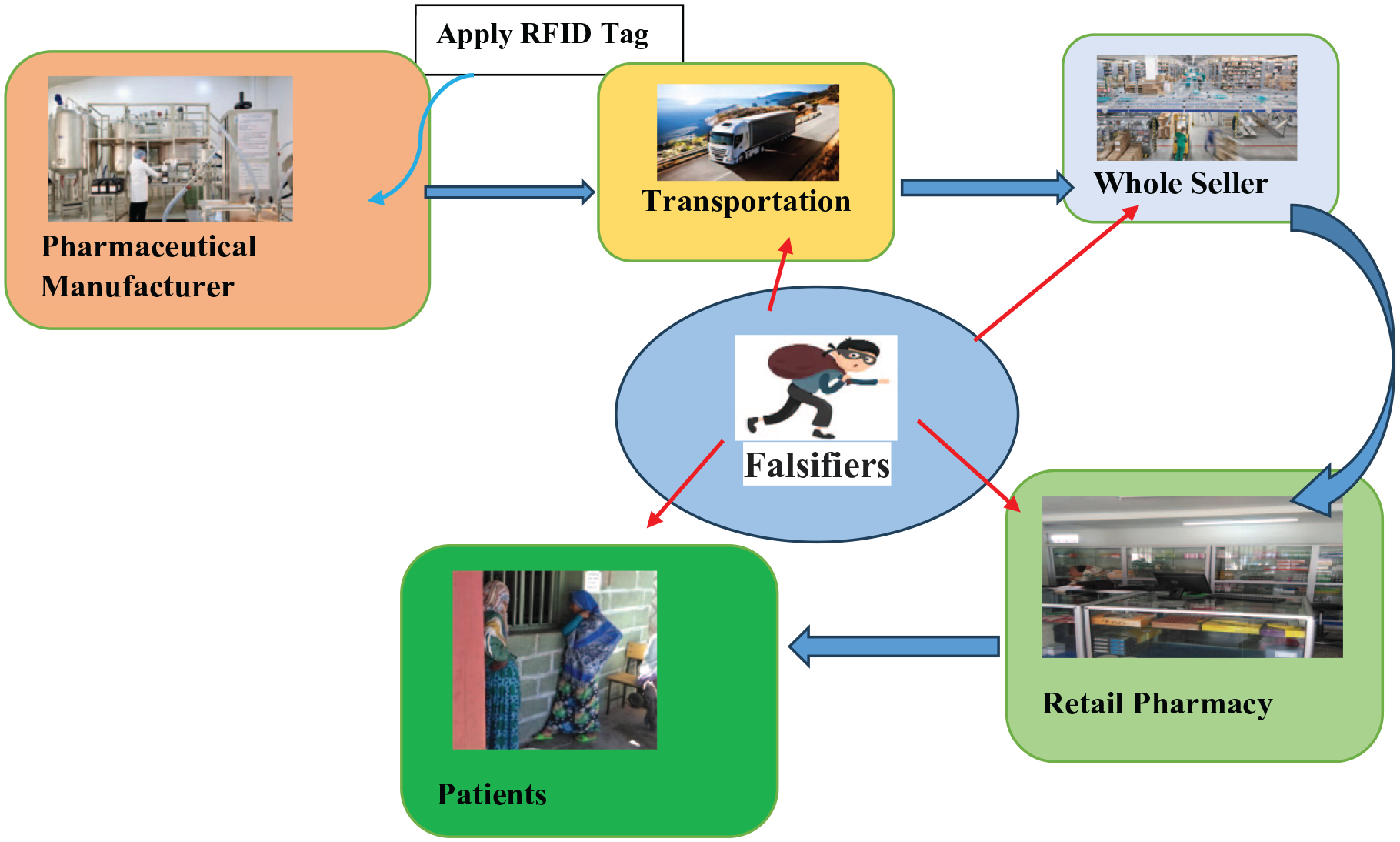
Detection process of FPPs using RFID system. Initially, the pharmaceutical manufacturers market different pharmaceutical products with varies batches containing unique bare codes. Then, the products are distributed to the wholesalers, retailers and patients locally and abroad; from every distribution drug supply chain falsifies could infiltrate FPPs in the market which is difficult to detect visually. Hence, if drug recall is there, scan with RFID tag and if it matches, the product is authentic and if not, manufacturer is not responsible.
The drug supply chain management cycle is complex so that FPPs can enter into market. In each step, it can be tracked by scanning RFID tag to obtain data inside the package. This data are then transmitted into a computer that can retrieve the data and match the item’s serial number at point of transaction. The product can pass if and only if it matches with manufactures serial number.10,17,61,63
RFID provides facility to the organizations to recognize and control their assets (pharmaceutical products). It has a wide range of applications in product tracking, starting from the production of the goods in the plant to postsales of the goods. The mobilization of pharmaceutical products may be regulated by RFID access control. 64 RFID tags can be applied on the commodities which are frequently lost or stolen. They can also be applied to those which are under maintenance. RFIDs also ensure the expiration dates and improve expiration management. It lessens the time spent in the recognition of products considered for recall. 65 The drug distributors can get an idea about the availability of drugs in their stores with the help of RFID systems and hence, they can have stocks of important drugs for emergency situations. If any person steals the product and leaves the manufacturing unit, then stolen goods could be traced even from outside the unit.65,66
During transport, if the products are not handled properly, they get spoiled. In such cases, the facilities have to maintain perishable inventory control to prevent additional cost of replacement and retain the goodwill of consumers. By using RFID technology for product identification, it is easier to assure the quality of such moving objects. The RFID system assists to follow well-defined issuing policies for the products depending on the requirements; for example, first-in-first-out policy for vegetables, and last-in-first-out policy for blood products. 67
Future perspective
This review studies the architecture of a RFID system to detect and combat pharmaceutical falsification. It is also better to predict the systems’ trend in the near future. RFID is a promising technology that, if efficiently implemented in drug supply chain cycle, will result in numerous benefits, including physicians, healthcare providers, patients and several other parties involved in the delivery and administration of healthcare services. However, several challenges still remain that must be addressed before RFID technology can be ubiquitously deployed in healthcare services.
The Electro Magnetic Interference (EMI) caused to the electronic biomedical devices by RFID wireless transmissions could critically affect their performances. Without careful investigations of the impact of such interferences, the introduction of RFID into detection of FPPs might threaten patient’s safety. Indeed, the EMI generated by RFID tags and readers can potentially disrupt the performance of electronic medical equipments. 68 A syringe pump or an external pacemaker that switches off in the proximity of RFID devices would directly endanger a patients’ health. In some cases, the EMI is due to different modulation rates. In addition to updating safety standards for electronic medical equipment, power control mechanisms should be considered as a solution to minimize the impact of EMI. 69
The main challenging issue in RFID information delivery is to guarantee security and privacy.70,71 Efficient authentication algorithm is needed to ensure the security of smart RFID systems, but the privacy issue is also important. For example, another malicious person may activate the RFID tag attached to a patient, access the electronic medical record and do something adverse to him/her. Thus, the code should only be delivered to trusted RFID readers. Basic RFID tags are vulnerable to simple counterfeiting attacks. They are also prone to be relayed or man-in-the-middle attacks. 72
RFID tags, by default, communicate with all surrounding devices, both good and malicious. Since they automatically respond to the interrogation from the reader without alerting the bearer/owner, a person carrying an RFID tag is prone to clandestine physical tracking. In addition, in supply chain applications, individually tagged objects in stores allow competitors to learn about the stock turnover rates (inventorying). 70
Since RFID tags have a small amount of storage and processing capabilities, innovative and efficient RFID authentication techniques are needed. 68 The issue of limited computational resources still needs further investigation to develop novel solutions that can implement highly efficient, secure solutions. In such a limited environment, it is important to have a clear and precise assessment of the security attacks that can be addressed. 71 Thus, in the near future, scientists could probably witness several RFID tags technology platforms for detection of FPPs.
Limitation
This review had limitations concerning the search strategies of the different literatures. There was no pre-defined question or search strategy. There were also some old published book chapters as reference. They lack structure and adhere to no set procedure. The reviewers will gain knowledge about the issue and have a thorough grasp of the state of the science.
Conclusion
In conclusion, during the last several decades, the quality of pharmaceutical products has not been a matter of much concern since pharmaceutical manufacturing is the more closely regulated industrial activities. Globalization had the potential to rapidly spread FPPs worldwide. The implementation of anti-counterfeit RFID technologies is the prominent preventive measure for ensuring the authenticity of drug products provided to consumers through retailers. RFID is new technology to track and/or detect pharmaceutical falsification. Popular drug products such as OxyContin and Sildenafil Citrate which are particular targets of counterfeiters had mandated the use of RFID technology.
Footnotes
Acknowledgements
We would also like to acknowledge all the authors of the references that we used. We are also ready to thank College of Medicine and Health Sciences, University of Gondar.
Author’s contributions
MCW contributed in designing, conceptualization and manuscript preparation. TD, MGF and YKA contributed in data extraction, in revision and had edited the drafted manuscript. All authors have agreed to journal to which the review be submitted.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Ethical consideration
Not applicable.
Consent for publication
Not applicable.
