Abstract
Objective:
To observe the efficacy of haploidentcial peripheral blood stem cell transplantation combined with a single unrelated cord blood unit for severe aplastic anemia patients with donor-recipient ABO incompatibility.
Methods:
This was a retrospective cohort study and data of 57 severe aplastic anemia patients underwent haploidentical stem cell transplantation from August 1, 2018 to February 28, 2022 in the First Affiliated Hospital of Xi’an Jiaotong University was retrospectively analyzed. All patients were divided into two groups, the donor-recipient ABO matched group (bone marrow+peripheral blood group) using haploidentical bone marrow and peripheral blood stem cells as grafts, and donor-recipient ABO mismatched group (cord blood+peripheral blood group), using unrelated cord blood and haploidentical peripheral blood stem cells as grafts. The differences of hematopoietic reconstitution, acute and chronic graft-versus-host disease, Cytomegalovirus (CMV) and Epstein-Barr virus (EBV) infection, and overall survival between the two groups were compared.
Results:
There were 30 cases in cord blood+peripheral blood group and 27 cases in bone marrow+peripheral blood group. One patient in bone marrow+peripheral blood group had primary graft failure, while other patients were successfully implanted. There were no significant differences of neutrophil and platelet recovery rates between two groups. The erythrocyte recovery time of cord blood+peripheral blood group was slower than that of bone marrow+peripheral blood group (
Conclusion:
These findings suggest that haploidentical peripheral blood stem cell transplantation combined with a single cord blood unit may be an alternative option for severe aplastic anemia patients with donor-recipient ABO incompatibility.
Keywords
Introduction
Allogeneic hematopoietic stem cell transplantation (allo-HSCT) is an effective treatment for severe aplastic anemia (SAA). Especially for young SAA patients without matched sibling or unrelated donors, haploidentical hematopoietic stem cell transplantation (haplo-HSCT) is an alternative selection. 1 The grafts of haplo-HSCT with the classic Beijing protocol for SAA are bone marrow stem cells combined with peripheral blood (PB) stem cells, and the incidence of graft versus host disease (GVHD) with bone marrow and PB stem cells is lower than that with PB stem cells alone.2,3 Although ABO incompatibility is not considered as a contraindication in allo-HSCT, its impacts on the clinical outcomes are still doubtful. Some reports that there was a correlation of slower neutrophil engraftment with major ABO mismatch and there was a correlation of delayed erythrocyte engraftment with ABO mismatch transplantation. Minor ABO incompatibility in patients with SAA had been reported to increase the risk of grade III–IV acute GVHD.4,5 While some reports that there was no impact of ABO incompatibility on neutrophil and platelet engraftment, and ABO incompatibility did not seem to have a significant effect on GVHD, relapse rate, transplantation related mortality and overall survival (OS). 6 For ABO incompatible, can we use unrelated cord blood (CB) instead of bone marrow stem cells? There were rare reports about umbilical CB combined with haploidentical PB stem cell transplantation for SAA patients with ABO incompatible. This article aims to observe the efficacy of CB combined with PB stem cells transplantation for SAA patients with donor-recipient ABO incompatibility.
Methods
Patients
This was a retrospective cohort study and patients with SAA who received haplo-HSCT based on the “Beijing Protocol” 7 at the First Affiliated Hospital of Xi’an Jiaotong University from August 1, 2018 to February 28, 2022 were included. Patients diagnosed with congenital bone marrow failure disease, patients who underwent allo-HSCT prior to this study, and patients who had a matched sibling or matched unrelated donor were excluded. All enrolled patients were divided into two groups based on donor-recipient ABO compatibility, the donor-recipient ABO matched group (BM+PB group) using haploidentical bone marrow and PB stem cells as grafts, and donor-recipient ABO mismatched group (CB+PB group), using unrelated CB and haploidentical PB stem cells as grafts. This study was approved by the ethics committee of the First Affiliated Hospital of Xi’an Jiaotong University (No. XJTU-2021N23).Written informed consent was obtained from all adult patients or legally authorized representative of underage patients in accordance with the principle of the Declaration of Helsinki.
Conditioning regimen
All patients were treated with fludarabine, rabbit anti-human thymocyte immunoglobulin, cyclophosphamide-based conditioning regimen consisted of fludarabine (30 mg/m2/day intravenously (i.v.) on days −5 to −2), cyclophosphamide (30 mg/kg/day i.v. on days −5 to −2), rabbit anti-human thymocyte immunoglobulin (thymoglobulin, Genzyme, Polyclonals S.A.S, Lyon, France) (2.5 mg/kg/day i.v. on days −5 to −2). For SAA patients with paroxysmal nocturnal hemoglobinuria cloning, gene mutations, chromosomal abnormalities, busulfan 3.2 mg/kg/day i.v. on days −7 to −6 was added.
GVHD prophylaxis and treatment
GVHD prophylaxis consisted of cyclosporin A (CSA) and a short course of methotrexate (MTX) plus mycophenolate mofetil (MMF). CSA was administered at a dose of 3 mg/kg/day on day −7 and then adjusted dosage according to the concentration and tapered within 12–18 months post-transplantation in the absence of GVHD. Intravenous MTX were given at 15 mg/m2 on day +1 and 10 mg/m2 on days +3, +6, and +11. MMF was administered orally (30 mg/kg/day for children and 1 g/day for adults) from days −1 to + 30 and a half dose was subsequently administered for 15 days. The first-line treatment for acute GVHD was intravenously administration of 1–2 mg/kg/day methylprednisolone, while the second-line treatment for steroid-refractory acute GVHD consisted of CD25 monoclonal antibody and ruxolitinib. Treatment of chronic GVHD included corticosteroids, ruxolitinib and MMF.
Stem cell collection and infusion
Recombinant human granulocyte colony-stimulating factors were subcutaneously injected on days −4 to 0 (10 μg/kg/day) to mobilize stem cells. Recipients were infused with grafts on one or two consecutive days. For BM+PB groups, on Day 1, the bone marrow stem cells were infused, while PB stem cells were infused on Day 2. For CB+PB groups, the unrelated CB unit was infused into recipient 8 h before the PB stem cells infusion on Day 1. The CB unit were obtained from the CB banks of Shandong Province in China, and the ABO blood type of CB was consistent with that of recipients and there were three human leukocyte antigen (HLA) loci matched with the recipients.
Engraftment criteria
Neutrophil engraftment was defined as the first day that the neutrophil count remained ⩾0.5 × 109 cells/L for three consecutive days. The first day that a platelet count of ⩾20 × 109 cells/L and hemoglobin was more than 80 g/L for seven consecutive days without platelet and red blood transfusion were defined as the platelet and erythrocyte engraftment time.
EBV and CMV infection
CMV and EBV were screened by polymerase chain reaction weekly after neutrophil recovery, bi-weekly until 100 days post-transplantation, and monthly thereafter. Acyclovir was administered for CMV prophylaxis or ganciclovir for pre-emptive treatment pre-transplantation. Pre-emptive therapy was started when two consecutive test results of CMV or EBV-DNA quantification were positive and then withdrawn after at least two negative tests.
Evaluation parameters and follow-up
The evaluation parameters included engraftment time, CMV and EBV infection, and the severity of acute and chronic GVHD. All the patients were followed up until February 2024. OS was also observed.
Statistical analysis
The expected day 30 engraftment rate was 85% in BM+PB group and 98% in CB+PB group. Thus, to have a power of 90% to detect the difference of 13% significantly, the number of patients needed would be 54. Taking into account a potential dropout rate of 5%, our targeted sample size would be 57. Engraftment times were compared by Mann–Whitney tests and baseline characteristics, GVHD occurrence rate, and relapse rate were compared by χ2 tests. OS was estimated by Kaplan–Meier analysis. The data were analyzed using SPSS for Windows, Version 19.0 (SPSS Inc., Chicago, IL, USA).
Results
Patient characteristics
Fifty-seven patients were enrolled at the First Affiliated Hospital of Xi’an Jiaotong University from August 1, 2018 to February 28, 2022 and were divided into the donor-recipient ABO matched group (BM+PB group,
Characteristics of SAA patients.
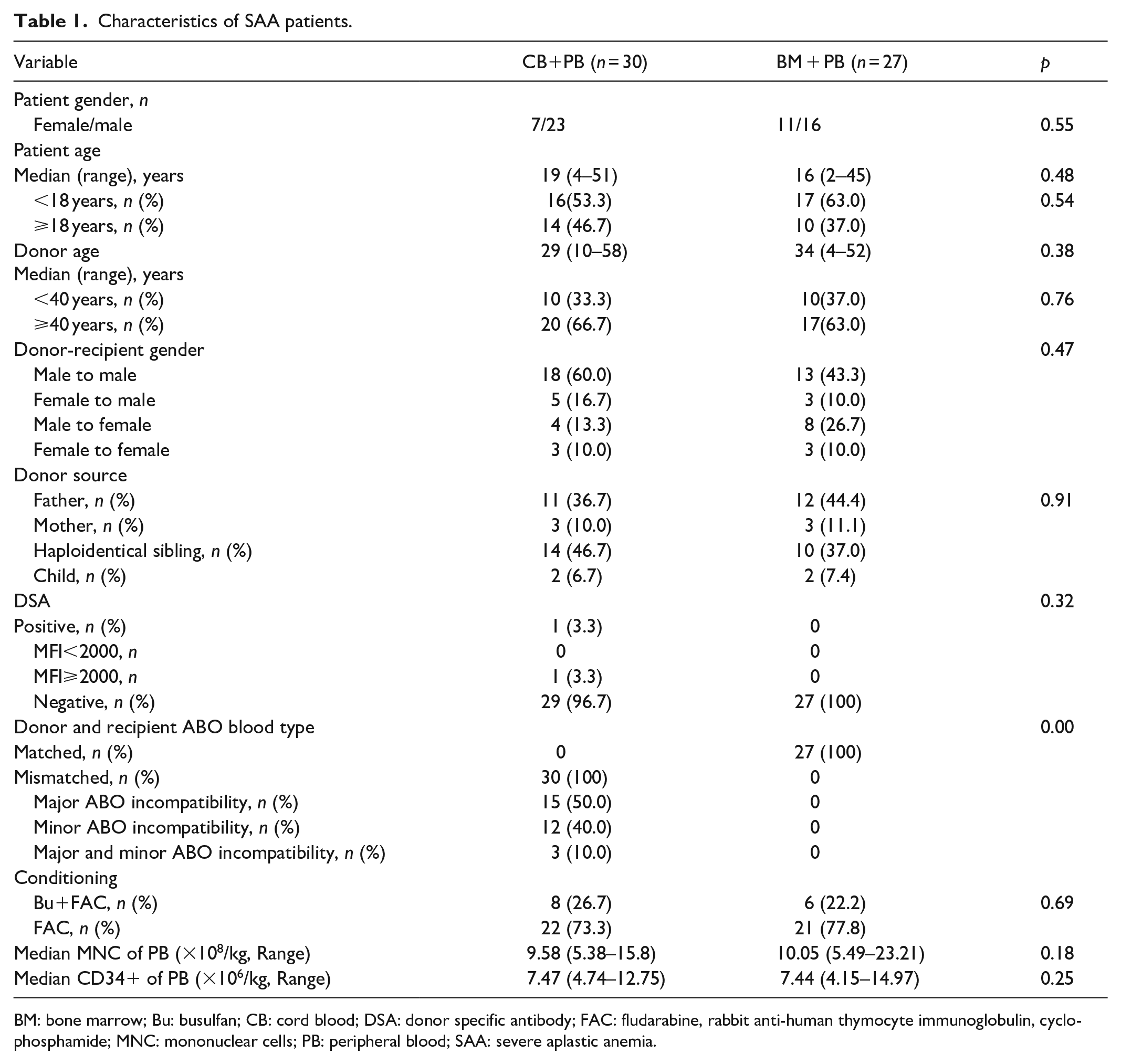
BM: bone marrow; Bu: busulfan; CB: cord blood; DSA: donor specific antibody; FAC: fludarabine, rabbit anti-human thymocyte immunoglobulin, cyclophosphamide; MNC: mononuclear cells; PB: peripheral blood; SAA: severe aplastic anemia.
Engraftment
Among all patients, only one patient in BM+PB group had primary graft failure and no one had pure red cell anemia (PRCA). The mean times to neutrophil engraftment in BM+PB and CB+PB group were 10.42 ± 2.19 and 10.53 ± 1.19 days, respectively (
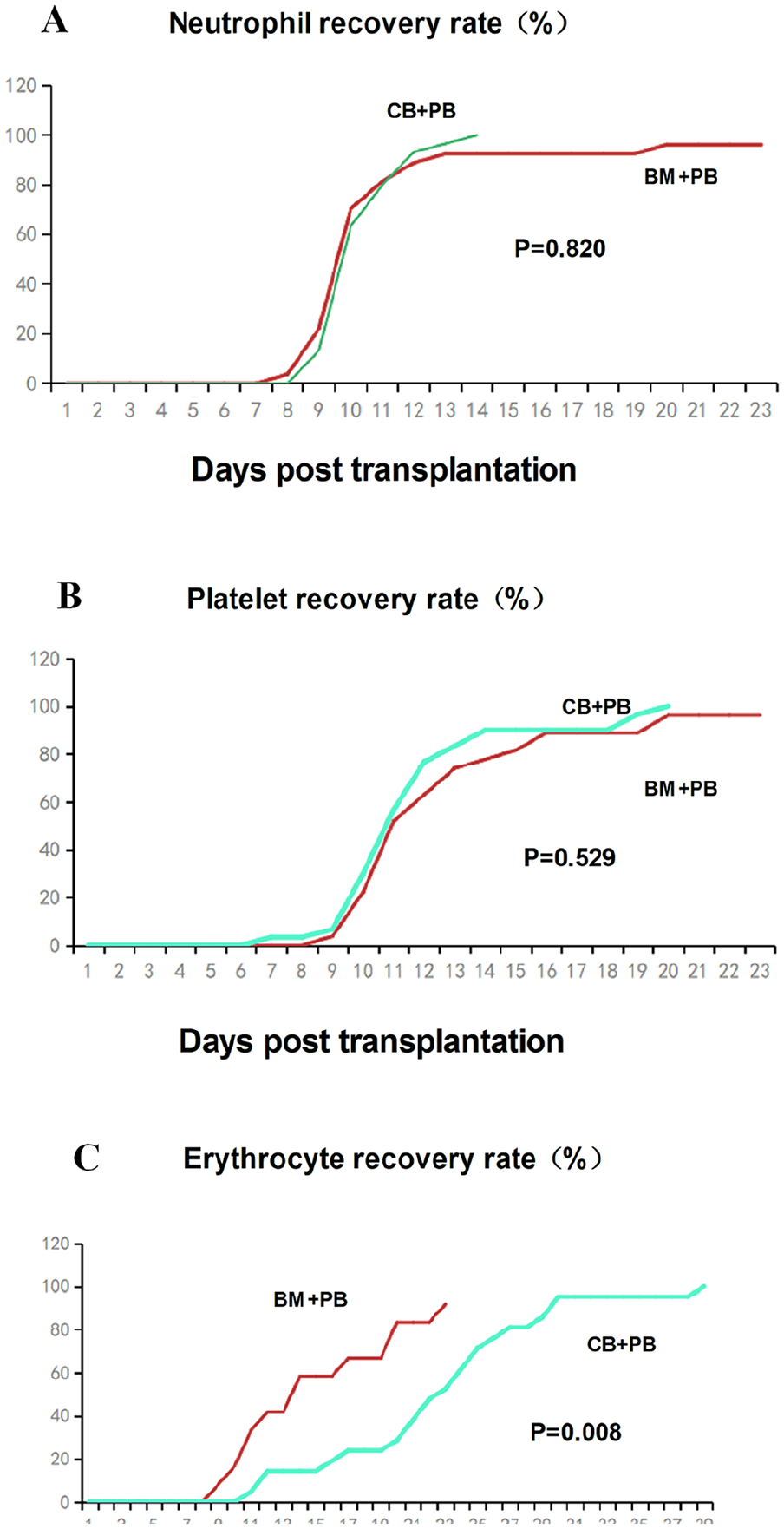
Neutrophil, platelet and erythrocyte recovery rates in BM+PB and CB+PB group.
Incidence and severity of GVHD
Grade II–IV acute GVHD occurred in 14 cases in CB+PB group, including six cases of skin and eight cases of gastrointestinal acute GVHD. Grade II–IV acute GVHD occurred in 13 cases in BM+PB group, including 12 cases of skin and 1 cases of gastrointestinal acute GVHD (Figure 2). The cumulative incidences of acute GVHD were 53.33% in CB+PB group and 51.85% in BM+PB group, and the difference was not significant (

Acute GVHD in BM+PB and CB+PB group.
Other complications
All recipients and donors were CMV seropositive (CMV-specific immunoglobulin G-positive) before HSCT. No patient developed EBV or CMV viraemia pre-HSCT. 60% (18/30) patients in the CB+PB group experienced CMV reactivation and developed CMV viraemia without CMV disease post-HSCT, and recovered after the administration of antiviral drugs. 51.85% (14/27) patients in BM+PB group experienced CMV reactivation and one died of CMV pneumonia.
Eleven patients in the CB+PB group developed EBV viraemia and four patients developed EBV-associated post-transplantation lymphoproliferative disorder (PTLD), and only one patient died of PTLD, others achieved complete remission after rituximab therapy. Seven patients in BM+PB group developed EBV viraemia, and six patients developed PTLD and all achieved complete remission after rituximab therapy. Two patients in CB+PB group and one patient in BM+PB group had hemorrhagic cystitis. One patient in BM+PB group had autoimmune hemolytic anemia. None of patients had sinusoidal obstruction syndrome.
Survival outcomes
All patients were followed-up until February 2024. Two patients in BM+PB group and three patients in CB+PB group died. The 1- and 3-year OS rates in the BM+PB group were 96.2% and 76.9%, respectively. There was no significant difference of OS between two groups (Figure 3).
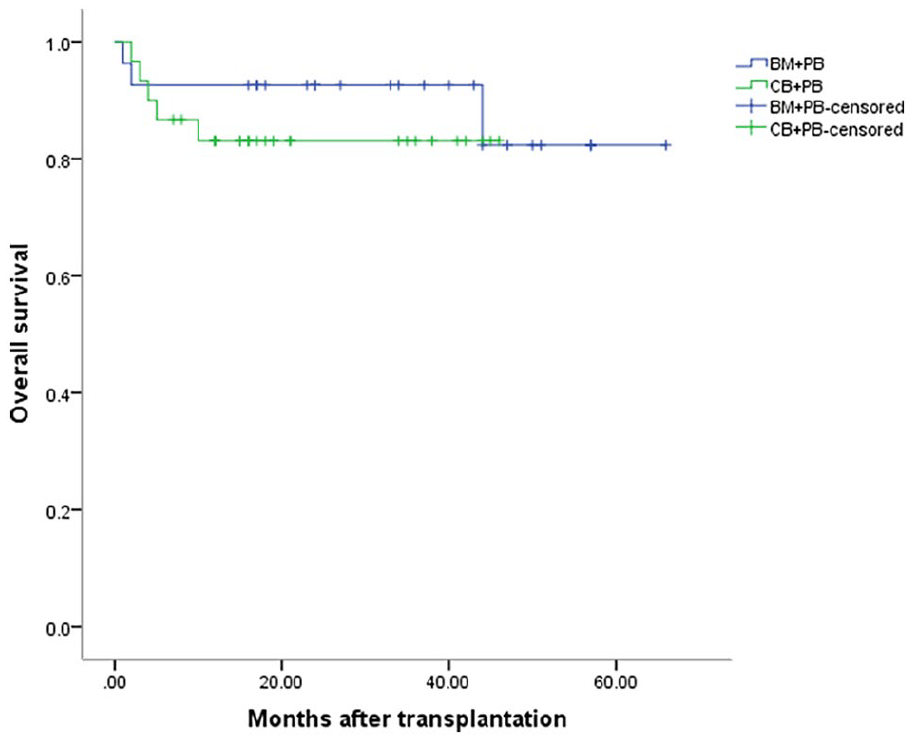
Overall survival of patients.
Discussion
Haplo-HSCT was an alternative selection for young SAA patients without HLA matched related or unrelated donors. The grafts of haplo-HSCT with classic Beijing protocol in China were a combination of bone marrow stem cells and PB stem cells. The engraftment time of recipients with PB stem cells alone as grafts was faster than that of recipients with bone marrow stem cells as grafts, but the risk of GVHD was increased. The incidence of acute GVHD and chronic GVHD in CB transplantation was the lowest compared with bone marrow transplantation and PB stem cell transplantation.
8
Because CB had a low number of initial and mature T cells, and was rich in Treg and mesenchymal stem cells. There were rare reports about haploidentical PB stem cell combined with a single unrelated CB unit for SAA patients. The impacts of combined with CB as grafts on the clinical outcomes of haplo-HSCT were still doubtful. The team of the First Affiliated Hospital of Soochow University reported that compared with haploidetical marrow and blood stem cell transplantation, combined with single unit CB as grafts could improve the OS and selection of HLA-A site compatible CB could further improve survival and reduce the incidence of GVHD.9,10 While Shaoyan Hu et al. reported that for children SAA patients underwent haplo-HSCT, the haplo-cord group with bone marrow, PB, and CB stem cells as grafts showed a lower incidence of II–IV acute GVHD than the haplo-group with bone marrow and PB stem cells as grafts. Incidences of chronic GVHD and moderate to severe chronic GVHD in the haplo-cord group were also lower than that in the haplo-group. Combined with CB stem cells as grafts was identified as the only factor associated with a lower incidence of II–IV acute GVHD and chronic GVHD.
11
In our study, the overall incidence of acute GVHD was 53.33% in CB+PB group and 51.85% in BM+PB group, but the difference was not significant (
Although ABO incompatibility is not considered as a contraindication in allo-HSCT, transfusion grafts of bone marrow stem cells with major ABO incompatibility were more complicated compared with PB stem cells, since grafts from bone marrow contained high amounts of red blood cells and antibodies and were needed for the removal of erythrocytes or plasma. 12 The impacts of ABO incompatibility on erytrocyte, granulocyte, and platelet engraftment in haplo-HSCT varied in different reports. Some reports that hemolysis reactions might cause PRCA in the majority of patients with major ABO-mismatched HSCT. It is more frequently observed in group A donors with group O recipients. 13 Several studies demonstrated that the presence of anti-donor isohemagglutinins could delay erythrocyte recovery and increased requirements of red blood cells transfusion post-transplantation. Studies of the Japan Marrow Donor Program (JMDP; 5549 patients included) and the Société Française de Greffe de Moelle et de Thérapie Cellulaire (SFGM-TC; 1108 patients included) showed that neutrophil engraftment of major ABO mismatch HSCT was slower than that of ABO matched HSCT.14,15 Lanping Xu et al. 4 reported that there were no significant differences of neutrophil and platelet engraftment in SAA patients underwent ABO-compatible or ABO-incompatible haplo-HSCT. Canaani Jonathan et al. 16 reported that major ABO mismatching was associated with inferior day 100 engraftment rate in acute myeloid leukemia patients receiving haploidentical transplants. Yibo Wu et al. reported that there were no significant differences in neutrophil and platelet engraftment, blood transfusion independence, and transfusion requirements at 30, 60, 90, 180, and 365 days post-transplantation among patients with ABO matching and those with minor, major, or bidirectional ABO incompatibility. Donor-recipient ABO matching did not differ significantly according to graft function (good versus poor). ABO incompatibility status has no major impact on engraftment in patients with hematologic malignancies undergoing ATG-based MAC haplo-SCT with PBSC-derived grafts. 17 In this study, we found that there were no significant differences in neutrophil and platelet recovery rates, and the erythrocyte engraftment time of patients with ABO incompatibility was slower than that of patients with ABO compatibility. Results of our study were different from those reported in the literature, and it may due to the differences of conditioning regimen and stem cell grafts in different studies.
The impacts of ABO incompatibility on acute and chronic GVHD were controversial. Lanping Xu et al.
4
reported that minor ABO-incompatiblity was identified as an independent risk factor for grade III–IV acute GVHD in SAA patients underwent haplo-HSCT. Canaani Jonathan et al.
16
reported bi-directional mismatching was associated with increased risk of grade II–IV acute GVHD and chronic GVHD rates were comparable between ABO-matched and -mismatched patients with AML underwent haplo-HSCT. But some reported that there was no significant association between ABO matching and grade II–IV acute GVHD, grade III–IV acute GVHD, and moderate and severe chronic GVHD in patients underwent ATG-based MAC haplo-HSCT with PBSC-derived grafts.
17
In our study, the incidence of acute GVHD was 53.33% in ABO-incompatiblity and 51.85% in ABO compatibility, and the difference was not significant (
Some studies showed ABO mismatch was correlated with decreased OS. Kimura et al. 14 reported a shorter OS for patients receiving a major ABO-mismatched graft compared to minor or bi-directional ABO-mismatched transplantation, whereas Michallet et al. 15 reported a lower survival rate for minor ABO-mismatched compared with ABO-matched cases. Canaani Jonathan et al. 16 reported AML patients with major ABO mismatching and bone marrow grafts had decreased survival after haplo-HSCT. Besides these large registry reports, various cohort studies including lower numbers of patients reported on conflicting results. The majority of authors did not observe an influence of ABO mismatch on survival. In our study, we also found that there was no significant difference in OS between the two groups, but it needs to further follow-up to identify the long-term influence.
Study limitations
There were some limitations to the study. It was a retrospective study, and the number of cases was small, requiring further expansion of the case study. We did not compare different outcomes between the major ABO-mismatched and minor ABO-mismatched groups.
Conclusion
In conclusion, haploidentical PB stem cell transplantation combined with a single CB unit may be an alternative option for SAA patients with donor-recipient ABO incompatibility.
Footnotes
Acknowledgements
None.
Author contributions
Xiaoning Wang: Data curation, Formal analysis, Investigation, Writing—original draft, Writing—review & editing; Yuqi Wang and Aiman Saulat: Data curation, Investigation; Ruimin Liu, Juan Ren and Huachao Zhu: Conceptualization, Resources; Mei Zhang and Pengcheng He: Methodology, Resources, Supervision, Validation.
Availability of data and materials
The datasets are not publicly available due to personal data protection reasons but are available from the corresponding author upon reasonable request.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by the Project of the First Affiliated Hospital of Xi’an Jiaotong University (2020HL-11), Medical Basic Clinical Integration Innovation Project of Xi’an Jiaotong Univeristy (YXJLRH2022007), the Key Research and Development of Shaanxi Province (2020SF-045) and National Key R&D Program of China (2022YFC2502700).
Ethics approval
This study was approved by the ethics committee of the First Affiliated Hospital of Xi’an Jiaotong University (No.XJTU-2021N23).
Informed consent
Written informed consent was obtained from all adult patients or legally authorized representative of underage patients in accordance with the principle of the Declaration of Helsinki.
Trial registration
Not Applicable.
