Abstract
Background:
Over the past few years, gestational diabetes mellitus has become more common. According to earlier research, gestational diabetes mellitus is linked to higher uric acid levels, and gestational hyperuricemia is linked to poor maternal and neonatal outcomes.
Aim and objectives:
To assess the influence of elevated uric acid >5 mg/dl on pregnancy outcomes such as neonatal weight, respiratory distress, preterm delivery, neonatal intensive care unit admission, premature membrane rupture, oligohydramnios, cesarean section, and intensive care admission for mothers.
Setting and methods:
It is a prospective cohort study, carried out at obstetric/family medicine outpatient clinics, Cairo University Hospitals on 221 pregnant females with gestational diabetes mellitus in their third trimester of pregnancy, selected by random sampling, performed structured interviews, and tested for serum uric acid level and were separated into two groups, those with elevated uric acid >5 mg/dl (121) and those without elevated uric acid ⩽5 mg/dl (100) patients and then followed up until delivery in Cairo University Hospitals for collecting pregnancy outcome data.
Results:
There were statistically significant differences between the antepartum uric acid and neonatal complications, maternal complications, preterm, macrosomia, neonatal intensive care unit admission, premature rupture of membranes, oligohydramnios, and cesarean section between both groups.
Conclusions:
This study showed that elevated serum uric acid >5 mg/dl can predict the incidence of maternal and neonatal problems in gestational diabetes mellitus including preterm, macrosomia, neonatal intensive care unit admission, premature rupture of membranes, oligohydramnios, and cesarean section.
Introduction
When glucose intolerance first appears or is initially diagnosed during pregnancy, it is known as gestational diabetes mellitus (GDM). it can lead to maternal and fetal complications including preeclampsia, polyhydramnios, cesarean section, congenital malformations, macrosomia, respiratory distress syndrome, and premature birth. 1 Therefore, it is essential to diagnose and treat GDM and early predict its complications to prevent undesirable pregnancy outcomes.
GDM incidence has dramatically increased in recent years as a result of the delaying of childbearing age and changes in dietary practices and lifestyles. 2 GDM is thought to be a risk for the later progression of cardiovascular problems and metabolic syndrome in postpartum women. 3
Uric acid is considered the product of purine metabolism, and its elevation has been linked to numerous unfavorable pregnancy outcomes. 3
There is no universally accepted definition of hyperuricemia during pregnancy, which makes treating it one of the challenges. Given that the urate’s solubility point is 405 mol/L (6.8 mg/dl), the serum uric acid concentration would appear to be more than this value. 4
Concentrations of serum uric acid in the blood greater than 7 mg/dl are considered to be hyperuricemia by the American College of Rheumatology.5,6
During early pregnancy, uric acid levels fall often to (2–4.2) mg/dl or below, this is usually due to the uricosuric effects from estrogen and the increase in renal blood flow. Uric acid levels increase during the third trimester, reaching levels of up to (3.1–6.3
A review of the literature did not yield enough data to make judgments about long-term maintenance therapy or treatment for hyperuricemia or elevated uric acid during pregnancy.
Few studies suggested treatment with allopurinol should be based on individualized risks and benefits, but Van Veen and Haeri suggest that gestational diabetes justifies its use in the second half of pregnancy only if presented with gout. 8
In addition to proteinuria, hypertension, and hyperinsulinemia, gestational hyperuricemia was found to be significantly connected to a high rate of pregnancy complications. Both of these conditions may independently be linked to a decrease in uric acid excretion through the urine. 9
Numerous investigations found that uric acid levels and pregnancy outcomes were linked with preeclampsia. GDM and preeclampsia have been linked to uric acid, a marker for the metabolic syndrome in non-pregnant females. Since prior research on serum uric acid metabolism in GDM had suggested a connection between uric acid levels and pregnancy outcomes in females with GDM, we found that only a small number of studies have attempted to unravel that connection. Even so, the relationship between uric acid levels in the third trimester and pregnancy outcomes is not frequently studied, and a reference point for what constitutes normal uric acid levels during pregnancy has not yet been established.
Concerning this we conducted this study aiming to assess the risk of elevated serum uric acid in GDM on pregnancy outcome.
Methods
Design
This prospective cohort study, which involved 240 pregnant females with GDM, sought to determine the potential effects of elevated serum uric acid >5 mg/dl on the course of the pregnancy in females with GDM. Our study was done at obstetric/family medicine outpatient clinics at Cairo University Hospitals.
Setting and population
Pregnant females already identified as having GDM between weeks 24 and 28 of pregnancy by an obstetrician according to American Diabetes Association Guideline criteria for the diagnosis of GDM 10 attending an obstetric clinic at Cairo University during the period from October 2020 to June 2022 and who agreed to participate and to be included in the study.
Inclusion criteria: Pregnant females with singleton pregnancy identified as having GDM between weeks 24 and 28 of pregnancy according to American Diabetes Association Guideline criteria for the diagnosis of GDM 10 by an obstetrician and have the intention to deliver their babies at Cairo University Hospitals and agreed to participate in the study.
Exclusion criteria: Pregnant females previously known to have chronic hypertension, diabetes mellitus, renal disease, or other systemic illness.
Sampling
As there is no clear universal definition of hyperuricemia in pregnancy, we use a cutoff point of 5 mg/dl as many studies state that the normal level of serum uric acid in normal pregnancy during the third trimester is around 5 mg/dl.11–13
Regarding that, all participants (240) were chosen by random sampling to prevent bias and then were split into two groups based on the level of antepartum uric acid at cutoff point 5 mg/dl to elevated uric acid group (121 participants) and non-elevated uric acid group (100 participants) after exclusion of drop out cases (19 participants).
Sample size
The sample size was determined using the Epi info version 7 program relaying on the previous study of Amini et al., 14 by comparing neonatal hypoglycemia, as one of the pregnancy outcomes, prevalence in hyperuricemic pregnancy was 32.2% and pregnancy with normal uric acid was 16.6%, a minimal sample size of 240 (including drop out) are required to achieve the study objectives under error 1%, confidence interval 95% by using openepi6 program with power 80%.
Sampling technique of uric acid test: A blood sample was taken from all subjects. Three milliliters was collected into sterile plane tubes and centrifuged at 300 g (gravitational force) for 5 min. Sera were separated immediately and stored at −20°C until the time of analysis.
Data collection tools and technique
The pregnant females were enrolled in the study when they came to the obstetric outpatient clinic, at Cairo University and were diagnosed with GDM according to the American Diabetic Association (ADA) which recommends the International Association of the Diabetes and Pregnancy Study Groups (IADPSG) criteria (“one-step strategy”) for diagnosis of GDM: Any one abnormal value of cutoff values after 75 g oral glucose tolerance test is enough for diagnosis.
Fasting > 92 mg/dl, 1 h > 180 mg/dl and 2 h > 153 mg/dl
10
Samples of their blood were drawn, and the levels of albumin, creatinine, estimated glomerular filtration rate (eGFR), and serum uric acid were measured.
The following steps were done for cases:
- Taking approval to a written informed consent from patients after ensuring the confidentiality of their personal information and medical data
- History taking including:
1. Personal history including name, age, address, consanguinity, and special habits such as smoking.
2. Menstrual history: The first day of the last menstrual period, regularity of menstrual cycles, and history of hormonal medications.
3. Obstetric history: Previous deliveries, mode of deliveries, number and sex of living children, date of last delivery, history of abortions, date of last abortion, puerperium, and history of complications.
4. Past history: Endocrine or systemic diseases such as hypertension, DM, renal diseases, and hyperthyroidism.
5. Medical history: Especially of hormonal medications and drugs known to elevate uric acid concentration in the blood such as phenothiazines.
6. Family history: of clinical importance like hypertension, DM.
- Clinical examination
General examination: vital signs (pulse, temperature, blood pressure, respiratory rate) body weight and chest, abdomen and heart examination, lower limb edema.
- Ultrasonography:
Obstetric ultrasound was used to confirm fetal viability, detect a number of fetuses, estimate gestational age, and check for other abnormalities.
- Laboratory tests:
Blood samples were taken for assessment of serum uric acid, creatinine, eGFR, and albumin concentration.
- Then we followed up with 221 patients until delivery (who delivered their babies in Cairo University Hospitals and 19 patients dropped out after delivery outside Cairo University Hospitals.
- We collected data on neonatal weight and incidence of respiratory distress, premature delivery, neonatal intensive care unit (NICU) admission, premature rupture of membranes (PROMs), oligohydramnios, caesarian section, and intensive care admission for mothers by reviewing medical records after delivery in Cairo University Hospitals.
Ethical considerations: The ethical and research committee of the family medicine department, as well as the research committee for the faculty of medicine at Cairo University, provided their permissions (Code: MD-274-2020), The researcher met privately with the subjects who agreed to take part in the study, the interview was done in a separate room to ensure patient privacy, and after ensuring them about the confidentiality of their personal information and medical data they signed the written informed consent (the participant was informed about the objectives, methods, and possible impact of the study).
Statistical analysis
Data were coded and entered using the statistical package for the Social Sciences (SPSS) version 28 (IBM Corp., Armonk, NY, USA). Data were summarized using mean and standard deviation for quantitative variables and frequencies (number of cases) and relative frequencies (percentages) for categorical variables. Comparisons between groups were done using unpaired t-test. 15 For comparing categorical data, a chi-square (χ2) test was performed. An exact test was used instead when the expected frequency was less than five. 16 Logistic regression was done to detect whether elevated uric acid acts as an independent predictor of different outcomes. 17 p-Values less than 0.05 were considered statistically significant.
Results
This study included 221 participants who split into two groups: the first represented 54.8% of participants (121) who had uric acid above 5 mg/dl and the second represented 45.2% (100 participants) who had antepartum uric acid below or equal to 5 mg/dl and for both groups, there were no cases of hyperuricemia (if defined as serum uric acid >7 mg/dl) between participants.
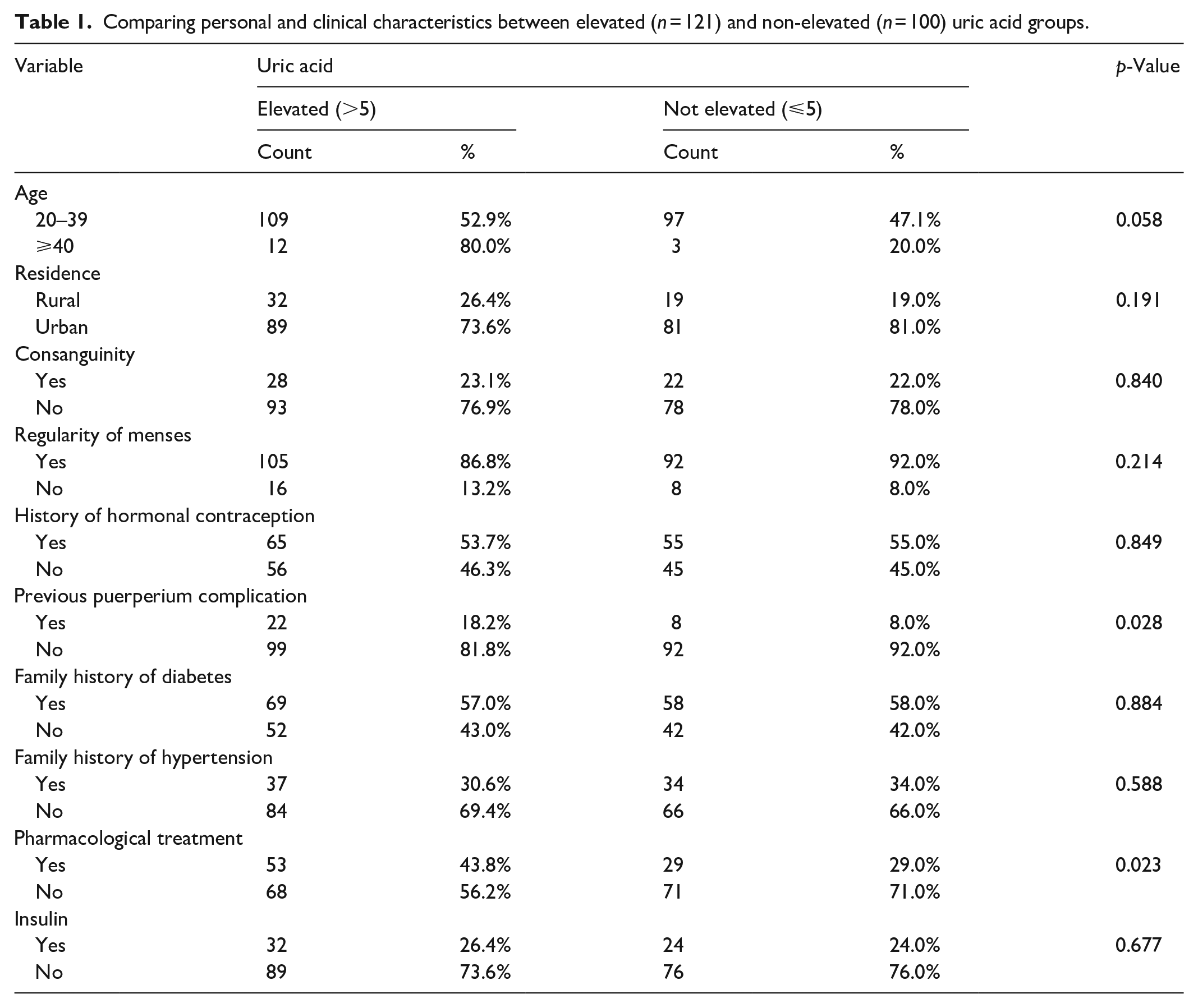
There was no statistically significant difference between the elevated and non-elevated uric acid groups regarding personal and clinical characteristics but there was a statistically significant difference between both groups regarding taking pharmacological treatment (insulin or oral metformin) with a p-value of (0.023) (Table 1).
Comparing personal and clinical characteristics between elevated (n = 121) and non-elevated (n = 100) uric acid groups.
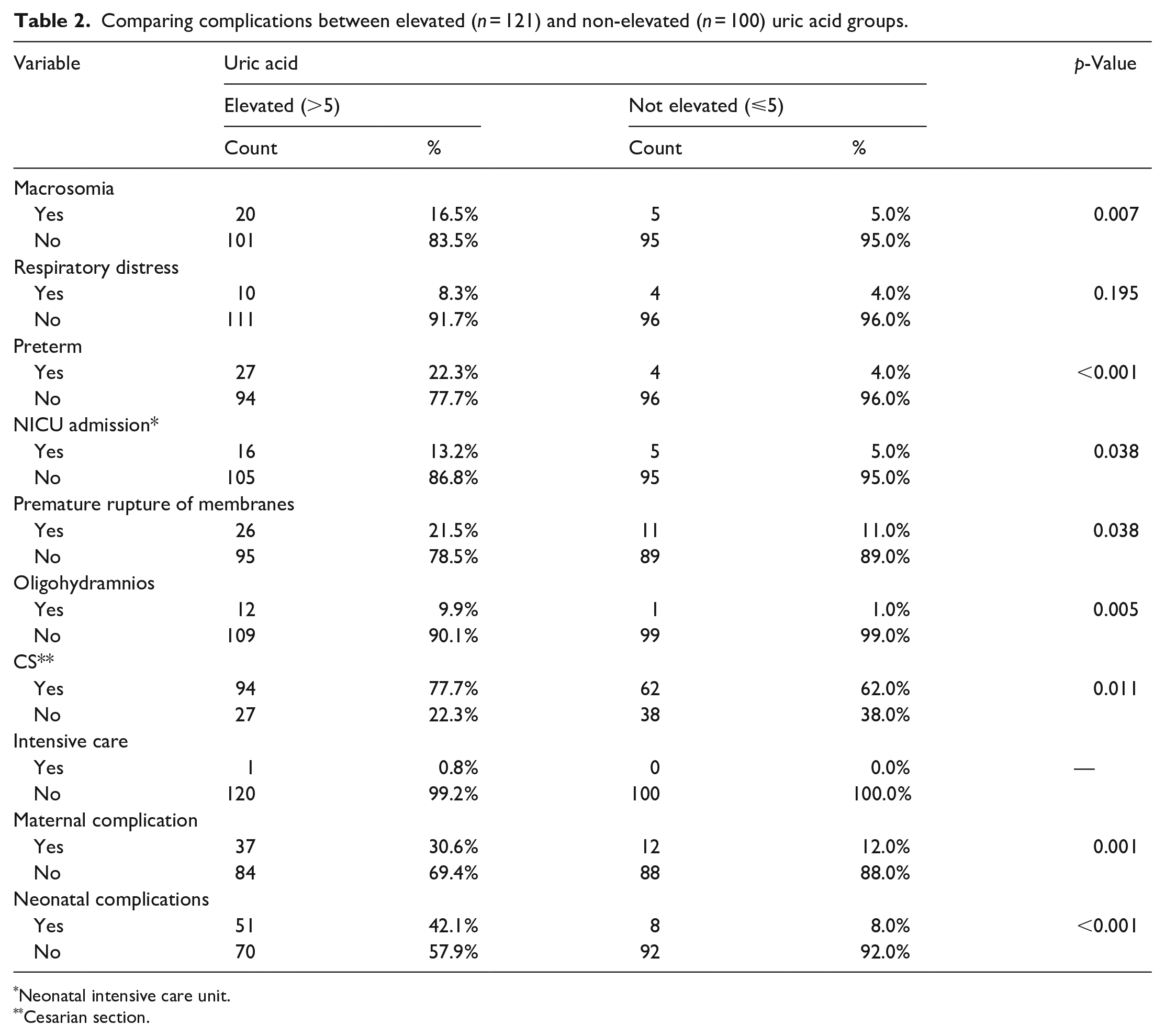
There were statistically significant differences between both elevated and non-elevated uric acid groups regarding neonatal complication with a p-value of (<0.001), maternal complications with a p-value of (0.001), macrosomia with a p-value of (0.007), preterm with a p-value of (<0.001), NICU admission with a p-value of (0.038), PROMs with a p-value of (0.038), and oligohydramnios with a p-value of (0.005) while there were no statistically significant differences regarding respiratory distress and intensive care admission of the mother (Table 2).
Comparing complications between elevated (n = 121) and non-elevated (n = 100) uric acid groups.
Neonatal intensive care unit.
Cesarian section.
After doing logistic regression for evaluating elevated uric acid (>5 mg/dl) as a predictor for complications, it shows that it is a significant predictor for incidence of macrosomia, preterm, NICU, PROMs and oligohydramnios, maternal complications, and neonatal complications (Table 3).
Logistic regression using elevated uric acid (>5 mg/dl) as a predictor for complications.

*Neonatal intensive care unit.
Discussion
This study finds that there were no cases of hyperuricemia (if defined as serum uric acid >7 mg/dl) among participants with 54.8% of participants having uric acid above 5 mg/dl and 45.2% having antepartum uric acid below or equal to 5 mg/dl and this is nearly similar to other researches by Kumar et al., 18 and Pleskacova et al., 19 who also found that there were no cases of antepartum hyperuricemia between pregnant females with GDM.
The prevalence of maternal complications with antepartum uric acid in the elevated uric acid group was 30.6% versus 12% in the non-elevated group and with a p value of (0.001), there were statistically significant differences between the two groups.
The prevalence of neonatal complications with antepartum uric acid in the elevated uric acid group was 42.1% versus 8% in the non-elevated group and with a p value of (<0.001), there were statistically significant differences between the two groups.
Hamed Khalil and colleagues in their study, “Hyperuricemia as a predictor of perinatal outcomes in pregnancy induced hypertension,” discovered that the link between harmful prenatal outcomes and high blood uric acid levels and pregnancy problems was direct. 20
The correlations of maternal hyperuricemia and its effects on gestational diabetes on short- and long-term newborn problems have not been examined in any prior research. It is unclear whether maternal hyperuricemia has any connections to metabolic abnormalities in newborns. In addition, because multiple gestations, preeclampsia, and hypertension can all affect pregnancy outcomes as well as uric acid levels, the majority of studies have been on how hyperuricemia affects pregnancy outcomes in these situations.
Our study finds that the prevalence of respiratory distress in the elevated uric acid group was 8.3% versus 4% in the non-elevated group and between the two groups, there were no statistically significant differences (p value, 0.195).
Unlike our study, Kumar et al. 21 evaluated a group of pregnant women with gestational hypertension who had healthy babies, the mean serum uric acid levels were (5.16 ± 1.74) mg/dl, and in the group of women who delivered distressed fetuses were (6.52 ± 2.31) mg/dl, which represents a significant relationship between uric acid and respiratory distress with a p value (<0.0001).
In our study, we discovered that the prevalence of macrosomia was 16.5% in the group with elevated uric acid compared to 5% in the non-elevated group. These findings were statistically different between the two groups with a p value of (0.007).
Conversely, another study observed no discernible difference in uric acid levels between the macrosomia group and the normal birthweight group when they investigated the combination of lipids and uric acid in the mid-second trimester to predict unfavorable pregnancy outcomes. 22
Other research, such as that by Zhou et al., 23 demonstrated a linear relationship between maternal blood uric acid concentrations during pregnancy and fetal development in large-for-gestational-age (LGA) babies. Studies indicate that oxidative stress, a biological condition assumed to be crucial to embryonic growth, is connected with both incredibly high and low uric acid values. Researchers observed that mothers of LGA and small-for-gestational-age infants had higher levels of oxidative stress. 23
According to this study, the prevalence of preterm birth was 22.3% in the group with raised uric acid compared to 4% in the non-elevated group, and the difference between the two groups was statistically significant, with a p value of (0.001).
In females with “benign” gestational hypertension, prematurity risk was found to be increased by high uric acid (OR: 3.2; 95% CI: 1.4–7.2), according to Hawkins et al.’s 24 study of plasma uric acid as a marker of poor outcome in hypertensive pregnancy.
Asgharnia et al. 25 discovered that in comparison to women who gave birth at term, preterm mothers’ mean uric acid levels were significantly higher (p = 0.0001) when she correlated preeclamptic women’s prenatal serum uric acid levels with maternal and neonatal problems.
Maternal hyperuricemia is a prenatal finding with ongoing perinatal and postnatal repercussions that was researched by Amini et al. 14 They discovered that it is linked to preterm birth (OR: 3.17; 95% CI: 2.1–4.79).
According to our study, the prevalence of NICU admission in the group with raised serum uric acid was 13.2%, compared to 5% in the group without an elevated level, and with a p value of (0.038), there were statistically significant differences between the two groups.
Prenatal evidence of maternal hyperuricemia in normotensive singleton pregnancy has persistent perinatal and postnatal effects, and according to research by Amini et al., 14 it is linked to NICU hospitalization (OR: 1.65; 95% CI: 1.12–2.94).
In a hospital-based cross-sectional study involving 200 women who are preeclamptic during pregnancy and have a gestational age of less than 34 weeks, Zangana and Hamadamen 26 discovered a statistically significant correlation between an elevated serum uric acid level and admission to a neonatal care unit.
According to our study’s findings on the connection between serum uric acid concentrations and PROM, the prevalence of PROM was 21.5% in the group with increased uric acid levels compared to 11% in the non-elevated group, and with a p value of (0.038), there were statistically significant differences between the two groups.
A total of 1602 singleton pregnant from Union Shenzhen Hospital of Huazhong University of Science and Technology participated in the study by Wu et al., 27 which examined the impact of serum urea nitrogen and uric acid levels as hazards for both maternal and fetal pregnancy outcomes. After adjustment, the researchers discovered that the risk of (PROM) increased by 48% (OR) in the third trimester when uric acid levels were at their maximum.
As opposed to Guo et al.’s 28 investigation, which examined the role of serum uric acid in protecting against premature membrane rupture in gestational diabetes, mild increases in uric acid levels between the 24th and 28th gestations may have a protective effect on PROM in GDM.
The prevalence of oligohydramnios in the group with elevated uric acid was 9.9%, compared to 1% in the group without elevated uric acid, and a p value of (0.005) indicated that there were statistically significant differences between the two groups.
In a study using severe preeclampsia as a model, Yassin Khalil and Saber Muhammed 29 looked at maternal serum uric acid as an indicator of perinatal complications. They discovered a positive and significant (p < 0.001) association between high serum uric acid levels and oligohydramnios.
However, Anil et al., 30 who studied uric acid levels as a sign of pregnant hypertension development, with a p value of 0.497, it was determined that there was no significant association between serum uric acid levels and oligohydramnios.
The prevalence of caesarian sections was found to be 77.7% in the group of participants with high uric acid compared to 62% in the non-elevated group, and with a p value of (0.011) indicating that there were statistically significant differences between the two groups.
Regarding other studies, Hawkins et al. 24 discovered in 2012 that cesarean deliveries were considerably more common in women with prenatal hyperuricemia (53% vs 40%; p < 0.001).
Limitations
1- Most of the females did not stick to their antenatal care visits, so it was difficult to follow up their weight gain during pregnancy, and the degree of controlling hyperglycemic state.
2- By revising the literature, there was no obvious definition or cutoff point of hyperuricemia during pregnancy or elevated uric acid, so we need more studies to figure out a reference value for hyperuricemia during pregnancy.
3- The results of this single institutional investigation cannot be generalized; therefore, a multi-center or community-based study will be required to validate these results.
4- This study did not assess the validity and reliability of methods and was not pilot-tested.
Conclusion
This study concluded that maternal serum uric acid >5 mg/dl in pregnant women with gestational diabetes can predict the incidence of neonatal and maternal complications including preterm, macrosomia, NICU admission, PROM, oligohydramnios, and cesarean section. Regarding that, this study recommends the evaluation of maternal serum uric acid as a part of routine workup during the third trimester in females with GDM to predict the incidence of complications and the need for closer follow-up visits.
Footnotes
Acknowledgements
The authors want to thank all pregnant females who agreed to participate and also the obstetric clinic staff who facilitated this work.
Author’s contribution
All authors contributed to the design and implementation of the research, the analysis, the results, and the writing of the manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from “Research Ethical Committee of the Faculty of Medicine Cairo University (Code: MD-274-2020).”
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
Not applicable.
