Abstract
Gouty tophus is an unusual cause of digital flexion contracture. Awareness of this pathophysiology will lead to more confidence in proper treatment and surgical management of this rare condition. This report describes a case of digital flexion contracture by tophaceous gout distinguished between trigger finger and locking of the metacarpophalangeal joint. We found the flexor tendon with a deposited chalky white substance suggestive of gouty tophus intraoperatively. We performed tenosynovectomy and removed the chalky white substance to the greatest degree possible. Histological findings confirmed the diagnosis of gout. Postoperatively, the patient recovered nearly to a full range of motion of the affected digits. When meeting with the patient who has had hyperuricemia and who is unable to extend the affected digits suddenly, one must keep in mind digital flexion contracture caused by tophaceous gout.
Introduction
Gout is an inflammatory arthritis caused by deposition of sodium urate crystals in joints and soft tissues when serum uric acid production exceeds its elimination. 1 Poor control of hyperuricemia can lead to gouty tophi in some areas.1–7,8 Gouty involvement of the flexor tendon has been reported in previous cases with related digital stiffness and triggering, carpal tunnel syndrome, and tendon rupture.1–7,8 Gouty involvement of the flexor tendon is not often considered in the differential diagnosis of trigger finger and locking of a metacarpophalangeal (MP) joint. This report describes an unusual case of flexor tendon tophaceous infiltration with locking of the MP joint at the right middle finger.
Case report
A 63-year-old man complained at our outpatient department of a sudden inability to extend the middle finger of his right hand (Figure 1). He noticed the symptom after waking up, but he did not observe the symptom the prior night. No indication of trauma history was found at the initial examination. The patient had a medical history of pyogenic spondylitis. Physical examination revealed that the MP joint of the middle finger was a rigid block to joint extension and flexion of the MP joint. The proximal interphalangeal joint was unaffected. Serum uric acid was 10.9 mg/dL, indicating hyperuricemia. Although C-reactive protein was 0.37 mg/dL and the erythrocyte sedimentation rate was 45 mm/min and increasing slightly, other laboratory findings including those of blood (red blood count = 4.39 × 106/μL; white blood count = 7200/μL; platelet count = 2.08 × 105/μL) and electrolytes (sodium = 141 mmol/L; potassium = 4.7 mmol/L; chloride = 102 mmol/L) were normal. Rheumatoid factor and anti-cyclic citrullinated peptide antibody were seronegative. Radiographs and computed tomography (CT) showed ulnar deviation of the left fingers and osteophytes on the radial lateral aspects of the metacarpal head of the right middle finger and osteoarthritic change of the distal radioulnar joint (Figure 2). He had no subjective symptom in any other joint and has never had a history of gout attack of any other joint in the past. We made a differential diagnosis between trigger finger and locking of the MP joint of the middle finger and performed digital block and manipulative reduction. Nevertheless, we were unable to reduce the flexion position of the MP joint of the middle finger.

Preoperative clinical sign showed trigger-like symptom and locking of the metacarpophalangeal joint of the left middle finger. Gouty tophus was deposited under the skin of the fingertip.
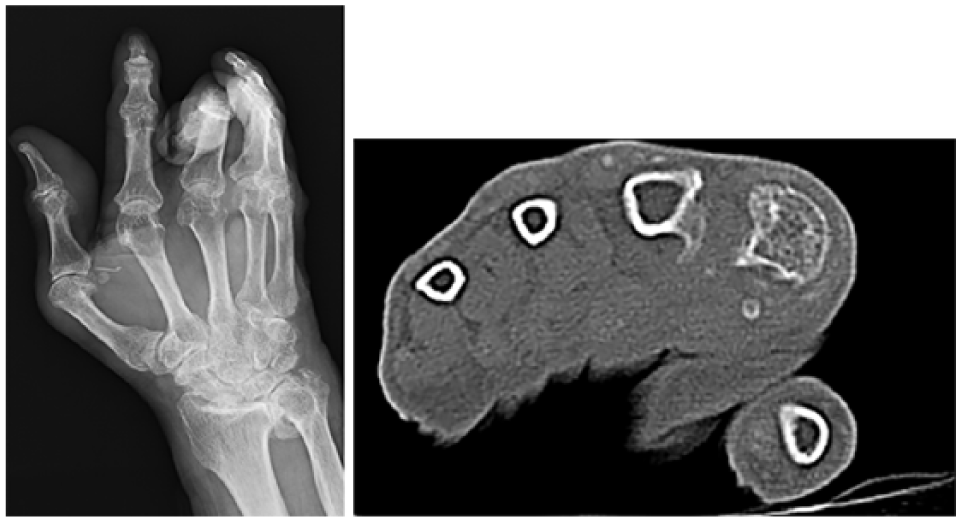
Radiograph and CT of the metacarpal head of the middle finger showing that the radial condyle tip had a sharp osteophyte.
He was treated surgically. Flexor tendon release was performed from the MP joint level to the wrist joint level. With carpal tunnel release, flexor digitorum superficialis (FDS) tendon and flexor digitorum profundus (FDP) of the middle finger were found with multiple involved white lesions (Figure 3). We performed tenosynovectomy and removed the chalky white substance suggestive of gouty tophus to the greatest degree possible and maintained the integrity of the FDS and FDP tendon. Histological findings confirmed the diagnosis of gout (Figure 4). Postoperatively, the patient recovered nearly to full range of motion of the affected digits. He had an arc of active motion of 80° (range = 0°–80°) of the MP joint and proximal and distal interphalangeal joints of the right middle finger. We administered febuxostat (10 mg/day) for hyperuricemia. The uric acid level 8 months after surgery was 6.0 mg/dL. Since surgery, the patient has had no recurrence of the symptom of sudden locking of the finger joint.

Intraoperative aspect of the flexor superficialis tendon of the middle finger showing the mass filled with a chalky white substance.

Histopathological evaluation of a section of resected tissue showing an eosinophilic amorphous gouty tophus deposit like needles surrounded by chronically inflamed cells, multinucleated giant cells, and fibrillations (hematoxylin–eosin stain 10×).
Discussion
We made differential diagnosis between trigger finger and locking of the MP joint. Our ultrasound assessment of the A1 pulley of the middle finger found thickness. Radiographs and CT of the metacarpal head of the patient’s middle finger showed that the radial condyle tip had a sharp osteophyte, which is associated with the captured volar plate and accessory ligament in locking of the MP joint in general. 9 We were unable to reduce flexion locking of the MP joint under general anesthesia, but the dynamic tenodesis effect was useful to assign the lesioned part causing the middle finger flexion contracture. Recognizing the possibility of finger flexion contracture by flexor tendon tophaceous infiltration is important.
Earlier reports of the relevant literature have described carpal tunnel syndrome caused by tophaceous gout in a flexor tendon.4,5 Some cases were complicated by a triggered wrist and digital flexion contracture associated with gouty tophus. 4 Another case was not complicated with such clinical signs. 5 Diagnosing carpal tunnel syndrome related with gouty tophus of the flexor tendon is difficult in such cases. Ultrasonography is useful for the diagnosis of carpal tunnel syndrome to detect enlargement of the median nerve cross-sectional area in patients with carpal tunnel syndrome. 10 Therimadasamy et al. 8 reported ultrasonographic detection of the flexor tendons tophaceous infiltration as a heterogeneous mass. In our case, we performed ultrasound assessment to distinguish the diagnosis of trigger finger at the MP joint level. Retrospectively, we were able to confirm increased diameter and volume of the flexor tendons and multiple hyperechoic foci with heterogeneous appearance within the tendon substance. Non-invasive and easy ultrasound assessment might contribute to a diagnosis of carpal tunnel syndrome associated with gouty tophus of the flexor tendon.
Tophaceous infiltration of the flexor tendon can cause flexor tendon rupture. 6 Options to restore flexor tendon rupture include side-to-side suture, tendon transfer, and bridge graft. In our case, tophaceous infiltration invaded the FDS and FDP tendon. Fortunately however, the flexor tendons were not ruptured. We removed the gouty tophus within the FDS and FDP tendon to the greatest degree possible and protected the FDS and FDP tendon integrity. Delays in diagnosis and treatment can cause functional damage. It is therefore important to devote attention to sudden finger contracture by gouty tophus of the flexor tendons in patients with hyperuricemia. Early diagnosis is necessary to prevent degeneration and rupture in patients with gouty flexor tenosynovitis.
Conclusion
We applied surgical treatment for a case of flexor tendon tophaceous infiltration in the finger, complicated by locking of the MP joint at the right middle finger. It must be recognized that gouty flexor tenosynovitis can cause the clinical sign of locking of the MP joint of the finger. Awareness of this pathophysiology will raise confidence in the proper treatment and surgical management of this rare condition.
Footnotes
Acknowledgements
The authors would like to thank Dr Hiromi Koshi in Department of Diagnostic Pathology, Gunma University Graduate School of Medicine and Dr Junko Hirato in Clinical Department of Pathology in Gunma University Hospital for pathological assessment.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
