Abstract
Objective:
Studies on the association of the aspartate transaminase-to-alanine transaminase ratio with the metabolic syndrome and its components among HIV patients were scarce. This study aims to determine the association between the aspartate transaminase-to-alanine transaminase ratio and the metabolic syndrome and its components in adult HIV patients on highly active antiretroviral therapy.
Methods:
This was a cross-sectional study conducted on 302 HIV patients from January 15 to June 30, 2021. Sociodemographic, clinical, and anthropometric data were collected using a structured questionnaire. The patient’s medical records were reviewed. Biochemical analysis was performed after 5 ml of venous blood was collected from each study participant. Metabolic syndrome was defined by the third report of the national cholesterol education program-adult treatment panel. Logistic regression was done to assess the association of MetS with the independent variables, and correlation analysis was performed to see the correlation of MetS components with the aspartate aminotransferase-to-alanine aminotransferase ratio.
Result:
302 HIV—positive patients on highly active antiretroviral therapy were included in this study, and 54.6% were female. The median and interquartile range of the age of the study participants were 41 (35–50) years. The prevalence of metabolic syndrome was 29.5% (confidence interval = 24.5–35.1). Chronic illness (Adjusted odds ratio = 4.8, confidence interval = 2.2–10.9) and aspartate aminotransferase-to-alanine aminotransferase ratio (adjusted odds ratio = 2.5, confidence interval = 1.4–4.4) were significantly associated with Metabolic syndrome among the study participants. The aspartate aminotransferase-to-alanine aminotransferase ratio was significantly correlated with blood pressure.
Conclusion:
This study found the existence of a significant association between the aspartate aminotransferase-to-alanine aminotransferase ratio and metabolic syndrome among HIV patients.
Introduction
Metabolic syndrome (MetS), also known as insulin resistance syndrome, is a constellation of several cardio-metabolic risk factors, which include hyperglycemia, obesity, insulin resistance, elevated blood pressure (BP), and dyslipidemia. The features of MetS among people living with HIV are common, with hypertension, dyslipidemia, and decreased high-density lipoprotein cholesterol (HDL-C) being the most prevalent components.1,2 The global burden of MetS among people living with HIV is high, ranging from 16.7% to 43.5% depending on the definition of MetS used.3,4 MetS can lead to abnormal hepatocyte metabolism, fat deposition, and necrosis. Injury or disease to the liver may lead to changes in hepatocyte function. The liver enzymes such as alanine aminotransferase (ALT) and aspartate aminotransferase (AST), which are normally found in plasma, will pathologically increase under liver stress such as liver injury and nonalcoholic fatty liver disease (NAFLD). 5
Several studies indicated that liver function tests such as ALT and AST were closely related to MetS. ALT and AST are utilized as indicators of hepatic dysfunction in a range of disorders, including NAFLD, which is strongly associated with MetS. 6 NAFLD is considered the hepatic manifestation of MetS, and it is linked to increased visceral fat, insulin resistance, obesity, dyslipidemia, and lipoatrophy. 7 The presence of MetS leads to NAFLD, and levels of NAFLD could be related to increased levels of AST and ALT. 8 A population-based cohort study done in Korean adults of age 40–70 found the aspartate transaminase-to-alanine transaminase (AST/ALT) ratio to be an independent negative predictor of the future development of MetS and its components. 9 The AST/ALT ratio is also considered a surrogate indicator of NAFLD, which may be considered an additional feature of MetS with specific hepatic insulin resistance.9,10
Although increased transaminase activity is a sign of liver illness, the AST/ALT ratio is linked to the severity and prognosis of liver injury. 5 A higher AST/ALT ratio implies alcoholic liver disease, whereas a lower ratio shows non-alcoholic steatohepatitis. 11 According to a recently conducted study, having a lower AST/ALT ratio was linked to the occurrence of MetS in the future.12,13 The availability of easily measurable serum biomarkers that are closely associated with MetS would be of great help for early recognition, and the AST/ALT ratio would be used as a marker of MetS. 14 Early detection of MetS is of great importance, and finding a clear association between MetS and the AST/ALT ratio may be helpful for the earlier prognosis of metabolic abnormalities. Nevertheless, there is limited evidence to show the association of the AST/ALT ratio with MetS and its components among people living with HIV.
To the best of our knowledge, no studies have been conducted on the association of the AST/ALT ratio with MetS among HIV patients in Ethiopia. Therefore, this study was aimed at assessing the association of AST/ALT with MetS and the correlation of MetS components with the AST/ALT ratio in adult HIV patients.
Methods
Description of study design and study area
This facility-based cross-sectional study was conducted at Hawassa University Comprehensive Specialized Hospital (HUCSH) and Yirgalem General Hospital’s (YGH) adjusted odds ratio (ART) outpatient department from January 15 to June 30, 2021. Hawassa, which is the capital city of Sidama Region, is located 275 km to the south of the capital city of Ethiopia, Addis Ababa. Currently, there are 7750 HIV patients on ART follow-up at HUCSH. Yirgalem town is located 260 km south of Addis Ababa and 40 km south of Hawassa. Currently, there are 1855 patients on ART follow-up at Yirgalem Primary Hospital.
Study subject
The study participants were selected from HIV patients attending the two ART clinics. Those HIV-positive patients on ART clinical care who signed informed consent were included in this study. We excluded all participants who refused to sign the informed consent, those who did not fast for the next appointment, and all with Hepatitis B or C co-infection.
Sample size and sampling procedure
The sample size was calculated using a single population proportion assumption: a 25% prevalence (p) of MetS among HIV patients in Ethiopia, 15 a 5% margin of error (d), and a 95% confidence interval (CI).
By applying Cochran’s correction formula to correct the sample size for the fact that the number of HIV patients was less than 10,000, the final sample size obtained was 278. By considering a 10% nonresponse rate, the final sample size for this study was 306.
Sampling procedure
The total number of HIV patients at Hawassa comprehensive specialized hospital and Yirgalem Hospital receiving ART clinical care was 9615. The sample size for the two study sites was allocated proportionally. Therefore, 247 and 59 study participants were selected from Hawassa and Yirgalem hospitals, respectively, using a systematic random sampling technique. The sample interval (K) was calculated using the formula N/n. The first participants were selected using lottery methods.
Data collection and measurements
After institutional review board approval, written informed consent was obtained from each study participant. Then, study participants were approached and recruited for the study during their routine medical checkups. Sociodemographic data such as age, sex, residence, occupation, educational status, monthly income; anthropometric data such as weight and height; clinical data such as BP, World Health Organization (WHO) clinical stage, and presence or absence of chronic illnesses, and information on current treatment information were collected using pretested structured questionnaires by trained nurses working at ART clinics. Then, the study participants were requested to give 5 ml of venous blood after overnight fasting for laboratory analysis of their lipid profile, liver enzymes, and blood glucose level. The patient’s medical records were reviewed to collect data related to the patient’s history, such as WHO clinical stage, HBV/HCV status, HAART duration, HAART regimen, and current CD4.
The height and weight of the study participants were measured according to the WHO’s stepwise approach. A digital electronic adult scale (ASTO) with a Height and weight scale was used to measure weight and height. Height was measured after the study participants removed their footwear and headgear. Then they stood with their feet together and against the background, knees straight, looking straight ahead, and not looking up. The height was then recorded in centimeters. Weight was also measured after the participants stood on the scale after their footwear was removed. Then it is recorded in kilograms. Both Height and weight were measured in duplicate, and a third measurement was taken if the first two height measurements differed by greater than 1 cm or greater than 200 g for height and weight, respectively. Body mass index (BMI) was calculated by dividing the weight (kg) of the study participants by their height squared (m2). The BMI obtained was then classified by the international conventions: BMI < 18.5 kg/m2 (underweight), BMI = 18.5–24.9 kg/m2 (normal weight), BMI = 25–29.9 kg/m2 (Overweight), and BMI >30 kg/m2 (Obese). In addition, waist circumference (WC) was measured according to the WHO stepwise technique, with the erect stand-up position following normal out breathing by non-stretching tape to the nearest 0.1 cm. Furthermore, systolic and diastolic BPs were measured using a digital electronic sphygmomanometer (Omron, Healthcare, Japan) after the patient rested quietly for 15 min with their legs uncrossed. For precision, three readings were taken within a 3-min difference, and the average BP was recorded. Alcohol consumption was assessed by asking the study participants if they had ever had an alcoholic drink.
The National Cholesterol Education Program-Adult Treatment Panel III (NCEP-ATP III) 2001 guideline was used to define MetS. Accordingly, it was defined as fulfilling three or more of the following criteria: WC >102 cm for men and >88 cm for women; triglycerides (TG) >150 mg/dl; HDL-c <40 mg/dl for men and <50 mg/dl in women; BP> 130/85 mmHg or hypertension treatment; and fasting plasma glucose (FBG) >110 mg/dl or being on diabetes treatment. Liver function abnormalities were confirmed if ALT and AST level were >33 Iu/l and >35 Iu/l, respectively. To determine the AST/ALT ratio, AST was divided by the ALT of the study participants, and the ratio was reported in Iu/L. An AST/ALT ratio of ⩽1 was defined as a low ratio, and an AST/ALT ratio of >1 was defined as a high ratio.
Blood sample collection and biochemical analysis
About 5 ml of venous blood was collected using a serum separator tube. The sample was then left at room temperature until it was completely clotted. The clotted sample was then centrifuged at 300 rpm for 10 min to obtain the serum. The serum was analyzed immediately or frozen at −20°C until analysis. Serum biochemical determination of lipid profile, total cholesterol (TC), HDL-C, low-density lipoprotein cholesterol (LDL-C), and TG analysis was performed by the enzymatic method. Liver enzymes (AST and ALT) were performed using the IFCC pyridoxal phosphate method, and FPG was assayed by the glucose oxidase method using the COBAS 6000 Random Access Chemistry instrument (Roche Diagnostics International Ltd, Rotkreuz, Switzerland. The hepatitis B and C results of the study participants were recorded from their medical records.
Data management and analysis
All data collected were coded, cleaned, and entered into Epi-Data version 3.1 and exported into SPSS version 20.0 (IBM Corp., Armonk, NY, USA) for analysis. Socio-demographic, clinical, and laboratory values were described using descriptive statistics such as frequency distribution. Continuous variables are expressed as median, Interquartile range (IQR), or mean (± standard deviation). Categorical data were expressed in percentages. The correlation between MetS components and the AST/ALT ratio was assessed using the Pearson correlation coefficient analysis. A binary logistic regression was done to assess the association between the prevalence of MetS and independent variables. Those variables with a p-value of less than 0.25 in the bivariate logistic analysis were fitted into multivariate logistic regression. The Mann–Whitney U-test was used to compare the median of MetS components by gender. A p-value of <0.05 was considered statistically significant.
Data quality
A pre-test was performed at Adare Primary Hospital on 5% of the sample size to check its consistency. Sociodemographic, anthropometric, clinical, and laboratory data were collected by trained nurses and laboratory technologists who have more than 3 years of experience. The supervisors performed manual data checks every day for the completeness of the questionnaire, unexpected discrepancies, and consistency. The weight scale was calibrated by using a known calibration weight, and zero adjustments were performed every day before the measurement started. The height scale was also calibrated by using Height calibration rods. A log of calibration results was maintained for both weight and height scales. Standard operating procedures were used during laboratory testing. Moreover, the function of laboratory instruments, laboratory chemicals, and procedural performance were checked daily by using commercially prepared quality control samples.
Results
Characteristics of the study participants
A total of 302 participants were included in our study (54.6% women and 43.4% men). One-third of the participants (36.4%) attended primary school, and 170 (56.3%) participants were married. The median (IQR) age of the study participants was 41 (35–50) years, and the age range was from 18 to 73 years. 54.3% of the participants were in the age range of 31–45 years (Table 1).
Distribution of the sociodemographic characteristic of the participants of adult HIV patients at HUCSH and YGH ART clinic.

Nearly all (99.3%) of the study participants were on WHO clinical stage I, and the mean (SD) duration of the study participants on HAART was 10 (3.7) years. The median (IQR) CD4 count was 574.5 (427.8–713.5) cells/mm3, and 51.4% of the participants had a current CD4 count greater than 350 cells/mm3. The prevalence of chronic illness among the study participants was 11.9%, and heart disease (6.3%) was the most common comorbidity. The majority (82.1%) of patients were on the TDF + 3TC + DTG ART regimen. The prevalence of overweight and obesity among the study participants was 27.2% and 18.2%, respectively. Regarding the AST/ALT ratio, 41.1% of the study participants have a low AST/ALT ratio (Table 2).
Clinical characteristics of HIV patients at follow-up in HUCSH and YGH ART clinic.
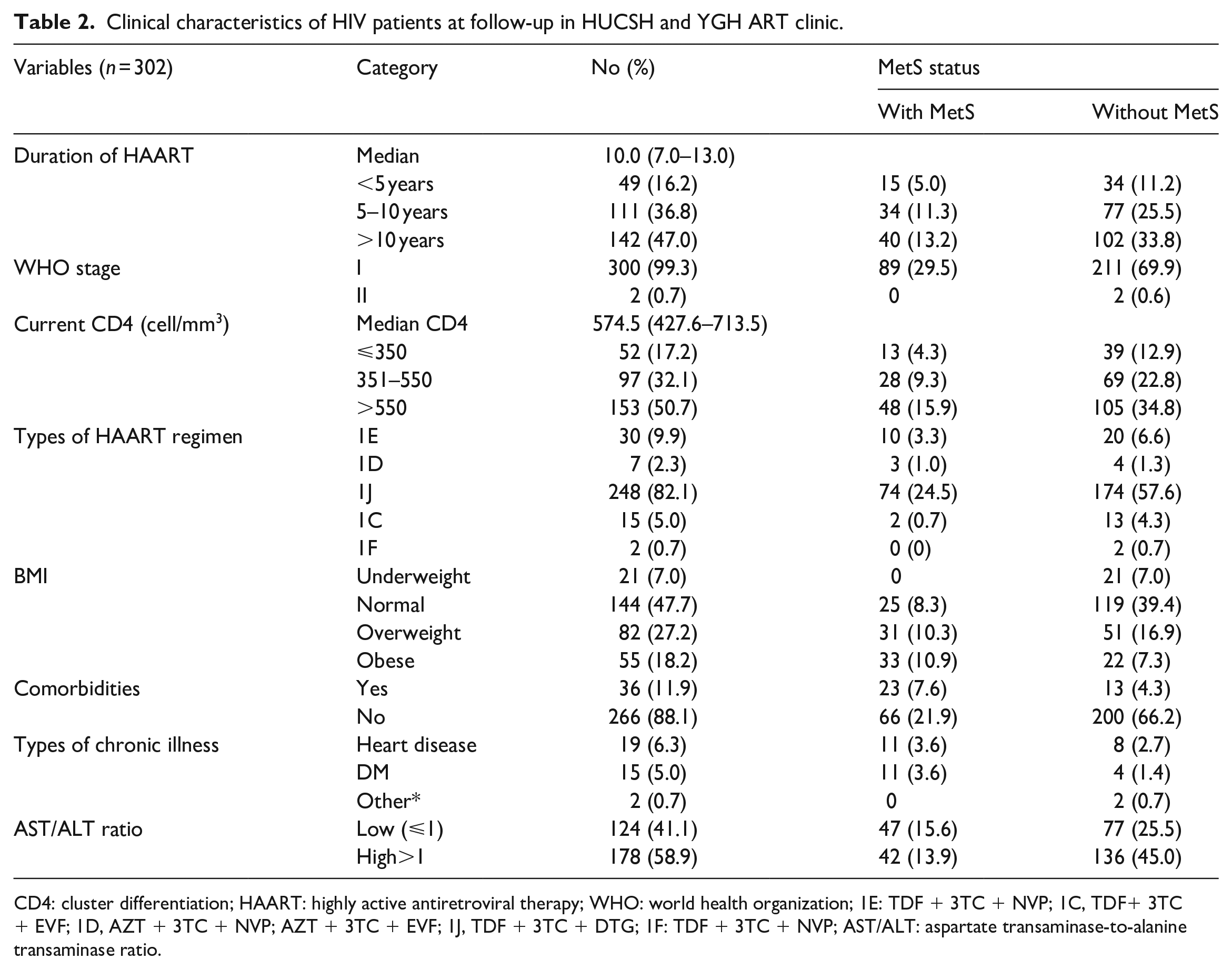
CD4: cluster differentiation; HAART: highly active antiretroviral therapy; WHO: world health organization; 1E: TDF + 3TC + NVP; 1C, TDF+ 3TC + EVF; 1D, AZT + 3TC + NVP; AZT + 3TC + EVF; 1J, TDF + 3TC + DTG; 1F: TDF + 3TC + NVP; AST/ALT: aspartate transaminase-to-alanine transaminase ratio.
Prevalence of MetS and its components among study participants
The overall prevalence of MetS among the study participants was 29.5% (CI = 24.5–35.1). The prevalence of MetS was higher among female participants than males, even though it was not statistically significant (18.2% versus 11.3%, p = 0.1). The MetS was also significantly high in the age range of 31–45 years old (Table 1). The prevalence of MetS also increased as the duration of study participants on HAART increased. Moreover, MetS prevalence was significantly higher among those study participants who had a lower AST/ALT ratio than their counterparts (15.6% and 13.9%, p = 0.007) (Table 2).
Distribution of components MetS among the study participants
The medians of TC, TG, HDL-C, LDL-C, FPG, and BMI of the study participants were 165.5 (136.0–189.5), 140.0 (105.0–201.0), 38.0 (31.2–48.0), 101.1 (80.8–120.0), 102.5 (92.0–113.6), and 23.7 (21.0–27.5), respectively. Seventeen (16.9%) of the patients had hypertension. Except for HDL-C, the medians of all MetS components were higher among study participants with MetS when compared with study participants without MetS (Table 3).
Distribution of MetS components among HIV patients with and without MetS in HUCSH and YGH ART clinic.

BMI: body mass index; DBP: diastolic blood pressure; FPG: fasting plasma glucose; HDL: high density lipoprotein-cholesterol; LDL: low density lipoprotein-cholesterol; SBP: systolic blood pressure; TC: total cholesterol; TGs: triglycerides; WC: waist circumference.
When compared to male participants, female participants had significantly higher medians of TC (170.6 versus 145.0, p ⩽ 0.001), HDL-C (41.0 versus 35.0, p ⩽ 0.001), LDL-C (108.8 versus 88.0, p ⩽ 0.001), and BMI (26.3 versus 22.0, p ⩽ 0.001) (Table 4).
Biochemical characteristics of HIV patients stratified by gender in HUCSH and YGH ART clinic.
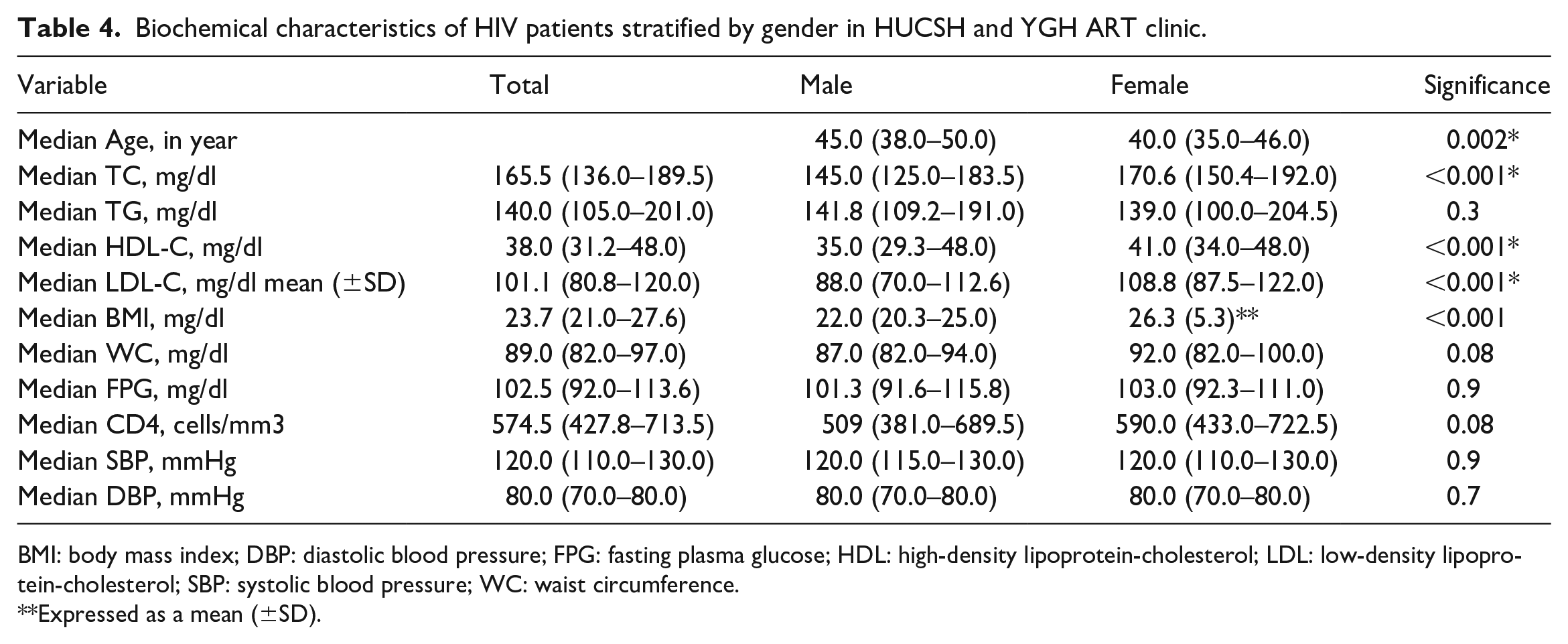
BMI: body mass index; DBP: diastolic blood pressure; FPG: fasting plasma glucose; HDL: high-density lipoprotein-cholesterol; LDL: low-density lipoprotein-cholesterol; SBP: systolic blood pressure; WC: waist circumference.
Expressed as a mean (±SD).
Characteristics of liver function among the study participants
The median (IQR) level of ALT and AST was 21.2 Iu/L (16.2–30.0) and 23.6 Iu/L (19.2–28.7), respectively. When stratified by gender, the median of AST among male participants was significantly higher than that among female participants (26.0 Iu/L versus 22.5 Iu/L, p = 0.002). Furthermore, the median ALT was significantly higher among male than among female study participants (26.0 Iu/L versus 19.0 Iu/L, p < 0.001). There is no significant difference in the medians of both AST (22.0 Iu/L versus 24.0 Iu/L, p = 0.3) and ALT (21 Iu/L versus 23.0 Iu/L, p = 0.4) among participants with or without MetS. A liver function abnormality among the study participants was 21.9% (CI = 17.2–26.5). The prevalence of liver function abnormalities was significantly higher among male study participants than female study participants (14.9% versus 7%, p < 0.001).
Prevalence of MetS according to the quartile group of the AST/ALT ratio
We divided the AST/ALT ratio into quartiles with a cutoff 0.9, 1.1, and 1.4. Study participants in the lower quartile have the highest prevalence of MetS, and the prevalence of MetS decreases with the increase of the quartile from quartiles 1 to 4 (Figure 1).
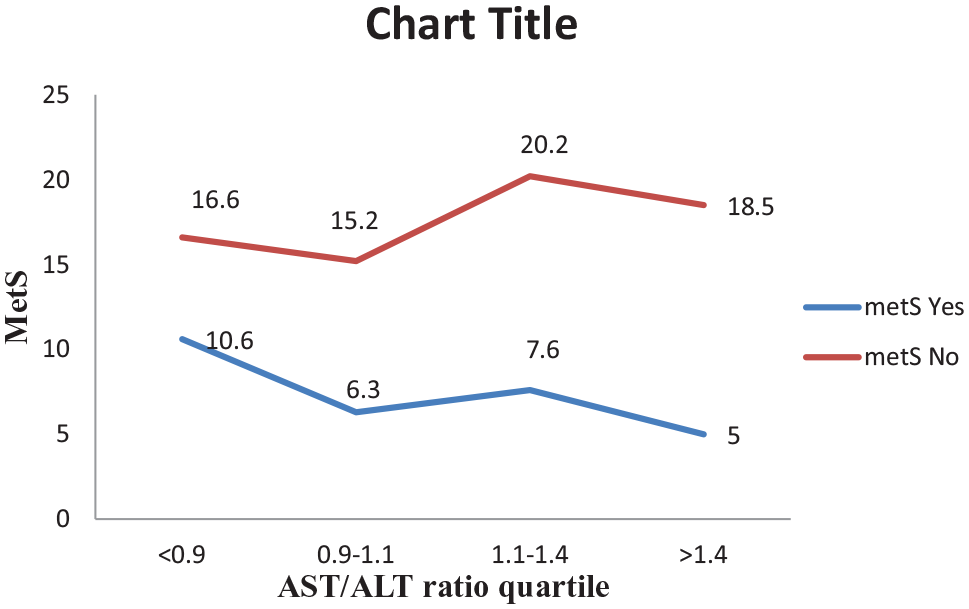
Prevalence of MetS according to quartile group of the AST/ALT ratio among patients at follow-up in HUCSH and YGH ART clinic.
Correlation of the AST/ALT ratio with individual components of MetS
From the MetS components, Pearson correlation analysis has indicated that only SBP has a significant correlation (p = 0.04) with the AST/ALT ratio. Although not statistically significant, the AST/ALT ratio has a negative correlation with FPG, BMI, WC, and TG (Figure 2).
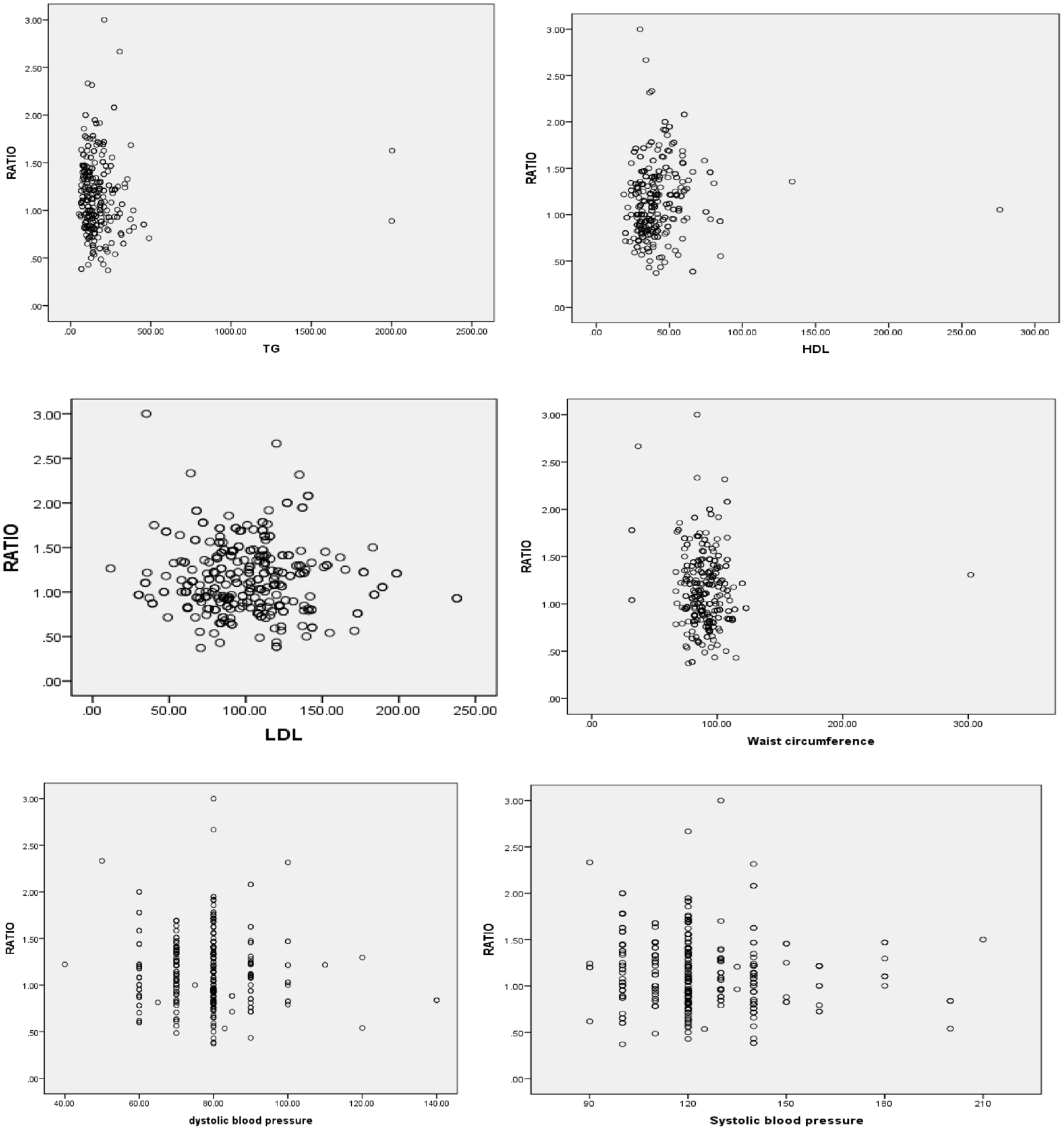
Scatter plot showing correlation of the AST/ALT ratio MetS components among HIV patients at follow-up in HUCSH and YGH ART clinic.
Predictors of the MetS
In the bivariate logistic regression, the AST/ALT ratio, sex, age, and chronic illness were candidate variables for multivariable logistic regression analysis (p < 0.25). In multivariable logistic regression analysis, chronic illness (AOR = 4.8, CI = 2.2–10.9), and AST/ALT ratio (AOR = 2.5, CI = 1.4–4.4) were associated with MetS (Table 5).
Predictors of MS among HIV patients at follow-up in HUCSH and Yirgalem Hospital ART clinic (bivariate and multivariate analysis), September 01 to June 30, 2020.

AOR: adjusted odds ratio; AST/ALT: aspartate transaminase-to-alanine transaminase ratio; CD4: a cluster of differentiation; CI: confidence interval; COR: crude odds ratio; ETB: Ethiopian Birr.
Discussion
In the present study, the overall prevalence of MetS was 29.5%. The AST/ALT ratio (AOR = 2.5, CI = 1.4–4.4) and chronic illness (AOR = 4.8, CI = 2.2–10.9) were the predictors of MetS. BP was the only component of MetS with a significant correlation with the AST/ALT ratio.
The prevalence of MetS in our study (29.5%) was in line with the studies conducted in Addis Ababa, Ethiopia (28.8%) 4 and Malaysia (26.5%), 16 and it is within the range of global prevalence (16.7–31.3%). 3 Our finding, however, was higher than studies from different regions of Ethiopia, where the prevalence of MetS was 24.6%, 15 18.1%, 17 and 21.1%. 18 The prevalence of MetS from the present study was also higher than the pooled prevalence of MetS in Ethiopia (21.7%), 19 sub-Saharan Africa (21.5%), 20 Lagos, Nigeria (1.3%), 21 Kampala, Uganda (13.9%), 22 Western Kenya (16.9%), 23 Darlinghurst (18%), 24 and Argentina (20.2%). 25 The observed MetS prevalence in our study, however, was lower than the study conducted in the Gedeo zone, Southern Ethiopia (43.4%), 26 and Southwester Uganda (58%). 27 The observed difference in MetS prevalence could be explained by variations in study design, sample size, population genetics, criteria used to classify MetS, duration of HAART, and treatment regimen.
MetS was more common among female (18.2%) study participants than male (11.3%) study participants, which was in line with studies done in Northwest Ethiopia, 18 Southwest Ethiopia, 28 and South Africa. 29 The observed higher prevalence of MetS among females in general might be due to biological and hormonal factors. 30
Regarding BP, 16.9% of the study participants had an elevated BP level, which was comparable to the study done in southern Ethiopia (15.9%) 31 but higher than the study done in Harar, Ethiopia (12.7%). 32 The prevalence of elevated BP, however, was lower than the studies conducted in Jimma referral hospital, Ethiopia (34.4%), 33 and Norway (21.0%). 34 The observed difference could be due to variations in gender, lifestyle, age difference, types of HAART, and duration of HIV infection and treatment.
The overall prevalence of liver function abnormalities among the study population was 21.9% (CI = 17.2–26.5). This finding was in line with similar studies done in Debre Tabor, Ethiopia (20.1%), Cameron (22.6%), South Africa (23.0%), and Brazil (19.7%).35–38 Direct infection and inflammation of liver cells through apoptosis, mitochondrial membrane permeability alteration that stimulates an inflammatory response, and mitochondrial dysfunction may be responsible for liver function abnormalities among HAART-experienced HIV patients. Being on ART for a prolonged period may also contribute to abnormal liver function levels.
In addition, patients with a low AST/ALT ratio were found to have 2.5 times higher odds of developing MetS than those with a higher ratio. This finding was in line with the studies conducted in Korea 9 and Thanjavur, Tamil Nadu, India 39 where the AST/ALT ratio was significantly lower in participants with MetS than those without MetS (p < 0.001). A study conducted in Mexico also observed a strong association between MetS and an AST/ALT ratio of less than one. 40
This study also found that study participants who had comorbidities were found to be five times more likely to develop MetS. Comorbidities such as hypertension, dyslipidemia, and type 2 diabetes mellitus could enhance the likelihood of MetS, and the association between these chronic illnesses and MetS was studied intensively by different scholars.4,41–43
The study participants’ characteristics, such as smoking habits, alcohol consumption, marital status, educational status, and residence, were not identified as being associated with MetS, which was consistent with studies from Hawassa University Referral Hospital and Jimma Town. 44 Furthermore, Age, BMI, WHO clinical stage, baseline CD4 count, current CD4 count, and duration of HAART regiment were also not associated with the presence of MetS, which is in line with other studies.17,44
According to this study, the AST/ALT ratio was significantly correlated with SBP. The correlation of the AST/ALT ratio with WC, TC, TG, FPG, and diastolic BP, however, is not statistically significant, which is in contrast to the study done in northeast China 45 and a retrospective follow-up study on the Chinese population. 46 This difference might be due to the total number of participants included in the study, the type of study population, or the type of study design employed.
Conclusion
Generally, this study demonstrated an elevated degree of MetS among people living with HIV. BP was the only component of MetS with a significant correlation with the AST/ALT ratio. MetS was associated with a low AST/ALT ratio and the presence of comorbidities. A global, multicenter study with a larger sample size and longer follow-up time should be conducted to determine the clinical utility of the AST/ALT ratio in predicting MetS and relatively elevated levels of MetS. For HIV patients, strategies for controlling components of MetS would be advantageous.
Limitations of the study
One of the limitations of the present study was that we did not screen the liver, gall bladder, and spleen by ultrasound. In addition, the elevation of AST and ALT levels caused by other reasons cannot be completely ruled out. Furthermore, the cross-sectional nature of the study design and small sample size would provide limited information about the association of the AST/ALT ratio with MetS and its correlation with MetS components.
Footnotes
Acknowledgements
First, we would like to acknowledge Hawassa University for facilitating this research. In addition, our appreciation goes to the data collectors, the study participants, and the staff who were working at the ART clinics of both Hospitals.
Author contributions
All authors made substantial contributions to the concept and design of the research work and the acquisition, analysis, and interpretation of data. All authors drafted the article and revised it critically for important intellectual content. All authors approved the version to be published.
Data availability
The data regarding this study can be made available upon reasonable request.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from IRB OF COLLEGE OF MEDICINE AND HEALTH SCIENCE OF HAWASSA UNIVERSITY (Ref. No. IRB/010/13).
Ethical considerations
First, an ethical approval letter for this study was obtained from the Institutional Review Board (IRB) of the College of Medicine and Health Science of Hawassa University (Ref. No. IRB/010/13), in line with the codes of the Declaration of Helsinki for Ethical Principles for Medical Research Involving Human Subjects (World Medical Association Declaration of Helsinki: November 27, 2013). Then, Support letters of collaboration were obtained from the HUCSH clinical and academic director offices and the YGH clinical director offices. In addition, written informed consent was obtained from each study participant before the start of data collection. Furthermore, the investigators had no access to information that could identify individual participants during or after data collection. Individuals were informed about their full right to withdraw from the study at any point in the study period.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study.
Trial registration
Not applicable.

