Abstract
Objective:
Patients with underlying conditions are predicted to have worse outcomes with COVID-19. A strong association between baseline cardiovascular disease and COVID-19-related mortality has been shown by a number of studies. In the current retrospective study, we aim to identify whether patients with pulmonary hypertension have worse outcomes compared with patients without pulmonary hypertension.
Methods:
Data from patients of ⩾18 years of age with COVID was retrospectively collected and analyzed (n = 679). Patients who underwent transthoracic echocardiography, at the discretion of the medical team, were identified and the transthoracic echocardiography was reviewed for the presence of pulmonary hypertension. Patient health parameters and outcomes were measured and statistically analyzed.
Results:
Of 679 consecutive patients identified with a diagnosis of COVID-19, 57 underwent transthoracic echocardiography, 32 of which were found to have pulmonary hypertension. Patients who underwent transthoracic echocardiography had a significantly higher intensive care unit admission rate (73.7% versus 25.4%, p < 0.001) and increased presence of acute respiratory distress syndrome (63.2% versus 21.6%, p > 0.001). These patients had longer intensive care unit length of stay, longer mechanical ventilation time, longer hospital length of stay, and a significantly higher mortality rate when compared to those not undergoing transthoracic echocardiography (59.7% versus 32.3%, p < 0.001). Among patients who underwent transthoracic echocardiography, those with pulmonary hypertension had significantly higher mortality compared to those without pulmonary hypertension (80% versus 43.8%, p < 0.01).
Conclusion:
COVID-19 in patients with pulmonary hypertension was associated with high in-hospital mortality even when adjusted for confounding factors. A number of mechanisms have been proposed for the worse outcomes in patients with pulmonary hypertension and right ventricular dysfunction, including right ventricle overload and indirect pro-inflammatory cytokine storm. Further, large-scale studies are required to evaluate the impact of right ventricular dysfunction in COVID-19 patients and to elucidate the associated mechanisms
Introduction
Since the emergence of SARS-CoV-2 in late 2019, the COVID pandemic has become a substantial cause of morbidity and mortality. As of May 1, 2022, over 500 million confirmed cases and over six million COVID-related deaths have been reported worldwide. 1 The main cause of morbidity and mortality has been attributed to respiratory complications such as severe pneumonia or acute respiratory distress syndrome. In addition to the direct alveolar involvement, investigations have also linked damage to lung microcirculation in COVID-19 patients.2–4 Autopsy studies have shown various degrees of pulmonary endothelial dysfunction and microvascular thrombosis.5,6 These conditions can lead to the development and complications of pulmonary pressure and right ventricle overload. The association between COVID-19 and cardiovascular diseases was recognized and addressed extensively. Cardiovascular diseases, including arrhythmias and myocardial dysfunction, have been recognized as key detrimental factors in COVID-19 patients.7–9 As early data have shown an increased mortality risk in patients with cardiovascular disease, 10 naturally there has been significant research focusing on cardiovascular complications of COVID-19 and predictors of mortality.
Pulmonary thrombosis associated with COVID-19-related ARDS can lead to thrombotic microangiography and cause pulmonary hypertension (PH), which can lead to right ventricular (RV) dysfunction 11 and increased mortality. 12 Increasing amount of imaging data has shown that the RV damage is more prevalent in COVID-19 patients than left ventricular damage.13,14 The right ventricle is directly and indirectly involved in the progression of COVID-19. The direct involvement is thought to be due to an increase in RV overload and decrease in RV contractility caused by various factors such as ARDS, pulmonary thrombosis, viral injury, and hypoxia. The indirect involvement may be due to inflammatory cytokine storm affecting the cardiovascular system caused by an increase in troponin levels 15 .
The use of transthoracic echocardiogram (TTE) in inpatient and critical care settings has been beneficial as it is a noninvasive, easily obtainable bedside procedure that can provide important clinical information. RV systolic pressure (RVSP) and function and pulmonary arterial systolic pressure (PASP) are frequently utilized echocardiographic parameters to evaluate the presence of PH and RV dysfunction without requiring invasive right heart catheterization. As the pandemic has progressed, there has been emerging data showing an association between findings of RV dilation with poorer clinical outcomes.16–18 We proposed that patients hospitalized with hypoxemic respiratory failure due to COVID-19 pneumonia that demonstrated elevated RVSP on echocardiogram had worse outcomes than patients without elevated RVSP.
Methods
Data from patients admitted to NewYork-Presbyterian Brooklyn Methodist Hospital from March to April 2020 with laboratory confirmed diagnosis of COVID-19 were retrospectively collected (n = 679). IRB approval was obtained prior to data collection (NewYork-Presbyterian Brooklyn Methodist Hospital Institutional Review Committee: IRB reference number 1589266). Inclusion criteria were all patients with a positive COVID-19 test result and inpatient admission and exclusion criteria was age of <18 years. All patients who underwent TTE (n = 57), at the discretion of the treating medical team, were identified and TTE was reviewed by a single cardiologist for the presence of PH. PASP was estimated by measurement of tricuspid regurgitation velocity and estimated right atrial pressure (RAP) based on the size and collapsibility of inferior vena cava with the formula PASP: 4v^2+ RAP. Patients with PASP > 35 mmHg were considered to have PH. Additional data collected included demographic data, Charlson Comorbidity Index, 19 intensive care unit (ICU) admission status, presence of ARDS, 20 and whether prone positioning was utilized. Chart review screened patients for a history of baseline PH prior to the current admission, of which only five patients were identified. Outcome measures were hospital length of stay (LOS), ICU LOS, total mechanical ventilation (MV) days, in-hospital mortality, and 30-day mortality.
Statistical analysis
We summarized the continuous variables using means and standard deviations and categorical variables as frequencies and percentages. We used multivariable linear regression for continuous outcomes (hospital LOS, ICU LOS, and total MV days) and multivariable logistic regression for categorical outcomes (in-hospital mortality). The final models included the outcome of interest as the dependent variable, presence of PH as the independent variable, and demographic variables as covariates. Other covariates did not improve the model fit and were not included. We examined the effect modification by including multiplicative interaction terms (PH versus demographics) but did not find any interaction. Stata/SE 15.1 (StataCorp LLC, College Station, TX) was used for all statistical analyses.
Results
Patients requiring TTE had worse outcomes
Baseline patient characteristics are shown in Table 1. We studied a cohort of 679 patients (mean age 64.6 ± 16.5) of whom 47.9% were female. Of these, 57 patients (mean age 67 ± 14.7 and 38.6 % females) underwent TTE; all patients in this sub-cohort had severe COVID, meeting the criteria of dyspnea, hypoxia, or >50% lung involvement on imaging within 24–48 h. Additionally, no patients in the cohort of those undergoing TTE had baseline PH. Other demographic variables (sex, body mass index, and race), smoking status, and presence of OSA did not show a significant difference between those who underwent TTE and those who did not. Aggregate markers of increased disease severity at baseline, both of chronic disease (Charlson Comorbidity Index) and acute disease (APACHE II Score 21 ), additionally did not show a significant difference between those who did and did not undergo TTE, although severe worsening of critical illness represented by the need for prone positioning during MV was higher in TTE versus no TTE patients (21.4% versus 3.6%, p < 0.001).
Baseline characteristics, total cohort (n = 679).

BMI: body mass index; CCI: Charlson Comorbidity Index; ICU: intensive care unit; ARDS: acute respiratory distress syndrome; LOS: length of stay; MV: mechanical ventilation.
Comparison of those with and without transthoracic echocardiogram.
Our data show that patients who underwent TTE had worse outcomes compared with the patients without TTE. Patients who underwent TTE had a significantly higher ICU admission rate (73.7% versus 20.9%, p < 0.001) and increased diagnosis of ARDS (63.2% versus 17.8%, p < 0.001). We also found that these patients had longer ICU LOS (23.0 ± 18.8 versus 12.4 ± 14.6, p < 0.0001), longer MV time (21 ± 11.2 versus 10.9 ± 10.4, p < 0.0001), longer hospital LOS (29.5 ± 19.7 versus 10.6 ± 11.9, p < 0.0001), and higher in-hospital death rate when compared to those who did not undergo TTE (59.7% versus 29.7%, p < 0.001). Mortality at 30 days showed similar findings to in-hospital mortality (43.9% versus 29.8%, p < 0.05).
The presence of PH increased the risk of mortality from COVID-19
We further investigated whether among those undergoing TTE, the presence of PH was associated with worse outcomes. Among patients who underwent TTE, 25 had PH confirmed by TTE and 32 did not (Table 2). We found no significant differences comparing PH versus non-PH patients with regard to ICU admission, presence of ARDS, ICU LOS, MV, and hospital LOS. There was, however, a significant increase in mortality in the PH group compared with the non-PH group (80% versus 43.8%, p < 0.01).
Baseline characteristics, patients with echocardiogram (n = 57).
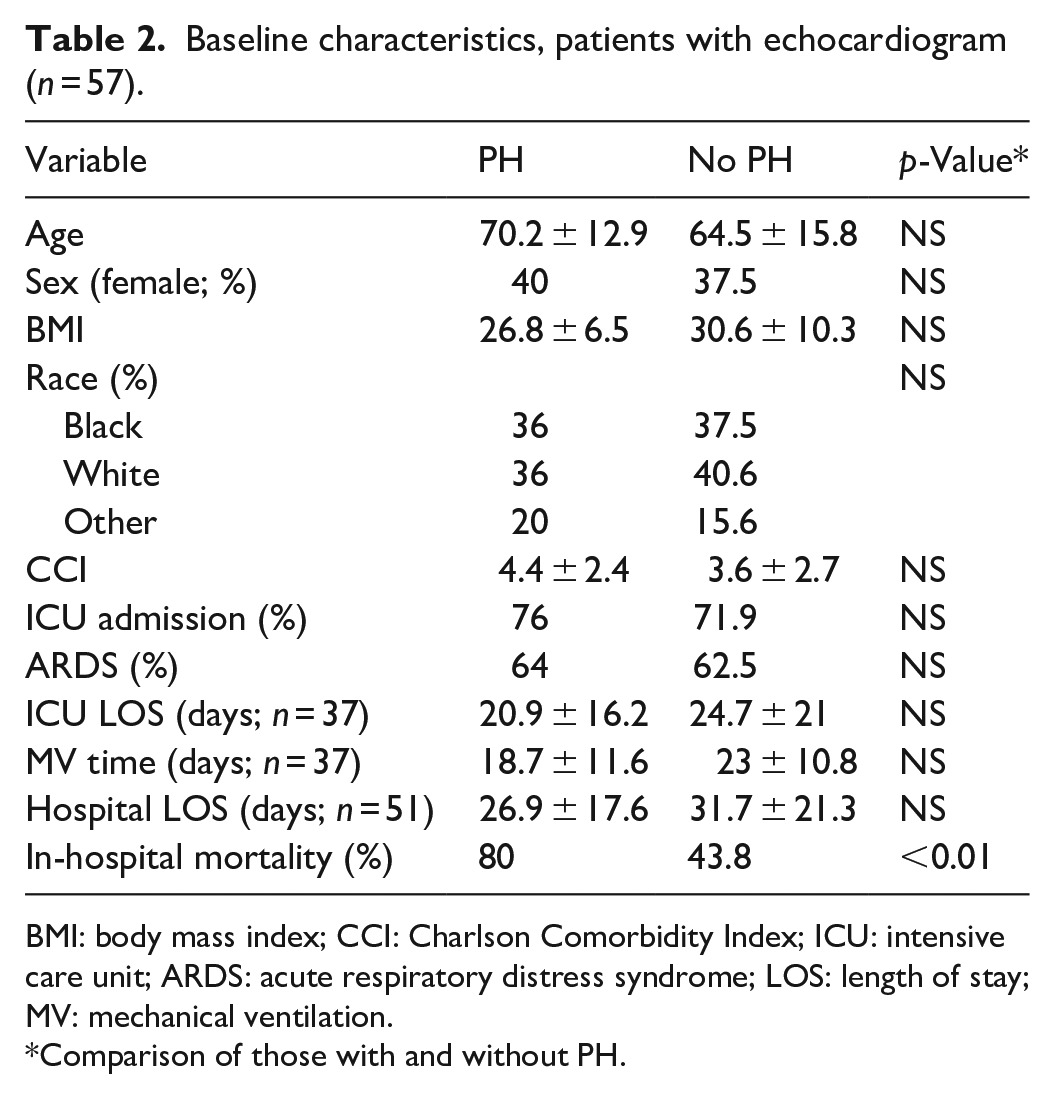
BMI: body mass index; CCI: Charlson Comorbidity Index; ICU: intensive care unit; ARDS: acute respiratory distress syndrome; LOS: length of stay; MV: mechanical ventilation.
Comparison of those with and without PH.
After multivariable adjustment via regression models, the findings remained consistent. Specifically, the presence of PH was not independently associated with ICU admission, presence of ARDS, ICU LOS, MV, and hospital LOS after adjusting for age, sex, and body mass index (Supplemental Table 1). PH was independently associated with higher mortality after adjustment for demographic variables (OR 8.9, 95% CI 1.6–49.2) (Supplemental Table 1).
Discussion
The emergence of SARS-CoV-2 and the subsequent COVID-19 pandemic has had major implications worldwide. With that, medical research has focused on elucidating pathophysiology of COVID-19 itself and the pathophysiology and predictors of its clinical sequelae and outcomes. The continuous flood of research studies has revealed patterns between risk factors and poorer outcomes in COVID-19 patients. Early in the pandemic, several studies found associations between chronic comorbidities and increased severity or complications in COVID-19. For example, obesity has been shown to have an increased risk of respiratory failure in patients with COVID-19. 22 Dessie et al. in a systematic review and meta-analysis of 42 studies, demonstrated that the presence of COPD, diabetes, hypertension, cancer, and obesity as well as male gender and older age were the clinical risk factors for a fatal outcome associated with coronavirus. 23 While the link between comorbidities and outcomes in COVID-19 is helpful, there is even greater utility in discovering an association between the measurable parameters during hospitalization and increased mortality. As bedside TTE is an easily obtainable and cost-effective procedure, it has become invaluable in diagnosing cardiac complications of COVID-19 patients and may provide prognostic value in hospitalized patients. Hence, the focus of this study was to determine whether PH alone was a predictor of mortality or worse clinical outcomes in patients with hypoxemic respiratory failure from COVID-19. In our cohort of patients hospitalized for COVID-19 in the first month of the pandemic, our data demonstrated a significant association between in-hospital mortality and the presence of PH in patients who underwent TTE during their hospital stay. We found that after multivariable adjustment for age, gender, and body mass index, the association of in-hospital mortality and PH remained while the univariate associations of PH with ICU admission, presence of ARDS, ICU LOS, MV time, and hospital LOS were no longer present.
Many complications including death in COVID-19 have been related to acute respiratory failure, with COVID pneumonia and ARDS being the most common pathologies encountered. While the respiratory system is the most commonly affected, emerging research has demonstrated significant effects on other organ systems; most notably, the cardiovascular effects of COVID-19 have been considerable and well documented. There have been reports of higher levels of serum cardiac enzymes in COVID-19, which in turn was associated with increased mortality. 24 A study by Lindner and colleagues evaluated the presence of SARS-CoV-2 in myocardial tissue from autopsy cases and found viral presence within the myocardium. 25 More systemic processes are also likely contributing, as several studies have implicated the significant inflammatory response caused by the virus promoting cytokine release. These pro-inflammatory cytokines have different effects on different tissues. From a cardiac perspective, there have been links between the cytokine cascade and ventricular remodeling, including hypertrophy and diastolic dysfunction. 26 It has also been suggested that this same mechanism contributes to pulmonary vascular remodeling, eventually leading to PH. 27
The pulmonary vasculature could also be affected via direct endothelial injury. Ackerman and colleagues compared the autopsy findings from patients with ARDS from COVID-19 versus influenza and showed that COVID-19-infected patients had significantly more pronounced endothelial injury with vascular changes including extensive thrombosis and microangiopathy compared to influenza-infected patients. 2 The proposed mechanism for endothelial damage is likely related to the inflammatory cytokine storm. These cytokines promote inflammatory cell involvement that can increase perivascular inflammation and vascular permeability, leading to fibrin and thrombi formation, which results in the proliferation of pulmonary arterial smooth muscle cells and consequently vascular remodeling.2,27 These factors can lead to the development of PH.
Hypoxic pulmonary vasoconstriction is also considered an important factor in the development of PH in COVID-19. Several studies report a possible long-term effect of hypoxemia sustained during COVID-19 infection on the degree of neurological sequalae from the disease.23,28 When hypoxia is sustained, as seen in COVID-19 pneumonia-associated ARDS, it could lead to prolonged pulmonary vasoconstriction and consequently increased pulmonary vascular resistance. 27 In combination with the inflammatory cytokine storm and endothelial damage observed with the infection, this further worsens the pulmonary hemodynamics and causes RV strain and elevated pulmonary arterial pressures as documented in our COVID-19 patient cohort. There have also been reports that this “protective” hypoxic pulmonary vasoconstriction can become impaired by the aforementioned inflammatory changes eventually worsening ventilation-perfusion matching and hypoxemia. 29
Altogether, these effects on the pulmonary vasculature lead to alterations in hemodynamics causing deleterious effects on the right ventricle. The latter is ultimately associated with worse clinical outcomes. Just as our study has highlighted, there has been developing research linking PH and RV dysfunction to increased mortality risk in COVID-19 patients.13–15 In particular, a recent meta-analysis showed a significantly increased mortality risk in patients with COVID-19 requiring respiratory support who had signs of RV dysfunction on TTE compared to those without. 30 Another study used tricuspid annular plane systolic excursion (TAPSE), a widely utilized parameter of RV function, coupled with PASP to predict mortality risk in patients with COVID-19-induced respiratory failure. A more recent investigation showed that PH in COVID-19 patients was associated with increased mortality. 31 A study by Hinojosa and colleagues showed that PH and COVID-19 patients mortality was not related to hemodynamic severity due to comorbidities. 32 Generally, as right-sided heart pressures increase, there is an associated decrease in the TAPSE, which can be measured on a bedside TTE. D’Alto et al. found that a lower TAPSE/PASP ratio was associated with increased mortality. 33 Clinically, lower PASP independently predicted survival in elderly patients admitted to the ICU due to COVID-19 pneumonia. 34 These studies and our own emphasize the utility of echocardiographic bedside diagnostics in predicting mortality in COVID-19 patients, specifically regarding parameters associated with PH and RV dysfunction.
There are several limitations present in the current study. As this was a convenience sample early in the pandemic, we were unable to characterize the echocardiographic findings of all patients with COVID-19. Our findings that there were worse outcomes in those who underwent TTE versus those who did not are to be expected; only the sickest patients in whom knowledge of cardiac function was sought underwent TTE. These findings were present despite no difference in the TTE versus no TTE groups in Charlson Comorbidity Index and APACHE II scores, as these are baseline measurements on admission to the hospital and admission to the ICU, respectively. However, the population selected for TTE were those who progressed rapidly during their hospitalization, creating a subset of very severely critically ill patients. The retrospective nature of data collection creates biases and prevented us from collecting additional information that may have elucidated the relationship between COVID-19, PH, and mortality. For example, additional patients with PH may have been missed due to the lack of TTE on all subjects included in this study. We did, however, perform sensitivity analyses including five patients identified via chart review with baseline PH prior to the current admission (who did not undergo TTE during the study admission), but these analyses did not alter our results. Additionally, this study was completed prior to the establishment of accepted treatments for COVID-19, as data was collected during the first 3 months of the pandemic; thus, external validity may be affected. Due to the retrospective nature of this convenience sample, a power analysis was not conducted to determine the required sample size.
Conclusion
COVID-19 has become a major cause of morbidity and mortality in hospitals across the globe. We demonstrated the independent association of PH and mortality from COVID-19 infection. Interestingly, other surrogate markers of disease severity based on hospital course (ICU LOS, hospital LOS, and MV) were not worse in those with PH. Additional studies are warranted to further evaluate the full impact of RV dysfunction and the presence of PH on morbidity and mortality from COVID-19 infection.
Supplemental Material
sj-docx-1-smo-10.1177_20503121231187755 – Supplemental material for Pulmonary hypertension predicts higher mortality in patients admitted with severe COVID-19 infection
Supplemental material, sj-docx-1-smo-10.1177_20503121231187755 for Pulmonary hypertension predicts higher mortality in patients admitted with severe COVID-19 infection by Ruth Minkin, Roger Hopson, Kumudha Ramasubbu, Mayel Gharanei and Jeremy A Weingarten in SAGE Open Medicine
Footnotes
Author contributions
The authors confirm contribution to the article as follows: study conception and design: RM and JW; data collection: RM, RH, and JW; analysis and interpretation of results: RM, RH, KR, MG, and JW; and draft article preparation: RM, RH, MG, and JW. All authors reviewed the results and approved the final version of the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Ethical approval for this study was obtained from NewYork-Presbyterian Brooklyn Methodist Hospital Institutional Review Committee: IRB reference number 1589266.
Informed consent
Requirement for written informed consent was waived off by Institutional Review Board and was not sought for the present study because this was a retrospective analysis study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
