Abstract
Introduction:
Globally, urothelial bladder carcinoma is a disease which carries a poor prognosis. There are various treatment modalities for urothelial bladder carcinoma with intravesical Bacillus Calmette–Guérin immunotherapy being the most efficacious intravesical therapy and the treatment of choice for patients with carcinoma in situ. A number of chemotherapeutic drugs are also available for the management of Ta/T1 tumors such as mitomycin C and epirubicin. However, relapse and progression is quite common. The optimal management of patients with Bacillus Calmette–Guérin-unresponsive disease remains to be a challenge. The purpose of this study was to conduct a systematic review on the treatment modalities available for the management of Bacillus Calmette–Guérin-unresponsive carcinoma in situ and urothelial bladder carcinoma in patients who are ineligible or decline radical cystectomy.
Methods:
Two authors independently searched three databases on the treatment modalities available for the management of Bacillus Calmette–Guérin-unresponsive carcinoma in situ and Bacillus Calmette–Guérin-unresponsive urothelial bladder carcinoma.
Results:
The systematic search resulted in 15 studies. We recommend the use of intravesical CG0070 adenovirus or hyperthermic intravesical chemotherapy mitomycin C in patients with carcinoma in situ only disease. In patients with carcinoma in situ ± Ta/T1 disease, we recommend the use of intravesical radiofrequency-induced chemohyperthermia or electromotive drug administration of mitomycin C. In patients who have Ta/T1 disease, we recommend the use of either hyperthermic intravesical chemotherapy epirubicin or electromotive drug administration mitomycin C followed by chemohyperthermia mitomycin C. If any of these second line therapies fail, an alternative regimen would be a combination of gemcitabine, cabazitaxel, and cisplatin.
Conclusion:
This recommendation is subject to the available resources and clinical expertise available in different hospitals. More studies using study designs such as randomized controlled trials comparing multiple drugs with larger sample sizes and regular follow-up intervals should be performed to accurately assess the different medications and aid in designing guidelines to guide the management of Bacillus Calmette–Guérin-unresponsive non-muscle invasive intravesical bladder cancer.
Keywords
Introduction
Globally, urothelial bladder carcinoma (UBC) is the tenth most commonly diagnosed cancer. 1 It is a disease of elderly with a median age at diagnosis of 69–71 years in both genders, 2 which carries a poor prognosis, with a median survival of 12–18 months for all ages. Approximately three-quarters of patients diagnosed with UBC have disease limited to the mucosa (stage Ta or carcinoma in situ (CIS)) or submucosa (stage T1) and in patients younger than 40 years of age this percentage is even higher. Flat, high-grade (HG) tumors confined to the mucosa are classified as CIS (Tis). 1 The gold standard for diagnosing CIS is a combination of cystoscopy, urine cytology, and histological evaluation. Diagnosis and resection of visible Ta/T1 tumors is done by transurethral resection of bladder tumors (TURBT).2,3
Guidelines are well established for management of non-muscle invasive intravesical bladder cancer (NMIBC) following staging. Intravesical Bacillus Calmette–Guérin (BCG) immunotherapy remains to be the most efficacious intravesical therapy and the treatment of choice for patients with CIS. A number of chemotherapeutic drugs are also available for the management of Ta/T1 tumors such as mitomycin C (MMC) and epirubicin. 2 80% of the patients treated initially with BCG show complete response. However, relapse and progression occurs in more than half of the patients. BCG-unresponsive (BU) tumors are defined as all BCG-refractory tumors and those with T1/Ta HG recurrence within 6 months of completion of adequate BCG exposure or CIS within 12 months of completion of adequate BCG exposure. 2 The optimal management of patients with BU disease remains to be a challenge, as there are few effective treatment modalities available apart from radical cystectomy (RC). 4 However, several novel strategies such as cytotoxic intravesical therapy, device-assisted instillation therapy, intravesical immunotherapy, systemic immunotherapy, and gene therapy have been studied and some have been approved by the Food and Drug Administration (FDA) in the management of BU NMIBC. 1 The purpose of this study was to conduct a systematic review on the available literature on the treatment modalities in the management of BU CIS and BU NMIBC in patients who are ineligible for or decline RC.
Materials and methods
Literature search
This study was designed as a systematic review based on the available literature and a comprehensive search of the literature following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines that was conducted on April 13, 2022 (Figure 1). 5

Preferred Reporting Items for Systematic Reviews and Meta-Analyses diagram.
Three databases were searched in this systematic review: PubMed, Scopus, and Cochrane library by two independent reviewers. The search strategy employed keywords with appropriate BOOLEAN operators such as: (“BCG”[All Fields] AND (“unresponsive”[All Fields] OR “unresponsiveness”[All Fields])) AND ((humans[Filter]) AND (english[Filter]) AND (2007:2022[pdat])) Translations
Duplicate articles and articles meeting the exclusion criteria were excluded. Two authors independently reviewed the abstracts of the studies and only studies eliminated in consensus were removed from the list. Two authors independently reviewed the full texts of the studies.
Inclusion criteria
Original articles within the last 15 years (written in English language)
Studies investigating treatment modalities other than intravesical BCG immunotherapy in patients with BU CIS and/or BU UBC
Exclusion criteria
Studies investigating treatment modalities other than intravesical BCG immunotherapy in patients with BCG-responsive CIS and/or BCG-responsive UBC
Studies investigating RC in patients with BU CIS and/or BU UBC
Case reports
Studies written more than 15 years ago
Review articles and meta-analyses
Book chapters
Conference abstracts
Expert opinions
Cadaveric or animal studies
Articles not written in English
We obtained 99 hits on PubMed, 120 hits on Scopus and 31 hits on Cochrane library. The systematic search resulted in 15 studies (Table 1). Articles in question were deliberated upon by the authors until a consensus decision was reached.
Summary of included studies.
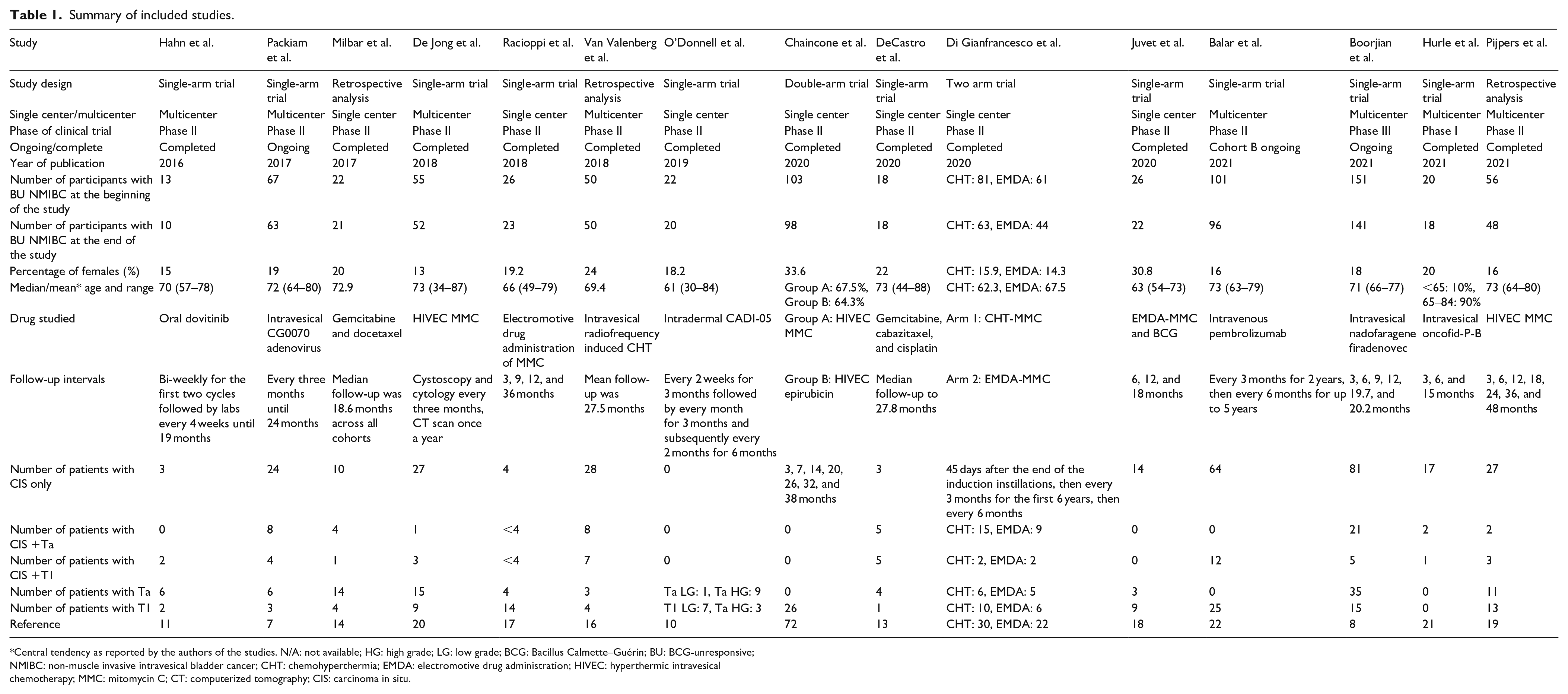
Central tendency as reported by the authors of the studies. N/A: not available; HG: high grade; LG: low grade; BCG: Bacillus Calmette–Guérin; BU: BCG-unresponsive; NMIBC: non-muscle invasive intravesical bladder cancer; CHT: chemohyperthermia; EMDA: electromotive drug administration; HIVEC: hyperthermic intravesical chemotherapy; MMC: mitomycin C; CT: computerized tomography; CIS: carcinoma in situ.
Data extraction
Two authors reviewed and extracted data from studies that satisfied the inclusion criteria. The following variables were extracted from each study: study design, whether the study was conducted in a single center or in multiple centers, phase of the clinical trial, whether the study is completed or ongoing, year of publication, number of participants at the beginning and end of the study, percentage of females in the study, mean/median age of the patients, drug investigated, duration of follow-up, number of patients with CIS only, CIS + Ta, CIS + T1, Ta, and T1 disease; the measure of treatment response, complete response rate (CR), disease-free status (DFS), recurrence-free survival (RFS), and common adverse events (AEs). A summary of the studies is mentioned in Table 1. Bias assessment was performed using the MINORS criteria for nonrandomized studies. 6 The study biases were summarized in Table 2. Below is a summarized description of each treatment modality.
Assessment of bias.

N/A: not applicable.
CG0070
Packiam et al. 7 designed a phase II trial involving 67 patients. BOND II (NCT02365818) evaluates the effectiveness of intravesical CG0070 adenovirus, in high-risk BU NMIBC patients who decline RC. Interim results of this trial at the 6-month period of enrollment were published in April 2017.
The interim data included 45 evaluable patients with CR as the primary outcome. Of the 45 evaluable patients, 24 had CIS only, 8 had CIS + Ta, 4 had CIS + T1, and 6 had Ta only and 3 had T1 stage disease. Overall, 6-month CR was about 47% (95% confidence interval (CI), 32%–62%). CR was 58% (95% CI, 37%–78%) for CIS only, 50% (95% CI, 33%–67%) for CIS ± Ta/T1, 33% (95% CI, 10%–65%) for CIS + Ta/T1, and 33% (95% CI, 8%–70%) for pure Ta/T1.
Three grade III AEs were observed. Treatment-related adverse events (TRAEs) which ranged from grade I to III were reported by 38 patients (66%). Commonly experienced side effects included bladder spasms (36%), blood in the urine (28%), painful urination (25%), and sudden urge to urinate (22%).
Intravesical nadofaragene firadenovec
One study by Boorjian et al. 8 assesses the efficacy and safety of intravesical nadofaragene firadenovec. It is an ongoing phase III trial (NCT02773849) with a planned 4-year treatment and monitoring phase. A total of 151 patients were classified as BU NMIBC. One hundred three patients were included in the CIS cohort and 48 patients were included in the Ta or T1 cohort.
At the 3-month efficacy analysis, CR was noted in 55 (53.4%, 95% CI, 43.3%–63.3%) patients in the CIS cohort. At the 12-month data cutoff, 25 (24.3%, 95% CI, 16.4%–33.7%) patients remained in HG-RFS. Seventy-three (71%) patients developed recurrent HG NMIBC, and five (5%) patients progressed to detrusor muscle invasion. Median duration of CR in patients with CIS was 9.69 months and median follow-up time was about 19.7 months. 24-month overall survival (OS) in this cohort was 91.2% (95% CI, 74.7%–97.1%).
In the HG Ta or T1 cohort, CR was provided by 35 (72.9%, 95% CI, 58.2%–84.7%) patients at the 3-month efficacy analysis. At the 12-month data cutoff, 21 (43.8%, 95% CI, 29.5%–58.8%) patients remained in HG-RFS. Twenty-three (48%) patients had HG recurrence. Three patients (6%) progressed to muscle-invasive bladder cancer (MIBC). Median follow-up time was 20.2 months and median duration of HG-RFS was about 12.35 months; 24-month OS was 93.5% (95% CI, 75.0%–98.5%).
Recurrences of any stage were observed in 104 (69%) patients, with HG NMIBC observed in 96 (61%) patients. Eight (5%) patients developed MIBC. One hundred ten (70%) patients had drug related complications. One hundred three (66%) patients reported grade I/II AEs and six (4%) patients reported grade III AEs. Most frequent TRAEs were grade I/II discharge around the catheter during instillation reported by 39 (25%) patients and grade I/II fatigue reported by 31 (20%) patients. Three patients discontinued treatment due to TRAEs. As of December 16, 2022, the FDA has approved nadofaragene firadenovec for adult patients with high-risk BU NMIBC with CIS with or without papillary tumors. 9
CADI-05
A phase II trial (NCT00694798) was performed by O’Donnell et al. 10 in which 22 patients were recruited with intermediate to high-risk BU or BCG-recurrent (BR) NMIBC. The participants were followed up for 15 months. The primary end point of the study was to determine the recurrence-free rate termed as durable response (DR) rate at 15-month period.
Efficacy analysis included 20 patients of which 8 patients had BU disease (6 patients with BCG-refractory and 2 patients with BCG-relapsing), and 12 patients had BR disease. At the end of 15-month period, 35% patients had no recurrence (seven patients). These patients with no recurrence had a persistence response till 40 months despite no treatment beyond the study follow-up period. The median time to recurrence and follow-up were 9 and 31.5 months, respectively. The DR rate was 66.67% (4/6) in patients with BCG-refractory disease, 62.5% (5/8) in the BU group, and 16.67% (2/12) in the BR group. The median RFS was 6 months in the BR group while it could not be achieved in the BU group.
Grade I/II high fever was observed in five (25%) patients and one patient experienced grade II increased urinary frequency. In addition, a total of 84 injection site reactions were reported in which 76 (90.5%), 7 (8.3%), and 1 were grade I, II, and III, respectively.
Dovitinib
Hahn et al. 11 conducted a pilot trial (NCT01732107) in BU NMIBC patients with FGFR3 gene mutations. In this study, 13 patients received a median of three prior intravesical regimens with all patients having received at least two prior BCG induction courses.
One patient (8%) showed CR, 11 patients (85%) showed no response, and 1 patient (8%) showed progression to MIBC. Dovitinib therapy was associated with frequent toxicity, and all the patients (100%) experienced at least one grade III or IV AE. The high rate of treatment-related toxicity led to frequent dose reductions. Importantly, this trial demonstrated that oral administration of dovitinib penetrated the urothelial tissue well. Therefore, dovitinib was discontinued due to drug-related toxicity rather than drug delivery failure. For this reason, further development of dovitinib in the NMIBC population is limited.
HIVEC epirubicin
Chaincone et al. 12 designed a trial to evaluate the results and side effects of Hyperthermic Intravesical Chemotherapy (HIVEC) with MMC and epirubicin for patients with NMIBC. Twenty-eight patients (27.2%) were treated with HIVEC using epirubicin (group B). Two patients were excluded from outcome analysis due to severe AEs. Histopathological data presented that 19 patients (73.1%) had tumor size <3 cm and 7 patients (26.9%) had tumor size ⩾3 cm. With regards to pathological state, 11 patients (57.7%) had TaG3 and 15 patients (42.3%) had T1G3. Five patients (19.2%) had concomitant CIS.
Maximum follow-up in patients receiving treatment with epirubicin was 32 months and DFS was about 21.9 months. Out of the 26 patients, 21 patients (80.7%) responded to the HIVEC regimen with epirubicin, 2 patients (7.7%) had HG recurrence, 1 patient (3.8%) had LG recurrence, and 2 patients (7.7%) developed disease progression.
Most common TRAEs were grade I pelvic pain (7.1%), and grade III urinary urgency (17.8%). Most AEs were mild and self-limiting. One patient was not able to complete the whole induction course due to grade II allergic reactions.
Gemcitabine, cabazitaxel, and cisplatin
DeCastro et al. 13 designed a phase I trial in which 18 participants underwent a regimen of sequentially administered cabazitaxel, gemcitabine (GEM), and cisplatin. Median follow-up time was about 27.8 months. Among the patients enrolled, three (16%) had CIS, eight (44%) had HG Ta + CIS, four (22%) had HG Ta/T1 disease, two (11%) patient had HG T1 + CIS, and one (5%) patient had HG T1 disease limited to prostatic urethra (PU) only.
CR rate found on initial evaluation after the 3-month period was 89% (16 patients), one patient (5.5%) responded partially while one patient (5.5%) with HG T1 + CIS limited to PU had recurrence revealed on biopsy. At 1-year, two more patients, one with HG-T1 + CIS in PU and other with HG-T1 + CIS in the bladder had recurrence shown on biopsy. RFS at one-year was 83% (recurrence noted in 3/18 patients with two having PU disease) while at 2-year it was 64%. Seven patients (39%) were noted to have disease recurrence at the last follow-up. Overall, median follow-up time of 21.8 months for patients with recurrence and 24.5 months for patients without recurrence were reported.
Twelve of the 18 patients experienced either grade I or II AEs with dysuria and urinary urgency being the most common side effects.
GEM and DOCE
Milbar et al. 14 described a retrospective study to determine the safety and efficacy of gemcitabine (GEM) and docetaxel (DOCE) sequential therapy in patients with BCG naïve, intolerant, and unresponsive NMIBC. In this study, 33 patients diagnosed with NMIBC who received intravesical GEM and DOCE were retrospectively analyzed. Twenty-two patients (66%) had BU disease and three (9%) had BCG-intolerant NMIBC. Both subgroups were analyzed together in the study.
The primary outcome of interest was recurrence of HG CIS (TIS), papillary tumor (HG Ta), lesions breaching lamina propria (T1) and structures beyond, and any-grade recurrence of low-grade papillary (LG Ta) or any other lesion included in the HG recurrence. HG-RFS and RFS were also determined.
Median follow-up time was about 18.6 months. Among the BU and intolerant patients, 13 (52%) patients had HG disease recurrence after administration of GEM/DOCE. Seven patients out of these had HG CIS, four had HG Ta, and two had T1 disease. For BU patients, median DFS and HG-RFS was about 6.5 months. One-year and 2-year DFS were found to be 38% and 24%, respectively. One-year and 2-year HG-RFS were found to be 49% and 34%, respectively. Only one patient had to discontinue the therapy because of intolerance. Majority of the side effects included urinary frequency (21%) and urinary urgency (18%).
Chemohyperthermia mitomycin C
A retrospective cohort study conducted by Di Gianfrancesco et al. 15 studied conservative device-assisted treatments with chemohyperthermia (CHT) and electromotive drug administration (EMDA) of MMC. In this study, 209 patients were analyzed. Sixty-three patients underwent CHT-MMC of which 10 patients had HG Ta, 30 patients had HG T1, 15 patients had CIS only, 2 patients had Ta (HG) + CIS, and 6 patients had T1 (HG) + CIS.
After a median follow-up of 59 months, HG DFS in the CHT subgroup was 65% in patients without CIS (at median follow-up time of 12 months) and 8.7% in patients with CIS (at median follow-up time of 14 months). PFS was 85% in patients without CIS (at median follow-up time of 22.5 months) and 26.1% in patients with CIS (at median follow-up time of 18.2 months). At the end of follow-up, HG DFS in patients receiving CHT were 70%, 63.3%, 6.7%, 0%, and 16.7% in patients with Ta stage disease, T1 stage disease, CIS only, Ta + CIS, and T1 + CIS, respectively. PFS were 90%, 83.3%, 26.7%, 50%, and 16.7% in patients with Ta stage disease, T1 stage disease, CIS only, Ta + CIS, and T1 + CIS, respectively. OS were 100%, 96.7%, 93.3%, 50%, and 66.7% in patients with Ta stage disease, T1 stage disease, CIS only, Ta + CIS, and T1 + CIS, respectively. The most common AEs included bladder spasm (22.2%), non-infective cystitis (19%), urinary frequency (19%), and urinary tract pain (19%).
Intravesical radiofrequency-induced chemohyperthermia
Van Valenberg et al. 16 conducted a retrospective analysis aimed to assess the effects of intravesical RF-CHT in CIS patients who were categorized as BU, other BCG-treated or BCG-naive. A total of 150 patients were enrolled for evaluation. Fifty patients were classified as BU of which 39 patients (78%) had CIS only, one patient (2%) had CIS + LG Ta, six patients (12%) had CIS + HG Ta, one patient (2%) had CIS + LG T1, and three patients (6%) had CIS + HG T1.
Mean follow-up of BU patients was about 27.5 months. CR after 6 months was about 46% (23/50). The 2-year recurrence rate of these patients was about 17% and the 2-year RFS was about 68.9%. At the final follow-up, 16% of BU patients had progressed to MIBC. OS was 76% and mean survival time was 79.7 months. Complications related to treatment were reported for the 150 evaluated patients. No specific reports were made for the 50 patients classified as BU. AEs included pain or spasms during an instillation in 7.8% of the patients, allergic reactions in 8.2% of the patients, and urinary frequency or urgency between instillations in 7.5% of the patients.
Electromotive drug administration of mitomycin C
Two studies discussed the clinical safety and efficacy of EMDA-MMC. A phase II trial was conducted by Racioppi et al. 17 to estimate the efficacy and toxicity of EMDA-MMC. In this study (EudraCT2017-002585-43), 26 consecutive patients who had BCG-refractory HG-NMIBC were selected. Four (15.4%) had TaG3, 14 (53.8%) had T1G3, 4 (15.4%) CIS, and 4 (15.4%) Ta/T1G3 + CIS with a median follow-up period of 36 months.
At the end of the induction course, CR rate was 73.1% at the end of the induction cycle and 61.5% after 3 years of follow-up, with a progression rate of 15.4%.
Stratifying patients on TNM classification (TaG3, T1G3, CIS, and Ta/T1G3 + CIS), HG-DFS rates were 75%, 78.6%, 75%, and 50%, respectively, at first follow-up; 75%, 71.4%, 75%, and 25%, respectively, at 9 months follow-up; and 75%, 71.4%, 50%, and 25%, respectively, at 12 follow-up and until the end of follow-up.
With regards to AEs, three patients (11.5%) had systemic events of hypersensitivity to MMC leading to discontinuation of therapy, and six patients (23.1%) had local side effects. The most common local side effects include pain, bladder spasm, urinary frequency/urgency in three patients (11.5%) each and dysuria in five patients (15.4%).
The second study by Di Gianfrancesco et al. 15 described the use of EMDA-MMC in 44 patients who were retrospectively analyzed. The median follow-up was 59 ± 5.3 months. Four patients experienced early systemic AEs which led to discontinuation of therapy. Ta disease was seen in six patients (13.6%), T1 disease in 22 patients (50%), CIS only in nine patients (20.5%), Ta + CIS in two patients (4.5%), T1 + CIS in five patients (11.4%).
At the end of the follow-up, in the Ta cohort, HG-DFS, PFS, and OS were 66.7%, 66.7%, 100%, respectively. In the T1 cohort, HG-DFS, PFS, and OS were 54.5 %, 72.7%, and 95.4%, respectively. In the CIS only cohort, HG-DFS, PFS, and OS were 22.2%, 44.4%, and 77.8%, respectively. In the Ta + CIS cohort, OS was 100%. In stage T1 + CIS, 20% and 80% in PFS and OS were observed, respectively. The most common side effects included bladder spasms, non-infective cystitis, hematuria, urinary frequency, and urinary tract pain in nine patients (20.4%) each.
EMDA MMC and BCG
A study by Juvet et al. 18 investigated the sequential administration of BCG and EMDA of MMC for the treatment of NMIBC following BCG failure. Twenty-six patients were considered BU. PFS was considered the primary outcome.
At a median follow-up of 871 days, 34.6% of patients (9 of 26) exhibited pathological progression, 11.5% (3 of 26) failed EMDA, necessitating additional treatment, and 73.1% (19 of 26) either experienced pathological progression or recurrence. Following BCG EMDA-MMC treatment, PFS rates were 58.3% (95% CI, 41.1–82.1) at 1 year and 48.9% (95% CI, 32.1–74.5) at 2 years from the date of induction. At 1 year, RFS was 41.9% (95% CI, 25.9–67.8), and at 2 years, it was 27.2% (95% CI, 13.6–54.4). Median survival time was 21.4 months for PFS and 9 months for RFS. At 6, 12, and 18 months, CR was observed in 16 (61.5%), 11 (44.0%), and 7 patients (30.4%), respectively. Dysuria (19.2%) and hematuria (19.2%) were among the most common side effects, all of which were grade I toxicities and were treated conservatively. Three patients (11.5%) with fever and possible bacteremia (grade III) required hospitalization and intravenous antibiotic therapy.
Hyperthermic intravesical chemotherapy administration of MMC
Three studies evaluated the effects of HIVEC MMC in patients classified as BU. The HIVEC induction regimen was identical in all three studies.
Pijpers et al. 19 conducted a retrospective analysis of 56 patients. Fifteen patients (27%) were classified as BCG-relapsing and 41 patients (73%) were classified as BCG-refractory. Eleven patients (20%) had Ta disease, 13 patients (23%) had T1 disease, 2 patients (4%) had Ta + CIS, three patients (5%) had T1 + CIS, and 27 patients (48%) had CIS only.
The median follow-up lasted 32.2 months. The HG-RFS at 1 and 2 years was 53% (standard error (SE) 6.8) and 35% (SE 6.9), respectively. In total, 64% of the patients experienced a recurrence. Patients with only papillary disease had a median HG-RFS of 15.6 months (SE 12.7) while those with concurrent CIS had a median HG-RFS of 11.9 months (SE 3.0). At 6, 12, and 18 months, the CR for patients with CIS was 70%, 45%, and 33%, respectively.
At 6, 12, 18, 24, 36, and 48 months, the cumulative incidence rate of HG tumor recurrence was 22%, 47%, 59%, 65%, and 88%, respectively. After a median interval of 11.9 months, there was disease progression in seven individuals (12.5%) at the time of the initial tumor recurrence. In total, eight individuals died, with bladder cancer being the cause of two deaths.
In total, 45 (80%) patients reported 62 AEs. Forty-four patients had grade I/II AEs and only one patient had a grade III AE. The most common TRAEs were bladder spasms (50%), urinary tract pain (16%), and urinary tract infection (14%). Two patients had to discontinue treatment due to TRAEs.
Another study was conducted by Chaincone et al. 12 as a phase II trial with 72 patients. Thirteen (18%) patients were BCG-intolerant, 31 patients (43%) were BCG-refractory, 16 patients (22.2%) were BCG-relapsed, and 12 patients (16.7%) were BU. Fifteen patients (79.2%) had Ta disease, and 57 patients (20.8%) had T1 disease. Eleven patients (15.3%) also had concurrent CIS.
The maximum follow-up period was 38 months and the mean DFS was 22.6 months. The CR rate was 70.8% (p-value = 0.47), HG recurrence rate was 19.4% (p-value = 0.28), LG recurrence rate was 4.2% (p-value = 0.61), and progression rate was 5.6% (p-value = 0.93).
Twenty-five patients (34.7%) reported TRAEs, and three patients (4%) had to discontinue treatment. Most treatment related AEs were grade I/II. Most commonly reported AEs included urinary frequency/urgency in seven patients (9.3%) and urinary tract pain in nine patients (12%).
The third study was conducted by de Jong et al. 20 as a post-hoc analysis and enrolled 55 patients. Fifteen patients (27%) had Ta disease, nine patients (16%) had T1 disease, one patient (2%) had Ta + CIS, three patients (6%) had T1 + CIS and 27 patients (49%) had CIS only. Thirty patients had concurrent CIS and 12 patients had very high risk T1 or T1 + CIS.
In total, 26 patients (50%) had DFS, 22 patients (42%) experienced recurrent disease, and 4 patients (8%) experienced progression to MIBC or metastatic disease. The median follow-up for all patients was 14 months.
The overall median DFS was 17.7 months (SE 6.72). The median DFS in patients with papillary disease only was 28.8 months (SE 11.9), while the median DFS in patients having concurrent CIS was 17.7 months (SE 6.1). The median DFS in very high risk BCG unresponsive patients was 12.1 months (SE 4.6). At 3 months, 21 (70%) concurrent CIS patients had a CR. Cumulative incidence rate of disease recurrence/progression at 3, 6, 9, and 12 months was 25%, 35%, 44%, and 53%, respectively.
Thirty-eight patients (69%) reported at least one AE during the installation course. The most common reported TRAE was urinary frequency/urgency in 19 patients (35%).
Oncofid-P-B
Hurle et al. 21 conducted a phase I trial (EudraCT 2016-004144-11) to evaluate safety and effectiveness of oncofid-P-B in patients with high risk BU NMIBC.
Of the 20 patients enrolled, 17 patients (85%) had CIS only, one had CIS + T1 (5%), while two (10%) had CIS + Ta disease. Eighty percent of the included patients were BU while 20% were BCG-intolerant.
The primary endpoint of the study was to test the safety and tolerability of the regimen. Secondary endpoints reported were CR rates at the end of intensive phase, at 6 months and at the end of maintenance period in addition to duration of response (DoR), time to progression and relapse. Evaluation for response was determined with cytology and cystoscopic biopsies of bladder at baseline, 4 weeks before institution of therapy, and at the 13–15 week interval of the study.
At the end of intensive phase, CR rate was 75% (15 patients). At 3-month of maintenance treatment, CR rate was 65% (13 patients) and at the end of maintenance phase CR rate was 40% (8 patients). Among the non-responders, four (20%) had persistent disease, four (20%) patients relapsed, two (10%) showed progression of disease, and two (10%) were not evaluated. Due to inadequate number of events median DoR could not be calculated, minimum DoR reported was three months while maximum was 12.4 months. Median time to failure was about 12.4 months.
During intensive phase, compliance rate was 91.6%. Eighteen patients (90%) reported AEs while three patients (15%) had TRAEs. Two patients (10%) experienced serious AEs. Grade III AEs or greater were experienced by six patients (30%) which included hematuria, proteinuria, nausea, and urticaria. One patient (5%) had an AE that led to discontinuation of therapy.
Pembrolizumab
Balar et al. 22 performed a phase II trial (NCT02625961) on pembrolizumab. Patients were divided into cohort A (with CIS) and cohort B (without CIS). Analysis of cohort A is complete while cohort B is still ongoing. One hundred one patients were enrolled in cohort A, out of which 96 patients were found eligible for the final efficacy analysis.
At the third month of enrollment, CR was noted in 39 patients (41%) and 18 patients (46%) were in CR for 12 months or longer with a median duration of about 16.2 months. Eleven patients (28%) had continued response beyond the data cutoff time (36.4 months) while 20 (51%) showed disease recurrence. OS was 98%, 95%, and 91% at 1, 2, and 3 years, respectively.
Thirteen patients (13%) experienced grade III or IV TRAEs. Three patients (3%) encountered immune-related AEs. TRAEs caused adjustment in pembrolizumab dosage in 13 patients (13%), and treatment withdrawal in seven patients (7%).
Discussion
For around four decades, intravesical BCG has been the standard choice for adjuvant treatment of UBC. 23 However, about 30%–77% patients experience recurrence within 5 years. 14 Furthermore, BU patients carry a 20%–40% risk for progression to MIBC within 5 years. 22 The standard of care for patients with high risk BU NMIBC is RC or additional intravesical BCG. Many patients refuse or are considered ineligible for RC. RC is also associated with significant morbidity and mortality that in turn lowers the quality of life. 8 This calls for a dire need of an effective salvage treatment.
In our systematic review, there was evidence of wide-ranging heterogeneity pertaining to drug employment, administration and dosage, follow-up period, and study end points. Additionally, different studies enrolled a variety of other groups under the umbrella of BCG failure. Hence, drawing a unanimous conclusion on every factor discussed in each individual study would be difficult.
Another potential bias includes the evaluation of response by urine cytology and cystoscopy. Urine cytology has low sensitivity in LG papillary disease, whereas certain flat lesions go undetected by cystoscopy. 24 In the study by Boorjian et al., five patients with HG recurrence would have been considered recurrence free based on urine cytology and cystoscopy. One solution to this problem is the use of advanced cystoscopy techniques such as blue light imaging which was able to detect 23% of the bladder lesions that could not be detected by white light in an evaluation of more than 3500 bladder lesions. 25
The stratification of patients based on histopathological data, a recommendation provided by the FDA, is of great importance in trials as it helps to identify higher efficacy of one subset over another subset in response to the study drug. 17 Packiam et al. showed that CIS patients had the highest CR which suggests that CIS may be the pathologic subset with the most favorable response to CG0070 adenovirus therapy. In contrast, Chaincone et al. demonstrated that CIS patients had a worse response to CHT. Boorjian et al. and DiGianfrancesco et al. showed that treatment with intravesical nadofaragene firadenovec and EMDA-MMC respectively, proved to be more efficacious in patients with papillary disease as compared to CIS patients.
In patients who decline RC, but are otherwise eligible for surgical management, patients need to be informed that the window period for RC or bladder preserving therapy may be narrowed, should they opt for nonsurgical options. 18 However, DiGianfrancesco et al. compared patients that received second line device assisted therapies using MMC (CHT and EMDA) to patients that received RC, which showed that the rates of OS and CSS were comparable between the two groups (91.6% versus 90.2%; 94.4% versus 95.1%, respectively). However, PFS was slightly lower in device-assisted patients as compared to patients that received RC (59.8% versus 75.5%).
Patient satisfaction is another parameter that was not extensively studied in any of the included studies. Some factors that could improve patient satisfaction and adherence would be less frequent instillations, orally administered drugs and less frequency and severe side effects.11,13,22
Cost is also an important factor in the decision making for the choice of drug to be used. The study by DiGianfrancesco et al. provided data regarding cost per patient which amounted to €15,000 for CHT, €6000 for EMDA, and €20,000 for RC. Hence, cost saving could only be expected in EMDA treatment. Another study by Milbar et al. reported that the cost of a six-instillation induction course of GEM/DOCE translates to roughly 125% the cost of a six-instillation induction course of BCG. What can be inferred from this is that although alternate drug regimens may be more expensive than BCG therapy, they are cheaper than RC and its associated costs.
The European Association of Urology 2021 guideline recommend patients who are ineligible or decline RC to be enrolled in clinical trials for novel drug therapies or to pursue other bladder preserving strategies. 1 Similar recommendations are offered by the American Urology Association guideline which recommend enrollment in clinical trials or intravesical chemotherapy. 26 Both guidelines don’t offer any specific recommendation to a specific drug therapy.
Our study has a few limitations. Almost all the included studies presented a very small sample size, were single arm trials and had some sort of selection bias. Half of the studies were also single center studies. These decrease the statistical significance of the results presented. We also included studies in English only. Although we aimed to include studies within the last 15 years, all our studies were published between 2016 and 2021, suggesting that limited data is available on these drugs in their management of BU NMIBC.
Conclusion
In conclusion, in patients who are BU and are ineligible for or decline RC, we have designed the following recommendation (Figure 2). We recommend the use of intravesical CG0070 adenovirus or HIVEC MMC in patients with CIS only subtype. In patients with CIS ± Ta/T1 disease, we recommend the use of RF-CHT or EMDA-MMC. In patients who have Ta/T1 disease, we recommend the use of either HIVEC epirubicin or EMDA-MMC followed by CHT-MMC. If any of these second-line therapies fail, an alternative regimen would be a combination of GEM, cabazitaxel, and cisplatin, regardless of disease subtype. This recommendation is subject to the available resources and clinical expertise available in different hospitals. More studies using study designs such as randomized controlled trials comparing multiple drugs with larger sample sizes and regular follow-up intervals should be performed to accurately assess the different medications and aid in designing guidelines to guide the management of BU NMIBC.

Recommendation designed for nonsurgical management of BCG-unresponsive NMIBC.
Supplemental Material
sj-doc-1-smo-10.1177_20503121231160408 – Supplemental material for A systematic review on the available treatment modalities for Bacillus Calmette–Guérin-unresponsive carcinoma in situ and tumors in patients who are ineligible for or decline radical cystectomy
Supplemental material, sj-doc-1-smo-10.1177_20503121231160408 for A systematic review on the available treatment modalities for Bacillus Calmette–Guérin-unresponsive carcinoma in situ and tumors in patients who are ineligible for or decline radical cystectomy by Saad Syed, Mansoor Rahman, Aisha Israr, Masroor Anwar, Sumalatha Khatroth, Danish Safi and Amir Kamran in SAGE Open Medicine
Footnotes
Acknowledgements
We would like to acknowledge Muhammad Hanif for his early contributions to the study.
Author contributions
SS drafted the study. SS, AI, MR, MA, and SK performed the systematic review and drafted the manuscript. DS and AK suggested the topic, supervised the study, and reviewed the manuscript. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Registration and protocol
The systematic review was not registered on any register. The review protocol can be made available by contacting the correspondence author.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
