Abstract
Ductal carcinoma in situ in men is incredibly rare and detection by conventional mammography and ultrasound is often challenging. We report an unusual case of a 50-year-old male, with no family history of breast cancer, who presented with an 8-year history of left-sided breast pain and recurrent bloody nipple discharge without any significant suspicious imaging features in mammography and targeted high-resolution ultrasound. Breast magnetic resonance imaging was performed as an adjunct modality. Magnetic resonance imaging findings revealed a suspicious retroareolar non-mass abnormality of segmental, linear and dendritic pattern, which was highly suspicious for a ductal carcinoma in situ. Stereotactic guided biopsy and subsequent mastectomy were consistent with pure high-grade ductal carcinoma in situ of the left breast. Overall, this case highlights the challenges in diagnosing ductal carcinoma in situ in men and demonstrates the importance for further investigating clinical suspicions of the male breast.
Introduction
Male breast cancer is extremely rare with an estimated incidence of approximately 1.1 per 100,000 a year. 1 About 1% of all malignant breast neoplasm cases occur in men. 2 Compared to its female counterpart, breast cancer in men shows differences in tumor characteristics. Moreover, there appears to be a markedly lower prevalence of invasive lobular carcinomas in men. 3 Most male breast cancer tumors are hormone receptor positive whereas HER2 positive or triple negative breast cancers are more frequent in women. 4 Concerning radiographic features, breast cancer in men can be masked by the presence of concurrent gynecomastia and tend to be fewer in number, coarser and less frequently rod-shaped than in female breast cancer. 5 There is a later age of onset, a more advanced stage at clinical presentation and an overall worse prognosis in men as compared with women.1,6,7
Pure ductal carcinoma in situ (DCIS) accounts for about 10% of all breast cancers detected in men. 8 To date, only few cases of pure DCIS in men have been reported in the literature. Respectively, diagnostic tools and treatment strategies are based on guidelines for women. Here, we report the case of a pure high-grade DCIS of the breast in a 50-year-old male patient, which has been depicted by magnetic resonance imaging (MRI) findings.
Case presentation
A Caucasian 50-year-old man was referred to our department with an extensive history of left-sided breast pain and recurrent bloody nipple discharge. The patient reported recurrent left-sided breast pain for 8 years. Cardiac diagnostics were always unsuspicious. Breast pain was assumed to be most likely of psychosomatic order. Two years ago, the initial left-sided bloody nipple discharge occurred. Mammography and targeted high-resolution ultrasound revealed gynecomastia vera of the left breast and pseudogynecomastia of the right breast. A suspected lesion was not detected. In February 2016, the patient reported to his primary care physician another instance of bloody nipple discharge. Due to high-normal prolactin blood levels and headache at presentation, a magnetic resonance tomography of the head was performed. Imaging results were unsuspicious. Subsequently, the patient was referred to our department for further diagnostics. There was no family history of gynecologic cancers. The patient was in good physical condition with a body mass index of 29. On physical examination, anisomastia with increased left-sided gynecomastia was present without any palpable masses, skin lesions or enlarged lymph nodes.
The patient underwent bilateral mammography and targeted high-resolution ultrasound. Mediolateral oblique and craniocaudal mammography showed bilateral retroareolar breast tissue which was consistent with gynecomastia. There was no evidence of any soft-tissue abnormalities. Furthermore, mammograms of both breasts showed benign, diffusely scattered microcalcifications. Mammograms were categorized according to the Breast Imaging Reporting and Data System (BI-RADS) lexicon as BI-RADS 2 (Figure 1). Subsequent targeted high-resolution ultrasound of the left breast raised suspicion for a lipoma (8 × 29 × 26 mm) without any sonographic explanation for bloody nipple discharge. Ultrasound of the right breast was unsuspicious without any masses, architectural disturbances or suspicious calcifications. Ultrasound findings were categorized as BI-RADS 0 (further imaging required) of the left breast and BI-RADS 1 of the right breast (Figure 2). Because of the equivocal findings in mammography and ultrasound, native as well as contrast-enhanced breast MRI was performed, using a 1.5T magnet. The T2 images were performed as TSE sequences (4 mm) and T1 images as fl3d sequences (2.5 mm). Native MRI confirmed bilateral retroareolar breast tissue. Contrast-enhanced MRI of the left breast revealed a suspicious retroareolar non-mass abnormality of segmental, linear and dendritic pattern, which was highly suspicious for a DCIS. Moreover, an enlarged axillary lymph node was visible. MRI findings of the left breast were categorized as BI-RADS 4 (suspicious retroareolar non-mass abnormality, biopsy should be considered). Contrast-enhanced imaging of the right breast revealed a small point-shaped contrast enhancement, probably benign, in the right retroareolar breast. Enlarged lymph nodes were not visible. MRI findings of the right breast were categorized as BI-RADS 3 (small point-shaped contrast enhancement, probably benign, short interval follow-up suggested) (Figure 3).
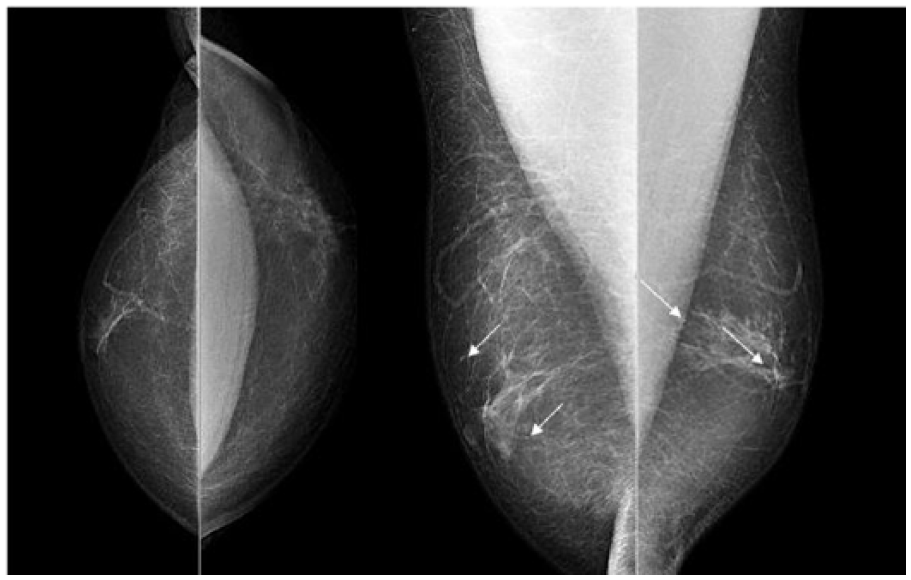
Mammography. Craniocaudal and mediolateral oblique views of the right and the left breast demonstrate bilateral flame-shaped gynecomastia. Disseminated microcalcifications are present in both breasts (arrows). A suspicious lesion is not visible. The mammograms were categorized as BI-RADS 2 (benign findings, diffuse distributed calcifications).
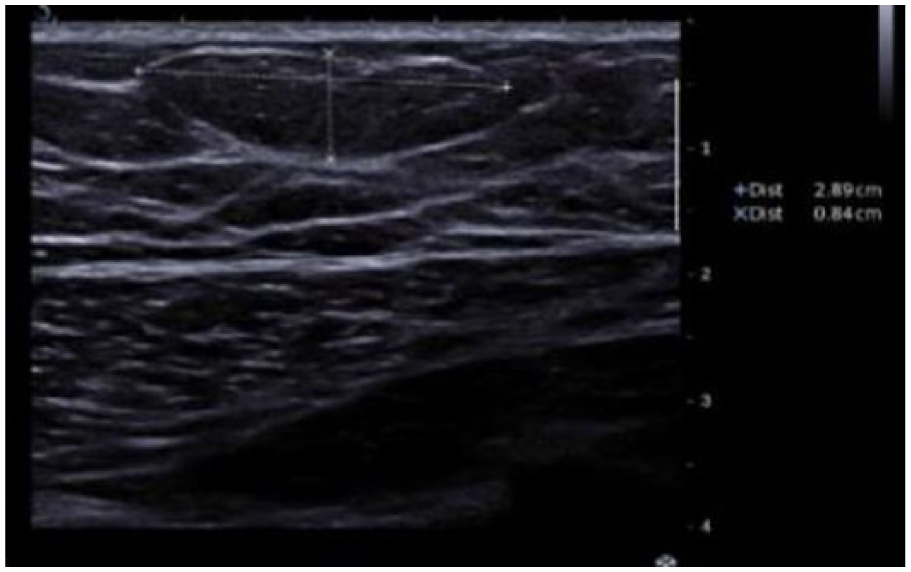
Ultrasound. Targeted high-resolution ultrasound of the left breast raised suspicion for a lipoma. Ultrasound findings of the left breast were categorized as BI-RADS 0 (further imaging is required).
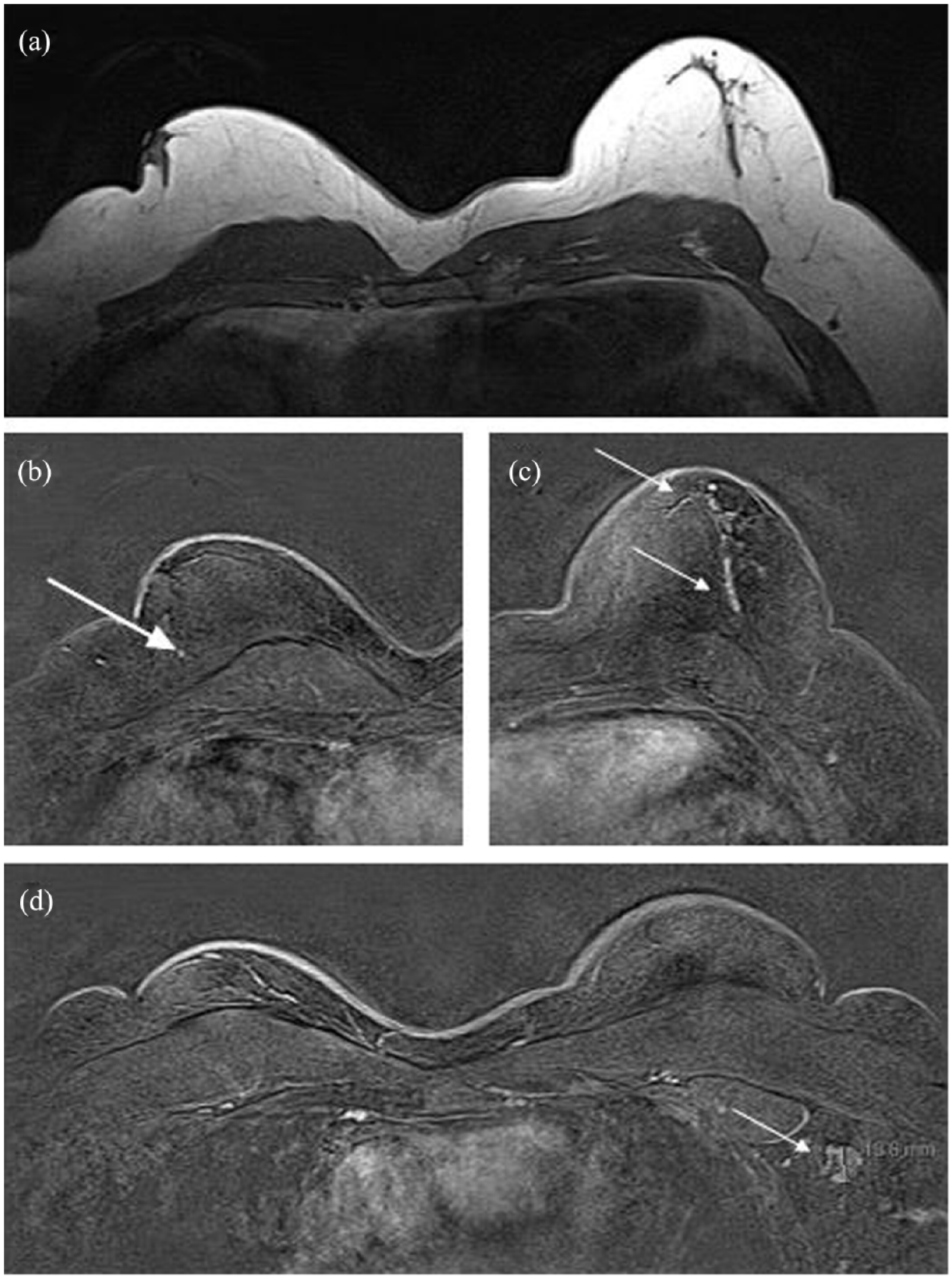
MRI findings; native (T2-TSE-tra-4 mm) or contrast-enhanced (T1-fl3d-tra-dyn-2.5 mm). (a) Native MRI shows bilateral gynecomastia. (b) Contrast imaging of the right breast demonstrates an unsuspicious retroareolar small point-shaped contrast enhancement (arrow). (c) A suspicious retroareolar non-mass abnormality of segmental, linear and dendritic pattern is visible after contrast-enhanced imaging of the left breast (arrows). (d) An enlarged lymph node of the left axillary is shown after contrast enhancement (arrow). Axillary lymph nodes of the right side are unsuspicious. MRI findings were categorized as BI-RADS 4 of the left breast and BI-RADS 3 of the right breast.
Expansion of the scattered microcalcifications in mammography was consistent with the suspicious findings in the MRI of the left breast. Therefore, the patient underwent stereotactic guided biopsy of the left breast (Figure 4). Pathologic examination revealed a DCIS of the left breast. Hormone receptor status was positive for estrogen and progesterone receptor. Invasive cancer was not detected.

Stereotactic guided biopsy of the left breast. Magnified specimen radiograph shows microcalcifications in core biopsies (arrows).
Subsequently, a simple mastectomy and sentinel lymph node excision of the left breast were performed. The sentinel lymph node showed no metastasis. The microscopic findings revealed a pure high-grade DCIS, measuring 3.5 cm at its largest diameter (3.5 × 1.8 × 1.4 cm). The tumor cells showed a papillary and cribriform growth pattern. Microcalcifications were present (Figure 5).

Histopathologic findings. (a) DCIS of papillary and cribriform pattern with microcalcifications (hematoxylin and eosin stain, 20×). (b) High-grade DCIS with big, pleomorphic cells and single-cell necrosis (hematoxylin and eosin stain, 400×).
A follow-up MRI after 6 months was bilaterally unsuspicious. The MRI showed a sclerosing lesion in the left breast which was consistent with the postoperative scar. The small point-shaped contrast enhancement in the right retroareolar breast was still detectable. The imaging was bilaterally consistent with BI-RADS 2 (Figures 6 and 7). Nipple-sparing mastectomy of the right breast was performed 6 months later due to anisomastia and asymmetry. Histological examination showed gynecomastia without any suspicious lesions. The patient has been disease-free to date.
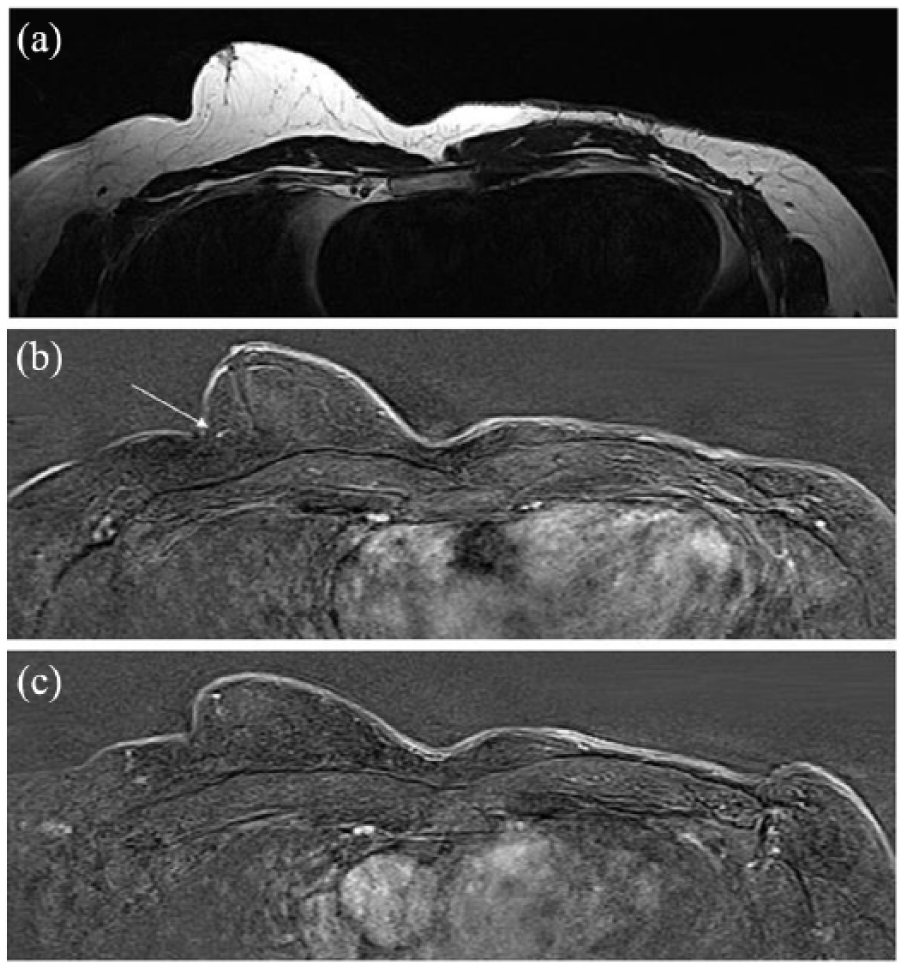
MRI findings in the follow-up native (T2-TSE-tra-4 mm) or contrast-enhanced (T1-fl3d-tra-dyn-2.5 mm). (a) Native MRI in the follow-up (6 months later) shows a sclerosing lesion in the left breast which is consistent with the postoperative scar. (b) Previous point-shaped contrast enhancement of the right breast is still visible (arrow). (c) Previous left-sided lymph node enlargement is not present anymore in contrast-enhanced imaging.
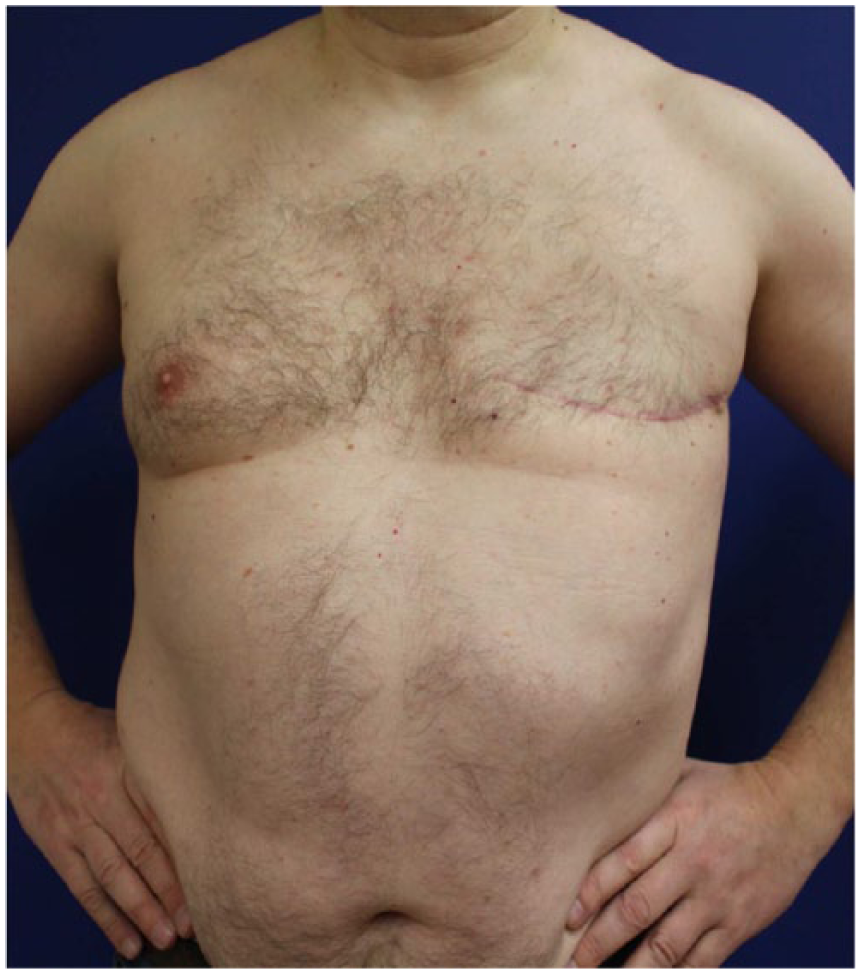
Postoperative result. Patient 6 months later after simple mastectomy and sentinel lymph node excision of the left side were performed.
Discussion
DCIS of the male breast is incredibly rare with a prevalence of approximately 0.1%. 8 Due to its rarity, diagnosis of male DCIS is a major challenge for physicians. This case is unique because of a pure high-grade DCIS in a male patient which was detected prior to progression to invasive cancer after an 8-year symptomatic left breast. Moreover, it demonstrates the value of additional MRI in men to overcome equivocal findings in routine breast imaging.
Compared to women, men with DCIS present at a more advanced stage and later age. Reasons might be a combination of no routine screening-detection, lower awareness by patients and physicians. 6 Men are less likely to assume symptoms and signs of breast cancer as palpable masses, skin lesions, lymphadenopathy, breast pain or nipple discharge, which can cause a delay in seeking treatment. Greater awareness among men and physicians is the most critical element of lowering the incidence of advanced stage breast cancer. Even in the absence of a positive family history for breast cancer, physicians should still keep it in mind. Hittmair et al. 9 showed that median duration of symptoms at presentation in pure DCIS is 2 months whereas for DCIS and invasive carcinoma, the time-span is 6 months. In the only pure high-grade DCIS case in current literature, the duration of symptoms is reported to be 6 months. 10 In our case, the duration of symptoms was 8 years. The most common sign at presentation is a retroareolar, firm, painless, palpable mass.9,10 In 39% of DCIS cases, bloody nipple discharge is the leading symptom. 8 Additional signs may be breast pain, skin lesions or lymphadenopathy.
Men with gynecomastia, obesity, diabetes, alterations of the estrogen–testosterone ratio, occupational hazards, prior radiation exposure, Klinefelter’s syndrome and BRCA1/BRCA2 mutations are at higher risk to develop breast cancer.11,12 It has been shown that in 35% of DCIS cases, gynecomastia was present. 8 Interestingly, incidental finding of DCIS in histologic analysis of gynecomastia is approximately 6%. 13 Analogous with previous reports in the literature, our patient was also having a bilateral history of gynecomastia. 8 Breast cancer in men is more likely than breast cancer in women to be related to an inherited or de novo gene mutation. 14 Up to 40% of male breast cancers may be related to BRCA2 mutations whereas only up to 10% of breast cancers in women are considered to be due to a gene mutation. For this reason, it is widely recommended that men diagnosed with breast cancer should be advised to undergo genetic testing. Studies on hyperprolactinemia as a risk factor for breast cancer are inconsistent. 15 Nevertheless, there is one case reporting about hyperprolactinemia and bilateral DCIS in men. 16 Okada et al. 16 emphasize that in DCIS patients with increased prolactin levels, the contralateral breast should also be observed carefully.
Radiologic imaging of the male breast is challenging because of its rarity and missing screening mammography. Imaging features are less familiar and diagnostic criteria are currently based on clinical studies in women. Doyle et al. 17 have demonstrated important radiological differences in mammography between breast cancer in men and women. Microcalcifications are less frequent in men and gynecomastia can mask underlying malignancy. 17 In previous studies, as well as in our case, mammography and ultrasound are often equivocal in men. 10 If mammography and high-resolution ultrasound are unrevealing, central duct excision (CDE) can be considered the gold standard in the management of bloody nipple discharge. 18 Although there is still inadequate scientific evidence, most authors agree that MRI should be performed in all patients with bloody nipple discharge and negative conventional imaging.18,19 Guidelines also recommend MRI as an additional problem-solving tool in women if clinical examination, mammography and targeted high-resolution ultrasound fail to yield a definite diagnosis. 20 This is the first male DCIS case, where native and contrast-enhanced MRI was performed. Sensitivity of MRI detection for DCIS in women ranges from 40% to 80%. 21 In our case, supplemental MRI could support the clinical suspicion for a malignant lesion. MRI findings revealed also an enlarged left-sided axillary lymph node. Consistent with MRI findings in the follow-up and pathologic examination of the sentinel node, lymph node enlargement was of reactive nature. Enlarged reactive lymph nodes could be frequently detected along the lymphatic drainage pathways, particularly of large or necrotic tumors. 22 Nevertheless, MRI findings have to be interpreted with caution, regarding false-positive and false-negative rates, especially in men.
The most dominant histologic subtype of pure DCIS in men is papillary with a superimposed cribriform pattern.8,9 In previous studies, histological grade of pure DCIS was always low to intermediate. 8 There is only one case with pure high-grade DCIS. 10 Analogous to our case report, the pathologic subtype was papillary and cribriform. 10 In previous studies of invasive breast cancer in men, a papillary histologic subtype was rare.17,23 Therefore, Cherrn et al. 24 suggest that papillary lesions in males are more likely to expand and remain in situ than other subtypes.
Currently, the standard treatment is mastectomy and sentinel lymph node excision for DCIS in men. In previous studies, simple, radical or radical-modified mastectomy with resection of the nipple–areolar complex was usually performed.8,25 Axillary dissection is not recommended anymore for pure DCIS. 6 In our case, the patient was treated with sentinel node excision followed by a simple mastectomy (Figure 4). Nipple-sparing mastectomy of the contrary breast was performed 6 months later due to consisting anisomastia and asymmetry. As in women, no adjuvant treatment is required after mastectomy in male. Concerning breast conserving surgery, there are ongoing trials to find out more about their role in male breast cancer. To date, there are no recommendations for breast conserving surgery in male patients.
In summary, early detection of DCIS in men is of great prognostic value. Due to the rarity of breast cancer in men, physicians and patients are less suspicious of the disease, which therefore might delay diagnosis and treatment to a great extent. A DCIS in a male patient should be considered whenever the clinical findings are consistent with common signs in presentation for DCIS in women. Any man with such symptoms as palpable masses, skin lesions, lymphadenopathy, breast pain or nipple discharge should obtain a detailed medical history, physical examination and be referred to an accredited cancer center. If subsequent mammography and targeted high-resolution ultrasound fail to yield a definite diagnosis, breast MRI should be performed in male patients as an additional problem-solving tool for further investigating clinical suspicions. To the best of our knowledge, this pure high-grade DCIS case of a male patient with an 8-year symptomatic medical history is the first report in current literature. Moreover, the case demonstrates the value of additional MRI in men to overcome equivocal findings in routine breast imaging.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient(s) for their anonymized information to be published in this article.
