Abstract
Objectives:
To compare Premaquick biomarkers (combined insulin-like growth-factor binding protein 1 and interleukin-6) and cervical length measurement via transvaginal ultrasound for pre-induction cervical evaluation at term among pregnant women.
Methods:
A randomized clinical trial of consenting pregnant women at the Nnamdi Azikiwe University Teaching Hospital, Nnewi, Nigeria. The women were randomized equally into Premaquick group (n = 36) and transvaginal ultrasound group (n = 36). The cervix was adjudged ‘ripe’ if the Premaquick test was positive or if the trans-vaginal measured cervical length was less than 28 mm. The primary outcome measures were the proportions of women who needed prostaglandin analogue for cervical ripening and the proportion that achieved vaginal delivery after induction of labour. The trial was registered in Pan African clinical trial registry (PACTR) registry with approval number PACTR202001579275333.
Results:
The baseline characteristics were similar between the two groups (p > 0.05). There was no statistically significant difference between the two groups in terms of proportion of women that required prostaglandins for pre-induction cervical ripening (41.7 versus 47.2%, p = 0.427), vaginal delivery (77.8 versus 80.6%, p = 0.783), mean induction to delivery interval (22.9 ± 2.81 h versus 24.04 ± 3.20 h, p = 0.211), caesarean delivery (22.2 versus 19.4%, p = 0.783), proportion of neonate with birth asphyxia (8.30 versus 8.30%, p = 1.00) and proportion of neonate admitted into special care baby unit (16.7 versus 13.9%, p = 0.872). Subgroup analysis of participants with ‘ripe’ cervix at initial pre-induction assessment showed that the mean induction to active phase of labour interval and mean induction to delivery interval were significantly shorter in Premaquick than transvaginal ultrasound group.
Conclusion:
Pre-induction cervical assessment at term with either Premaquick biomarkers or transvaginal ultrasound for cervical length is effective, objective and safe with similar and comparable outcome. However, when compared with women with positive transvaginal ultrasound at initial assessment, women with positive Premaquick test at initial assessment showed a significantly shorter duration of onset of active phase of labour and delivery of baby following induction of labour.
Keywords
Introduction
Induction of labour is an efficient and safe method for vaginal delivery. However, it is not free of complications. It is associated with increased risk of failure and caesarean delivery especially when performed on an unripe cervix. 1 When compared to spontaneous onset of labour, induction of labour is associated with increased risk of caesarean delivery and perinatal morbidities. 2
In an attempt to reduce the risk of caesarean section as a result of failed induction, a cervical scoring system was developed by Bishop and later modified to evaluate the readiness or inducibility of the cervix for induction of labour.3,4 However Bishop score is subjective and not reproducible with high intra- and inter-observer variations and has shown to be a poor predictor of outcome of labour induction. 5 This has led to development of other newer and more modalities or methods like transvaginal ultrasound (TVUS) measurement of cervical length and checking for the presence of phosphorylated forms of insulin-like growth factor-binding protein-1 (IGFBP-1), fetal fibronectin, interleukin-6 (IL-6) or their combination in cervico-vaginal fluid at term. 6
Previous studies have shown that TVUS cervical length measurement is a precise and objective method of cervical evaluation when compared to Bishop score because the supra-vaginal portion of the cervix which make up about 50% of the entire cervical length is very difficult to evaluate digitally especially when the cervix is closed.7,8 Additionally, relying on Bishop score may lead to overtreatment of patients initially considered to benefit from administration of prostaglandins as a result of low Bishop score. 8
A phosphorylated form of IGFBP-1 is predominantly produced by human decidual cells and it is present between the chorion and decidual. 9 The chorion detaches from the decidua as pregnancy advances toward labour and releases phosphorylated form of insulin-like growth factor-binding protein-1 (phIGFBP-1) into cervico-vaginal secretions. Thus, the presence of phIGFBP-1 in cervico-vaginal secretion is an indication of decidual activation and dilatation of the internal cervical os. 9 This may signify imminent onset of labour. The detection of phIGFBP-1 in cervico-vaginal secretions has been shown to be associated with increased risk of preterm labour, and when present at term, it is an indication that the cervix is ripe for induction of labour. 10 Thus, the presence phIGFBP-1 is about four times in a ripe cervix than unripe ones. 11 phIGFBP-1 when compared to other chorio-decidua cell adhesion molecules like fetal fibronectin is cheap, and the result is not easily affected by contamination by urine or recent sexual intercourse and the test is rapid. 12
Inflammatory cytokines like IL-6 have also been implicated in the aetiology of labour through their effect in synthesis of prostaglandins. 13 Previous study has revealed that maternal serum IL-6 level increases before spontaneous onset of labour at term and the concentration is said to be significantly higher in serum of women that will have spontaneous labour in 48 h than those women who do not go into spontaneous labour at ⩾14 days. 14 Hence detection of IL-6 in serum or cervico-vaginal secretions at or before term may likely indicate that labour is imminent.15–17
Newer available bedside-friendly, point-of-care, rapid test kits have been developed to detect those labour biomarkers from cervical secretions that can signal the imminent onset of labour. Premaquick, developed by Biosynex SA (Strasbourg, France), is one of such point-of-care rapid tests. Premaquick biomarker is a trio of IGFBP-1 (native/intact form, total/cleaved form and IL-6). The native IGFBP-1 corresponds to the full-length protein sequence and the total IGFBP-1 corresponds to IGFBP-1 native and/or N-terminal fragment. 18 IL-6 is a glycosylated protein with the typical four-helix bundle structure. 19
Although, most of the prior studies that compared TVUS measurement of cervical length and Bishop score for pre-induction cervical assessment at term have shown that cervical length measurement by TVUS is superior to Bishop score.20,21 Premaquick in a randomized study has demonstrated to be superior to Bishop score for pre-induction cervical evaluation. 6 Since newer studies have demonstrated the superiority of either TVUS measurement of cervical length or Premaquick over Bishop score, which is an age-old method of pre-induction cervical assessment, it will be necessary to compare these two superior methods in a randomized clinical trial.
We therefore hypothesize that cervicovaginal fluid Premaquick biomarkers predictive of labour may be similar in the term labour setting and will be superior to transvaginal ultrasonographically measured cervical length (TVUS) in pre-induction cervical assessment. We also understand that neither test (Premaquick or TVUS) is part of standard care; a recent Cochrane systematic review indicated that direct comparisons via randomized control trials between modalities of assessing pre-induction cervical assessment such as phIGFBP-1, vaginal fetal fibronectin or TVUS or Bishop score for assessing pre-induction cervical ripening among parturients at term gestations was yet to be carried out. 22 The use of IGFBP-1 or IL-6 could permit the feasibility of self-sampling and self-assessment, and the use of TVUS can reveal other clinical useful information before pre-induction cervical ripening. Additionally, a recent randomized control trial by Eleje et al. that compared Premaquick biomarkers with Bishop score for pre-induction cervical assessment at term, the authors stated as one of their limitation that their study did not evaluate the performance of Premaquick compared to TVUS for cervical length assessment, and the authors recommended that such comparison could be a subject for future studies. 6 Although Bishop score compares favourably with TVUS, the major problem of TVUS is that both the equipment and skill are not readily available in low-income settings and there could be clear cut-off cervical length value that is most likely to indicate benefit from a cervical ripening agent prior to induction of labour.6-8 Hence our present study is intended to fill the research gap by comparing Premaquick versus TVUS assessment.
This study is therefore aimed to compare combined IGFBP-1 and IL-6 (Premaquick biomarkers) versus transvaginal ultrasonographically measured cervical length for pre-induction cervical evaluation for pregnant women admitted at term for induction of labour.
Methods
Study setting
The study was carried out at Nnamdi Azikiwe University Teaching Hospital (NAUTH), Nnewi. The NAUTH is a tertiary health institution situated in Nnewi, Anambra state, South-East Nigeria. The study was conducted from August 2020 to February 2021
Study population
It involved consented pregnant women that presented at term (37–42 weeks), prepared through and admitted into the labour/delivery ward via the antenatal ward of the hospital.
Study design
This was a randomized prospective clinical study. The eligible participants were randomly allocated into two equal groups for pre-induction cervical assessment viz Premaquick group and TVUS group using computer-generated random numbers. The randomization was performed by the principal researcher using randomly permuted blocks (blocks of four, allocation ratio 1:1) with the software available online (http://www.randomization.com). The randomization in blocks of four included one from the Premaquick or transvaginal group.
All nulliparous women at term (37–42 weeks) with singleton and cephalic fetuses, intact fetal membranes and no contraindications to vaginal delivery were eligible for the study. Exclusion criteria were women with previous caesarean or uterine scar, preterm pregnancy, fetal abnormal lie/presentation, multiple gestation, pre-induction fetal heart rate abnormalities and intrauterine fetal death. The data was collected using a structured proforma which captured information on the biodata of the patient, clinical presentations/clinical findings, interventions and outcome measures. All the consenting pregnant women at term for cervical ripening and induction of labour who met the inclusion criteria were enrolled into the study. Relevant history was obtained from the antenatal cards of the patients followed by general physical examinations and obstetrics examination. The sample size (n) for each group was calculated to be 36, based on a power of 80, 5% error margin and estimated attrition rate of 10%.
The primary outcome measures were proportion of women who needed prostaglandin analogue for cervical ripening after pre-induction cervical assessment by each method and proportion of women who achieved vaginal delivery after induction of labour in each group. The secondary outcome measures were as follows: mean induction to delivery interval (in hours) after induction of labour in each group, proportion of women who were delivered by caesarean section after induction of labour in each group, the mean total doses of prostaglandins administered for pre-induction cervical ripening in each group, proportion of neonates with birth asphyxia (Apgar score less than 6 in 1 min) in each group as well as proportion of neonates that required admission into the new born special-care baby unit in each group.
Sample size calculation
The sample size was obtained using the formula n = {u√[π1(1−π1) + π0(1−π0)] + v√[2π m (1−π m )]}2/(0.64−0.25)2
Where n = required minimum sample size, u = a power of 90% is assumed for the study corresponds to 1.28, π0 = proportion of patients that had adequate labour induction at Bishop score of ⩽4 = 0.25, 8 π1 = proportion of patients that had adequate labour induction at TVUS length of ⩾28 mm by Park et al. (which is a similar randomized clinical trial as the index proposed study) = 0.64, 8 π m = mean of the two proportion = 0.45, v = percentage of the normal distribution corresponding to the required (two-sided). Therefore, a minimum sample size of 64 was obtained and rounded up to 72 (36 in each group) to cater for 10% attrition.
Study procedure
Eligible participants in the antenatal clinic scheduled for cervical ripening and induction of labour who met the inclusion criteria were informed of the study and written consent obtained. The consent was confirmed at a presentation in the labour ward. Eligible unbooked parturient that presented in the labour ward for the first time was also recruited. The recruited participants were equally and randomly allotted into the Premaquick group and TVUS group using computer-generated random numbers. Group assignment was predetermined and allocation concealment was performed using serially numbered sealed non-transparent envelopes with a piece of paper inside that bore the label ‘Premaquick’ or ‘transvaginal ultrasound’. The sealed envelopes were stored and opened by independent staff (nurse midwife) of the hospital. Participants’ allocation never changes after the envelope has been opened. A detailed history was obtained and case file reviewed, a general physical examination and obstetric examination were performed. Routine Bishop score was done and documented. A pre-induction CTG was performed and a transabdominal scan done to rule out contraindications to vaginal delivery. Further intervention performed was based on the group the patient belonged.
Premaquick test was done for Premaquick group. Having placed the patient in dorsal position and posterior vaginal fornix exposed with a self-retaining Cusco speculum. The Premaquick kit (containing sterile Copan flocked swab, tube containing 1 ml of buffer solution and a cassette) was opened by the assistant. The sterile swab was introduced into the exposed posterior vaginal fornix to absorb cervico-vaginal fluid/secretions for about 15–30 s and allowed for optimal absorption. The sterile swab was removed and placed in the buffer solution. Rotating for about 10 s in the buffer solution, two to three drops of the already mixed buffer solution with cervico-vaginal fluid were placed into each well of the three biomarkers (IGFBP-1 N, IGFBP-1 T and IL-6) and the result was interpreted and recorded in the patient’s chart. The cassette has C and T letters that correspond to Control and Test lines. The presence of three control lines (C) was important for validation. The test was said to be positive when two or three T lines of the biomarkers were present. The negative test was when the three T lines were absent or when there was the presence of only one T line.6,23
For participants in the TVUS group, TVUS measurement of cervical length was done using an ALOKA GmbH Prosound scanner (model SSD-3500 with a 5.0/6.0-MHz vaginal probe made in Japan). The internal os of the cervix was located as a dimple against the hypoechoic background of the amniotic fluid. The external cervical os was found by following the distal contour of the posterior cervical lip. In the absence of uterine contractions, the cervical length in mm was measured from the internal to the external cervical os through the endocervical canal. Three different measurements were obtained and the average was taken for the study.24–26 A cervical length of less than 28.0 mm was considered favourable (ripe) while length ⩾28.0 mm was said to be unfavourable (unripe) for labour induction. 8
For women that were not contracting, both Premaquick and transvaginal cervical length were measured. The measurement was repeated every 6 h until the time of induction of labour or transition into labour, whichever comes first. The obstetric care provider on duty in the labour ward who was not blinded to the Premaquick or TVUS results to enable him or her to know when to commence oxytocin continued to manage the patients in accordance with the NAUTH, Nnewi, Nigeria labour ward protocol. 26 The cervix was considered unripe when Premaquick was negative or when transvaginal cervical length was greater than or equal to 28.0 mm. 8
In participants with an unripe cervix, 50 μg of misoprostol (cytotec Pfizer comprimidos misoprostol, lot b132221) was given intravaginal every 6 h. A maximum of four doses of misoprostol were administered. No further oxytocics were administered to the woman when uterine contractions reached a frequency of 3 in 10 min. For failed cervical ripening, caesarean section was performed after the fourth dose of misoprostol. In participants with a ripe cervix (i.e., positive Premaquick or TVUS cervical length of <28.0 mm), oxytocin infusion was commenced using standard induction of labour protocol of the hospital. Thus intravenous oxytocin (10 IU in 1000 ml of 5% dextrose in water) was administered using gravity-fed manual drip set at 10 drops per min and increased by 10 drops per min every 30 min, up to a maximum of 60 dpm or adequate uterine contractions were obtained.6,27,28 Amniotomy was done when the presenting part was at station 0, dilated at least 4 cm and the fetal head was well applied to the cervix.
Caesarean section was performed for women that have not reached active phase (at least 4 cm cervical dilatation) by the end of this period. For progress of labour halted at <1 cm cervical dilatation per hour during the course of induction of labour, oxytocin infusion in incremental doses, as per protocol above was commenced. However, oxytocin augmentation was delayed until the duration from the last dose of misoprostol administration was at least 4 h to minimize the risk of hypertonus or hyperstimulation syndrome. Continuous electronic fetal heart rate monitoring with cardiotocography was used in all cases and partograph to monitor labour events.
Successful induction of labour was defined as the ability to achieve vaginal delivery. Failed induction was defined as inability to achieve active phase of labour within 24 h of commencement of induction of labour. 25 Failure to progress was defined as the absence of cervical dilatation during the active phase of labour for the last 2 h or inability of the presenting part to descend while in the second stage of labour for at least 1 h in the presence of adequate uterine contractions. 25
The study was approved on 28 May 2019 by the Ethics Review Committee of NAUTH, Nnewi, Anambra State, Nigeria, with reference number: NAUTH/CS/66/VOL 12/024/2018/027; The trial was registered in PACTR registry with approval number PACTR202001579275333. The study was conducted based on ethical principles for medical research involving human subjects according to Helsinki declarations.
Statistical analysis
Statistical Package for Social Science computer software version 16 was used for the data analysis. Continuous and categorical data were compared using Student’s t test and Fisher’s exact test, respectively. Considering that all tests were two-sided, statistical significance was considered to be at a probability value of ⩽0.05.
Results
Of the 83 women that were assessed for eligibility for the study, 4 declined to participate, while 7 did not meet the inclusion criteria. Therefore, 72 nulliparous women who underwent induction of labour that met the inclusion criteria were randomized into the Premaquick group (n = 36) and TVUS group (n = 36). A flow diagram describing the women’s flow through the study is shown in Figure 1. Tables 1 and 2 summarized the socio-demographic and clinical characteristics of both groups. There was no significant difference in the baseline socio-demographic and clinical data of the two groups (p > 0.05).

Flow chart of the participants in the study.
Participants socio-demographic of both groups.
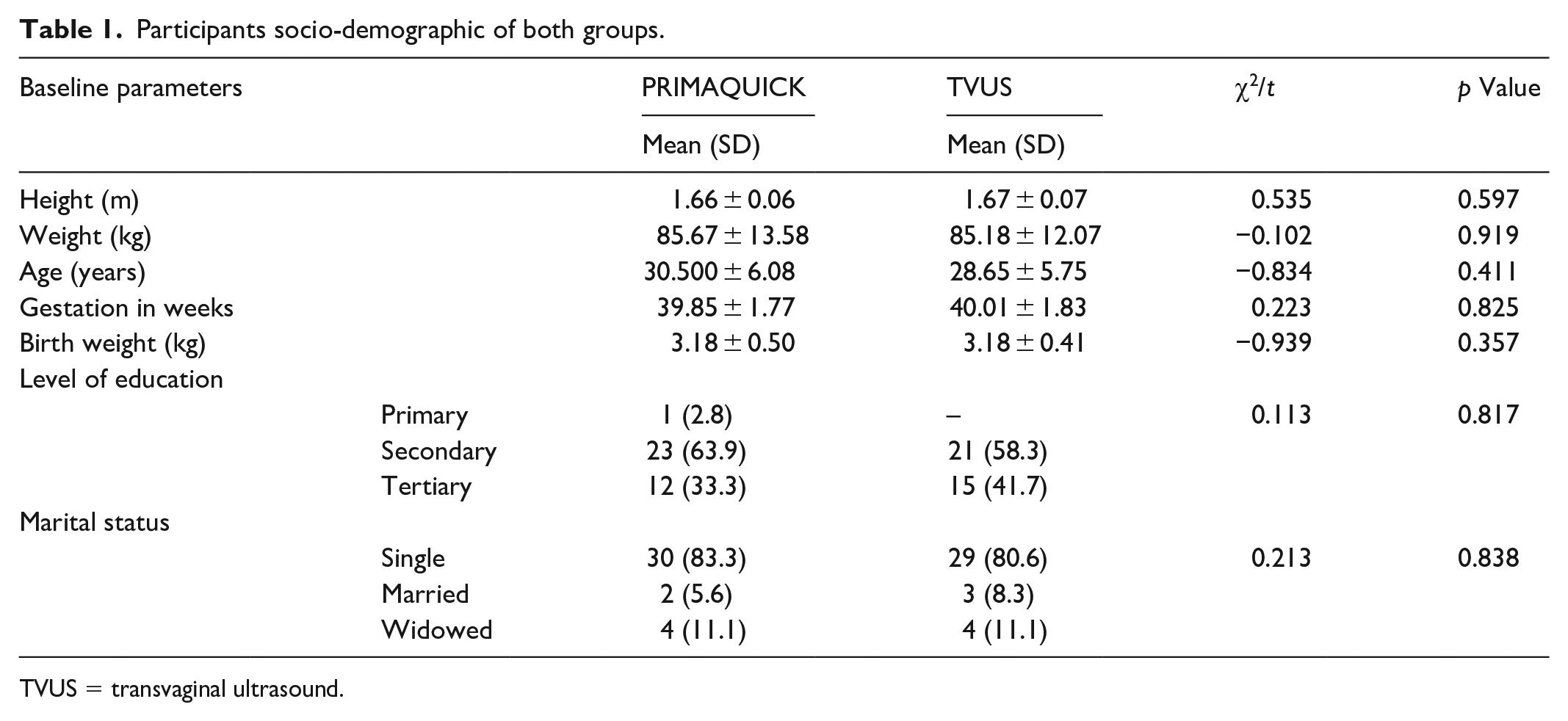
TVUS = transvaginal ultrasound.
Clinical characteristics of both groups.

TVUS: transvaginal ultrasound.
For the Premaquick group, 21 (58.3%) women tested positive for the Premaquick biomarkers, while 15 (41.7%) women tested negative for Premaquick biomarkers at initial pre-induction cervical assessment. The Premaquick positive participants (i.e., those with ‘ripe’ cervix) were commenced on straight induction of labour with oxytocin while the Premaquick negative women (i.e., those with ‘unripe cervix’) had cervical ripening/priming with synthetic prostaglandin E1 analogue (misoprostol (cytotec) by Pfizer comprimidos misoprosol, lot b132221). In the TVUS group (n = 36), 19 women (52.8%) had cervical length of <28 mm (‘ripe cervix’ or positive TVUS) while 17 women (47.2%) had cervical length ⩾28 mm (i.e., ‘unripe’ cervix or negative TVUS; see Figure 1, the flow chart).
At study recruitment of participants, the studied base line parameters, including the height, weight, age, gestational age in weeks, level of education, Bishop score and other parameters, did not show a significant difference between the two groups (p > 0.05, for all). This is shown in Tables 1 and 2. The indications for induction of labour were compared and found to be similar in both groups. As shown in Table 3, the commonest indication for induction of labour in both groups was post-datism. The other indications included hypertensive disorders in pregnancy, diabetes mellitus in pregnancy, previous immediate neonatal death and maternal wish (see Table 3).
Indications for induction of labour in both the Premaquick and TVUS group.

Post-datism is defined as pregnancy lasting beyond expected gestational age of 40 weeks +0 day. Chronic hypertension in pregnancy means hypertension that occurred prior to pregnancy or before 20 weeks of gestation. Pregnancy induced hypertension is hypertension that occurs in a pregnant woman after 20 weeks of gestation in a previously normotensive pregnant woman. TVUS: transvaginal ultrasound.
Fisher’s exact test other Pearson’s χ2 tests was used.
For the total participants enrolled in Premaquick group (n = 36), 15 women (41.7%) tested negative to the Premaquick biomarkers and hence required synthetic prostaglandins E1 analogue (misoprostol) for cervical ripening and induction of labour while 17 (47.2%) women in the total TVUS group (n = 36) had cervical length ⩾28 mm and needed cervical ripening and induction of labour with misoprostol (p = 0.427). The mean dose of misoprostol in microgram required for cervical ripening and induction of labour was more in TVUS group (150.0 ± 30.0) than the Premaquick group (140.0 ± 40.0), However, the difference was not statistically significant (p = 0.501). This is shown in Table 4.
Comparison of the primary outcome measures in both Premaquick and TVUS group.

TVUS: transvaginal ultrasound.
For number of spontaneous vaginal deliveries, 28 (77.8%) women in the Premaquick group had successful vaginal delivery while 29 (80.6%) of the total TVUS group achieved successful vaginal delivery (p = 0.783). The mean induction to delivery interval in hours was 22.9 ± 2.18 h in the Premaquick group while the TVUS group spent 24.04 ± 3.2 h (p = 0.211). The caesarean section rate did not differ in the two groups (p = 0.783). Details are as shown in Table 4.
A total number of six babies (16.7%) in the Premaquick group were admitted into the special care baby unit (SCBU), while five babies (13.9%) in the TVUS group were admitted into the SCBU (p = 0.872). The incidence of birth asphyxia was also comparable between the two groups (p = 1.00). Details are as shown in Table 5.
Comparison of the secondary outcome measures in both Premaquick and TVUS groups.

TVUS: transvaginal ultrasound; SCBU: special care baby unit.
Out of the total number of twenty-one women (n = 21) that had positive Premaquick at the point of recruitment, all of them had vaginal delivery 21 (100.0) and all the nineteen women (n = 19) with cervical length less than 28 mm (n = 19) also had vaginal delivery 19 (100.0) with (p = 0.317) as shown in Table 6. The induction to active phase of labour interval in hours in the positive Premaquick group and positive TVUS group were 8.63 ± 1.77 and 9.7 ± 2.73, respectively (p = 0.049), while the induction to delivery interval in hours in both groups were 16.3 ± 2.82 and 18.9 ± 3.82 (p = 0.031), respectively. This is shown in Table 6. There was no difference in the incidence of admission into SCBU or birth asphyxia (p > 0.05). Details are as shown in Table 6.
Comparison of participant with positive Premaquick and positive TVUS (cervical length <28 mm) group.

TVUS: transvaginal ultrasound; SCBU: special care baby unit.
For the negative Premaquick group (n = 15) at the point of recruitment, the mean dose of misoprostol in micrograms required for cervical ripening and induction of labour was 140.0 ± 40.0 while that of TVUS ⩾28 mm (n = 17) was 150.0 ± 30.0 (p = 0.501). This is shown in Table 5. The mean induction to active phase of labour interval in the negative Premaquick group and negative TVUS group (cervical length ⩾28 mm) were 13.63 ± 1.07 and 14.87 ± 1.47 (p = 0.313), respectively.
In both groups, the induction to delivery intervals were 28.27 ± 1.31 and 29.17 ± 1.51 (p = 0.656), respectively. There was also no difference in the incidence of admission into SCBU or birth asphyxiation (p > 0.05). There was no recorded case of uterine rupture during the trial. Details are as shown in Table 7.
Comparison of participant with negative Premaquick versus negative TVUS (cervical length ⩾28 mm).

TVUS: transvaginal ultrasound; SCBU: special care baby unit.
Figure 2 shows the relationship between proportion of women who participated in the study and duration of labour in the two arms.

The relationship between proportion of women who participated in the study and duration of labour in the two arms.
Discussion
The ‘take home message’ or principal findings of this study are that the number of women that required prostaglandins for pre-induction cervical ripening were the same for women receiving Premaquick and those receiving TVUS. Also, no differences were observed for vaginal delivery rate, average induction to delivery time, cesarean delivery rate, number of babies with birth asphyxia and those admitted into the SCBU. Therefore, both methods or agents for preparing the cervix before induction of labour are effective, objective and safe.
The requirement for Prostaglandins for pre-induction cervical ripening in the Premaquick and TVUS group was 41.7 and 47.2%, respectively. This observed need for prostaglandin for cervical ripening was similar to Eleje et al.’s findings, which stated that the need for misoprostol for cervical ripening was 44.7% in women following Premaquick testing. 6 With regards to the TVUS group, this finding is similar to a previous randomized control study by Park et al. which revealed that prostaglandin was needed in 36% of the nulliparae for cervical ripening. They concluded that TVUS for pre-induction cervical assessment can reduce the need for prostaglandin administration by approximately 50% without affecting the outcome of induction of labour in nulliparae at term if the cut-off value used was ⩾28.0 mm. 8 Similarly, in another randomized control trial by Bartha et al., 50% of women required prostaglandin following pre-induction cervical assessment in TVUS group.
Interestingly, in this study, there was no statistically significant difference in the proportion of women that had successful vaginal delivery in both Premaquick and TVUS groups (77.8 versus 80.6%, p = 0.783). Similarly, Park et al. reported a vaginal delivery success rate of 77.0% in TVUS group. 8 This finding is also similar to a previous randomized control trial on nulliparous women by Eleje et al., which reported a vaginal delivery success rate of 78.9% in Premaquick group. 6 The findings are interesting as a greater number of women had successful vaginal delivery following cervical ripening and induction of labour in either method. This finding further suggests that both methods of pre-induction cervical assessment are effective and safe.
When the subgroup analysis was considered, it was observed that the mean induction to active phase of labour interval and the mean induction to delivery interval in those with positive Premaquick (8.63 ± 1.77 versus 9.7 ± 2.73; p = 0.049) and positive TVUS <28 mm (16.3 ± 2.82 versus 18.9 ± 3.82; p = 0.031) were statistically lower in Premaquick group. This finding agrees with the previous study report on Premaquick biomarkers by Eleje et al., where the induction to the active phase interval was 9.2 ± 4.1 h, and the induction delivery interval was 14.2 ± 7.9 h. 6 For studies on TVUS, Bartha et al. observed an induction to active phase of labour interval of 5.7 h and Park et al. observed 5.7 ± 3.4 h.8,29 The lower interval observed in these studies on TVUS might be due to different definitions of active phase labour in those studies. The above findings were also similar to the findings by Valikkannu, where they concluded that IGFBP-1 is a stronger independent predictor of success of induction of labour and vaginal delivery within 24 h than TVUS in nulliparous women and bed side testing of IGFBP-1 may help in making decision concerning induction of labour in nulliparous women. 30 Also the present findings are similar to EL Mekkawi et al. study that revealed that the induction to delivery interval was significantly shorter in women with cervical length <28 mm than those with cervical length ⩾28 mm (p = 0.02; 95% confidence interval: 4.9, 8.4). 26 Similarly, Dogl observed that IGFBP-1 test (a component of Premaquick test) is an independent predictor for successful induction within 24 h in post term pregnancies. 31
Regarding the proportion of neonates with birth asphyxia (8.30 versus 8.30%, p = 1.00) and proportion of neonates admitted into the SCBU (16.7 versus 13.9%, p = 0.872). There was no statistically significant difference between the two groups studied. These findings were similar to Eleje et al.’s findings on women assessed using the Premaquick for pre-induction cervical assessment. Similarly, Park et al. and Bartha et al. concurred with the findings of TVUS group. Although, their incidence of neonatal admission into the SCBU was lower. The difference could be due to the environment of practice, as ours were done in low- income settings where the problem of birth asphyxia and neonatal admission is expectedly higher.
In women with positive Premaquick and those with positive TVUS <28 mm at the time of initial evaluation, it was observed that out of 21 women that had positive Premaquick test and 19 women that had cervical length <28 mm, all the women in both groups had vaginal delivery (21 (100.0%) versus 19 (100.0%), p = 0.317) with no recorded case of caesarean delivery in both groups. There were also no recorded cases of uterine rupture. This finding is also interesting and further suggests that both methods are safe and effective. According to Kosinska-Kaczynska et al.’s findings, phIGFBP-1 may be a useful predictor of spontaneous onset of labour within 24 hs and successful vaginal birth but its sensitivity is comparable with TVUS cervical length measurement. 32 Their findings were comparable in multiparous women, but in nulliparous women, only phIGFBP-1 and TVUS measurement of cervical length were comparable and significantly sensitive in predicting spontaneous onset of labour and vaginal delivery. 30 This is also similar to the findings from the index study. Premaquick biomarker which contains phIGFBP-I is a simple to use, bed side objective point of care test that can be used to predict the success of induction of labour either alone or in combination with TVUS cervical length assessment. Also, in another study, it was observed that antenatal measurement of cervical length may be of great importance in counselling women on the mode and gestational age of delivery as women with short cervices were more likely than those with long cervices to have successful vaginal delivery. 33 However, elongated cervices have no effect on the duration of labour. All of the women with cervical length <28 mm in the current study had vaginal delivery further stressing the significance of cervical length assessment on the outcome of labour.
From this study, TVUS cervical length has also demonstrated to be effective as Premaquick. However, considering the cost of TVUS measurement of cervical length, limited availability of skills, Premaquick is less expensive, easy to use and does not require specialized training to perform and this may be of great benefit for pre-induction cervical evaluation especially in low resource poor settings. Also in some clinical settings where route of delivery is equivocal like clinically stable women with preeclampsia where there is need for delivery and other clinical conditions, Premaquick biomarker test results or TVUS for pre-induction cervical assessment might guide clinical decisions for proper patient management during cervical ripening and induction of labour. However, one of the problem with the use of TVUS measurement of cervical length for pre-induction cervical assessment is the lack of consensus cut-off as previous different studies have suggested different cut-off but for the purpose of this study, a cervical length cut-off of 28 mm similar to that done by Park et al. was used. 8
The clinical relevance of prostaglandin use as a primary outcome was due to the fact that these were outcomes evaluated in previous randomized control trials on similar topics. For example, a randomized control trial by Park et al. aimed at comparing sonographically measured cervical length with the Bishop score in determining the requirement for prostaglandin administration for pre-induction cervical ripening in nulliparae at term, revealed that the use of sonographic cervical length for assessing the cervix prior to induction of labour can reduce the need for prostaglandin administration by approximately 50% without adversely affecting the outcome of induction. 8 In another randomized controlled trial by Bartha et al., it was concluded that the use of TVUS instead of Bishop score for pre-induction cervical assessment to choose an induction agent significantly reduces the need for intracervical prostaglandin treatment without adversely affecting the success of induction. 29 The assessment for the need for prostaglandin administration was largely due to international comparisons. Additionally, included the need for prostaglandin administration as a primary outcome of interest in our present trial because in the previous Cochrane review by Ezebialu et al., only two trials reported on it. 22 The authors of the Ezebialu’s Cochrane review reported that the need for misoprostol for cervical ripening was more frequent in the TVUS group compared to the Bishop score group. Thus, the clinical relevance of prostaglandin use as a primary outcome is very interesting because needless use of prostaglandin agents could trigger uterine hyperactivity and uterine hyperstimulation, which can jeopardize the perinatal life, increase operative intervention, uterine rupture risks and hospital costs. Moreover, prostaglandin use (misoprostol) causes both cervical ripening and uterine contractions. As a result, more women progress through to vaginal birth without requiring an oxytocin infusion, which has potential safety benefits in the use of Premaquick for pre-induction cervical assessment.
Another clinical relevance of our study findings is that term birth prediction could have huge benefits to counselling women regarding likelihood of vaginal delivery – for example, in vaginal birth after cesarean section, or management of high risk women who wish to avoid delivering outside the hospital – for example, placenta praevia or unstable lie. However, such patients will have repeat elective cesarean section (vaginal birth after CS) or elective caesarean section (placenta praevia) instead of induction of labour. Additionally, another clinical relevance of our study findings is that the new methods (Premaquick) involve testing for the presence of IGFBP-1 and IL-6, their combination in cervicovaginal fluid. However, term and preterm labour events share common pathways of cervical ripening, even though the instigating prompts may differ. The detection of IGFBP-1 in the cervicovaginal fluid indicates a disruption of the choriodecidual interface, which can occur in cases of preterm and term labour events. This appears to illustrate the reason why the positive Premaquick group compared to the CL <28 mm group had a shorter induction to delivery or active phase interval. The 1–2 h difference might be important because it is enough for fetal distress to develop and time for resuscitation to be embarked on.
The strength of the study is that the study was randomized and conducted among nulliparous women only. Hence, previous vaginal delivery as a confounding variable was eliminated. The subjects were recruited strictly in accordance with the inclusion and exclusion criteria, thereby safeguarding the outcome against confounders. Additionally, to the best of the knowledge of the researcher, the researcher could not find out from existing literature any study that has compared IGFBP and IL-6 biomarkers versus cervical length measured using transvaginal ultrasonography for pre-induction cervical assessment at term. Previous studies have either compared IGFBP-1 and IL-6 with other techniques or agents for pre-induction cervical assessment. Notwithstanding, one of the limitations of the study is the single centre population which may deter generalization of findings. We could also not determine or compare the cost-effectiveness of the two methods. Further study involving a larger sample size and multicentre randomized studies will help to further validate our above findings. A cost-effectiveness analysis is also needed in future studies so as to evaluate their potential utility in low-income settings. Additionally, our study did not evaluate the three-armed comparison including Bishop score performance with Premaquick and TVUS for cervical length assessment. Such a three-arm comparison could be a subject for future studies. The study did not isolate the exact concurrence rates of the tests, and it is subject to future trials. We kept the discussion rather restricted to the outcomes of interests because a full discussion of the confounders such as chorioamnionitis, meconium and fetal distress, which necessitated operative delivery, was not potentially exhaustive.Also, we have not fully channelled our discussion in line with the ARRIVE Trial (a Randomized Trial of Induction Versus Expectant Management) and needs to be balanced against the risk of stillbirth from continuing pregnancy in pregnancy induced hypertension or post-dates. 30 This is because, in our study participants, majority (more than 60%) of the participants were at postdate gestation (more than 40 weeks gestation) at the start of the trial. A recent study concluded that the results of the ARRIVE trial should be carefully evaluated in different demographic and clinical settings and cannot be extended to the general population. 34
Conclusion
In conclusion, pre-induction cervical assessment with either Premaquick or TVUS for cervical length at term is effective, objective and safe, and both gives similar and comparable outcomes. However, when compared with women with positive TVUS at initial assessment, women with positive Premaquick test at initial assessment showed significantly shorter duration of onset of active phase of labour and delivery of baby following induction of labour.
Footnotes
Acknowledgements
The authors are grateful to everyone that contributed to the success of this study most especially the pregnant women that took part in the study, NAUTH Nnewi and Biosynx France. To God be the glory.
Author contributions
CGO and GUE contributed to conceptualization of the study, design, manuscript writing and revision. JI Adinma, EOU, COO, JII, GOU, BON, EOU, COE, OSU, CBO, CJO, CBO, CAO, ECE, KNO, TKN and JOU contributed to manuscript writing and revision. CGO, CCO, ROE, CPI, ABO, CCO, HIO, CCU, JEJ, PCO, AOI, LUO and LIE contributed to manuscript writing, data collection and analysis.
Disclosure statement for publication
All the above named authors contributed immensely towards: conceptualization and design of the study, or collection of data, or analysis/interpretation of data, drafting and thorough review for final approval of the submitted version. An abstract/E-poster of this work was presented at the FIGO international world congress in October 2021 and published in international journal of Gynaecology & Obstetrics/volume 155, issue52/p 127–532. ![]() . The full manuscript has not been submitted for publication in another journal.
. The full manuscript has not been submitted for publication in another journal.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The principal investigator bore the cost of transvaginal ultrasound scan and misoprostol (cytotec by Pfizer). Biosynex France provided the Premaquick Kits free of charge. There was no financial inducement of any sort from Biosynex.
Consent to participate and ethical approval
All the participants gave written informed consent to participate in the study. The study was approved on 28 May 2019 by the Ethics Review Committee of NAUTH, Nnewi, Anambra State, Nigeria with approval number: NAUTH/CS/66/VOL 12/024/2018/027. The study was conducted based on ethical principles for medical research involving human subjects according to Helsinki declarations.
Informed consent
All the participants gave written informed consent to participate in the study.
Trial registration
The trial was registered in PACTR registry with approval number PACTR202001579275333.
