Abstract
Objective:
Urinary tract infection has catastrophic health outcome among diabetes mellitus patients. This study was conducted to investigate prevalence of bacterial uropathogens, their antibiogram, and associated factors among diabetes mellitus patients in Kombolcha town.
Method:
Cross-sectional study was done from February to April 2020. A total of 282 study participants were involved and simple random technique was applied for enrollment of subjects. Well-constructed and pre-tested questionnaire was utilized to collect the data. Five to ten milliliters of mid-stream urine specimen was collected for microbiological data. Each sample was inoculated into cystine lactose electrolyte deficient medium, incubated overnight in aerobic atmosphere at 37°C for 24 h and finally biochemical tests were carried out. According to Kirby–Bauer disk diffusion method, the antimicrobial susceptibilities pattern of the bacteria was performed on Mueller–Hinton Agar. The data were analyzed with Statistical Package for Social Sciences version 23. Descriptive statistics, bi-variable, and multivariable logistic regression analyses were performed. For statistical significance association,
Results:
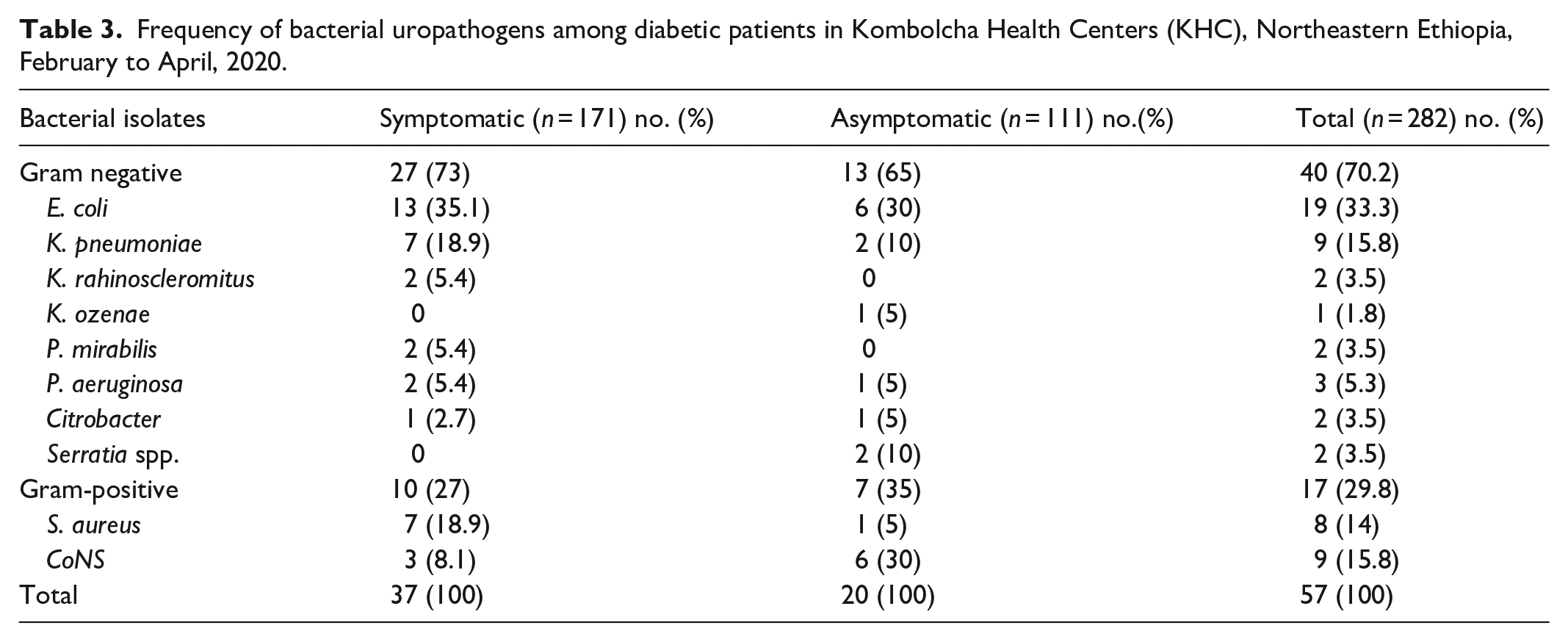
The general prevalence of urinary tract infection among study participants was 20.2% (57/282). Being female and having merchant occupation had a statistically significant association. Gram-negative bacteria accounted for 70.2% (40/57) and the leading isolate was
Conclusion:
The general prevalence of significant bacteriuria was found to be consistent with similar studies conducted in different areas. The higher multidrug resistance rate was observed for the isolated bacteria. Health-care professionals should give due attention and follow rational antibiotic prescription practices to treat these infections.
Introduction
Urinary tract infection (UTI), which is the most common cause of morbidity in the general population and in the health facilities,1–3 is defined as the presence of significant bacteria in urine specimen. 4 It is a clinical condition, which ranges from the presence of bacteria in urine without showing any symptom up to serious symptomatic situations.5,6 For example, significantly higher prevalence of pyelonephritis had been shown due to bacterial infection of the urogenital area. 7 Uropathogen, which is the causative agent of UTI, imposes formidable challenges on the clinical and economical aspects of the patient, particularly if the pathogens are developing resistance against the common antimicrobial agents. 8
The global incidence, prevalence, death, and disability-adjusted life-years (DALYs) associated with diabetes will become higher and higher within a few years in the near future. 9 Diabetes mellitus (DM) patients have shown higher opportunity of developing genitourinary tract infections. 10 These infections among DM are related to immune system dysfunction and variation to different biochemical substances. Elevated glycosylated hemoglobin (HbA1c) has predisposed diabetics to UTI 7 and also lower urinary cytokine and leukocyte concentrations have been demonstrated among DM patients than non-DM study participants. 11
The isolation of uropathogen from the urine culture varies in different localities with wide-ranging prevalence.12–16 Some of the familiar uropathogen species isolated from urine culture were
Similar studies were conducted among pregnant women, DM, human immune-deficiency syndrome (HIV) patients, and pediatric in different parts of Ethiopia,2,3,12,14,22,23 which were targeting populations in the hospital set-up. Studies in the assessment of UTI among DM patients in south wollo zone were limited, particularly there was no study conducted so far in health centers. Thus, this study was conducted to find out the prevalence of bacterial uropathogen, their antibiogram, and the associated factors among symptomatic and asymptomatic DM patients attending in two health centers in Kombolcha town, South Wollo Zone, Northeastern Ethiopia.
Materials and methods
Study design, area, and period
A health facility–based cross-sectional study was conducted from February to April, 2020 at Kombolcha Health Centers, Kombolcha Town, Ethiopia. The study was conducted in two health centers (Kombolcha Health Center 03 and 05) in Kombolcha town. Kombolcha is located in the Northeastern part of Ethiopia, 378 km north of the capital city, Addis Ababa.
Eligibility
Diabetic patients, 18 years of age and above, with and without symptoms of UTI were included in the study. Diabetic patients who did not give consent to participate in the study and who were taking antibiotics for the last 2 weeks were not included in the study.
Sample size determination and sampling techniques
To calculate the minimum required number of study population, a single population proportion formula was used by taking into account the following considerations: 95% confidence interval (Zα/2 = 1.96), 22.6% proportion of previous work, 18 and a 5% margin of error. The calculated sample size was 269 and by adding 5% non-response rates, a total of 282 study participants were involved in the study. To recruit the estimated sample size, we have used simple random sampling technique.
Data and specimen collection
A validated and pre-tested questionnaire and also patient’s card were used to obtain data related to demographic characteristic and associated factors. Appropriate container, with appropriate collection instruction was provided to each study participant to collect freshly voided 5–10 mL of mid-stream urine (MSU) specimen. Then, the specimen was transported to Medical Microbiology Laboratory using (0.1 g) boric acid as preservative. Urine specimens were processed in the laboratory within 2 h of collection and specimens that are not processed within 2 h were kept refrigerated at 4°C until analyzed.
Isolation and identification
Each sample was inoculated into Cystine Lactose Electrolyte Deficient medium (CLED; Oxoid Ltd, UK) and incubated overnight in aerobic atmosphere at 37°C for 24 h. Then, colonies were counted to decide whether the bacterial growth from each medium was significant or not. The culture of ⩾100 colonies of one type (number of bacteria is ⩾105 cfu/mL) is considered as a cutoff for UTI. 24 Colonies from CLED were sub-cultured into MacConkey agar (Oxoid, Ltd), blood agar plates (Biomark Labs), and Mannitol salt agar, and then incubated at 37°C for 24 h. The gram-negative bacteria were identified by indole production, H2S production in Kligler Iron Agar (KIA) agar, citrate utilization, urease test, motility test, oxidase and carbohydrate utilization tests.The gram-positive bacteria were identified using catalase and coagulase tests.
Antimicrobial susceptibility testing
Based on the Clinical and Laboratory Standards Institute (CLSI) guideline, 25 we performed the antimicrobial susceptibilities of all identified bacterial isolates according to Kirby–Bauer disk diffusion method on Mueller–Hinton Agar. Sufficient amount of colonies was taken from the pure culture growth and transferred to a tube containing 5 mL of normal saline. To make the transferred colony a homogeneous suspension, it was mixed gently. The turbidity of the suspension was adjusted to get bacterial inoculums equivalent to 0.5 McFarland turbidity standards and finally it was swabbed on Mueller–Hinton agar medium using a sterile cotton swab.
The inoculated plates with appropriate antibiotic disk were incubated at 35°C–37°C for 18–24 h. The diameters of the zone of inhibition around the disks were measured using a caliper. The following antimicrobials were used with their respective concentration: ampicillin (AMP, 10 μg), penicillin (PEN, 10 μg), augmentin (AUG, 20/10 μg), nitrofurantoin (NIT, 300 μg), trimethoprim–sulfamethoxazole (SXT, 25 μg), gentamicin (GN, 10 μg), ceftriaxone (CRO, 30 μg), ciprofloxacin (CIP, 5 μg), amikacin (AM, 30 μg), tazobactum (TZ, 10 μg), imipenem (CTZ, 30 μg), tetracycline (30 µg), and cefoxitin (CF, 30 μg). These antimicrobial drug disks are selected based on CLSI and also by considering the availability and frequent prescriptions of these drugs for the treatment of UTIs in the study area.
The interpretation of the results of the antimicrobial susceptibility tests was based on the standardized table supplied by the national committee for CLSI criteria as sensitive, intermediate, and resistant.
Data quality assurance
The questions in structured questionnaire were prepared by adopting from different published articles in Ethiopia and elsewhere.13,18,26–28 Prior to the actual data collection time, it was translated into local language (Amharic). Then it was pre-tested on 5% DM patients at Kombolcha Kebele 02 Health Centers, Kombolcha. The data collectors were trained and after data collection, the entire questionnaire was checked by the researchers for its completeness. The sterility of culture media was checked by incubating 5% of the batch at 35°C–37°C overnight and it was evaluated for possible contamination (for any growth). Reference strains of
Statistical data analysis
The collected data were entered into the Microsoft Excel spread sheet 2010. Then the data were exported and analyzed by Statistical Package for Social Sciences (SPSS) version 23. Descriptive statistics were computed and data were presented using tables. To examine any statistical association between variables, binary logistic regression was computed. Multivariable analysis was performed to identify factors that are independently associated with dependent variable. The variables that had shown
Results
Demographic features
Of the total study participants, 171 (60.6%) were with and 111 (39.4%) were without UTI symptoms. The age of the study participants ranged from 20 to 81 years, with a mean age of 48.9 ± 14.0 years. Nearly, 84 (29.8%) of the study participants were in the age group of 46–55 years (Table 1).
Socio-demographic characteristics of the study participants (

Prevalence and associated factors of UTI
Out of 282 participants, significant bacteriuria (SB) was detected in 57 DM patients (37 from symptomatic and 20 from asymptomatic), which give the general prevalence of 20.2%. In multivariable analysis, females had 3.2 times increased odds to develop UTI as compared to males (adjusted odds ratio (AOR) = 3.218, 95% CI = 1.057–9.794,
Logistic regression analysis among diabetic patients (

SB: significant bacteriuria; AOR: adjusted odds ratio; COR: crude odds ratio; 1*: reference category; 95% CI: 95% confidence interval; DM: diabetes mellitus; UTI: urinary tract infection; NA: not applicable; BMI: body mass index.
Statistically significant at
Bacterial uropathogen isolates
Of the total study participants, 10 different uropathogen species were isolated from the collected urine sample. A total of 57 bacterial isolates were identified, and it was revealed that gram-negative bacteria were more prevalent, 40 (70.2%), than gram-positive bacteria, 17 (29.8%). Overall, the leading isolate was
Frequency of bacterial uropathogens among diabetic patients in Kombolcha Health Centers (KHC), Northeastern Ethiopia, February to April, 2020.
Antimicrobial susceptibility pattern of bacterial uropathogens
In this study, majority of isolated gram-negative uropathogens showed resistance for ampicillin and only 19 (40%) were showing susceptibility for imipenem.
Antimicrobial susceptibility pattern of gram-negative bacteria (
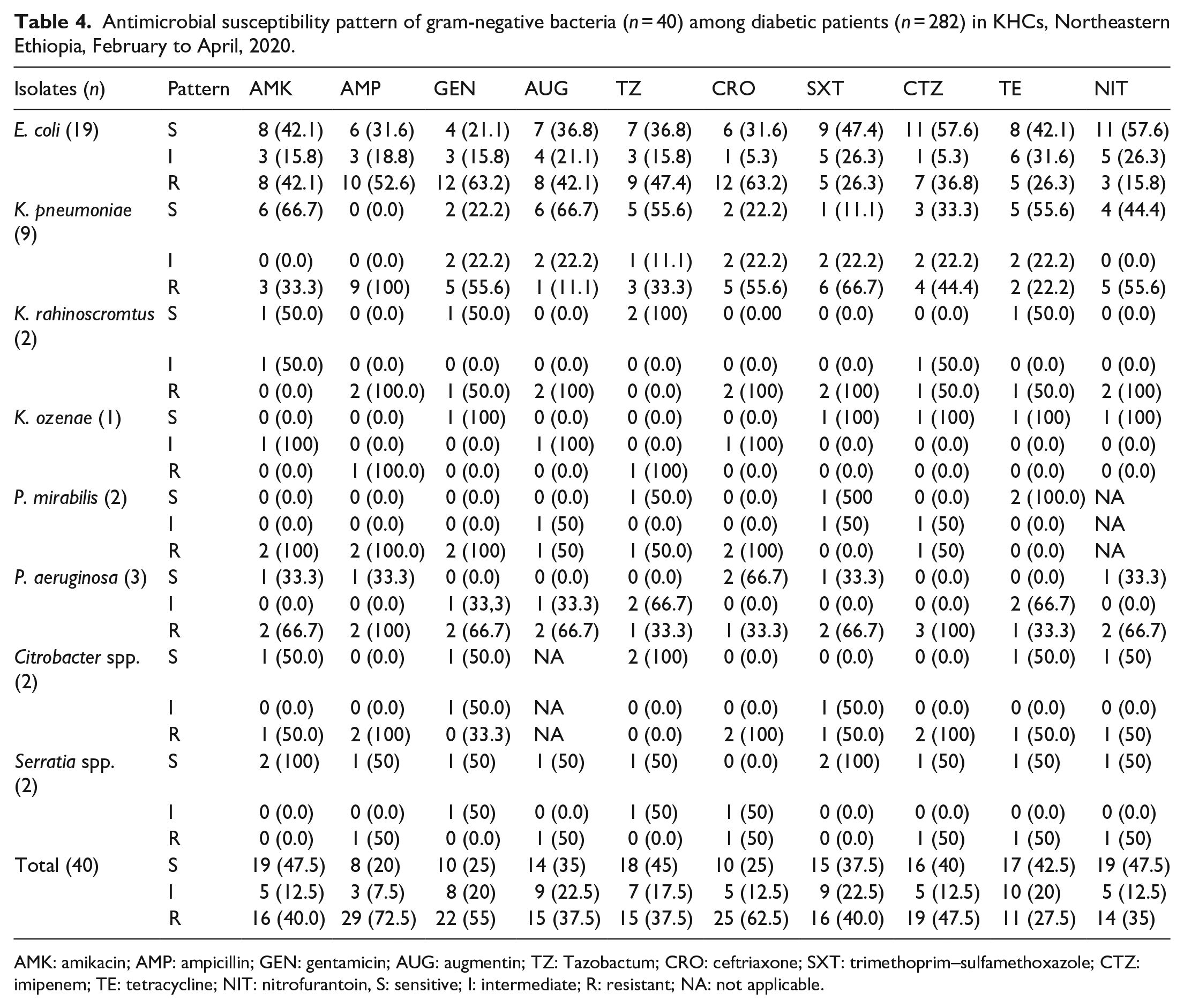
AMK: amikacin; AMP: ampicillin; GEN: gentamicin; AUG: augmentin; TZ: Tazobactum; CRO: ceftriaxone; SXT: trimethoprim–sulfamethoxazole; CTZ: imipenem; TE: tetracycline; NIT: nitrofurantoin, S: sensitive; I: intermediate; R: resistant; NA: not applicable.
Gram-positive uropathogen showed high-degree resistance to penicillin, 11 (64.7%) and gentamicin, 10 (58.8%). On the contrary, they showed relatively higher sensitivity to nitrofurantoin, 8 (47%) and tetracycline, 8 (47%). All isolates of
Antimicrobial susceptibility pattern of gram-positive bacteria (

GEN: gentamicin; CF: cefoxitin; SXT: trimethoprim–sulfamethoxazole; TE: tetracycline; NIT: nitrofurantoin; CIP: ciprofloxacine; PEN: penicillin; S: sensitive; I: intermediate; R: resistant.
MDR patterns
Overall, 47 (82.4%) bacterial isolates were found to be resistant to a minimum of one antimicrobial agent while 29 (50.88%) bacterial uropathogens were found to be resistant for more than three antimicrobial agents from different classes. About 57.5% of gram-negative and 35.3% of gram-positive bacteria showed MDR pattern (Table 6).
Multidrug resistance patterns among diabetic patients (

R0: no antibiotic resistance; R1: resistance to one; R2: resistance to two; R3: resistance to three; R4: resistance to four; ⩾R5: resistance to five and more drugs; MDR: multidrug resistant.
Percentage is computed from total number of isolates, based on which MDR definition is applied.
Discussion
Several infections, particularly UTI, are a common phenomena that can occur among DM patients 29 and DM is an important factor for the occurrence of UTI.29,30 There might be factors that facilitate microbial adherence to uroepithelial cells and that lead diabetic patients to be vulnerable to UTI.31–33
The general prevalence of SB in the current research was 20.2%. Analogous results have revealed from earlier published studies: Addis Ababa (22.6%), 18 Gondar (17.8%), 15 Metu (16.7%), 14 Nekemete (16.5%), 34 Sudan (19.5%), 19 and Uganda (22.0%). 35 However, this finding was relatively lower as compared to the studies conducted in Arba Minch, Ethiopia (33.8%), 12 in Addis Ababa (32.1%), 36 Nigeria (40%), 37 Nepal (54.76%), 38 and Pakistan (49.2%). 39 On the contrary, our finding is higher than other studies conducted in Addis Ababa (10.9%) 21 and Dessie (11.6%). 16 The finding of this study is still higher than a study which was conducted among UTI-suspected individuals in India. 40 The possible justification for the variance might be difference in population, study period, and study design.
Similar to studies conducted in Addis Ababa and Hawassa,13,21 the percentage of bacterial uropathogen isolate among DM patients with symptomatic UTI was higher than that of asymptomatic UTI. Whereas, a study conducted in Sudan reported contradicting result. 19 The reported bacterial prevalence of symptomatic UTI in this study (21.6%) was comparable with studies conducted in Hawassa (21.7%) 13 and Sudan (17.1%), 19 but it was lower than studies conducted in Arba Minch (64.1%), 12 Addis Ababa (49.3%), 18 Nigeria (35%), 37 and India (49.15 %). 41 This variation might be attributed to difference in the study setting, study population and the study period in different localities.
In other way, the bacterial rate of asymptomatic UTI among participants (18%) was higher than a study conducted in Addis Ababa (10.4%), 21 Nigeria (12.5%), 42 and in a systematic review report (12.2%), 43 but it was lower than studies conducted in Hawassa (78.3%), 13 Gondar (91.7%), 15 Addis Ababa (49.3), 18 Arba Minch (35.8%), 12 Nigeria (35%), 37 and Sudan (20.9%). 19 This discrepancy also may be as a result of variation in risk factors with geographical areas, number of study participants, the study population, and use of varied methodologies.
In this study, the proportion of
The higher proportion of uropathogen that was identified in this research was gram-negative bacilli (70.2%), which was similar to reports from preceding studies in Dessie, Addis Ababa, Sudan, and Kuwait.16,18,19,45 Among gram-negative bacilli,
In this study, SB was statistically associated with the sex of the study participant; female showed 3.2 times increased odds of acquiring UTI in comparison with male study subjects. This finding is similar to previous studies that have been conducted in Ethiopia12,14 and a study conducted in Saudi Arabia also indicates that females have the highest risk of acquiring UTI. 48 This is probably due to the higher level of recurrence of UTI in women. 49 It might be due to the anatomy and reproductive physiology that females are more prone to the pathogenicity of uropathogens. The possible entrance of bacteria to the female bladder can be driven by different situations like short urethra, urethra closer to peri-rectal area, the absence of bacteriostatic prostatic secretions, and sexual intercourse. 50 But according to studies in Dessie, Addis Ababa, and Romania, there was no statistical association between study participants’ sex and SB.16,18,28 This study also revealed that merchant occupation was positively associated with uropathogens. Up to 28.4% of the isolates were from participants with merchants compared with participants with other occupations. So, this study contradicts to previously done research findings in Arba Minch, Debre Tabor, and Metu12,14,17 in which there was no statistical association between merchants and bacteriuria.
In this study, variables such as age, type of DM, duration of DM, blood glucose level, previous history of UTI, glucose urea and body mass index (BMI) did not show a statistical association with the incidence of bacteriuria. But a study conducted in Saudi Arabia indicated that elevated BMI had associated with increased risk of bacteriuria. 48 Similar to the current one, few studies indicate that age did not influence the incidence of bacteriuria.13,16,48 In contrast, age of the study participants was found to be one of the associated factors for UTI in a recent study conducted in Addis Ababa. 18 Previous history of UTI had associated with SB and it was revealed in a few studies in Ethiopia.13,14
In this study, varied patterns of antimicrobial resistance was observed among the isolated bacterial species. Unlike few other studies,12,13 in this study, there was no antimicrobial agent that showed either full resistance or susceptible against all the isolated gram-positive and gram-negative bacteria. The majority of the isolated gram-negative uropathogens showed resistance for ampicillin and it was a comparable finding with the studies conducted in Ethiopia and Nepal.13,38 On the contrary, gram-negative isolates showed relative sensitivity to nitrofurantoin, 19 (47.5%) and amikacin, 19 (47.5%). Nitrofurantoin drug was found fully susceptible against gram-negative bacteria in a study conducted in Ethiopia. 13
Gram-positive uropathogen showed resistance to penicillin, 11 (64.7%) and gentamicin, 10 (58.8%). In agreement with this study, penicillin was found to be resistant against gram-positive bacteria.
12
A contradictory result was reported from a study conducted in Hawassa, Ethiopia, in which majority of the gram-positive isolates was found to be susceptible against gentamicin.
13
On the contrary, gram-positive bacterial isolates showed relatively higher sensitivity to nitrofurantoin, 8 (47%) and tetracycline, 8 (47%). This finding disagrees with a study done in Ethiopia in which majority of the gram-positive isolates showed resistance to both antibiotics.
14
Similar to studies conducted in Ethiopia,12,13,34 in this research work it was confirmed that all isolates of
Overall, 47 (82.4%) bacterial isolates were resistant to at least one antimicrobial agent, which is somewhat lower (94.9%) than a study conducted in Dessie town.
16
Whereas, in this study, 29 (50.88%) isolates were resistant to ⩾3 antimicrobials, which is comparable finding in a study conducted in Dessie.
16
On the contrary, different research works in Ethiopia have been reporting higher proportion of MDR bacterial isolate.13,15,18,21,26 Such variations are mainly due to the difference in the operational definition of MDR. In most of the previous studies, MDR was defined as resistance to two or more antimicrobial classes. About 57.5% of gram-negative and 35.3% of gram-positive bacteria showed MDR for the tested antimicrobial drugs. A high proportion of
Limitation of the study
The study was carried out in two health centers that are found in the town of Kombolcha, which is not epidemiologically well represented. The laboratory works were performed by well-experienced personnel in a well-organized laboratory set-up. One of the limitation of the study is it focuses only one bacterial pathogen, including others pathogen such as viral, fungal, and protozoan might strengthen the research. It would be very comprehensive if we were including some important clinical and laboratory tests like HbA1c estimations, cytokine profile, and WBC counts and analyzing with the prevalence of uropathogen. Besides, this study did not provide information about ESBL production and
Conclusion
The general prevalence of bacterial uropathogen in this study was found to be consistent with some other studies.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221139149 – Supplemental material for Bacterial uropathogens, their associated factors, and antimicrobial susceptibility pattern among adult diabetic patients in two health centers at Kombolcha town, Northeastern Ethiopia
Supplemental material, sj-docx-1-smo-10.1177_20503121221139149 for Bacterial uropathogens, their associated factors, and antimicrobial susceptibility pattern among adult diabetic patients in two health centers at Kombolcha town, Northeastern Ethiopia by Ousman Oumer, Yeshi Metaferia and Daniel Gebretsadik in SAGE Open Medicine
Footnotes
Acknowledgements
The authors would like to acknowledge Wollo University and the two health centers in Kombolcha town for their valuable technical support during the whole research activity. They would like to thank all the data collectors, particularly the clinicians who are working in the two health centers and study participants for their cooperation during data collection.
Author contributions
All the three authors made an equally weighted contribution to the article submitted, starting from the commencement of the proposal development up to the finishing point of the research work. All authors impose considerable role to the gap identification, background assessment of the study area, setting of objectives, data collection material preparation, study design, execution, acquisition of data, analysis and interpretation, or in all these areas; took part in drafting, revising, or critically reviewing the article; gave final approval of the version to be published; have agreed on the journal to which the article has been submitted; and agree to be accountable for all aspects of the work.
Availability of data and materials
All data underlying the findings are fully available without restriction. All relevant data are within the article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval and consent to participate
After evaluating the procedures, appropriateness of the method applied in this research work, the ethical approval committee of College of Medicine and Health Sciences, Wollo University, Dessie, Ethiopia has approved the study (CMHS 1137/2020). In addition, official cooperation letters to Kombolcha Health Centers were obtained from Wollo University, department of medical laboratory science. Furthermore, after explaining the importance, purpose, and procedure of the study briefly, a written consent was obtained from each study participant and from the legally authorized representative of the subject with no formal education. Any study participant who was found to be infected with the bacteria was referred to physician for treatment.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects and from the legally authorized representative of the subject with no formal education before the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
