Abstract
Objective:
We sought to determine predictors, incidence, and interventions required for patients who developed barotrauma. Pneumothorax, subcutaneous emphysema, and pneumomediastinum have all been reported as complications related to COVID-19-positive patients requiring invasive mechanical ventilation.
Methods:
In this retrospective study, clinical and imaging data from COVID-19 patients were collected and reviewed by two independent intensivists between January 4, 2020 and January 10, 2020. Data were used to identify COVID-19-positive patients requiring invasive mechanical ventilation and the incidence of barotrauma. Two separate cohorts were created as non-injured (no barotrauma) and injured (barotrauma present). We then sought to identify the risk factors for barotrauma in the non-injured cohort on Days 0, 7, 10, and 14 after intubation and day of injury in the injured cohort.
Results:
Of the 264 patients with COVID-19, 55.8% were African American. The non-injured group was older (60 ± 15 versus 49 ± 16, p = 0.006), with male predominance in the injured group versus non-injured group (75% versus 55%). A total of 16 (6.5%) patients developed one or more complications of barotrauma, defined as subcutaneous emphysema, pneumothorax, or pneumomediastinum. Length of stay was longer for the injured group versus non-injured group (47 versus 25 days). Plateau pressure (p = 0.024), fraction of inspired oxygen (p < 0.001), and driving pressure (p = 0.001) were statistically significant in injured cohort. Mortality rate in non-injured versus injured was 49.4% versus 69%. Using random effect model, fraction of inspired oxygen (p = 0.003) and mean airway pressure (p = 0.010) were significant at the time of injury. When comparing alive versus deceased in the injured cohort, thoracostomy placement in alive versus deceased was 80% versus 54.5%.
Conclusion:
COVID acute respiratory distress syndrome patients requiring invasive mechanical ventilation had a higher rate of barotrauma and were younger than those who did not develop barotrauma. Possible interventions to be considered to decrease barotrauma are decreased driving pressure goal and universal use of esophageal balloon manometry.
Keywords
Introduction
In December 2019, severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) which causes coronavirus disease 2019 (COVID-19) was first identified in Wuhan, China. 1 Clinical manifestations of COVID-19 can vary in infected patients from those with minimal symptoms to those who progress to COVID acute respiratory distress syndrome (C-ARDS). 2 Patients who advance to C-ARDS often require mechanical ventilation. Mechanical ventilation may be lifesaving; however, it can also lead to serious complications such as ventilator-induced lung injury (VILI). 3 VILI can occur due to high transpulmonary pressures (PLs) which can lead to barotrauma. In C-ARDS patients, barotrauma in the forms of subcutaneous emphysema, spontaneous pneumothorax, and pneumomediastinum has been reported. 4 While ventilated, patients may experience these forms of barotrauma due to increased pressure in the lungs and subsequent tears of the parenchyma. 3 Other proposed mechanisms for VILI in addition to barotrauma are volutrauma, atelectrauma, ergotrauma, and biotrauma with biotrauma related to COVID-19 infection and profound cytokine release.3,5,6 Additional explanations may be related to Macklin effect, which was first described in 1939. The Macklin effect occurs when air dissects along sheaths surrounding pulmonary vasculature, leading to pneumomediastinum, without the presence of pneumothorax. 7 Lastly, there have been reports of pneumomediastinum related to type I and type II pneumocyte injury related to COVID-19 infection. 8 In addition, barotrauma has been associated with high rates of mortality and prolonged hospital stays. 9 Outside the setting of COVID-19, VILI and barotrauma have been thoroughly investigated and occur at a rate of 2.9% to as high as 15% in mechanically ventilated patients but varied based on the reason for invasive mechanical ventilation (IMV; 2.9% for chronic obstructive pulmonary disease, 6.3% for asthma, 10.0% for chronic interstitial lung disease, 6.5% for traditional ARDS, and 4.2% for pneumonia).9–13 This variability in incidence is likely due to an underlying lung disease. An alternative explanation for increased VILI while requiring IMV is lung recruitment maneuvers with escalating positive end-expiratory pressure (PEEP), as demonstrated by the Alveolar Recruitment for ARDS Trial (ART). 13 In this study, escalating PEEP in the form of recruitment maneuvers resulted in 28/501 (5.6%) versus 8/509 (1.6%) incidences of VILI. 13 Based on ART and common indication for IMV, it is possible that at-risk lung parenchyma plus escalating PEEP could lead to increased incidence of VILI in traditional ARDS patients. Although recruitment maneuvers are not routinely used at our institution, it is recognized that certain clinical scenarios may still warrant its use. Our single-center retrospective study sought to determine the incidence of barotrauma and mortality of C-ARDS patients and compare these rates to traditional ARDS patients. Our primary goal is to identify the incidence and risk factors for barotrauma and potentially identify changes in the management of COVID-19 patients that may prevent the future development of VILI.
Methods
The data were extracted with a Structured Query Language (SQL) server using a linked server to our electronic medical record, Cerner®. All confirmed COVID-19-positive patients were queried against ventilator events and admitted to the intensive care unit (ICU) between January 4, 2020 and January 10, 2020. The resulting subset of only COVID-19-positive patients on IMV was subsequently analyzed. The diagnosis of barotrauma was defined as pneumomediastinum, subcutaneous emphysema, or pneumothorax, which are the commonly used terminologies to describe barotrauma in radiology reports/literature. These diagnoses were then queried against radiology reports of patients identified by the SQL server search. Confirmation of the presence of barotrauma was accomplished by reviewing radiology reports and direct visualization of computed tomography of chest or chest X-ray, by two independent intensivists. Patients were excluded if barotrauma was present before initiation of IMV or due to iatrogenesis, that is, central line placement or a chest tube place with subsequent development of barotrauma. To further evaluate non-injury versus injury, two cohorts were created, based on the absence or presence of barotrauma, respectively. The first cohort of patients was chosen to represent the control group at Days 0, 7, 10, and 14 and served as a convenience sample of patients and not randomization, as this cohort of patients did not experience barotrauma (non-injured) requiring IMV. Days 0, 7, 10, and 14 were selected specifically to maximize the statistical probability of identifying modifiable risk factors related to C-ARDs and the development of VILI. The second cohort of patients served as the experimental group as this cohort of patients experienced barotrauma (injured) requiring IMV. The time interval between intubation and barotrauma in the injured cohort was recorded and analyzed over time. The purpose of these time intervals was to determine the direction of change, as potential indicators of risk of developing barotrauma in the non-injured cohort at IMV Days 0, 7, 10, and 14. Confirmation of COVID-19 in both cohorts was confirmed by PCR for SARS-CoV-2.
Data collected and outcomes measured
Data collected for two cohorts included incidence of barotrauma, age, race, sex, weight (kg), height (cm), pH, arterial carbon dioxide (PaCO2), arterial oxygen tension (PaO2)/Fraction of inspired oxygen (FiO2) (mmHg), date of intubation, mode of ventilation, mode of ventilation at the time barotrauma was identified, set tidal volume (mls), exhaled tidal volume (mls), PEEP (cmH2O), total PEEP, plateau pressure (cmH2O), plateau pressure at the time of barotrauma was identified (cmH2O), peak inspiratory pressure (PIP) (cmH2O), PIP at the time of barotrauma was identified (cmH2O), set PIP (cmH2O), set PIP at the time of barotrauma identified (cmH2O), mean airway pressure (MAP) (cmH2O), MAP at the time of barotrauma was identified (cmH2O), static compliance (ml/cmH2O), static compliance at the time of barotrauma was identified (ml/cmH2O), exhaled minute ventilation (VE) (L/min), and VE at the time of barotrauma was identified (L/min), type of barotrauma (pneumothorax, pneumomediastinum, subcutaneous air), intervention required to treat barotrauma (if applicable), hospital LOS, and date of death or discharge (if applicable).
Statistical analysis
Patient characteristics among the two cohorts without barotrauma (control/non-injured) and the cohort with barotrauma (injured) were presented using the mean, standard deviation for continuous variables, frequency, and proportions for categorical variables. A comparison of means of groups for continuous variables was conducted using ANOVA. In cases of highly skewed variables, data were presented using median, 25th and 75th percentiles, and comparison was made using the Kruskal–Wallis test. Proportions were compared using the chi-squared test or Fisher’s exact test in case of small sample sizes. 14 A p value less than 0.05 was considered significant.
Primary endpoint: Mortality rates and hospital LOS were compared among each cohort using the chi-square test or Fisher’s exact test in case of small cell sizes. Mortality rates and LOS values were presented using frequency and proportions in each group. Multivariate logistic regression was conducted to control for the impact of demographics, specifically related to age at intubation, sex, race, weight, and height. Survival for discharge/expired cohorts was estimated using the Kaplan–Meier analysis. 14
Secondary endpoint: Changes in pH, arterial carbon dioxide (PaCO2), arterial oxygen tension (PaO2)/FiO2 (mmHg), date of intubation, mode of ventilation, mode of ventilation at the time barotrauma was identified, set tidal volume (mls), exhaled tidal volume (mls), set PEEP (cmH2O), total PEEP, plateau pressure (cmH2O), plateau pressure at the time of barotrauma was identified (cmH2O), PIP (cmH2O), PIP at the time of barotrauma was identified (cmH2O), set PIP (cmH2O), set PIP at the time of barotrauma identified (cmH2O), MAP (cmH2O), MAP at the time of barotrauma was identified (cmH2O), static compliance (ml/cmH2O), static compliance at the time of barotrauma was identified (ml/cmH2O), exhaled minute ventilation (VE) (L/min), and VE at the time of barotrauma was identified (L/min), type of barotrauma (pneumothorax, pneumomediastinum, subcutaneous air), intervention required to treat barotrauma (if applicable), hospital LOS, and date of death or discharge (if applicable) were compared between the groups using repeated-measures ANOVA. Day 0 of mechanical ventilation was treated as a dependent variable and comparison between groups and time was used as the independent variable. Descriptive statistics were presented using mean, standard deviation, median, 25th and 75th percentiles, and range (min, max). If the dependent variable was skewed and model residuals did not satisfy normality assumption, then appropriate transformation was used such as log transformation before conducting repeated-measures ANOVA. Multivariate repeated-measures ANOVA were constructed to control for baseline patient characteristics such as age, gender, race, and so on. In addition, alive versus dead were used as independent variables and were compared between the groups using repeated-measures ANOVA.
Lastly, a separate analysis of the two data sets over time was performed, using a random effect model. This separate analysis intended to provide a direct comparison between two data sets to identify the direction of change, while determining whether differences across data sets have some influence on dependent variables, such as risk for developing barotrauma. 15 All comparisons for follow-up time were made with Day 0 in the injured cohort (day of injury). Uninjured comparisons were made on Days 0, 7, 10, and 14. All other analyses were done using R version 4.0.0 except repeated-measures ANOVA, which was done using SAS version 9.4.
Results
Between January 4, 2020 and January 10, 2020, 264 patients with PCR-confirmed COVID-19 and requiring IMV were analyzed, of which 55.8% were African American. The non-injured group was older at the time of intubation (60 ± 15 versus 49 ± 16, p = 0.006), with a higher occurrence of males in the injured group versus non-injured group (75% versus 55%), inclusion and exclusion criteria are depicted in Figure 1.

Flowchart of patients analyzed.
Injured patients were younger compared to the non-injured cohort (49 ± 16 versus 60 ± 15, p = 0.006). More injured patients were males (75% versus 55%). On average, non-injured patients were hospitalized for a shorter period and also required fewer days mechanically ventilated for 24.9 ± 26.6 days, requiring IMV 14.9 ± 24.8 days versus injured patients were hospitalized for 47 ± 4 days, requiring IMV for 34 ± 29 days. Patient demographics are summarized in Table 1.
Baseline characteristics of non-injured versus injured cohort.

AC, assist control; FiO2, fraction of inspired oxygen; ICU, intensive care unit; LOS, length of stay; PEEP, positive end-expiratory pressure; SD, standard deviation; VC, volume control.
Injured patients were intubated for 20 ± 16 days before barotrauma developed. MAP was elevated in the injured cohort versus non-injured cohort (19 ± 6 versus 16 ± 6, p = 0.005). FiO2 was statistically significant (p < 0.001), likely representing the development of chronic lung disease. In addition, driving pressure was elevated in the injured cohort (18 ± 7 versus 15 ± 5, p = 0.001). No statistical difference in mortality between the two cohorts was identified. Patient demographics are summarized in Table 1 and mortality is summarized in Figure 2.

Kaplan–Meier curves comparing the overall probability of survival in non-injured versus injured patients with COVID-19 infection on invasive mechanical ventilation. Patients with and without barotrauma had no difference in overall survival are represented by red and blue curves, respectively (p > 0.196).
In all, 16 of the 248 patients had at least one barotrauma event (6.5%). Of the 16 patients in this cohort, 11 died, with the majority being African American (50%). PIP at the time of injury was lower in the deceased injured cohort (median [interquartile range, IQR]: 30.50 [28.25, 33.25] versus 32.00 [27.00, 36.00], p = 0.010), with elevated FiO2 requirement at the time of injury in the deceased injured cohort (median [IQR] 0.55 [0.43, 0.90] versus 0.40 [0.40, 0.40], p = 0.020). Lastly PCO2 was elevated overall in deceased injured cohort (median [IQR] 46.00 [39.00, 57.00] versus 43.00 [40.00, 47.00]), with a p value of 0.027 and then normalized at the time of injury (median [IQR] 43.00 [38.50, 50.50] versus 38.00 [32.00, 44.00], p = 0.571). No statistical significance was noted for the laterality of barotrauma, or if intervention was required in 10 of the 16 patients. Patient demographics are summarized in Table 2.
Baseline characteristics (injured), alive versus deceased.

IQR, interquartile range; LOS, length of stay; PEEP, positive end-expiratory pressure.
Uninjured cohort comparisons were made on Days 0, 7, 10, and 14. Interestingly, driving pressure was statistically significant on Days 7 and 14 (p = 0.042 and 0.031, respectively), along with PaO2 on Days 7, 10, and 14 (0.0003, <0.0001, and <0.0001, respectively), which correlated to FiO2 significance on Days 7, 10, and 14 (0.007, 0.13, and 0.011, respectively). Static compliance was statistically significant on Days 7 and 10 (p = 0.013 and 0.037, respectively). MAP was statistically significant on Day 14 (p < 0.0001). PEEP total was statistically significant on Days 7, 10, and 14 (p = 0.003, 0.0004, and 0.0007, respectively), which also correlated to PEEP set on Days 7, 10, and 14 (p = 0.0002, <0.0001, and <0.0001, respectively). Patient demographics are summarized in Table 3.
Comparison of select variables on Days 0,7,10, and 14, relative to the date of intubation (non-injured).

DF, degrees of freedom; PEEP, positive end-expiratory pressure.
Injured cohort comparisons were made at the time of injury. FiO2 was statistically significant on the day of injury (p = 0.003) along with MAP (p = 0.010). Of note, the FiO2 value was decreasing, and MAP was increasing at the time of injury. Patient demographics are summarized in Table 4.
Comparison of select variables at day of injury, relative to date of intubation (injured).

DF, degrees of freedom; PEEP, positive end-expiratory pressure.
Discussion
In this study, we observed a higher incidence of barotrauma within the C-ARDS cohort (6.5%) versus traditional ARDS (2.9%). When compared to other studies of incidence of barotrauma in patients with C-ARDS, the incidence of barotrauma ranges from 4.74% to as high as 21%.16–18 One possible explanation for this difference in incidence is the widely accepted and use of esophageal balloon manometry, to guide PEEP and DP. Optimization of PEEP and DP with esophageal balloon manometry will be discussed later. Our study represents a twofold increase in barotrauma as compared to traditional ARDS, which translates into approximately 22 additional days in the hospital and approximately 19 additional days requiring mechanical ventilation. To compare C-ARDS studies and barotrauma, our incidence is on the lower side, but within the cited range of 4.74–21%.16–18
In our study, patients with COVID-19 requiring IMV and barotrauma were younger than those without barotrauma, possibly suggesting an age-related risk of developing barotrauma. Younger age, higher maximal peak inspiratory airway pressures (PIP), and higher levels of maximal PEEP have previously been associated with barotrauma in ICU patients. 18 We did not identify a relationship between mode of ventilation and risk for development of barotrauma. The majority of non-injured patients were supported with assist control/volume control (AC/VC; 70%), and 40% of injured patients supported with AC/VC. These findings would suggest that barotrauma may be inherent to disease process and possibly be related to patient-self-inflicted lung injury (P-SILI). To mitigate VILI risk, all patients in our cohort were placed on lung-protective strategies (LPS). Additional strategies used to potentially decrease the risk of injury from P-SILI, was to deeply sedate, in an effort to maintain LPS volumes. One of our goals was to evaluate C-ARDS patients requiring IMV and exclude all other patients. Review of the recent meta-analysis by Belletti et al., found that the incidence of barotrauma was 16% and mortality in C-ARDS patients. 19 Our goal was similar to Belletti et al., to determine the incidence of barotrauma in C-ARDS patients. On the review of the Supplemental Material, 7 of the 13 studies reported initial IMV settings, with many patients breathing spontaneously, or using noninvasive ventilation or high-flow nasal cannula. Of the IMV parameters cited, Tidal volume (Vt), plateau, PIP, and Respiratory rate (RR), only three studies reported these IMV parameters. In our study, the IMV parameters set and measured were similar to those three studies reporting full details. Excluding spontaneously breathing C-ARDS patients may have contributed to lower incidence of barotrauma in our study. Based on our analysis, we found that the following variables were significant in the injured and the non-injured cohorts: DP, MAP, FiO2, P/F ratio, and PEEP, which could potentially be used to monitor for increased risk of developing barotrauma. Our study was able to show trends over Days 0, 7, 10, and 14 in the non-injured cohort, but determining when a patient crosses over from non-injured to injured is a potential for further research.
Notably, patients were younger in the injured cohort with increasing MAPs (p = 0.010) and decreasing FiO2 (p = 0.003) at the time of injury. FiO2 was the only variable that was statistically significant (p = 0.020) when compared across the alive versus deceased injured group. A slight decrease in PIP at the time of injury was noted in the deceased injured group and was statistically significant (p = 0.010). These trends may represent the transition from the exudative phase to the fibrosing-alveolitis phase, as seen in end-stage ARDS. 20 The fibrosing-alveolitis phase has been thought to be mediated by proinflammatory mediators such as interleukin (IL)-1 and other cytokines such as tumor necrosis factor alpha (TNF-α).21,22 Our study represents the period before the widespread use of dexamethasone, as recommended in the RECOVERY trial. 23 Although steroids have been trialed in traditional ARDS, with limited improvement in mortality, the use of anticytokine medications, such as tocilizumab (monoclonal antibody against IL-6 receptor), combined with dexamethasone, showed improvement in overall mortality in severe cases of C-ARDS requiring IMV. 24 Possible benefits and decreased mortality related to the use of IL-6 receptor monoclonal antibody combined with steroids blunt the immune response and decrease fibrosis via minimizing the effects of TNF-α and cytokines. 24 Due to the lack of universal use of steroids and tocilizumab at the time of our study, this could be considered a limitation of our study, as related to increased mortality, but unrelatable to the development of barotrauma.
Although our study did not demonstrate statistical difference in mortality between the two cohorts (p = 0.196), a larger percentage of injured cohort died, when compared to non-injured group (69% versus 49%, respectively). This would suggest a relationship between development of barotrauma and increased mortality. 19 The lack of statistical significance between the two cohorts is likely multifactorial, but highly relatable to the increased age of non-injured cohort, possibly reflecting frailty and higher risk for death. 25
In addition, our study demonstrated that injured patients were more likely to require an overall longer hospitalization (47 days versus 25 days). This may not be necessarily related to IMV, as the injured group required 35 days of IMV and the non-injured group required 36 days of IMV.
Average time to injury after intubation was 20 days in the injured group and approached statistical significance (p = 0.051). Time to injury was similar to other recent studies, ranging from 2.5 to 19.6 days with a pooled mean time of 3.7 days. 19 Another variable identified in our study as being significant is the higher DP in the injured group versus the non-injured group (18 versus 15, respectively). This finding could also translate into higher mortality, when unable to maintain DP between 13 and 15 mmHg.26,27 Higher PEEP has also been shown to be correlative with a higher risk for barotrauma when DP is also elevated.26,27 Plateau pressure set with ARDSnet guidelines suggests that the goal plateau pressure should be less than 30 to avoid barotrauma, but this value was not exceeded in either cohort. The non-injured versus injured plateau was 25 versus 26, respectively, and the p value was 0.025, which leads to the question whether DP is a better predictor of injury, or just increased mortality when exceeded.
Risk factors for the development of barotrauma have been identified as P-SILI, 28 due to an increase the PL and overdistension, 29 even in the setting of LPS. 30 P-SILI often occurs more frequently in younger patients. 31 The ability to correctly identify when P-SILI is violating set Vt and plateau, one would need to consider utilizing esophageal balloon manometry. Incorporating esophageal balloon manometry into clinical practice could potentially provide reliable estimates of respiratory effort, by measuring esophageal pressure (Pes) and airway opening pressure, which can be used to calculate PL.32,33
Possible targets to consider when trying to minimize the risk of injury in severe C-ARDS patients requiring IMV are as follows: Consideration of lower DP target and early institution and use of esophageal balloon manometry monitoring. This consideration may be especially useful in the P-SILI population. Although controversial, P-SILI is thought to be related to high respiratory drive resulting in strong inspiratory efforts, which then lead to overdistension, pendelluft, atelectrauma, increase in vascular transmural pressure, and transpulmonary pressure. This deleterious effect can lead to increased respiratory workload injury and potentially the development of pneumothorax or pneumomediastinum. 34 Adequate sedation could potentially lead to the prevention of injury but needs to be carefully balanced, so as not to invoke iatrogenesis by way of oversedation, lack of mobility, and increased risk for tracheostomy due to prolonged intubation.35,36 Once patients are intubated and adequately sedated, total PEEP measurement could be more accurately measured with the placement of esophageal balloon manometry, to estimate Pes and pleural pressures.32,33 Another potential target to minimize the risk of injury is to potentially consider trying to achieve a lower DP than 14. The original article by Amato et al. found that an increase above 7 cmH2O of DP trended toward increased mortality (relative risk, 1.41; 95% confidence interval, 1.31–1.51; p < 0.001). 26 This was seen even in patients placed on LPS, controlling for tidal volume and plateau pressure. 26 Amato et al. also went on to postulate that DP is a surrogate marker for cyclic strain (respiratory compliance) which was a better predictor of lung injury when compared to tidal volume. 26 With the use of an esophageal balloon manometry, one could measure transpulmonary driving pressure (DPL) and target a DPL less than 10–12 cmH2O. DPL is thought to correlate better cyclic strain, due to the variability in chest wall and lung parenchyma. 37 Measuring DPL may ultimately be a better way of preventing barotrauma. 37 In addition, maintaining PL less than 15–20 cmH2O may also prevent lung injury. 37 Our institution followed these expert opinions when placing and measuring pressures with an esophageal balloon manometry. An adopted algorithm from our institution outlines how to obtain these values and goals, as depicted in Figure 3.
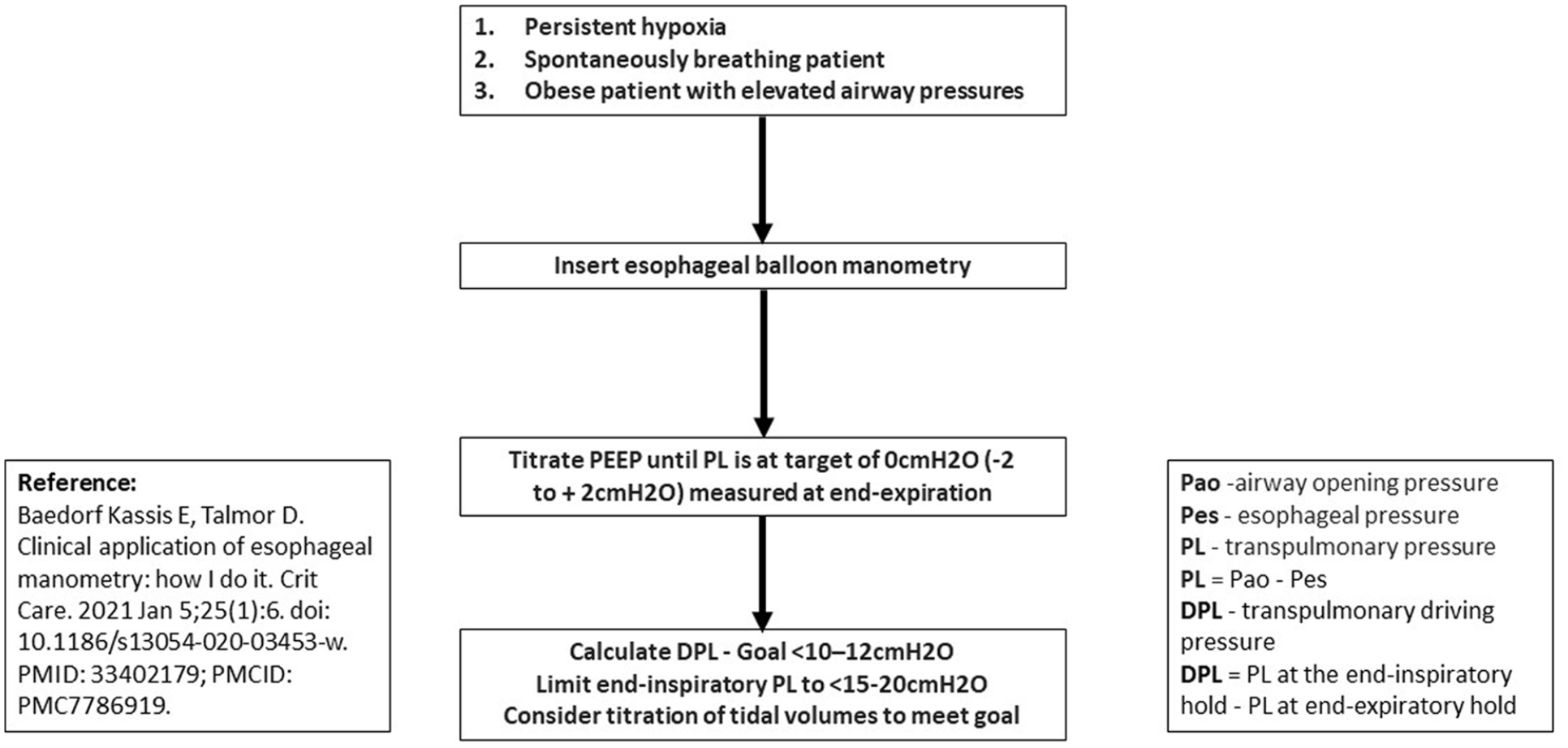
Simplified esophageal balloon manometry algorithm (adopted from MedStar Washington Hospital Center).
Possible interventions to consider when barotrauma occurs are to decrease PEEP and Vt as low as possible, consider the incremental decrease in DP less than 14 cmH2O, with the assistance of esophageal balloon manometry. Thoracostomy placement is required, when conservative management of pneumothorax is failing, as lung parenchyma is friable and may lead to a persistent air leak and bronchopulmonary fistula, possibly requiring surgical intervention. If tension pneumomediastinum develops and unable to ventilate the patient, consultation with a cardiothoracic surgeon may be necessary, specifically considering the need to perform a Gills procedure. 38
Our study has several limitations. Many of these are related to the inherent bias unintentionally introduced when retrospectively collecting data. An effort to mitigate bias was carried out by analyzing consecutive patients and having two independent intensivists review and confirm the presence of barotrauma related to COVID-19 while excluding iatrogenic causes of barotrauma. Each patient’s chart and radiograph were carefully reviewed for any evidence of barotrauma; however, it is conceivable that the method used to detect barotrauma may have resulted in an overestimation or underestimation of the incidence of barotrauma. To avoid this unintentional error, all patients were sampled on Days 0, 7, 10, and 14, to capture all incidences of barotrauma. An additional limitation related to the retrospective nature of study is the inability to calculate power analysis. This source of error was mitigated by limiting inference to predictors of mortality, as a larger sample size would have been needed to make a conclusion related to mortality associated with barotrauma.
Lastly, with the inability to directly compare the non-injured group (control) to the injured group, identifying predictors was not possible, but identifying trends may provide clinicians with possible signals to be aware of when managing C-ARDS patients. Understanding the mechanism between COVID-19 and barotrauma is imperative for preventative measures to be developed. Furthermore, being aware of the incidences of barotrauma in C-ARDS patients across different hospital settings (tertiary to community based) should make clinicians more vigilant in search of causes of worsening respiratory status.
Conclusion
Barotrauma occurred more often in younger patients and also represented an increased need for intervention. Identifying risk factors related to a younger population with COVID-19 and requiring IMV may help to mitigate and minimize the risk of developing barotrauma. Possible interventions to be considered to decrease barotrauma are to decrease the DP goal and the universal use of esophageal balloon manometry.
Footnotes
Acknowledgements
Sameer Desale and Eshetu Tefera for statistical analysis.
Author contributions
SND, SS, EE, MH, SY and PC contributed to the conception and design of this work, drafted and revised the manuscript and approved the submitted version.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the MedStar Health Research Institute Institutional Review Board on an expedited basis (IRB#STUDY0003701).
Informed consent
Informed consent was not sought for this study because of the retrospective nature of the analysis. Waiver of consent was granted on the basis that no prospective intervention or alteration of therapy was performed and use of existing data.
