Abstract
Objectives:
We aimed at investigating the iridocorneal angle, anterior segment structural differences, and their relationship in pediatric anisohyperopic amblyopic eyes.
Methods:
This study was designed as an observational case-control study. We prospectively evaluated the pediatric patients who were newly diagnosed with hyperopic anisometropic amblyopia. Iridocorrneal angle in four sectors, axial length, and anterior segment structures were compared to emmetropic fellow and control eyes using Pentacam and IOLMaster 700 devices. The anisohyperopia and iridocorneal angle related factors were determined in hyperopic anisometropic amblyopic eyes.
Results:
Forty-three hyperopic anisometropic amblyopic eyes, 43 fellow eyes, and 44 control eyes were included. The mean axial length, anterior chamber volume, and anterior chamber depth were significantly reduced in the study eyes compared to the fellow (p < 0.001, p = 0.021, and p = 0.045, respectively) and control eyes (p < 0.001, p = 0.001, and p = 0.009, respectively). The average iridocorneal angle was significantly lower in the study eyes than in the fellow and control eyes (p < 0.001 and p = 0.018, respectively). The iridocorneal angle correlated with anterior chamber depth (r = 0.49, p = 0.013), anterior chamber volume (r = 0.42, p = 0.038), the anterior radius of curvature (r =−0.54, p = 0.005), the posterior radius of curvature (r =−0.58, p = 0.002), and lens thickness (Rho =−0.41, p = 0.033) in the study eyes. Anisohyperopia was related to the interocular difference in axial length (Rho = 0.53, p = 0.005), anterior chamber depth (Rho = 0.53, p = 0.005), and anterior chamber volume (Rho = 0.42, p = 0.031).
Conclusion:
Pentacam and IOLMaster 700 are suitable for non-contact imaging of iridocorneal angle and anterior segment structures in the pediatric age. These biometric differences and their relationship should be kept in mind whenever anterior or posterior segment surgery is planned for pediatric anisohyperopic amblyopic eyes.
Keywords
Introduction
Determining the iridocorneal angle (ICA), axial length (AL), and anterior segment biometric parameters is essential in planning anterior or posterior segment surgery, and predicting glaucoma progression. Pentacam and IOLMaster 700 are suitable and easy to use for non-contact imaging of ICA and anterior segment structures. The ICA, AL, and anterior segment biometric differences in anisohyperopic amblyopic eyes should be kept in mind when anterior or posterior segment surgeries are planned for congenital or acquired pathologies in pediatric age.
Anisometropia (spherical equivalent (SE) difference = dSE) is defined as one or more diopter (D) differences in the SE between two eyes. It is derived from anisomyopia, anisohypermetropia, or anisoastigmatism based on the refractive difference between the eyes. Anisohypermetropia and anisoastigmatism are more susceptible to evolving into amblyopia than anisomyopia. 1 Anisohypermetropic amblyopia is characterized by a decrease in best-corrected visual acuity (BCVA) of at least two acuity test lines in the affected eye due to anisohypermetropia. 2
Microvascular and structural changes in the fovea, choroid, optic nerve head, lateral geniculate nucleus, and visual cortex related to anisometropic amblyopia have been reported in many studies.3 –7 It is controversial whether hyperopic anisometropic eyes have some anterior segment structural changes.8,9 We aimed at investigating variations in anterior segment structures and ICA in four sectors and determining the association among these structural changes and dSE in hyperopic anisometropic eyes using Pentacam (OCULUS Optikgeräte GmbH, Wetzlar, Germany) and IOLMaster 700 (Carl Zeiss Meditec AG, Jena, Germany).
Materials and methods
This study was designed as an observational case-control study and carried out between December 2019 and October 2020. We prospectively evaluated pediatric patients aged between 6 and 16 years who attended Muğla Sıtkı Koçman University Training and Research Hospital, Department of Pediatric Opthalmology, for routine ocular examination.
In the first examination run, a routine ocular examination for pediatric patients, including uncorrected visual acuity, cover–uncover test, ocular motility tests, intraocular pressure (IOP) with Topcon TRK-2P (Topcon Medical Systems, Inc., Tokyo, Japan) air puff tonometer, cycloplegic refractive error, and anterior and posterior segment investigation with slit-lamp biomicroscopy, was carried out. Cyclopentolate 1% (Cycloplegin; Abdi Ibrahim, Istanbul, Turkey) was applied three times at intervals of 5 min to obtain a full cycloplegic refractive error. Patients then waited for about 45 min to reach complete cycloplegia, which resulted in dilated pupils that did not react to intense light. The evaluation was performed with a Topcon TRK-2P (Topcon Medical Systems, Inc., Tokyo, Japan) keratorefractometer. A standard refractometer model was used, and the mean spherical and cylindrical powers (S and C, respectively) of three consecutive measurements were recorded. If three straight measurements differed by >0.50 D, repeated evaluations were performed until the variations decreased to <0.50 D. The SE was calculated using the S + C / 2 formula. 10 After the first examination, patients were sent for a second examination to define BCVA with cycloplegic refraction results and scan non-dilated imaging of the anterior segment parameters and ICA with the Pentacam and IOLMaster 700.
In the second examination run, corneal thickness (CT) at the pupil center, horizontal white-to-white diameter (WTW), corneal volume (CV) in a 10 mm zone around the anterior corneal apex, anterior chamber volume (ACV) in a 12 mm zone around the anterior corneal apex, internal anterior chamber depth (int ACD), the anterior and posterior radius of curvature (ARC, PRC), pupil size, and ICA in four sectors were imaged with Pentacam Scheimpflug camera under 150–200 lux photopic condition with a non-dilated pupil. The photopic condition and non-dilated pupil were used to show the ICA properly. The Pentacam Scheimpflug non-contact camera rotates around the eyes, generates radially oriented images, creates a three-dimensional model of the entire anterior segment structures, and automatically measures those structures and the ICA in many sectors. The ICA was recorded in four sectors (superior, inferior, nasal, and temporal). The average ICA (aICA) was calculated as the arithmetic mean of the four quadrants. The automated release mode was used, and repeated measures were obtained until the image quality specification showed an OK sign. It means that the pictures have sufficient resolution for a complete imaging of anterior segment structures and reliable measurements. Images with a quality score of <95% and with no visibility of the peripheral part of the iris and base of the ICA in all quadrants were not included. To reduce upper and lower lid originated artifacts and to image the ICA completely in four sectors, an assistant fully opened the eyes at least 1 millimeter (mm) distance from the limbus (Figure 1).

Anterior segment pictures collected with Pentacam and IOLMaster 700. (a) and (b) pictures show superior, inferior, nasal, and temporal ICA and ACD in a patient’s right eye. The AL and LT were measured with IOLMaster 700 (Picture (c)).
AL and lens thickness (LT) were automatically measured with IOLMaster 700 optical biometer in the same room and under the same light conditions. This device measures the mean values of three scans and gives a standard deviation (SD) result. The measurement was accepted as reliable if the SD was under 27 and 38 µm for AL and LT, respectively. All measurements were obtained by an expert who was blinded to the study (Figure 1).
The SE difference between the two eyes of both study and control groups was computed as dSE. Anisohyperopia was diagnosed as at least 1.5 D hyperopic differences in SE between two eyes for cycloplegic refraction. Hyperopic anisometropic amblyopia was defined as at least two or more line difference in BCVA with cycloplegic refraction on the Snellen chart. Differences in AL (dAL), ACD (dACD), ACV (dACV), CT (dCT), ARC (dARC), PRC (dPRC), WTW (dWTW), LT (dLT), and ICA (dICA) between study and fellow eyes were computed from the mean of the values.
Pediatric patients who had hyperopic anisometropic amblyopia were included in the study group, and their amblyopic and non-amblyopic eyes were enrolled as study and fellow eyes. In the control group, one emmetropic healthy eye without any congenital or acquired anterior and posterior segment pathology was randomly selected from age- and sex-matched volunteers whose uncorrected visual acuity was 1.0 decimal and S and C power < 1 D in the cycloplegic condition. Patients with other amblyopia types (strabismic, stimulus deprivation, other refractive amblyopias) and involuntary eye movement, a history of ocular surgery, peripheric anterior synechiae or other iridocorneal abnormalities that do not allow for reliable ICA imaging, corneal, lens, vitreous, retinal, or optic nerve head pathologies, ametropia (bilateral ⩾ 5 D S power, ⩾ 2 D C power), anisomyopia, anisoastigmatism, and unreliable results were not included. Patients who did not come for the second examination were also excluded.
Statistical analysis
The Statistical Package for the Social Sciences (SPSS) version 21.0.0.0 (IBM Corporation, 2012, Armonk, NY, USA) was used for data collection and statistical analysis. After testing normality with the Shapiro–Wilk test, a paired-sample t-test, or Wilcoxon signed-rank test was applied to compare study eyes with fellow eyes, and a Student’s t-test, or Mann–Whitney U test was used to compare study eyes with control eyes. Pearson’s or Spearman’s rank correlation coefficient was used for assessing associations. A chi-square test with Yates’ continuity correction was used to compare gender. A value of p < 0.05 indicated statistical significance.
The study followed the tenets of the Declaration of Helsinki’s principle and was approved by the Mugla Sıtkı Kocman University Clinical Research Ethics Committee with a 12/Ⅱ decision number. All patients and parents or legally authorized representatives were informed, and written consent was obtained from the child and his or her parents or legally authorized representatives.
Results
Eighty-six eyes (43 study and 43 fellow eyes) of 43 patients (20 (46%) female, 23 (54%) male) in the study group and 44 eyes of 44 patients (19 (43%) female, 25 (57%) male) in control group were included. The mean ± SD of age was 9.78 ± 3.43 years (range 6–16 years) and 11.00 ± 2.83 years (range 6–16 years) in the study and control groups, respectively. The groups were similar for age and gender (p = 0.104 and p = 0.734, respectively). The mean IOP in the study eyes was similar to the mean values of fellow and control eyes (p = 0.561 and 0.126). The S and C powers and SE were considerably higher in the study eyes than fellow eyes (p < 0.001, p = 0.002, and p < 0.001, respectively) and control eyes (p < 0.001, p < 0.001, and p < 0.001, respectively). The mean ± SD of dSE was 2.50 ± 0.65 D (range 1.5–3.88 D) in the study group. The mean ± SD of dSE (selected and unselected eyes of the same participants) in the control group was 0.04 ± 0.13 D (range 0.13–0.38 D) and not clinically important. The mean ± SD of BCVA in study eyes was 0.48 ± 0.29 decimal and significantly lower than in fellow and control eyes (p < 0.001 and < 0.001, respectively). The AL, ACV, and ACD values were considerably lower in the study eyes than the fellow eyes (p < 0.001, p = 0.021, and p = 0.045, respectively) and control eyes (p < 0.001, p = 0.001, and p = 0.009, respectively). On the contrary, CT, LT, WTW, pupil size, CV, ARC, and PRC in study eyes were similar to values in the fellow and control eyes (all these results are reported in Table 1).
Comparison of the two groups and eyes in terms of gender, age, dSE, AL, BCVA, refractive error, and anterior segment structures.

A paired t-test or Wilcoxon signed-rank test, and student t-test or Mann–Whitney U test were applied, respectively, to compare the study eyes with fellow and control eyes after testing normality using Shapiro–Wilk test. Chi-square test with Yates’ continuity correction was used to compare gender. The significant p values are shown in bold.
D: diopter; F: female; M: male; BCVA: best-corrected visual acuity; AL: axial length; SE: spherical equivalent; dSE: anisohyperopia (spherical equivalent difference); S: spherical; C: cylindrical; CT: corneal thickness at the pupil center; IOP: intraocular pressure; LT: lens thickness; WTW: white-to-white diameter; CV: corneal volume; ACV: anterior chamber volume; ACD: anterior chamber depth; ARC: anterior radius of curvature; PRC: posterior radius of curvature; SD: standard deviation; n: number.
Indicates the values that are not distributed normally.
In general, the aICA was significantly lower in the study eyes than in fellow and control eyes (p < 0.001 and p = 0.018, respectively). In the sectoral analysis, the ICA was reduced in all sectors in study eyes although the decrease was considerable in the superior and nasal quadrant (p = 0.003 and 0.041, respectively) compared to the fellow eyes and only in the nasal (p = 0.003) sector compared to the control eyes (Table 2). The study eyes had a narrower and wider ICA in the sectoral analysis of the superior and temporal quadrants, respectively. In addition, in the study eyes, the aICA correlated with ACD (r = 0.49; p = 0.013), ACV (r = 0.42; p = 0.038), ARC (r = −0.54; p = 0.005), PRC (r = −0.58; p = 0.002) and LT (Rho = −0.41; p = 0.033) as shown in Figures 2–4, respectively. The dSE (Rho = 0.23; p = 0.278), WTW (Rho = 0.21; p = 0.189), AL (Rho = 0.07; p = 0.678), CT (Rho = −0.05; p = 0.953), and SE (Rho = 0.20; p = 0.221) did not significantly affect the aICA (Table 3).
Comparison among average values (mean) for the ICA in four quadrants.

Due to the normal distribution of all values in this table, paired t-test and Student t-test were applied, respectively, to compare the study eyes with fellow and control eyes. The aICA was calculated as the arithmetic mean of the four quadrants. The significant p values are shown in bold.
aICA: average iridocorneal angle; SD: standard deviation; n: number.

Relationship between aICA and ACD, ACV in the study eyes. The aICA positively correlated with ACD (r = .49; p = .013) and ACV (r = .42; p = .038).

Relationship between aICA and ARC, PRC in the study eyes. The aICA negatively correlated with ARC (r =–.54; p = .005), PRC (r =–.58; p = .002).
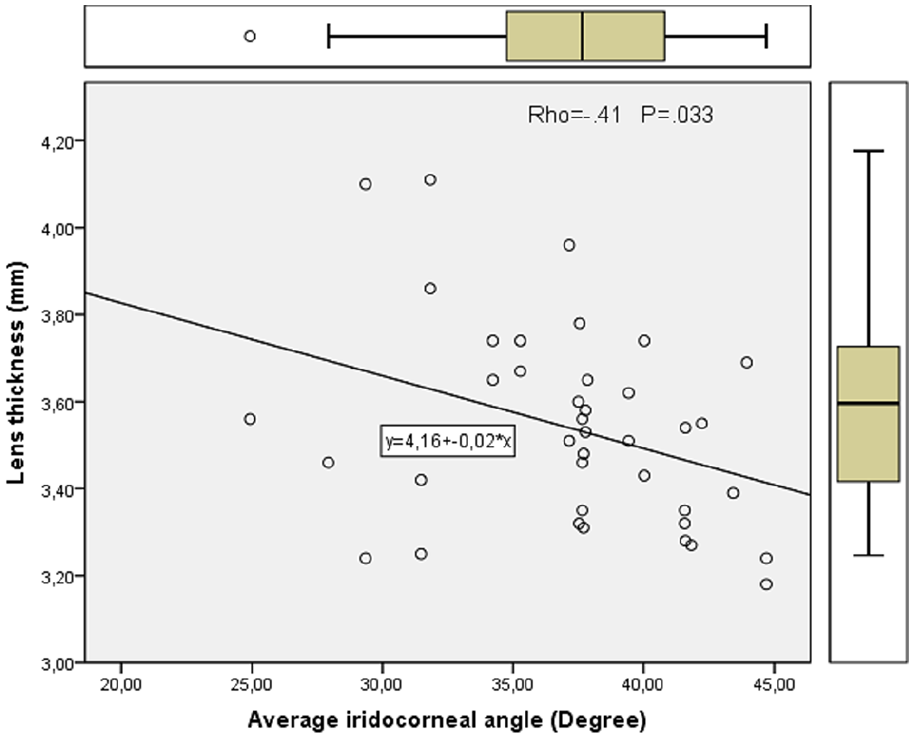
Relationship between aICA and LT in the study eyes. The aICA negatively correlated with LT (Rho =–.41 p = .033).
Correlation between aICA, dSE, SE, AL, and anterior segment structures in study eyes.

The aICA is negatively correlated with LT, ARC, and PRC. Moreover, it is positively associated with ACD and ACD. The relations between aICA and dSE, AL, and SE are all not significant. We used the Pearson’s or Spearman’s rank correlation coefficient depending on the normal distribution of the values. The significant p values are shown in bold.
Rho: Spearman’s rank correlation coefficient; r: Pearson’s correlation coefficient; AL: axial length; SE: spherical equivalent; dSE: anisohyperopia (spherical equivalent difference); LT: lens thickness; ACV: anterior chamber volume; ACD: anterior chamber depth; ARC: anterior radius of curvature; PRC: posterior radius of curvature; WTW: white-to-white diameter; aICA: average iridocorneal angle.
In addition, in the study group, dSE was related to dAL (Rho = 0.53; p = 0.005), dACD (Rho = 0.53; p = 0.005), and dACV (Rho = 0.42; p = 0.031) as shown in Figures 5 and 6, respectively. Corneal (dARC (Rho = −0.04; p = 0.832), dPRC (Rho = −0.10; p = 0.631), dWTW (Rho = −0.22; p = 0.261), dCT (Rho = 0.02; p = 0.932)), and lenticular (dLT (Rho = −0.24; p = 0.227)) structural differences were not linked to dSE (Table 4).

Relationship between dSE and dAL in the study group. In the study group, dSE was related to dAL (Rho = .53; p = .005).

Relationship between dSE and dACD, dACV in the study group. In the study group, dSE was related to dACD (Rho = .53; p = .005) and dACV (Rho = .42; p = .031).
Correlation between dSE and ocular structural differences between the study and fellow eyes.

dSE was correlated with dAL, dACD, and dACV, and not correlated to dLT, dARC, dPRC, dWTW, dCT, and dICA. The significant p values are shown in bold. This table shows that AL, ACV, ACD differences between anisohyperopic and fellow eyes increase as anisohyperopia increases.
Rho: Spearman rank correlation coefficient; dSE: anisohyperopia (spherical equivalent difference); dAL: axial length difference; dLT: lens thickness difference; dACV: anterior chamber volume difference; dACD: anterior chamber depth difference; dARC: anterior radius of curvature difference; dCT: corneal thickness difference; dPRC: posterior radius of curvature difference; dWTW: white-to-white diameter difference; dICA: iridocorneal angle difference.
Discussion
This study is unique in investigating the ICA variations in four sectors and their relation to anterior segment structures in hyperopic anisometropic amblyopic eyes. We found that the aICA was lower in the study eyes than in fellow and control eyes. The mean ICA was reduced in all sectors, although ICA was significantly decreased in the nasal quadrant only than the fellow and control eyes. In the study eyes, the aICA was affected by LT, ARC, PRC, ACD, and ACV but not by AL, WTW, SE, CT, and dSE. Moreover, dSE was correlated with dAL, dACD, and dACV but not to dLT, dARC, dPRC, dWTW, dCT, and dICA.
Determining ICA is essential in planning cataract or refractive surgery and predicting glaucoma progression.11 –13 Gonioscopy is a standard method for ICA analysis, although it is semiquantitative, self-dependent, and not appropriate for uncooperative patients, such as children. Nowadays, ultrasonic biomicroscopy (UBM), anterior segment optic coherence tomography (AS-OCT), and Scheimpflug imaging are commonly used to image the ICA.
Pentacam uses Scheimpflug imaging cameras and captures three-dimensional pictures of anterior segment structures and ICA with no contact with the patients’ eyes. Although it cannot show the innermost details of the ICA structures as much as UBM and AS-OCT, sufficient correlation with gonioscopy and high repeatability has been reported.7,14,15
Onay et al. 16 evaluated the ICA in pediatric subjects using Pentacam and observed significantly different results between right and left eyes. Moreover, they reported that children had wider ICA than adults. Nevertheless, their patients had healthy eyes rather than hyperopic anisometropic amblyopic eyes, and the control group consisted of adult participants. Bozkurt et al. 17 reported a decrease in ICA in hyperopic anisometropic amblyopic eyes in pediatric subjects compared to fellow and control eyes. Their study design and results were similar to ours, except for the sectoral analysis of ICA. They did not compare the ICA in four quadrants and did not evaluate its relation with dSE, AL, and other anterior segment structures.
We found that the aICA was negatively correlated with LT, ARC, and PRC, and positively correlated with ACD and ACV in the study eyes. Likewise, Xu et al. 18 related narrow ICA with shallow ACD, decreased ACV, and increased LT in a population-based study. Schuster et al. 19 explained the antagonistic relation between ICA, ARC, and PRC as the peripheral cornea shapes the frontal part of the ICA. As the cornea flattens and the lens thickens, the iridocorneal width and ICA decrease. Moreover, we found no association between aICA and AL, CT, SE, WTW, and dSE. In contrast, some studies have reported that shorter AL was related to narrowed ICA.13,20 However, their patients had angle-closure glaucoma rather than hyperopic anisometropic eyes. Some studies have shown that ICA is related positively to myopia, negatively to hypermetropia, and not to CT.19,21,22 Nevertheless, they did not compare anisohyperopic eyes with emmetropic eyes.
Decreased AL in hyperopic anisometropic eyes is a well-known finding reported in several studies.23,24 Patel et al. 25 found that anisohyperopic amblyopic eyes had shorter AL than healthy fellow eyes. In addition, we found a positive correlation between dAL and dSE in hyperopic anisometropic amblyopic eyes, that is, the AL difference between anisohyperopic and fellow eyes increases as anisohyperopia increases.
Different results have been reported for anterior segment structural changes in hyperopic anisometropic eyes. Similar to our results, Yüksel et al. also reported no significant differences in ARC, PRC, CV, and pupil diameter in hyperopic anisometropic amblyopic eyes. In addition, we also found that dSE did not correlate with the differences in dARC, dPRC, dCT, and dWTW in patients with anisohyperopic eyes.
Varied findings were shown in the comparison of ACD and ACV in hyperopic anisometropic amblyopic eyes. Similar to our results, Demircan et al. 23 also reported decreased ACD and ACV in hyperopic anisometropic amblyopic eyes. In distinction, Wang and Taranath 26 reported that ACD and ACV in hyperopic anisometropic amblyopic eyes were not different to those in fellow eyes. In our opinion, their study had some limitations. Their sample size was relatively small, and spherical refractive error was more hyperopic in fellow eyes. It seems that fellow eyes have an ametropic amblyopic pattern. Moreover, they did not compare the hyperopic anisometropic amblyopic eyes with emmetropic controls.
We found that the ACD and ACV were significantly reduced in hyperopic anisometropic amblyopic eyes than in fellow and control eyes. In addition, dACD and dACV were positively correlated with dSE in the study group. We interpreted this finding as increased anisohyperopia produced more significant interocular differences for ACD and ACV, resulting in shallower ACD and decreased ACV in more hyperopic eyes.
Our study had some limitations. The LT, ACD, ACV, and other values may be affected by accommodation because the measurements were carried out without cycloplegia. We included only anisometropic amblyopic eyes. Thus, the findings could not be validated for other types of amblyopia. All patients were newly diagnosed and had not been treated for amblyopia. The effects of patching or other treatment strategies and visual acuity changes on these structures are still unexplained. In addition, the study’s sample size and power analysis were not performed.
Conclusion
To conclude, Pentacam and IOLMaster 700 are suitable and easy to use for non-contact imaging of ICA and anterior segment structures in pediatric subjects. The ICA, AL, and anterior segment biometric differences in anisohyperopic amblyopic eyes should be kept into account when anterior or posterior segment surgery is planned for congenital or acquired pathologies in pediatric subjects. Long-term prospective studies are needed to clarify the effect of amblyopia treatment and visual acuity improvement on these structures.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Mugla Sıtkı Kocman University Clinical Research Ethics Committee with a 12/II decision number. In this study’s procedures, we followed the local institutional research committee’s ethical standards and the 1964 Helsinki declaration of ethical standards. The Local Ethics Committee arranged ethical approval.
