Abstract
Objective:
The burden of pulmonary fungal infection is increasing and often misdiagnosed as pulmonary tuberculosis in developing countries where the prevalence of pulmonary tuberculosis is high. |Therefore, the purpose of this study is to determine the spectrum of potential pulmonary fungal pathogens and the prevalence of the association between pulmonary tuberculosis and potential fungal pathogens.
Materials and methods:
A cross-sectional study was conducted between October 2018 and May 2019. Sputum was collected from 636 study participants. Part of the sputum was inoculated onto Brain Heart Infusion agar, and fungi were identified following standard microbiological procedures. The remaining part of the sample was used for the investigation of pulmonary tuberculosis.
Results:
Among 636 sputum samples, 75.9% (483) and 25.6% (163) were positive for potential fungal pathogens and pulmonary tuberculosis, respectively. The prevalence of the association between pulmonary tuberculosis and potential fungal pathogens was 20.0%. Of fungal isolates, 81.4% were yeasts. The remaining 128 (18.6%) isolates were molds. The isolation rate of fungi was higher in males (51.6%) than in females (48.4%). There was no statistically significant association between the prevalence of potential pulmonary fungal pathogens and sex (
Conclusion:
High prevalence of potential pulmonary fungal pathogens and the association of tuberculosis and potential fungal pathogens recorded in this study will enforce health personnel to pay due attention to these conditions and arise the interest of researchers to conduct further work on the burden of the association between tuberculosis and potential fungal pathogens. Our study also revealed the need to employ conventional microbiology tests along with clinical and radiological evidence since clinical manifestations and radiological pictures of tuberculosis mimic that of pulmonary fungal infection.
Introduction
Although the true burden of fungal respiratory tract infection is elusive, the frequency of the infection has been increasing in the last few decades. 1 Pulmonary tuberculosis (PTB), HIV/AIDS, chronic obstructive pulmonary disease, and extensive use of immunosuppressive drugs are incriminated for such an increase. 2 Globally, it is predicted that 1.2 million individuals live with chronic pulmonary aspergillosis (CPA) as a sequel to PTB, in which the prevalence of CPA is the highest in Africa, Western Pacific, and Southeast Asia. 3 The chronic nature of PTB along with prolonged chemotherapy with or without corticosteroids resulted in immune suppression in PTB patients eventually leading PTB patients susceptible to fungal infection. 4
Pulmonary fungal infection is an infectious disease of the lungs that is caused by fungi. The infection develops after the colonization of the lungs by fungi or their spores through inhalation, or the reactivation of latent infection, or via hematogenous dissemination. Fungi or their spores are abundant and exist virtually everywhere in the human environment.5,6 Given the ubiquitous nature of fungi and their spores, colonization or infection of the lungs is unavoidable, but mechanisms of differentiating fungal colonization from fungal infection are not well established, and hence the subject remains a serious challenge. In this article, isolation of fungi alone or in association with
PTB is principally a disease of poverty, with 95% of cases and 98% of deaths occurring in developing countries. 12 Ethiopia stands 10th among the 30 high TB burden countries with an estimated incidence rate of 151/100,000. 13 The high rate of co-infection of pulmonary fungal infection with PTB further compounded the burden of PTB in these countries as the association of the two infections is responsible for a high rate of morbidity and mortality. 3 Therefore, proper diagnosis of fungal pathogen especially in PTB patients is critical. 14 To this end, the spectrum of pulmonary fungal infections and their association with PTB in Ethiopia is lacking.
Furthermore, similarities in clinical and radiological presentation of pulmonary fungal infection and PTB have made definite diagnosis between these two infections difficult. Persistent cough for more than 3 weeks is a common symptom of pulmonary disorders caused by
Methods
Study area and design
The present study is a hospital‑based, cross‑sectional study conducted at the Saint Peter’s Specialized Tuberculosis Referral Hospital located in Addis Ababa Administrative region, the capital city of Ethiopia between October 2018 and May 2019.
Population
Source of population
The source of population is those people seeking health service at Saint Peter’s Specilized Tuberculosis Referral Hospital and those referred from other health institutes.
Study population
The population of this study is those patients who were assessed clinically and by radiological procedures by attending physicians in the hospital.
Inclusion and exclusion criteria
Patients with clinical manifestation of pulmonary infection (particularly of those with a persistent cough for more than 3 weeks and with radiological characteristics mimicking that of PTB) were included in the study. Healthy individuals with no clinical symptoms, no radiological picture mimicking of PTB, and those who were under antifungal treatment were excluded from the study.
Sample size determination
The minimum sample size of this study was determined based on a single population proportion formula,
Sampling method
A convenient sampling method was utilized to achieve the estimated sample size. All PTB presumptive patients visit Department of Microbiology Laboratory of Saint Peter’s Specialized Tuberculosis Referral Hospital within the specified time of the study.
Data collection procedure
Demographic data
The sociodemographic data—age, gender, and clinical symptoms—were obtained from a standard laboratory request form completed by physicians.
Data quality assurance quality control
The quality of sputum samples, GeneXpert kit and reagents, and mycological culture media were inspected before they are used. The sterility of culture media and the growth performance of each culture medium were evaluated as per standard procedures.
Statistical analysis
All data from the investigation were coded, double entered, and analyzed using SPSS version 20. Descriptive statistics and logistical regressions were used to estimate the crude ratio with a 95% confidence interval to the different variables. The
Laboratory investigation
Sputum collection
Patients were instructed to wash their mouth gently with tap water prepared for this purpose more than once and then purulent sputum was collected by sterile falcon tube by breathing deeply three times. Part of the early morning sputum was used for the detection of
Fungal isolation and characterization
Unprocessed sputum was inoculated directly onto duplicate Brain Heart Infusion agar tubes supplemented with chloramphenicol (Oxoid, Basingstoke, UK) under safety cabinet level II at Saint Peter’s Specialized Tuberculosis Referral Hospital. All inoculated tubes were then transported to the Department of Medical Laboratory Science, College of Health Science, Addis Ababa University. One of the tubes was incubated at 25°C while the other one was incubated at 37°C aerobically for up to 4 weeks. Culture plates were examined twice a week for any fungal growth.
Identification
Mold identification
Mycelia fungi were identified by studying their microscopic and macroscopic characteristics. Pigmentation of the front and the reverse side, texture, topography, and rate of growth of each culture were considered for macroscopic identification. Diagnostic microscopic features of mycelial fungi were determined by using a Lactophenol Cotton Blue (LPCB) staining procedure. Briefly, a drop of LPCB stain was placed on a clean glass slide. A piece of fungal culture was placed on clean glass slides containing LPCB for the staining process. A stained preparation was then covered by a cover slide and examined for microscopic characteristics such as macro- and micro-conidia, chlamydospores, the morphology of reproductive structures, and the nature of hyphae by using 10× and 40× objectives of the microscope. Features seen in the stained slide were compared with established characteristics of fungal features using mycology atlases.16,17
Yeast identification
Yeasts were identified by employing an array of standard biochemical and assimilation test procedures, 18 germ tube production, and using CHROMagar Candida culture medium (Becton Dickinson, Paris, France) as per the instruction of the manufacturer.
Detection of M. tuberculosis
Detection of
Results
Gender, age, and clinical symptoms of study participants
As shown in Table 1, the current study enrolled 636 study participants of which 327 (51.4%) were males and 309 (48.6%) were females. The ages of the study participants varied from 1 to 94 years with a mean age of 41 years. Table 1 also shows the percentage of patients with each symptom. The most common presenting symptom was cough (97.95%), followed by expectoration (78.45%) and weight loss (43.86%).
Demographic profile and clinical symptoms of study participants.

Distribution of fungi with gender and age
As shown in Table 2, out of 636 sputum samples collected and analyzed, fungal species were recovered from 483 samples. Among 483 culture-positive sputum samples, 51.6% (249/483) and 48.4% (234/483) were collected from male and female study participants, respectively. Consequently, the isolation rate of fungi was higher in male than in female study participants. However, there was no statistically significant association between the prevalence of pulmonary fungal isolation rate and the sex of patients (
Distribution of fungal isolates against gender and age.

Statistical association as determined by χ2 test.
The spectrum of fungal isolates
In the present study, of a total of 636 sputum cultures, 483 were positive for potential fungal pathogens out of which 180 samples yielded more than one fungal species. A total of 690 fungal isolates were recovered. Among the isolates, 562 (81.4%) were yeasts comprising of
Spectrum of fungal isolates in presumptive pulmonary tuberculosis patients (

NAC: non albican candida.
TB and fungal co-infection
Among 636 study participants, 75.9% (483/636) were positive for potential fungal pathogens, and 25.6% (163/636) were positive for PTB. The association between PTB and potential fungal pathogens among the study participants (636) was 20% (127/636). With regard to PTB patients, the association between PTB and potential fungal pathogens was seen in 77.9% (127/163) of study participants (Table 4). The association between PTB and potential fungal pathogens was not statistically significant.
Prevalence of tuberculosis and the association between tuberculosis and potential fungal pathogens among study participants (

Statistical association as determined by χ2 test.
As shown in Table 5, about 183 potential fungal pathogens were recovered from PTB and potential fungal pathogens associated study participants of which 148 (80.9%) were yeasts while 35 (19.1%) were mycelial fungi.
Spectrum of fungi in fungal–tuberculosis associated patients (
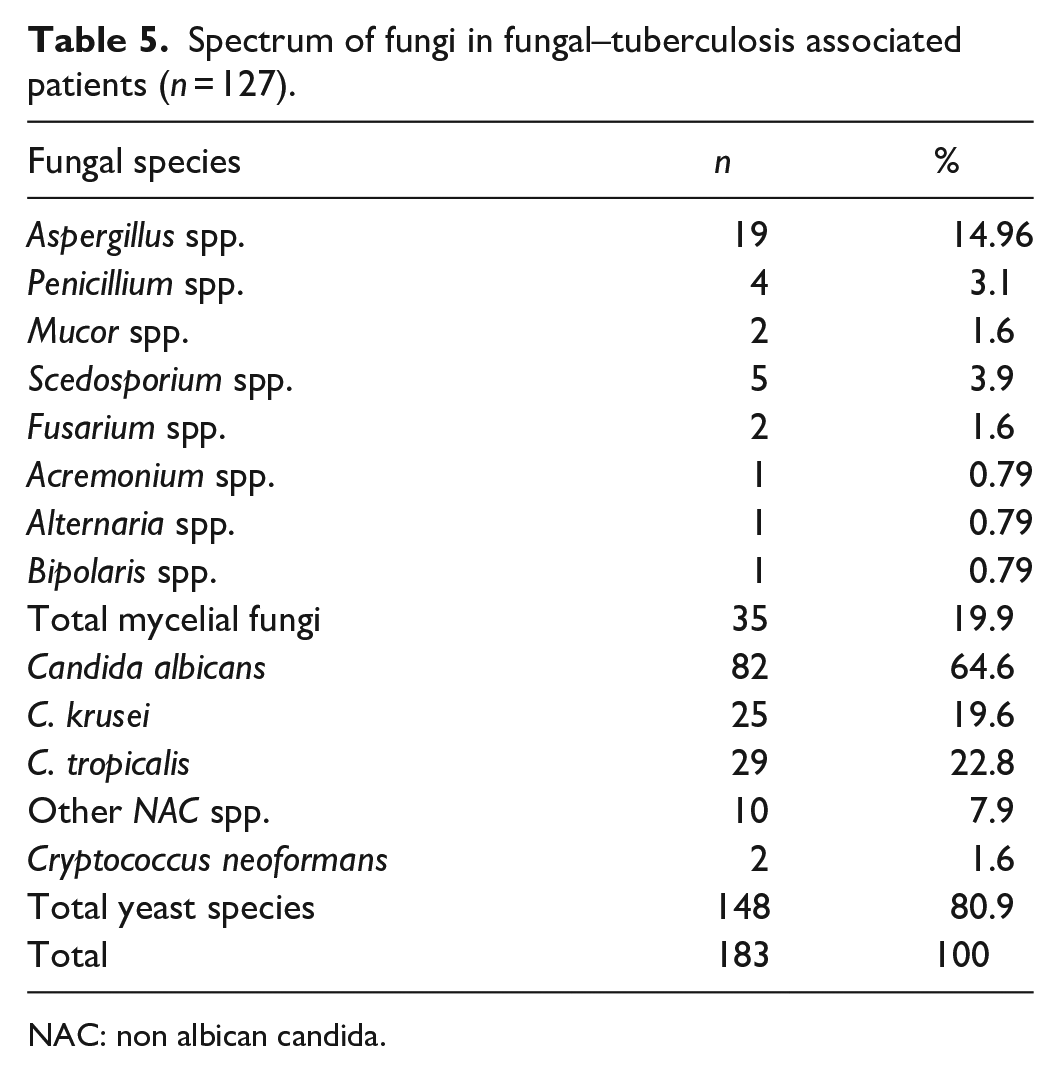
NAC: non albican candida.
Discussion
The global burden of pulmonary fungal infections caused by opportunistic fungal pathogens is increasing. 19 Sustaining of patients by drugs, chemicals, and mechanical processes that compromise physical barriers to infection, suppress immune mechanisms, or upset the balance of normal flora are responsible for rendering hosts more susceptible not only to pathogenic fungi but also to all fungi with which they come in contact. The increased age of the world population that resulted in more chronic diseases with their debilitating effects is also another attributing factor for an increase in fungal lung infection by an opportunistic fungal pathogen. 20 The impact of these factors may explain for the high prevalence of the association between potential fungal pathogens and TB reported in the present study.
Our result regarding the prevalence of PTB and potential pulmonary fungal pathogens was consistent with the findings of Sani et al., 21 but PTB potential fungal pathogen association in our study was threefold (20.0% against 6%) from that of their report. On the contrary, PTB potential fungal pathogen association in the range of 18% to 40% was reported by other similar studies.22,23 The prevalence of PTB potential fungal pathogen association in this study was also high. This may support that preexisting or residual cavity produced following TB infection are frequent places of fungal colonization, and the chromic nature of PTB along with prolonged chemotherapy makes PTB patients more susceptible to fungal infection. 4 The high prevalence of PTB and potential fungal pathogen association exhibited in this study may worsen the existing burden of PTB and, hence, due attention should be given.
Lower respiratory tract fungal infection (pulmonary fungal infection) such as aspergillosis frequently occurs in middle-aged to an elderly individual and is more commonly reported in male patients.24,25 Our finding was in line with the findings of Kosmidis and Denning
24
and Kohno et al.
25
as the isolation rate of potential fungal pathogens was higher in patients above 35 years than younger age groups, and male than female study participants. Other studies,26,27 however, demonstrated that the age group of 20 to 34 years is most affected by fungal pathogens. Certainly, old age is a known risk factor for pulmonary fungal infection probably due to diminishing immune function as one gets aged.
20
The association of potential pulmonary fungal infection with age and sex in our study was not statistically significant with respective
In the present study, many species of mycelia fungi and yeasts were recovered from sputum. Among fungal isolates,
Fungal infections of the respiratory tract by large are considered to be identical with invasive pulmonary infections caused by
Pulmonary fungal infections have long been recognized as a significant complication and are mainly caused by
In our study,
Limitations of the study
The increased prevalence of fungal lung infections now than in the past is largely related to increased numbers of immune-compromised human hosts. Lack of information about the immune status of study participants was the major limitation of the study as most of human fungal pathogens are opportunistic. Due to lack of facilities and resources, unable to determine the antifungal susceptibility profile of the fungal isolates in the present study was another limitation of our study.
Conclusion
High prevalence of potential pulmonary fungal pathogens in our study, especially in cases where sputum for
Footnotes
Acknowledgements
The authors would like to thank the Department of Medical Laboratory Science, College of Health Sciences, Addis Ababa University, Ethiopia, and Saint Peter’s Specialized Tuberculosis Referral Hospital for allowing the authors to use laboratory facilities for free. The authors also would like to acknowledge study participants for their participation in the study. They are also very grateful to the medical laboratory technologists for their help during sample collection.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval of this study was obtained from the Internal Review Board (IRB) of the Department of Medical Laboratory Science, College of Health Sciences, Addis Ababa University (Protocol Number: DRER/393/19/MLS/2019).
Informed consent
Written informed consent from all participants or their legally authorized representatives was obtained prior to study initiation.
