Abstract
Objectives:
Implant failure leading to revision total joint arthroplasty can occur through a variety of different mechanisms which are typically associated with a soft tissue response adjacent to the implant that provide insight into the underlying etiology of implant failure. The objective of this study was to elucidate mechanisms of implant failure as they relate to histological classification and findings of adjacent periprosthetic tissue.
Methods:
Histological analysis of soft tissue adjacent to the implant was performed in 99 patients with an average age of 64 years old and grouped into four categories based on the study conducted by Morawietz et al.:
Type I (N = 47) Wear particle induced type
Type II (N = 7) Infectious type
Type III (N = 19) Combined type I and II
Type IV (N = 26) Indeterminant type
Modes of failure were categorized into five groupings based on the study conducted by Callies et al.: Instability (N = 35), Aseptic Loosening (N = 24), Hardware and/or Mechanical Failure (N = 15), Septic (N = 13), and Other failures (N = 12). We calculated odds ratios and conducted regression analysis to assess the relationship between modes of failure and histological findings as well as modes of failure and comorbidities.
Results:
Hardware/mechanical failure was independently correlated with histological findings of anucleate protein debris, histiocytes, Staphylococcus epidermidis, and synovitis. Furthermore, hardware/mechanical failure was independently correlated with osteosarcoma as a co-morbidity. Septic failure was associated with histological findings of Enterococcus, granulation tissue, and tissue necrosis as well as comorbidities of Crohn’s disease, deep venous thrombosis, lung disease, and rheumatoid arthritis. Infection was 5.8 times more likely to be associated with Type II histology. Aseptic loosening was associated with histologic findings of synovitis.
Conclusion:
Our findings support the existing literature on periprosthetic tissue analysis in revision total joint arthroplasty which may improve surgeon understanding of the patholophysiological mechanisms that contribute to implant failure and revision surgery.
Keywords
Introduction
Total joint arthroplasty (TJA) is one of the most successful surgical procedures for pain relief and functional improvement in patients with arthritic joints. The number of surgical procedures for primary total joint replacement and subsequently revision total joint replacements continues to increase each year. 1 The mode of failure that leads to revision TJA varies from case to case though there are several predominant mechanisms. 1
Implants may fail or become painful due to implant wear with or without aseptic loosening, septic loosening or deep infection, mechanical failure, soft tissue imbalance or instability, and malalignment of components. 2 With these modes of failure, there is an associated soft tissue response adjacent to the implants that may occur to a significant degree resulting in an unfavorable surgical outcome.3,4 In the past, microscopic analysis of the soft tissue response in patients with implant wear and/or aseptic component loosening has revealed a proliferative infiltration of macrophages with a foreign body reaction.3,5–14 Many times, these modes of failure lead to revision of the artificial joint.
There are approximately 100 revision TJAs performed at our institution by one surgeon each year. Surgical indications include implant wear (with or without associated osteolysis), aseptic loosening of implants, septic loosening of the implant with deep joint infection, soft tissue imbalance and/or instability, and malalignment of implants. Implant designs that utilize alternative bearing surfaces such as metal-on-metal chrome cobalt articulations, ceramic-on-ceramic implants, and modifications of the standard polyethylene articular implants create distinct soft tissue responses adjacent to the implant.1,11,14–16 At our institution, a patient presented for a revision surgery of a hip replacement and microscopic analysis of the adjacent soft tissue revealed a proliferative eosinophilic reaction consistent with an allergic response which prompted the current study of 99 patients who required arthroplasty revisions.
Our observational study was designed to evaluate the relationship between histological findings, mechanisms for failure, and co-morbidities in patients undergoing revision TJA. We hypothesized that specific histological findings and comorbidities would be associated with particular modes and/or categories of failure. These data may provide researchers and surgeons actionable information to guide future studies and patient management prior to implant revision.
Materials and methods
Population
Our study was a single-center prospective analysis of 99 soft tissue specimens over a 1-year time period from January 2016 to January 2017 that were debrided from areas adjacent to the implant of total joint revision, including synovial tissue. Inclusion criteria included all patients with an arthroplasty who underwent revision over the course of 1 year. There were no exclusion criteria within these patients. We did not conduct a power analysis for this study.
Tissue collection
The soft tissue specimens collected were debrided from bone and synovial tissue adjacent to the implants and sent to the pathologist for histological analysis, specimen culture, and hematoxylin and eosin (H&E) stain to categorize the data based on the modified scale from Morawietz et al. With respect to bacterial collection in culture, collection of these specimens within the implant–tissue interface is the most accurate method for pathological analysis. Collection of synovial and periprosthetic tissue is an established method used for histological classification.10,17,18
Histological tissue classification
To better analyze the data set, histological findings were grouped in a simplified but similar manner to the classification of periprosthetic tissue format laid out originally by Morawietz et al. 10 and validated by Bemer et al. 17 Type I histological findings indicate the presence of one or multiple of the following criteria related to granulomatous inflammation: Anucleate Protein Debris, Calcification, Histiocytic Infiltrate, Fibrous Tissue, Foreign Body, Giant Cell, Histiocytes, Hyperplasia, and Macrophages with Abundant Cytoplasm (Epithelioid Cells). 19 Type II histological findings indicate the presence of one or all of the following pathogenic organisms, including their clinical course: Diptheroids, Enterococcus, Granulation tissue (infection-related), Lymphocytic Infiltrate (without giant cell reaction), Soft-Tissue Necrosis, and Staphylococcus epidermidis.10,17 Type III histological findings indicate a combination of Type I and Type II histology whereas Type IV histology is a histologic finding that does not meet Type I or Type II histology.
Mode of failure classification
Modes of failure were categorized into five groupings. Pain, Instability (Primary or Secondary), Aseptic Loosening, Hardware and/or Mechanical Failure (which included one or all of the following: Arthrofibrosis, Eccentric wear, Hardware failure, Malalignment, Periprosthetic fracture, Reduced function), Septic Failure, and Other failures. (Other failures include one or more of the following: Unspecified Knee Pain, Crepitus, Deteriorated bone, Conversion of Hemi-arthroplasty to Total, High Grade Aseptic Lymphocyte-Dominated Vasculitis-Associated Lesions (Pseudotumor), Wound dehiscence.)20–22 Our failure classification was derived from an etiological study of revision total knee arthroplasties conducted by Calliess et al. 23
Statistical analysis
Medians and associated interquartile ranges were used to summarize demographic data. Frequencies and percentages were used to describe our sample in terms of operations, histology, and modes of failure (Table 1).
Description of study sample.

IQR: interquartile range.
All data are n (%) unless otherwise specified.
Type I: Wear particle induced type: Anucleate protein debris, calcification, histiocytic infiltrate, fibrous tissue, foreign body, giant cell, histiocytes, hyperplasia, and macrophages with abundant cytoplasm (epithelioid cells).
Type II: Infectious type: Diptheroids, Enterococcus, granulation tissue (infection related), lymphocytic infiltrate without giant cell reaction, soft-tissue necrosis, and Staphylococcus epidermidis.
Type III: Combined Type 1 and 2.
Type IV: Indeterminant type.
Odds ratios
Odds ratios were calculated using logistic regression analysis to determine whether different histological categories were associated with increased odds of certain categories of failure (Table 2). Given the broad confidence intervals associated with these findings, the decision was made to perform further analysis to allow for a more complete picture of the data and its implications.
Odds ratios for modes of failure given histology type classification.

Type I: Wear particle induced type: Anucleate protein debris, calcification, histiocytic infiltrate, fibrous tissue, foreign body, giant cell, histiocytes, hyperplasia, and macrophages with abundant cytoplasm (epithelioid cells).
Type II: Infectious type: Diptheroids, Enterococcus, granulation tissue (infection related), lymphocytic infiltrate without giant cell reaction, soft-tissue necrosis, and Staphylococcus epidermidis.
Type III: Combined Type I and II.
Type IV: Indeterminant type.
p-value < 0.05.
Beta regression coefficients
Logistic regression analysis was performed for histological categories and all comorbidities to further delineate their correlation with individual modes (rather than categories) of failure. Subsequent regression analysis was performed to determine the correlation of individual histological findings and comorbidities with categories of failure. Independent histological findings and comorbidities correlating with individual modes of failure were also identified.
ANOVA
Correlation coefficients (R) and adjusted R2 values were calculated using logistic regression analysis to assess the percent of variance in mode of failure categories that could be accounted for by each histological type and all comorbidities.
Results
Cohort descriptive statistics
Median time from primary TJA to histological sampling was 73 months, and median age of our cohort at time of revision TJA was 64 years. Knee arthroplasties accounted for 58.6% of cases, with hip and shoulder arthroplasties representing 39.4% and 2%, respectively (Table 1). Frequencies and percentages of histology types and modes of failure are presented in Table 1.
Histology
Odds ratios, beta regression coefficients
Only Infection was significantly associated with any histological category (p ⩽ 0.05). Patients with noted Infection had an odds ratio of 5.84 for Type II histology (Table 2). Anucleate protein debris, histiocytes, S. epidermidis, and synovitis were found to be significant, independent predictors of Hardware/Mechanical failure (Table 3). Enterococcus, granulation tissue, and necrosis were independent predictors of Septic failure. Synovitis was the only independent predictor of Loosening failure, and synovial cell hyperplasia was the only predictor of Other failure (Table 3). A more detailed breakdown of correlations between specific histological findings and specific modes of failure can be found in Table 4.
Independent histological findings—regression analysis of categorized modes of failure.

SE: standard error.
p ⩽ 0.05 = significant correlation between histologic finding and mode of failure. Beta = strength of correlation (Beta = 1 for two identical sets of values). Negative values indicate significant inverse relationship.
Only significant correlations (p ⩽ 0.05) shown.
Independent histological findings—regression analysis of independent modes of failure.

SE: standard error.
p ⩽ 0.05 = significant correlation between histologic and specific modes of failure. Beta = strength of correlation (Beta = 1 for two identical sets of values). Negative values indicate significant inverse relationship.
Only significant correlations (p ⩽ 0.05) shown.
Histology—ANOVA
Type II Histology accounted for 29.7% of variance in Periprosthetic Fractures and 13.0% in failure due to Infection. Type III Histology accounted for 6.8% of Malalignment failures. Type IV Histology accounted for 5.7% of failures with Deteriorated Bone or Pseudotumor (Table 5).
Histological categories and all comorbidities—ANOVA regression analysis of independent modes of failure.
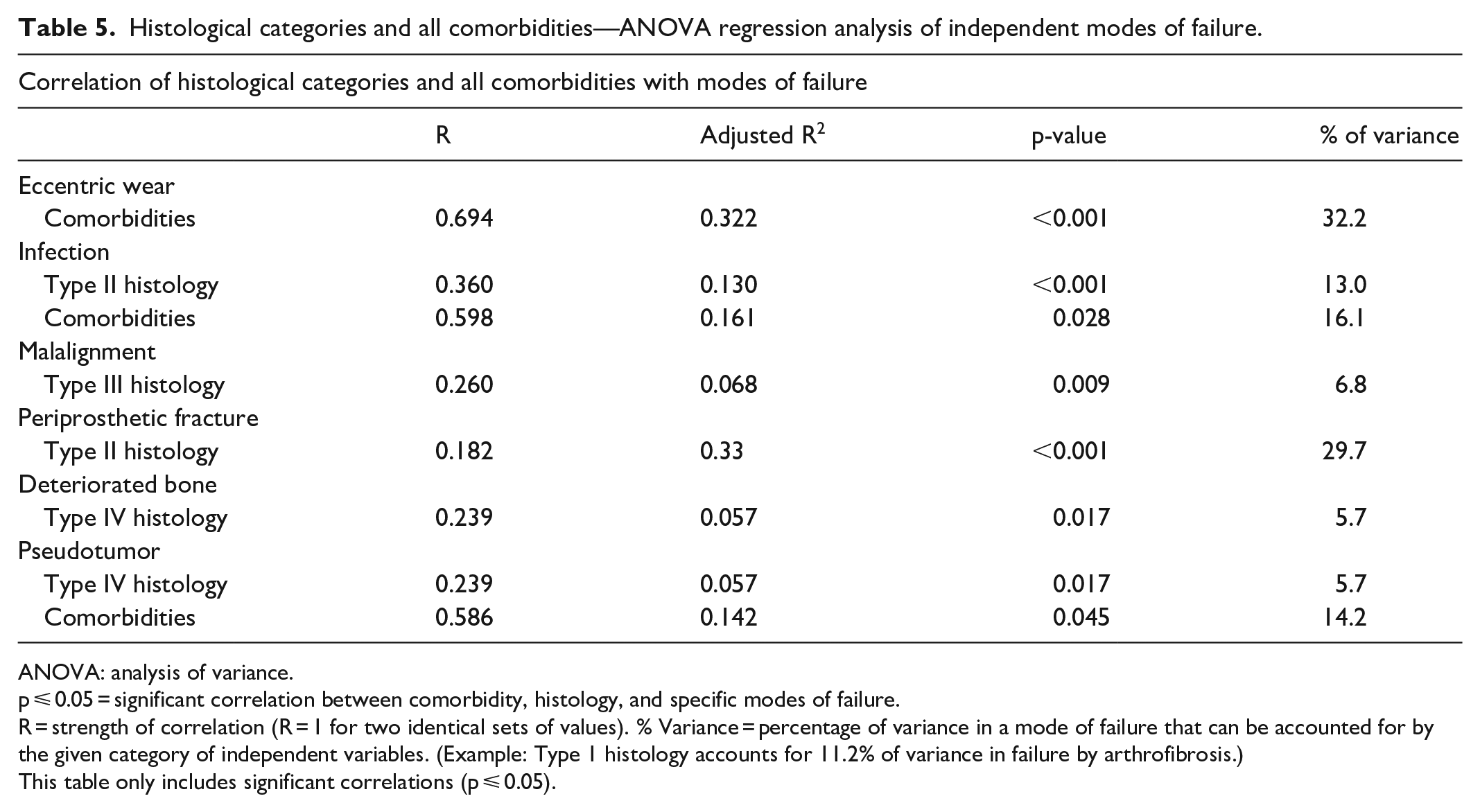
ANOVA: analysis of variance.
p ⩽ 0.05 = significant correlation between comorbidity, histology, and specific modes of failure.
R = strength of correlation (R = 1 for two identical sets of values). % Variance = percentage of variance in a mode of failure that can be accounted for by the given category of independent variables. (Example: Type 1 histology accounts for 11.2% of variance in failure by arthrofibrosis.)
This table only includes significant correlations (p ⩽ 0.05).
Comorbidities
Beta regression coefficients
Osteosarcoma was the only comorbidity that was an independent predictor of Hardware/Mechanical failure (Table 6). Crohn’s disease, deep venous thrombosis (DVT), lung disease, and rheumatoid arthritis were all significant, independent predictors of Septic failure (Table 6). A more detailed breakdown of correlations between specific comorbidities and specific modes of failure can be found in Table 7.
Independent comorbidities—regression analysis of categorized modes of failure.

SE: standard error.
p ⩽ 0.05 = significant correlation between specific comorbidities and mode of failure. Beta = strength of correlation (Beta = 1 for two identical sets of values). Negative values indicate significant inverse relationship.
This table only includes significant correlations (p ⩽ 0.05).
Independent comorbidities—regression analysis of independent modes of failure.

SE: standard error.
p ⩽ 0.05 = significant correlation between comorbidity and specific modes of failure. Beta = strength of correlation (Beta = 1 for two identical sets of values). Negative values indicate significant inverse relationship.
Only significant correlations (p ⩽ 0.05) shown.
ANOVA
Comorbidities accounted for 32.2% of failures attributed to Eccentric Wear, 16.1% of failures via Infection, and 14.2% with Pseudotumor (Table 4).
Discussion
When evaluating possible pathologic changes associated with revision arthroplasty, histological analysis is a promising next step.24,25 This study sought to determine whether periprosthetic histological findings during revision arthroplasty and/or comorbidities were associated with particular modes of prosthetic failure. Our results indicate that histological profiles do differ between mechanisms, and certain mechanisms were associated with particular pathological results and/or comorbidities.
Frequently, these associations seemed to fall into an intuitive pattern. Type I findings represent pathological analysis associated with foreign bodies—likely microscopic pieces from the aged prosthesis. Type I histology showed increased odds of failure by Instability and Loosening. The discovery that Instability and Loosening, as modes of failure, are associated with foreign body-based histology supports our current understanding of prosthetic breakdown. Interestingly, Type II histology (indicative of infection) was associated with decreased odds of failure by Instability and Loosening. While many of the connections found in our study were expected, some may warrant further investigation.
Synovium hyperplasia-pain/fracture
Our data showed a significant correlation between synovium hyperplasia and patient-reported knee pain. Pain is the most common indicator of implant failure. 26 When patients present with extra-articular pain post-implant, synovial hyperplasia should be considered within the differential diagnosis. 27 From a treatment perspective, synovial hyperplasia may be managed with arthroscopic debridement of the extensor mechanism of the hyperplastic synovium with modest pain-relieving effects.26–28
Synovitis with TJA loosening
The proliferation of synovial and interfacial membranes is caused by over-expression of transforming growth factor-α (TGF-α), transforming growth factor-β (TGF-β), and epidermal growth factor (EGF).29,30 All three of these cytokines have been found to stimulate bone resorption.29,30 The combination of bone resorption and profibrotic nature of these growth factors is thought to cause loosening in TJA. In TJA implants with polyethene components, particle-induced synovitis is characterized by frond-like appearance on magnetic resonance imaging (MRI).31,32 Interestingly, our results contradict those of a case series which reported five patients (six knees) with persistent synovitis following cobalt-chromium total knee arthroplasty (TKA), none of whom demonstrated component loosening intraoperatively or on MRI. 33 The mechanism of failure in these cases appeared to be metal hypersensitivity rather than loosening.
Osteosarcoma and hardware failure
Osteosarcoma was the only comorbidity that independently predicted Hardware and/or Mechanical failure in our study. When compared to conventional patients receiving an arthroplasty, patients with osteosarcoma are reported to have a significantly higher rate of failure (25%–92%) with the mechanisms of failure demonstrating heterogeneity—ranging from infection to mechanical failure and aseptic loosening. 34 While the literature describing this specific phenomenon is limited, cancer patients are generally thought to have a lower tolerance for hardware. 35
Bone necrosis and loosening in the joint
The results of our study showed that osteolysis (bone necrosis) was associated with loosening of the joint. This aligns with previous findings in the literature related to component loosening in arthroplasties.36,37
Lymphocytic infiltrate and arthroplasty malalignment
Although lymphocyte-driven hypersensitivity reactions have been implicated as a potential mechanism for arthroplasty failure, histopathologic findings of nickel lymphocyte transformation testing (LTT) have not confirmed TJA failure due to immune reaction. 38
Our data, however, showed a significant correlation of lymphocytic infiltrate with eccentric failure. As previously mentioned, microscopic analysis in one of our cases revealed a proliferative eosinophilic reaction to an alternate bearing articulation total hip replacement consistent with a lymphocyte-driven hypersensitivity reaction. Further research is needed to evaluate the potential confounders related to lymphocytic infiltrate that may drive a lymphocyte reaction in the area adjacent to the prosthesis.16,38
Staphylococcus epidermidis and periprosthetic fractures
Our data indicated that S. epidermidis (coagulase-negative staphylococcus) was related to a periprosthetic fracture. Current literature has yet to elucidate the contribution of infection to implant loosening and its role in subsequent periprosthetic fracture. 39 However, microbiology literature suggests that osteoclastic lineage is favored over the osteoblast lineage via the RANK-RANKL mechanism during active infection with acute phase reactants and cytokines in the localized area of the prosthesis. 40 This promotes bone resorption and may destabilize the bone matrices. 40
Limitations and future implications
One challenge this study faced was limited power. With only 99 total participants, our data set could not be expected to definitively parse out the nuances between every group, yet we were still able to achieve significance for many correlations. While common modes of failure were reasonably represented in our study, a larger sample size is needed to assess those that occur less frequently. Related to modes of failure, the Other category is a heterogeneous catch all group established by Calliess et al. This classification is broad as in our study it included hemi to total conversion to symptomatic findings that resulted in revision surgery. Given how broad these classifications are within this group, there is heterogeneity that makes it difficult to analyze how this category influences histologic findings. Future research should focus on adding to the study by Calliess et al. to delineate this broad catch-all group further into more specific categories. Another limitation is in the fact that we did not conduct a power analysis given the variables we selected to analyze. This type of study requires a power analysis and a larger patient population in the future.
The separations between groups of both failure and pathological analysis are also imperfect. The modes of failure were grouped by similar mechanism, but similar does not mean exactly the same and overlap between the modes of failure groups was inevitable. The same could be said for the histological analysis. By using the histological grouping paradigm laid out by Morawietz et al., 10 we were able to organize the histological types. These were not exact matches. The pathology reports could include identifiers for multiple groups in a single patient, and each patient may qualify for multiple types of histological findings. Additionally, a cohort study of both revision and non-revision prostheses may allow for more direct comparisons.
Most of our findings related to histologic changes that occur between implant and bone interface in patients requiring arthroplasty revision support data seen within the literature. However, our findings regarding S. epidermidis on histology and its association with periprosthetic fracture was a finding that has not been well studied in vivo and the mechanism of infection and periprosthetic fractures still remains theoretical. Future studies evaluating periprosthetic–bone interface interventions (i.e. vancomycin-tobramycin impregnated cement) and the incidence of periprosthetic fractures in patients with adjacent bacterial colonization could provide meaningful data to a contentious intervention and devastating complication in arthroplasty.
Conclusion
As our patients live longer and are more active post-arthroplasty, it is increasingly necessary to understand the long-term implications of prostheses at a histological level. Many of our findings support the existing literature on periprosthetic tissue analysis in revision TJA and further validate potential pathological mechanisms of implant failure. The results of our study also add to domains in which the literature is limited, providing insights for future investigations by addressing the predictive value of pathological analysis and comorbidities in patients requiring TJA revisions. These data may also improve surgeon understanding of the mechanisms that contribute to implant failure and revision surgery.
Footnotes
Acknowledgements
The authors thank Nancy Swinford, CCRC, Clinical Research Coordinator, Texas Tech University Health Sciences Center for assisting with IRB submission.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from Lubbock/Odessa Institutional Review Board (#L12-048).
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects before the study.
