Abstract
Vascularized composite allotransplantation represents the final level of the reconstructive ladder, offering treatment options for severe tissue loss and functional deficiencies. Vascularized composite allotransplantation is particularly susceptible to ischemia–reperfusion injury and requires preservation techniques when subjected to extended storage times prior to transplantation. While static cold storage functions to reduce ischemic damage and is widely employed in clinical settings, there exists no consensus on the ideal preservation solution for vascularized composite allotransplantation. This review aims to highlight current clinical and experimental advances in preservation solution development and their critical role in attenuating ischemia–reperfusion injury in the context of vascularized composite allotransplantation.
Introduction
Vascularized composite allotransplantation (VCA) is the simultaneous transplant of units composed of several different tissues. The field of VCA has seen remarkable growth since 1998, when the first successful hand 1 and laryngeal transplantation 2 were performed. Advances in immunologic control, which aim to prevent acute and chronic allorejection, 3 have been responsible for the progress in the field of VCA, and have facilitated successful expansion into lower extremity, 4 facial, 5 penile, 6 and uterine transplantation. 7 Despite these advancements in the field of VCA, acute rejection within the first year continues to occur in over 80% of all upper extremity and face transplants, thus highlighting the infancy of the field and the importance of continued development to reduce these effects and improve graft functionality to facilitate widespread use of VCA. 8
VCA is particularly susceptible to ischemia–reperfusion injury (IRI) since it involves the transplantation of highly metabolically active tissues. This is known to be one of the primary determinants of tissue survival in VCA. IRI triggers an inflammatory cascade which ultimately results in tissue dysfunction, poor graft function, and survival. There is no effective treatment for IRI, and thus efforts center around prevention and minimization by reducing ischemia time and the metabolic activity of tissue during storage. Static cold storage (SCS) and limiting ischemia time to a maximum of 4–6 h are two such ways in which this is undertaken.9–12 While many different preservation solutions exist, each with theoretical advantages, there is no gold standard preservation solution for use in experimental or clinical studies of VCA. 13 This review aims to highlight current clinical and experimental advances in preservation solution development and their critical role in attenuating IRI.
Cellular and molecular events during IRI
IRI is considered the first insult in VCA, with a complex molecular and cellular pathophysiology. Mechanisms of IRI in skin, 14 skeletal muscle, 15 uterus, 16 and penis 17 have been previously described with differing injury responses related to unique properties of the individual tissues. Among the various tissue types contained in an allograft, muscle tissue is the most metabolically active, and is thus highly susceptibility to IRI. 11 Events during ischemic and reperfusion injuries are summarized in Figures 1 and 2, respectively.
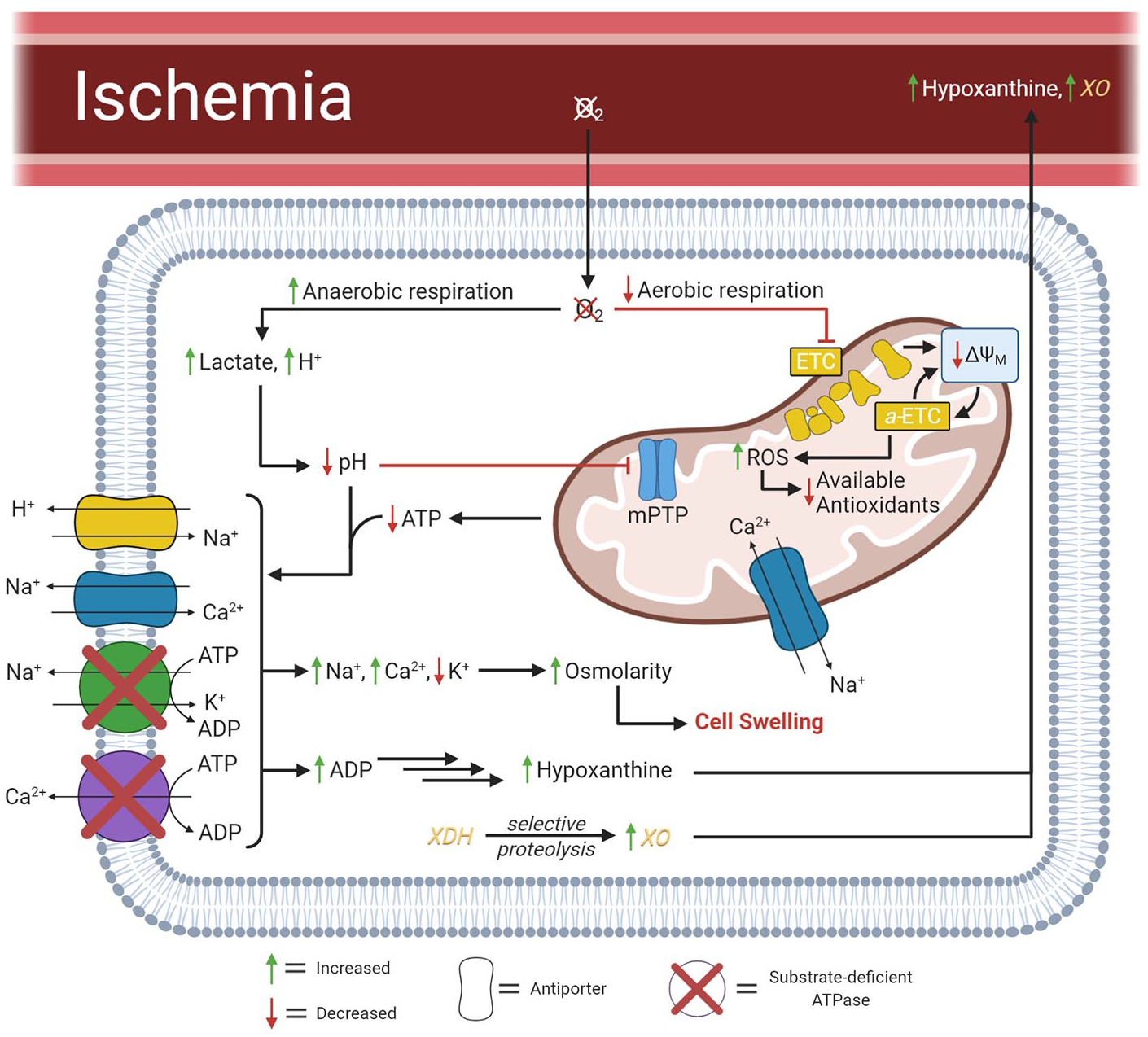
Pathophysiological events of ischemic injury.

Pathophysiological events of reperfusion injury.
Ischemic injury
Ischemia indicates a loss of oxygenated blood supply resulting in a necessary transition from aerobic to anaerobic metabolism. 18 Due to shunting of pyruvate to the homolactic fermentation pathway, an excess of lactate and protons are generated within the cytoplasm. 18 In muscle tissue, it has been shown that lactate production occurs continuously for up to 6 h, after which exhaustive depletion of adenosine triphosphate (ATP) results in irreversible tissue death.18,19 The limited ATP produced from lactic acid fermentation is primarily used for maintaining ion concentration gradients and membrane potential.15,18 Under normal aerobic conditions, adenosine diphosphate (ADP) is reused for ATP production through oxidative phosphorylation. However, during prolonged ischemia, ADP accumulation results in an equilibrium shift toward synthesis of hypoxanthine, a substrate that contributes to reactive oxygen species (ROS) production during reperfusion. 20 Low cytosolic ATP eventually results in altered ion transporter function, leading to increased intracellular Na+, Ca2+, and H+ concentrations.21–23 The high osmolarity buildup caused by intracellular Na+ and Ca2+ results in an influx of water that causes cell swelling. 18 In ischemic tissues, high intracellular Ca2+ also contributes to the irreversible conversion of xanthine dehydrogenase (XDH) to xanthine oxidase (XO).24,25 XO is considered to be critical in the generation of ROS upon reperfusion, using hypoxanthine and xanthine as a substrate. 24 In the event that reperfusion does not occur, accumulation of Ca2+ can cause degradation of important cellular enzymes, such as phospholipases, lysozymes, and proteases, which contribute to cell death. 18
In the mitochondria, inhibition of the electron transport chain (ETC) caused by anoxia leads to a reduction in mitochondrial membrane potential (ΔΨM).26,27 Through a positive feedback loop, the lowered ΔΨM further inhibits the Complex III of the ETC, producing superoxide (O2• −) molecules and other derived ROS.18,24,28,29 The O2• − molecules are further oxidized to hydrogen peroxide (H2O2) by superoxide dismutase or spontaneous disproportionation. 30 These ROS are degraded by non-enzymatic antioxidants, such as glutathione, depleting available antioxidants, and making cells more vulnerable to oxidative stress from ensuing reperfusion.18,31
The mitochondrial permeability transition pore (mPTP) is hypothesized to transiently open during ischemia to protect cells by reducing cytosolic Ca2+32 Opening of the mPTP further dissipates ΔΨM, impairing ATP production. The mPTP is regulated by many different signals, including pH, Ca2+, ROS, and ATP, however, the specific mechanisms by which these affect the mPTP remain unknown.18,33 Although buildup of ROS and Ca2+ in mitochondria and decreased available ATP prime the mPTP for opening, low cellular pH prevents the pore from fully opening.34,35
Reperfusion injury
Reintroduction of O2 at the onset of reperfusion is the primary source of ROS. 18 In the presence of O2, XO catalyzes the conversion of hypoxanthine to xanthine and xanthine to urate in the extracellular environment and on the cell surfaces of endothelial and skeletal muscle cells. 21 The ROS by-products of this reaction further convert nearby XDH to XO. 25 In this way, ROS represent a feed-forward mechanism by which XDH–XO conversion occurs and accounts for further ROS generation. 24 Urate, a type of damage-associated molecular pattern (DAMP), induces a sterile inflammatory response through binding to intracellular protein complexes known as inflammasomes.36,37 Inflammasomes mediate cytokine production and in the context of IRI can trigger a cytokine storm.15,36 In addition, DAMPs are recognized by toll-like receptors, a type of pattern recognition receptor on cell surfaces of innate immune cells. 11 Through this mechanism, and by chemokine signals provided by resident macrophages and endothelial cells, neutrophils are recruited and activated at the site of ischemic tissue. 38 Activated neutrophils are responsible for a significant burst of ROS and are implicated in the development of the no-reflow phenomenon, in which capillaries fail to reperfuse due to plugging by leukocytes. 39 In this way, tissues serviced by small blood vessels do not reperfuse and consequently do not receive O2, making them more susceptible to cell death. Furthermore, DAMP-mediated activation of antigen-presenting cells results in an increase in effector T-cells. 40 Thus, reperfusion following prolonged ischemia enhances the adaptive immune response, contributing to both acute and chronic rejection of the VCA graft.15,36,41
At the time of reperfusion, restoration of blood supply washes out accumulated H+ in the intravascular space resulting in normalization of the extracellular pH. 18 The removal of extracellular ions creates an osmotic gradient, with high intracellular osmolarity caused by the Na+ and Ca2+ that accumulated during ischemia. Consequently, further cell swelling induced by hypoosmotic stress leads to water influx and irreversible damage to the plasma membrane, organelle breakdown, and cell membrane rupture. 42
While the return of oxygenated blood results in a transition from anaerobic back to aerobic respiration pathways, mitochondria damaged during ischemia continue to produce ROS due to aberrant oxidative phosphorylation caused by the altered-ETC (a-ETC). The ROS cannot be eliminated because the non-enzymatic antioxidant defenses are greatly reduced or entirely depleted during ischemic generation of ROS. 18 Although the mitochondrion is not the predominant source of ROS, these ROS contribute to secondary tissue damage and inflammation.18,43
After normalization of intracellular pH at the onset of reperfusion, the mPTP opens and allows cytosolic Ca2+ to enter the mitochondria. 18 This results in uncoupling of oxidative phosphorylation and further reduction of ΔΨM. 44 In addition, water influx into the mitochondria, caused by the established osmolar gap, leads to mitochondrial swelling and rupture. 44 In this way, opening of the mPTP in a single mitochondria places additional strain on remaining mitochondria to meet the cellular energy demand. 44 ROS and cytosolic Ca2+ continue to increase due to other enzymatic activity, resulting in a vicious cycle in which total cellular dysfunction occurs after mitochondrial failure.18,33 As such, opening of the mPTP represents an important step in tissue death following IRI. 44
Components of preservation solutions
Preservation solutions are formulated based on three broad principles: (1) providing a physical and biochemical environment that maintains viability of cells during ischemia, (2) minimizing damage associated with reperfusion injury, and (3) providing a temperature for preservation at which the solution does not freeze or coagulate.45,46 Preservation solutions are commonly differentiated based on their Na+/K+ ratio, wherein high Na+ corresponds to an extracellular-type solution (ETS) and high K+ corresponds to an intracellular-type solution (ITS). An ITS maintains intracellular ion concentration by creating a temporary cation equilibrium that compensates for the lack of active transport during ischemia. 47 Euro-Collins solution (ECS) is one such ITS that was historically used widely in solid organ transplantation (SOT).45,48 High K+ ITS perfusates were found to induce vasoconstriction and enhance the no-reflow phenomenon during IRI, and have thus fallen out of favor for use in some SOT. 49 ETS was developed to reduce vascular endothelial cell damage and creates a postischemic environment that enhances recovery of the Na+/K+–ATPase in ischemic cells.45,48 ETS are most commonly used for tissues that are more tolerant to ischemic damage, wherein stabilization of cation flux is not as critical. 45 While ETS and ITS each have strengths and limitations, no conclusion on superiority of one over the other can be made as the protective effects are specific to tissue/organ type.45,48 This trend is observed and particularly important in VCA. The susceptibility to IRI is dependent on the specific tissue composition of the whole graft and also of each component within the VCA. 11
Other ions
Calcium in preservation solutions has been shown to have varying effects on organ injury and survival based on the length of preservation time and type of tissue. 50 More specifically, Ca2+ addition to University of Wisconsin solution (UWS) showed improved preservation of rat liver that was preserved for 24 h, but no improved protection in liver stored for 48 h. 50 Magnesium demonstrates a protective effect on endothelial cells through preservation of membrane hyperpolarization and reversal of K+-induced membrane depolarization in smooth muscles.51,52 Anions, including Cl−, PO43−, HCO3−, and SO42−, act as counterions and buffers. Buffers are crucial in maintaining physiological pH and thereby preserving cell membrane integrity and protein conformations.45,48
Saccharides
Saccharides are used in some preservations solutions to act as osmotic barriers, thereby reducing cell edema during ischemia. 53 Glucose was initially used in early preservation solutions; however, slow penetration of glucose into the cell when added in large quantities exacerbates acidosis and is therefore no longer added to preservation solutions. 45 Sucrose, trehalose, raffinose, lactobionate, and mannitol are used as substitutes to glucose as they have all proven to be more effective impermeant agents.45,54 Furthermore, raffinose and lactobionate are metabolically inert, thereby reducing contribution to lactate buildup during ischemia. Mannitol also acts as an indirect antioxidant by upregulating levels of catalase in tissue under oxidative stress.55,56 Catalase catalyzes the breakdown of H2O2 to H2O and O2. 56 In general, antioxidants reduce oxidative stress in ischemic tissue and are key contributors in attenuating IRI.
Amino acids and derivatives
Amino acids are important metabolic substrates during glucose deprivation and are thus incorporated in some preservation solutions to serve as an alternative energy source in graft tissue. 53 Histidine is a commonly employed buffer in medicine and is also added to preservation solutions for this purpose. N-acetyl-L-histidine is used in place of histidine in newer perfusates as histidine was found to produce cell injury through iron-dependent ROS production.57,58 Aspartate, α-ketoglutarate, and glutamate are important metabolites in the citric acid cycle. 48 Arginine has been shown to improve endothelial function of ischemic grafts through nitric oxide-mediated protection against IRI.58,59 Experimental data have shown that glycine and alanine provide minor protection against hypoxic injury; however, the mechanism of their action remains unknown.48,60 Partially oxidized tryptophan metabolites, such as serotonin and melatonin, have also been shown to have ROS scavenging capacity. 61
Colloids
Colloids act as extracellular oncotic agents with the primary function of reducing cell swelling. The size of colloids used in preservation solutions is optimized to maximize surface area for H2O adsorption and minimize clearance and entry into the lymphatic system. 62 Early preservation solutions derived from whole blood used human serum albumin (HSA) as the primary oncotic agent; however, with the clinical success of UWS, pentafraction replaced HSA as the primary colloid of choice. 63 Pentafraction is a solution of biodegradable hydroxyethyl starch macromolecules with molecular weights between 10 and 100 kDa. Alternative colloids, such as dextran 40, a 40 kDa dextran solution, gluconate, and polyethylene glycol-20/35, a 20 or 35 kDa polyethylene glycol solution, have been shown to successfully substitute pentafraction in experimental models and clinical settings.54,63,64 In addition, dextran 40 and gluconate are less expensive to prepare and are thus incorporated in newer perfusates. 63
Other additives
Heparin is a linear highly sulfated glycosaminoglycan which serves as an anticoagulant in preservation solutions. Reduced glutathione is a sulfur-based endogenously found non-enzymatic antioxidant. Allopurinol is a hypoxanthine structural isomer that competitively inhibits binding of hypoxanthine and xanthine to XO, thereby reducing ROS generation. 18 Deferoxamine and LK-614 are iron chelators that also reduce the generation of ROS as caused by the Fenton reaction. 65 In the Fenton reaction, free Fe2+ catalyzes the breakdown of H2O2 to hydroxyl radicals, which greatly increases oxidative stress of ischemic tissue. 66 Deferoxamine and LK-614 are used in combination as deferoxamine has poor membrane permeability compared with LK-614, but is a stronger iron chelator. 65 Finally, adenosine is added in preservation solutions to serve as a precursor for ATP synthesis.
pH and osmolarity
Preservation solutions are maintained at or near physiological pH with added buffers to compensate for acidosis during ischemia. 15 Osmolarity of perfusates vary based on the oncotic agents present. Generally, newer preservation solutions maintain a higher osmolarity than blood to prevent cellular edema in ischemic tissue.15,18
History of preservation solutions in SOT
Significant strides have been made in the research of preservation solutions as they relate to SOT. While these organs have very different needs than VCA, much can be learned about techniques employed in SOT. Historically, solid organs were preserved at room temperature by dynamic perfusion with oxygenated blood; 67 however, this method was complex and was not economically feasible, which limited its availability and transportability. 46 In 1969, Collins et al. 47 developed SCS when they described successful preservation of canine kidneys using a hypothermic saline solution which proved to be far simpler and more cost-efficient for organ transportation. Today, SCS is a commonly employed preservation technique prior to transplantation. 68 The primary mode of action of SCS is to induce hypothermia of graft tissue to reduce cellular demand. As much as two-fold reductions in metabolic activity per 10°C reduction in temperature below 37°C have been reported. 68 In general, lower organ temperatures maintain reserves of ATP and its derivatives more effectively. 69 Notably, temperatures below 2°C significantly increase the risk of deleterious cold injury which incites protein denaturation and is thus detrimental to graft survival. 69 In contrast, temperatures above 8°C greatly increase metabolic activity, which can lead to a faster time to onset of irreversible hypoxic injury. 69 Thus, the ideal temperature range used for SCS is between 2°C and 8°C, and in most cases chosen to be 4°C.
Although there are a multitude of preservation solutions described and there is regional variation in protocols for organ transplant, common practices have been established in transplantation for organs, such as kidneys, liver, and lung.
In kidney transplant, UWS, histidine–tryptophan–ketoglutarate solution (HTKS), and Celsior solution have all demonstrated significantly lower rates of delayed graft function when compared with ECS.53,70,71 Randomized clinical trials comparing these three solutions have demonstrated equivalent results. 72 In the setting of kidney transplantation, these solutions are often administered through hypothermic machine perfusion rather than SCS alone.
The advent of UWS in 1987 also resulted in tremendous benefit to liver transplant outcomes. 73 This solution has been adopted as a gold-standard cold storage solution in liver transplant as it has resulted in improved cold ischemia tolerance.73,74
In lung transplant, experimental and clinical models have demonstrated improved transplant outcomes with the use of Perfadex solution as compared with ECS.75–77 Lung tissue has a unique property in that inflated lungs can continue to maintain aerobic metabolism during cold ischemia. 76 This means that the primary obstacle in successful lung transplant is not only IRI as is the case in the setting of most other organ transplant, and also a challenge of attaining adequate PaO2 in donor patients to meet the criteria for lung transplant. 78 In this patient group, gas exchange is often impeded by clinical variables, such as excessive crystalloid administration during organ harvest, reduced oncotic pressure stemming from hemodilution, and the development of pulmonary edema.78,79 To combat this obstacle, ex-vivo lung perfusion with Steen solution, a hyperoncotic serum that is designed to dehydrate the lungs, has been implemented. In the setting of lung transplant, edematous lungs that would otherwise be unsuitable for transplantation are effectively rehabilitated. This technique requires that the lungs be immersed in Steen solution while topical extracorporeal membrane oxygenation is undertaken around the lungs. Ex-vivo lung perfusion has been effectively implemented in a clinical setting to facilitate transplantation of donor lungs that would otherwise have been deemed unsuitable for transplant. 79
Preservation solutions used in VCA
To date, there have been no preservation solutions that have been specifically designed or studied for use in VCA. The preservation solutions that have been studied experimentally and employed clinically for use in VCA were initially designed for SOT. A detailed breakdown of the composition of the most commonly used preservation solutions in SOT and those used in VCA can be found in Table 1.
Composition of preservation solutions used in animal models and clinical practice for VCA.

VCA: vascularized composite allotransplantation; E: extracellular; EC: Euro-Collins; ETK: extracellular-trehalose-Kyoto; HTK: histidine–tryptophan–ketoglutarate; HTK-N: histidine–tryptophan–ketoglutarate-new; I: intracellular; IGL-1: Institut Georges Lopez-1; KH: Krebs–Henseleit; PEG-20/35: polyethylene glycol-20/35; UW: University of Wisconsin.
All units are in mmol/L except Dextran 40, pentafraction, and PEG-20/35, which are in g/L.
× 10−3.
Animal models
Over the past four decades, innovations in VCA animal models have helped expand the geographic boundaries of the VCA donor pool, reduce occurrence of transplant rejection, and improve long-term graft functionality. Many early IRI studies in animal models utilized replantation and syngeneic transplantation to simulate allotransplantation without the need for immunosuppression. These studies provided detailed procedures of surgical technique, useful histopathology and immunohistochemistry grading scales, and upper limits on critical ischemia times; the last of which are clinically relevant for advising policies on maximum ischemia times today.80–84 However, recently published studies on mechanistic progression of IRI provide new insights on the enhanced IRI effect observed in allografts versus autografts, isografts, and syngrafts.85–88 Thus, studies using autograft, isograft, and syngraft animal models do not provide a complete picture of IRI in VCA and were excluded from this review. Studies focused on developing and evaluating the feasibility of new human VCA analogs primarily contribute to advances in surgical technique and have minimal ischemia time.89,90 As such, these were also excluded from this review. Given these constraints, this section of the review focused on advances in VCA research described in studies using allograft VCA animal models to assess the effect of preservation solution and ischemia time on graft outcome. The specific search criteria and PRISMA flow diagram used for this section are provided in Table 2 and Figure 3, respectively.
Search strategy used for “Animal models” section. The search was run using Embase, Medline, and Ovid, and was performed on 1 August 2020. MeSH terms are marked with the syntax [MeSH].


The PRISMA flow diagram for “Animal models” section detailing the database searches, the number of abstracts screened, and the full texts retrieved.
In 1994, Strome et al. 91 published the first laryngeal allotransplant model that survived an ischemic interval longer than 3 h subject to SCS. Here, the efficacy of heparinized saline solution (HSS) and UWS on mitigating IRI were compared using histopathology after 24-h sacrifice. 91 The authors demonstrated superior preservation qualities of UWS versus HSS, as most transplanted larynxes preserved for 20 h in UWS remained fully histologically viable and none of the 6-h specimen preserved in HSS showed signs of viability. 91 In 2005, Cendales et al. 92 compared ECS and UWS for SCS preservation of an osteomyocutaneous radial forearm flap transplanted orthotopically in cynomolgus monkeys. The grafts were subject to 3 h of ischemia, and all but two animals recovered fully and had full active range of motion of upper extremities. 92 Histopathological markers of IRI were not noted, and no difference in preservation quality between ECS and UWS was observed. 92 The conclusion from this study was that the 3-h ischemia time was too short to assess the superiority of ECS or UWS. Pradka et al. 93 studied transplantation of vascularized rat epigastric flaps. Flaps were flushed with HSS and exposed to either 1 or 3 h of room temperature ischemia. 93 Higher rejection scores were noted in the 3 versus 1-h ischemia group with histopathological assessment. 93 The authors noted that skin histopathological scores did not convey the status of other tissues of the composite allograft or predict the graft’s eventual outcome. 93 They also found that muscle and fat were found to be highly susceptible to IRI. 93
There is a paucity of studies assessing IRI in VCA that utilize allograft animal models. In contrast, studies using autograft, isograft, and syngraft animal models have shown the usefulness of certain preservation solutions in attenuating IRI, such as Institut Georgez Lopez-1 (IGL-1), Perfadex, histidine-tryptophan-ketoglutarate-new, TiProtec, extracellular-trehalose-Kyoto, Krebs–Henseleit solutions.10,49,65,81 To truly validate the utility of these solutions in preclinical studies, further studies should be conducted using allograft animal models.
Applications in clinical practice
While VCA has been successfully performed in the clinical setting, little attention has been paid to preservation solutions used in VCA. The International Registry on Hand and Composite Tissue Allotransplantation has reported over 100 cases of upper extremity and face allotransplants that have been conducted up to May 2017. 84 UWS, IGL-1, HTKS, Celsior solution, Scot solution, and heparinized saline, have all been reported as perfusates used for these surgeries, with UWS used in 70% of cases. 84 Many case reports demonstrate recognition of the importance of minimizing cold ischemia through a described focus on the transport of donor and recipient to the same site prior to transplant as the primary strategy by which to reduce cold ischemic time, but do not provide information on why a particular preservation solution was chosen. The following review focuses on articles published after May 2017. The specific search criteria and PRISMA flow diagram used for this section are provided in Table 3 and Figure 4, respectively.
Search strategy used for “Applications in clinical practice” section. The search was run using Embase, Medline, and Ovid, and was performed on 16 September 2020.


The PRISMA flow diagram for “Applications in clinical practice” section detailing the database searches, the number of abstracts screened, and the full texts retrieved.
Two face transplant articles were identified with successful outcomes. Both reports outlined that UWS was the preservation solution of choice, but provided no rationale for their selection.94,95 Similarly, in the hand transplant literature, two papers were identified wherein preservation solution was described. One hand transplant technique paper from the University of Utah with a series of two bilateral and five unilateral hand transplants describes an SCS preservation technique with the use of three liters of HTKS. 96 This article did not provide a rationale for the use of HTKS over other solutions. 96 Another case report describes the first bilateral hand transplant in the Netherlands where UWS at 4°C was used. 97 Again, this technique article did not provide a rationale for the selection of UWS over other preservation solutions. 97
Although the benefits of preservation solutions for decreasing IRI are widely accepted, there is a lack of consensus on the optimal preservation solution for use in VCA. Our review finds that UWS is most commonly used in the clinical setting; however, there is currently no large study comparing the effect of different preservation solutions on the short- or long-term outcomes of VCA. This provides an opportunity for further research to determine the ideal preservation solution for use in clinical VCA, with the potential to increase the critical ischemia time and expand the donor pool greatly.
Future strategies
Although SCS is commonly used for VCA graft preservation in clinical settings, a number of limitations exist, including: (1) extensive tissue damage after prolonged hypothermic preservation, (2) difficulty in assessing graft viability prior to transplantation, and (3) limited preservation time, wherein most VCA grafts are limited to less than 6 h of SCS.46,98,99 These inherent limitations of SCS have encouraged research into new technologies that would allow for better graft preservation. Emerging techniques in VCA preservation can be broadly classified into two categories: (1) machine perfusion and (2) cryopreservation.
Machine perfusion
The goal of machine perfusion is to preserve organ viability through provision of oxygen and nutrients, and removal of metabolic by-products. 100 In this way, grafts are preserved extracorporeally for longer periods of time, thereby greatly increasing their geographic accessibility. 101 Grafts can be preserved under a variety of a perfusion temperatures; these include hypothermic (0°C–12°C), mid-thermic (13°C–24°C), sub-normothermic (25°C–34°C), and normothermic (35°C–38°C) conditions.102,103 Studies on both small and large animal VCA models have demonstrated successful preservation of graft tissue for up to 24 h using machine perfusion.104–106 In 2017, Werner et al. 107 applied sub-normothermic machine perfusion to preserve human limbs for 24 h with a plasma-based perfusate. The grafts remained viable after 24 h of preservation, showing no signs of myocyte injury as well as continued response to neuromuscular electrostimulation. 107 Although recent studies on viability of machine perfusion in preserving graft tissue demonstrate extended preservation times, limitations include: (1) few studies utilizing allografts, (2) no studies investigating long-term follow-up, and (3) a lack of consensus on ideal temperature or perfusate for use in a clinical settings.99,100
Cryopreservation
Cryopreservation aims to slow deterioration of graft tissue by reducing the rate of metabolism. This requires freezing of the graft to temperatures below 0°C and offers the possibility of storage for many weeks. 108 Several techniques of cryopreservation exist, wherein different temperatures are used for biological tissue preservation.99,109 These are broadly classified as: (1) sub-zero and high sub-zero temperatures suitable for supercooling (< 0°C to −30°C), (2) temperatures suitable for directional freezing of water (−80°C to −196°C), and (3) temperatures suitable for vitrification of water (< −137°C).99,110 Each technique requires meticulous control on the rate of cooling and addition of cryoprotectants to prevent formation of intracellular ice crystals, which is detrimental to tissue viability. 110 Studies on cryopreservation of single-cell systems, blood vessels, cutaneous tissue, bone, and nervous tissue have been previously described. 111 In 2008, Rinker et al. 112 successfully cryopreserved rat epigastric flaps at −140°C for 2 weeks followed by isotransplantation. Flaps remained viable for up to 60 days after transplantation, retaining normal pigmentation and hair growth, and showing no histological signs of inflammation or necrosis. 112 In 2017, Friedman and Gur 113 were the first to apply directional freezing and vitrification for preserving a syngeneic heterotopic rat hindlimb for 7 days. The authors observed histologically viable myocytes, blood vessels, and skin layers three days after transplantation. 113 Currently, there exist no studies measuring the effects of cryopreservation on human VCA grafts. Although cryopreservation provides the opportunity for long-term VCA graft storage, the complex and variable response of different tissues to freezing, thawing, and cryoprotectants render a unified protocol difficult to obtain today. 99
Conclusion
Experience and focused research in the field of SOT has facilitated an understanding of the pathophysiology of cold IRI.53,72 This understanding has in-turn facilitated advances in clinical outcomes stemming from technical modifications, including adaptations to preservation solutions that aim to mitigate the deleterious effects of IRI.53,72 VCA often includes tissue that is highly metabolically active and thus particularly susceptible to the effects of reperfusion injury. Despite this, preservation solutions in the setting of VCA have been largely overlooked without a consensus on the optimal solution to improve outcomes and accommodate the specific needs of these tissues. 114 This can be attributed to the complexity of IRI in composite tissue and the multiple mechanistic pathways that must be targeted. In addition, given the infancy of VCA in the clinical setting, large-scale clinical trials that assess the efficacy of various preservation solutions are yet to be conducted.
Given the current theoretical understanding of IRI pathophysiology, we hypothesize that an ideal preservation solution for attenuation of IRI in VCA will allow for protection of the microvasculature and reduction of ROS generation. Dextran 40 has been used effectively as a colloid agent to reduce cellular edema and protect the endothelial cells following reperfusion. In addition, deferoxamine, LK-614, and allopurinol are important for reducing production of iron-mediated and XO-mediated ROS. Finally, glutathione is an important antioxidant to reduce the cellular ROS load. Moreover, the ideal preservation solution depends on the type of VCA and its unique composition. Focused research is required to facilitate thoughtful selection of preservation solutions with the potential to improve clinical outcomes in VCA.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
