Abstract
Background:
Fluid overload is associated with poor outcomes, but mitigating its occurrence poses significant challenges.
Objective:
This study sought to assess the impact of hidden fluid volume on fluid overload.
Methods:
This study was a multi-center, retrospective evaluation of adults admitted to a medical or surgical intensive care unit for at least 72 h. Patients were divided into tertiles (low, moderate, and high) based on the hidden fluid volume received. Hidden fluids were defined as intravenous medications, line flushes, blood products, and enteral nutrition. The primary outcome was the incidence of fluid overload at intensive care unit (day 3). Secondary outcomes included mechanical-ventilation free days and association of hidden fluid volume with fluid overload, length of stay, and mortality.
Results:
A total of 219 (73 per tertile) were included, with hidden fluid volume comprising ⩽2500, 2501–4400, and >4400 mL in the low, moderate, and high tertiles, respectively. Incidence of fluid overload was significantly different across groups (low: 3%, moderate: 14%, high: 25%; p < 0.001). No difference existed in mechanical-ventilation free days or in-hospital mortality across tertiles. In binary logistic regression, hidden fluid volume received at 3 days was independently associated with fluid overload (odds ratio = 1.40, 95% confidence interval = 1.15–1.70).
Conclusion:
The volume of hidden fluid volume administered by intensive care unit day 3 independently predicted development of fluid overload.
Introduction
Fluid stewardship provides the four rights construct of pharmacotherapy management to promote optimal clinical decision-making regarding fluid therapy. 1 Increasing evidence suggests potential toxicities associated with use and misuse of fluids, including multi-organ damage, electrolyte derangements, new onset atrial fibrillation, coagulation abnormalities, increased duration of hospital stay, and impaired mobility, hospital disposition, and mortality.2,3
“Hidden” fluids, such as intravascular medications, line flushes, and enteral nutrition, contribute to total fluid administration. 4 However, the association of these fluids with the presence of fluid overload (FO) is unclear. 4 Even less clear is the clinical impact of these hidden fluids on patient outcomes. Furthermore, fluid stewardship has become a necessary concept secondary to the 2017 national shortage affecting nearly all intravenous fluids (IVFs) that prompted health systems to re-evaluate the management of IVF use. 5 The purpose of this study is to describe the relationship of hidden fluid volume (HFV) received with the development of FO and to identify sources of fluid input that could be reduced to decrease the incidence of FO. We hypothesize that cumulative HFV at 3 days is independently associated with the development of FO.
Methods
This retrospective cohort evaluation was conducted across three centers between January 2015 and December 2017. Patients were eligible for inclusion if they were at least 18 years of age and admitted to a medical or surgical intensive care unit (ICU). Given the retrospective nature of this research, the sample size was determined by the patients meeting inclusion criteria in the time period for which data were available. Patients were excluded if they transferred from an outside hospital, had end stage renal disease, received total parenteral nutrition, received a diagnosis necessitating maintenance IVF, stayed in the ICU less than 72 h, had do not intubate/do not resuscitate orders on admission, or were pregnant. The primary outcome measure was the percentage of patients with FO based on volume of hidden fluid administered at 3 days. To do this, patients were divided into tertiles based on HFV received: low, moderate, and high. Secondary outcomes include quantifying fluid administered by source up to day 7 of ICU admission and assessing the relationship of HFV as a percentage of total volume administered at 3 days with clinical outcomes including length of stay, mechanical ventilation-free days at day 28, new onset atrial fibrillation, new onset dialysis, and all cause in-hospital mortality. This study was approved by the institutional review board with waived informed consent.
Hidden fluids were defined as fluids in continuous and intermittent IV medications, line flushes, blood products, and enteral nutrition. Sources of fluid were defined as the individual hidden fluid sources listed above, resuscitation fluid (defined as fluid administered at a rate ⩾500 mL/h), and maintenance fluid (defined as fluid administered at a rate <500 mL/h). Percentage of hidden fluid was calculated by dividing the volume of hidden fluid administered by the volume of total fluid administered in a given time period (e.g. day 1). FO was defined as a positive cumulative fluid balance that would be expected to produce weight gain over 10% from baseline (e.g. fluid balance ⩾ 7 L in a 70-kg patient).6,7 Diagnoses necessitating maintenance of IVF included diabetic ketoacidosis, diabetes insipidus, cerebral salt wasting, high output fistula (>500 mL per day), high output drains (>500 mL per day), tumor lysis syndrome, and rhabdomyolysis (CPK > 5000). These disease states were agreed upon a priori by consensus of the investigators based on their potential to be impacted by indiscriminate fluid administration. Due to the sample size, only a limited number of covariates could be included in the model.
Statistical analyses
All statistical analyses were performed using SPSS Version 25, and statistical significance was assessed using an alpha level of 0.05. Due to small sample size, data were assumed to have a non-parametric distribution. Descriptive analyses were used for demographic, clinical, and outcome variables. As such, continuous variables were described as median and interquartile range and were compared using the Kruskal–Wallis test. Categorical variables were described as number and percentage and were compared using the chi-squared test. Binary logistic regression was applied to the primary outcome to determine factors associated with FO. Covariates for the model were agreed upon by all investigators and included: age, sex, weight, HFV, number of comorbidities, and severity of illness.
Results
A total of 219 patients were included and stratified evenly into tertiles based on HFV (low HFV: ⩽2500 mL, moderate HFV: 2501–4400 mL, and high HFV: ⩾4400 mL). Groups were mostly similar at baseline except for some differences in comorbidities. The low HFV group was older than the other two groups. Demographic characteristics are summarized in Table 1.
Baseline characteristics.

SOFA: sequential organ failure assessment; LVEF: left ventricular ejection fraction.
Values presented as number (percentage) or median (interquartile range).
The incidence of FO was 3%, 14%, and 25% in the low, moderate, and high HFV groups, respectively (p = 0.001). There were no significant differences in fluid intake on individual ICU days (Table 2). The low, moderate, and high HFV groups also received an increasing volume of total fluid intake over the first three ICU days. Patients in the high HFV group received a higher absolute volume of hidden fluids, but lower amount relative to total fluid intake on ICU days 2 and 3 (Figure 1). Hidden fluids comprised over 40% of cumulative fluid intake over the first 3 days in all groups. By ICU day 7, many patients received 100% of their total fluid intake as hidden fluids (Figure 2).
Fluid administration.
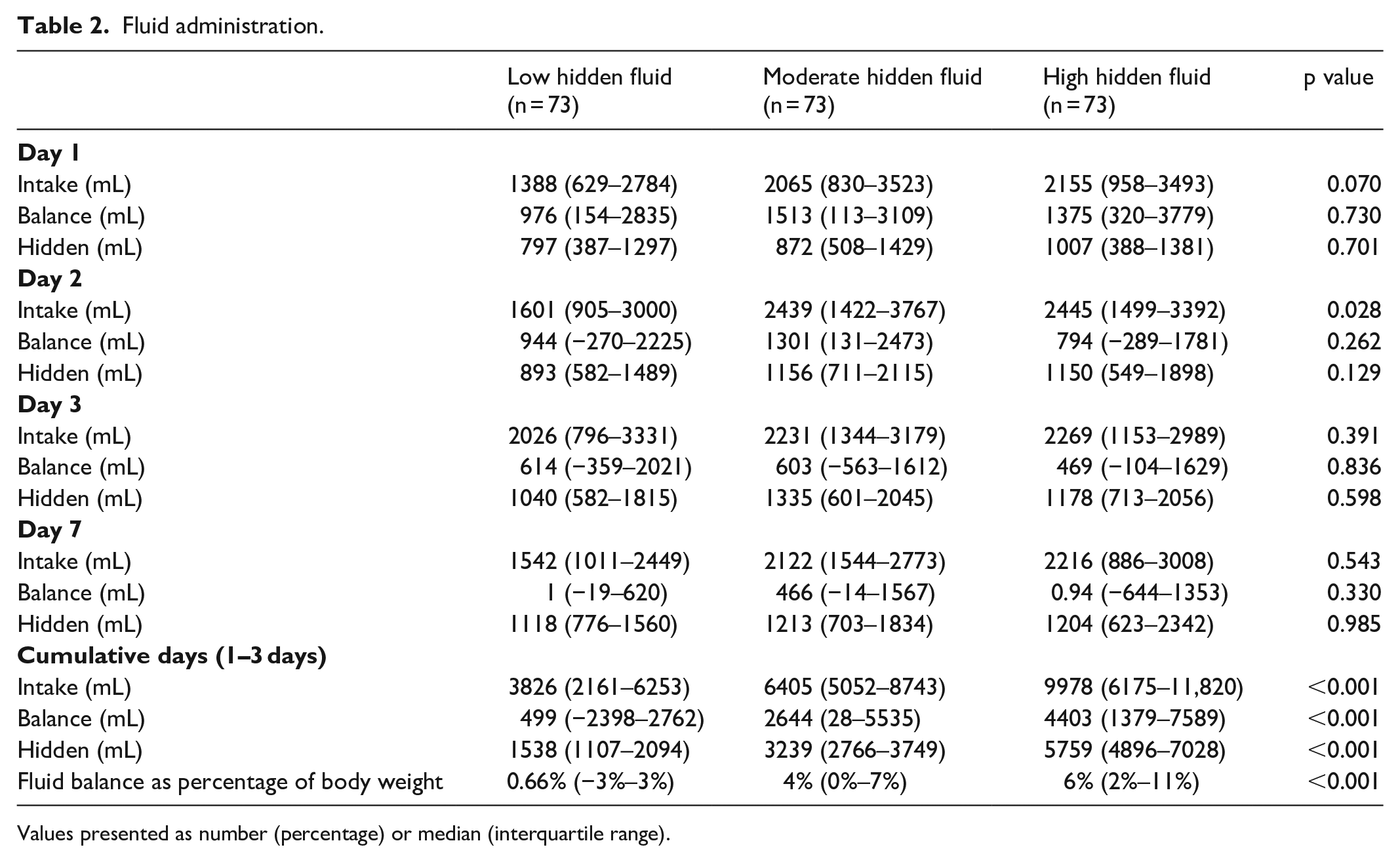
Values presented as number (percentage) or median (interquartile range).

Amount of hidden fluid (all values reported as median).

Percentage of hidden fluids (all values reported as median).
Clinical outcomes are described in Table 3. There were no differences in mechanical ventilation-free days, ICU length of stay, or mortality among patients who received low, moderate, or high volumes of hidden fluid.
Clinical outcomes.

FO: fluid overload; MV: mechanical ventilation; ICU: intensive care unit; LOS: length of stay; RRT: renal replacement therapy.
Values presented as number (percentage) or median (interquartile range).
Binary logistic regression was performed to identify factors associated with the development of FO (Table 4). In the first model, cumulative HFV over the first three ICU days was associated with an increased risk of developing FO. In a second model, HFV on day 1 was associated with a decreased risk of FO. In both models, male gender was associated with a decreased odds ratio (OR) of developing FO.
Factors associated with fluid overload.

CI: confidence interval; SOFA: sequential organ failure assessment.
Hosmer–Lemeshow chi-square = 5.584 (p value = 0.694) and Nagelkerke’s R Square = 0.182.
Discussion
The percentage of patients with FO increased with each tertile, but clinical outcomes, such as in-hospital mortality, mechanical ventilation-free days, new onset atrial fibrillation, and length of stay, were similar between groups. Much of the previous literature evaluated clinical outcomes related to FO at ICU discharge, whereas our study evaluated outcomes at the 3-day mark. This may suggest that predicting patient outcomes or prognosis based on FO in the first 3 days of ICU admission may be premature. However, evaluation of FO at this time point may be beneficial to identify patients for early de-resuscitation such as initiation of diuresis. An additional explanation for not seeing clinical outcomes related to FO could be the inherent bias favoring the low HFV group. These patients in the lower tertile were likely more prone to complications of fluid therapy, given the increased history of chronic lung, heart, and kidney disease.
Positive fluid balance is associated with new onset atrial fibrillation, specifically hypervolemia. 8 It is difficult to determine from the existing literature if the positive fluid balance is an independent predictor or if the positive fluid balance is a surrogate indicator for a higher severity of illness. In this study, we found no difference in the incidence of new onset atrial fibrillation between groups. This may be explained by a similar severity of illness observed across groups or the smaller volume fluid balance seen each day compared to previous studies.
An interesting finding in our results is the discordance observed in the OR associated with incidence of FO with the HFV administered at day 1 and at day 3. At day 1, the HFV administered had a negative association with FO. At day 3, the HFV administered over the first three days was associated with increased OR of FO. We are unable to pinpoint an exact rationale to explain these results. An important data point for future exploration would be the sources that account for HFV (e.g. antibiotics, vasopressors, and enteral feeds). It may be that in the first 24 h, patients with a higher severity of illness may receive only critical medication therapies, whereas patients with a lower severity of illness may see an earlier initiation of enteral feeds or maintenance medications. We attempted to account for this difference by controlling for severity of illness, but sequential organ failure assessment (SOFA) score alone may not be the best predictor for overall illness severity.
Several limitations are important to note for this study. First, stratifying the cohort into tertiles for data analysis is not as clinically sound as establishing volume thresholds for the low, medium, and high hidden fluid groups a priori. Furthermore, a sample size was not pre-determined. However, this is the first study to evaluate hidden fluids so determining a priori volume stratifications for the different tertiles would have been arbitrary. Instead, the results from our study should be considered similar to a dose finding study to evaluate the safety profile of inadvertent quantities of additional fluid administration. Second, a limitation is a standard definition of FO. In this study, we defined FO as a positive cumulative fluid balance that would be expected to produce weight gain over 10% from baseline. However, FO may be defined differently based on gender, severity of illness, baseline comorbidities, and volume status on presentation (e.g. hypovolemic versus euvolemic). In addition, because hidden fluids are not always seen as active fluid administration, it may be difficult to quantify the exact volumes of hidden fluid types. Many hidden fluid sources may not be actively captured in the electronic medical record or have a standard administration volume (medication flushes, diluents, fluids to keep lines patent or flush). Finally, it may be controversial to include administration of enteral nutrition as fluid administration and whether to include the total amount administered as contributing to fluid intake or only the portion of enteral feeding that constitutes free water as fluid intake. Even more challenging to quantify is the contribution to HFV from oral intake, including oral diets, ice chips, or sips of fluid with oral medication administration.
Conclusion
Increased HFV is associated with increased development of FO in ICU patients at day 3, but negative clinical ramifications of FO were not observed in this study. Recognizing patients at risk for FO sooner may help to identify patients who may benefit from early de-resuscitation before the consequences of FO manifest and warrants further research. In addition, classifying the most common hidden fluid sources is an important next step to help develop strategies to mitigate their contribution to overall fluid intake. Converting IV medications to an enteral route or utilizing more concentrated medications or nutrition when feasible are potential strategies to decrease the hidden component of IVF administration. As increased attention is applied to fluid stewardship in the ICU, one important component is recognizing the contribution of hidden fluid administration in overall fluid intake.
Footnotes
Acknowledgements
Amia Ghil assisted with data collection in this project and this did not warrant authorship on this article.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Dr. Newsome has received research funding through the National Center for Advancing Translational Sciences (NCATS) of the National Institutes of Health (NIH) under Award Numbers UL1TR002378 and KL2TR002381.
Ethical approval
Ethical approval for this study was obtained from Augusta University IRB (No. 1214309-6).
Informed consent
Informed consent was not sought for this study because it was waived per the IRB for being a retrospective study.
