Abstract
A dysregulated host response to infection resulting in life-threatening organ dysfunction defines the onset of sepsis. Unfortunately, sepsis is common, costly, and deadly. The Surviving Sepsis Campaign publishes regularly updated, evidence-informed, detection, and treatment guidelines culminating in time-sensitive care “bundles.” The goal of these bundles is to expedite sepsis recognition because it is widely held that early treatment is life-saving. Hospitals are mandated to publicly report their bundle compliance, and this will soon be tied to hospital reimbursement. For these reasons, hospitals are creating sepsis emergency response teams which are a form of a rapid response team consisting of dedicated medical professionals who evaluate patients with suspected sepsis and initiate therapy when appropriate. Evidence to date support sepsis emergency response teams as a mechanism to improve bundle compliance, and potentially, patient outcome. Nevertheless, some elements of bundled sepsis care are controversial (e.g., intravenous fluid administration) as some argue that mandated treatment precludes personalized care. Herein, we briefly describe general sepsis emergency response team structure, review evidence supporting sepsis emergency response teams to improve bundle compliance and patient outcome and report our unique experience incorporating point of care ultrasound—to guide intravenous fluid—into a nursing-led sepsis team. We propose that our sepsis emergency response team approach allays concern that sepsis care is either bundled or personalized. Instead, incorporating point of care ultrasound into a nursing-led sepsis emergency response team increases bundle compliance and individualizes care.
Keywords
Introduction
Sepsis is life-threatening organ dysfunction due to a dysregulated host response to infection. 1 Though the nomenclature has changed in recent years, herein “sepsis” refers to infection associated with organ dysfunction, similar to the former definition of “severe sepsis.” Sepsis and septic shock are common, highly mortal conditions. 2 Indeed, there were between 300 and 1000 cases of sepsis per 100,000 people in the United States (US) in the first decade of the 21st century, and that number is increasing 3 ; in-hospital mortality for sepsis ranges between 20% and 40%.4 –6 From a global perspective, approximately 1-in-5 deaths are attributed to sepsis. 7 Additionally, sepsis is costly. In the United States in 2013, sepsis care cost approximately $23 billion 8 ; unsurprisingly, sepsis is associated with a 75% longer inpatient stay. 9 Thus, it is widely accepted that prompt recognition and treatment of sepsis is imperative to reduce its human and financial burden.10 –12
Therapy for sepsis and septic shock continues to evolve. Since Rivers and colleagues published the landmark Early Goal Directed Therapy (EGDT) protocol in 2001, 13 the Surviving Sepsis Campaign (SSC) has published numerous guidelines on sepsis and septic shock management. 14 As a result of the 2012 SSC update, the New York State Department of Health (NYSDOH) mandated that all hospitals in New York State follow evidence-based protocols for identifying and treating sepsis and septic shock. 10 The protocols included a 3-h bundle consisting of: (1) blood cultures before antibiotics, (2) serum lactate level, and (3) broad-spectrum antibiotics, all completed within 3 h. Furthermore, these protocols also required a 6-h bundle that includes an intravenous (IV) bolus of 30 mL/kg of body weight of crystalloid in patients with hypotension or a serum lactate level of 4.0 mmol or more. 15 The 6-h bundle also included vasopressors for refractory hypotension, and repeat serum lactate within 6 h of protocol initiation. Despite mixed evidence regarding efficacy of these bundles,6,11 (especially with respect to IV fluid resuscitation), 10 the SSC has recently mandated 1-h completion of the aforementioned.16 –19 Furthermore, bundle compliance must be reported publicly by all hospitals in the United States and will be tied to reimbursement beginning in 2026 as a part of the Centers for Medicare and Medicaid Services (CMS) value-based care initiative. Given the clinical and financial importance of recognizing and treating sepsis expeditiously, interest in sepsis emergency response teams (SERTs) is growing. A SERT is similar to rapid or pulmonary embolism response team (i.e., RRT, PERT), however, focused on identifying and treating sepsis and septic shock.20,21
Over the last 2 decades, observational studies on the efficacy of SERTs have been published and reviewed. These studies often showed that SERT implementation associates with reduced sepsis mortality and, almost ubiquitously, increases guideline or bundle compliance. As outlined by both Funk et al. 22 as well as Bloos, 20 a SERT is characterized by its organization, trigger, response (including personnel and equipment) and quality improvement mechanism. Using this framework as a point-of-departure, we review and summarize basic elements of a SERT, give an overview of previously reported SERT investigations and describe our unique experience led by nurses trained in point-of-care ultrasound (POCUS). 23 We argue that nurses using POCUS can improve bundle compliance and individualize IV fluid therapy early in the care of sepsis and septic shock.
A brief overview of sepsis response team structure
Early identification of patients with sepsis and septic shock by a “sepsis emergency response team” (SERT) is believed to improve outcomes.20 –22,24 A SERT is defined as a form of a rapid response team (RRT) consisting of dedicated medical professionals who clinically evaluate patients suspected to have sepsis and initiate therapy when appropriate. 21 Fundamentally, for a SERT to be successful it should contain four basic parts22,25,26 (Table 1).
Basic SERT components.

SERT: sepsis emergency response team.
Afferent arm
Although an exhaustive review of SERT triggers is beyond the scope of this manuscript, a brief explanation is warranted. Both Bloos and Uffen and colleagues have provided up-to-date overviews on SERT alerts.20,21 Usually, the goal of a screening tool is to be overly sensitive (i.e., minimize false negatives) while accepting diminished specificity (i.e., tolerate more false positives). The downside to decreased specificity is that too many false positive alerts can cause “alarm fatigue.”27 –30 In general, in the emergency department, the presence of at least two markers of the systemic inflammatory response syndrome (SIRS) has good sensitivity but variable or poor specificity for predicting life-threatening organ dysfunction due to infection. 21 This explains why the presence of SIRS has only a weak association with mortality.31,32 Another clinical score used to predict sepsis is a derivative of the SOFA score, the “quick” SOFA or qSOFA. 32 The ability of qSOFA to detect sepsis suffers from the “opposite” problem of the SIRS criteria. That is, qSOFA of at least two has a very good specificity (i.e., there are fewer false positives, which reduces alarm fatigue) and is a good predictor of death or ICU admission. 33 Unfortunately, qSOFA can have quite low sensitivity.34,35 In other words, a clinically significant proportion of patients in the ED who go on to develop life-threatening organ dysfunction do not have at least two qSOFA early in their care. Given that screening tests should minimize false negatives (i.e., have high sensitivity), qSOFA is a less-appealing trigger.36,37
Efferent arm
There is great heterogeneity in SERT composition. The efferent arm (i.e., the personnel and equipment brought to the patient) may be composed of trainees (e.g., residents or fellows), critical care physicians, nurses and nurse practitioners, infectious disease specialists, phlebotomists, lab runners, radiology technologists, respiratory therapists, pharmacists, and bed coordinators. Their equipment and resources include, but are not limited to antibiotics, fluids, venous access tools, vasoactive medications, respiratory support, 15 and portable ultrasound technology for hemodynamic assessment, as described below.
SERT implementation: Effects on guideline compliance and hospital mortality
As this was a literature review, approval by a research ethics board and patient consent were unnecessary. We conducted a PubMed search for SERT studies published through December 31, 2023. Additionally, we screened all the of the references of the investigations and reviews that we identified. Only full manuscripts (i.e., not abstracts) in the English language were considered further. We included investigations with a dedicated SERT (i.e., additional personnel with or without specialized equipment presenting to the patient’s bedside), and that reported the effect of SERT implementation on guideline (or bundle) compliance and/or in-hospital mortality compared to no SERT. If the study reported only single elements of contemporary SEP-1 bundle compliance (e.g., time to antibiotics, lactate draw, cultures, etc.), it was included.
SERTs are itemized below based on whether they were led by nurses or nurse practitioners, what the afferent and efferent arms were, and whether POCUS was used to individualize resuscitation. We identified 34 studies and 26 are summarized below. The reasons for excluding the eight investigations are elaborated in the supplementary material.38 –45 Changes in guideline or bundle compliance or mortality are listed based upon statistical significance (i.e., p < 0.05 or less), unless otherwise stated.
Table 2 shows heterogeneity among the studies. This is best illustrated by the variation in baseline mortality rate (i.e., mortality prior to implementation of SERT or mortality rate of non-SERT treated patients) which ranged from 4% to 68% (median 25%, IQR: 13%–37%). Differences in patient population and time may explain this dissimilarity. For example, the study with the highest baseline mortality rate studied patients between 2005 and 2007, required organ dysfunction for SERT activation and included inpatients, 46 while the study with the lowest baseline mortality rate took place between 2016 and 2018, could have included patients meeting only two SIRS criteria (i.e., without organ dysfunction), and was restricted to the ED (i.e., early in disease course). 67 The median mortality change was an 8% reduction (i.e., −8%; IQR: −5% to −18%), though not all of the studies reporting reduced mortality were statistically significant. Of the 26 studies in Table 2, 13 (50%) found a statistically significant reduction in mortality associated with SERT implementation. The remaining 13 studies (50%) either found no significant change (i.e., 9 studies) or did not report or perform a statistical evaluation (i.e., 2 studies reported mortality change without statistical analysis and two studies did not report mortality numbers). When comparing hospital mortality prior to SERT implementation to after SERT implementation for the 24 studies that reported mortality, we found a significant decrease in associated mortality by student t-test (Figure 1); all data had normal distribution by the Kolmogorov–Smirnov test.
Overview of included SERT studies.

RN: registered nurse; NP: nurse practitioner; POCUS; point of care ultrasound; SSC: surviving sepsis campaign; ICU: intensive care unit; SIRS: systemic inflammatory response syndrome; MD: medical doctor; RT: respiratory therapist; SICU: surgical intensive care unit; PA: physician assistant: MAP: mean arterial pressure; CVP: central venous pressure; ED: emergency department; HR: heart rate; RR: respiratory rate; bpm: beats or breaths per minute; EWRS: early warning and response system; SWAT: sepsis workup and treatment; RRT: rapid response team; MEWS-SRS: modified early warning signs-sepsis recognition score; SBP: systolic blood pressure; °F: degrees Fahrenheit; CCRN: critical care registered nurse.
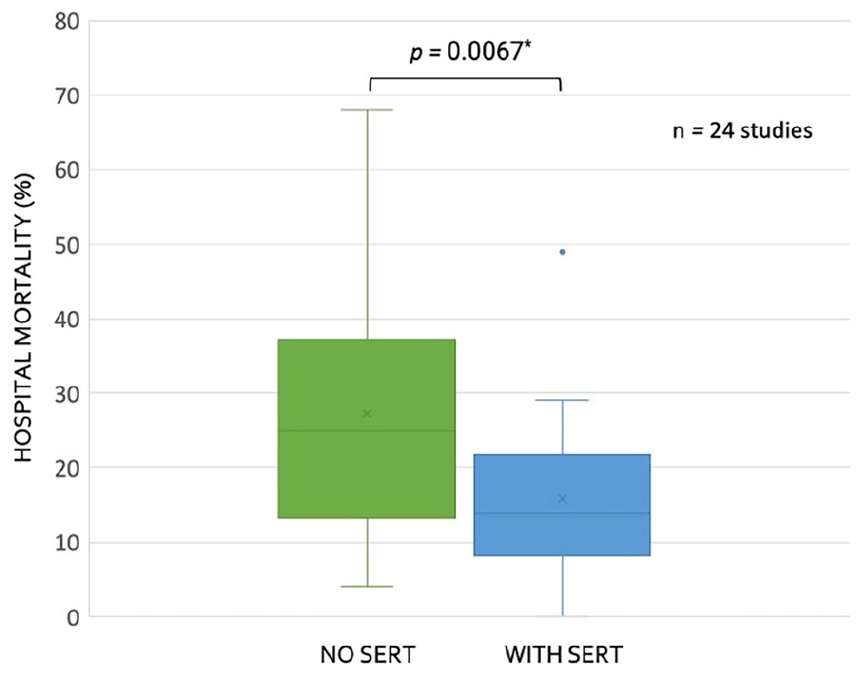
Box and whisker plots for reported mortality with and without sepsis emergency response team (SERT) implementation for studies (n = 24) reporting this data in Table 2.
Compliance with contemporary SSC guideline elements was ubiquitously improved by SERT as compared to no SERT. Because the definition of bundle compliance has changed over the last 20 years, we looked for whether SERT implementation associated with a statistically significant improvement in any or all of: antibiotic administration, blood culture or lactate collection, intravenous fluid provision or MAP goals for patients with sepsis or septic shock. 15 All studies that we located showed clinically and statistically significant improvement in at least one of these elements when SERT was implemented.
Limitations
Although these findings are noteworthy, causal inference cannot be drawn from the investigations reviewed in Table 2. As mentioned by Bloos, 20 before-and-after studies are quasi-experimental and at high risk of bias.20,72 Similar association between traditional RRT implementation and reduced mortality was observed, however, a randomized trial did not show significant benefit. 73 Furthermore, our review was not systematic, and we did not perform a funnel plot, so our search methodology as well as publication bias could partially account for these results. Thus, the effect on mortality we report should be viewed as hypothesis-generating and warrant more rigorous study.
Sepsis emergency response team structure: Our experience
Our SERT is composed of 14 RNs with specific training in POCUS and sepsis resuscitation. The SERT RNs work in collaboration with the primary care team and present the data collected including POCUS exam imaging and fluid responsiveness. The SERT is managed by the sepsis coordinator who also employs three RN data registrars. The data registrars audit each sepsis activation, afford feedback to care providers, and present the current data to the sepsis committee.
There is a total of three SERT RNs on duty per 12-h shift. All SERT nurses have worked in the ICU or ED for a minimum of 2 years prior to joining the team; their certifications include advanced cardiac and basic life support (i.e., ACLS, BLS), and pediatric advanced life support (PALS). SERTs may be initiated anywhere in the hospital as soon as the patient meets the internal activation criteria. The internal activation criteria utilized by our institution is defined as the presence of two or more SIRS with an end organ dysfunction and a known or suspected source of infection (Table 3).
SIRS criteria and end-organ dysfunction.

Mmol: millimole; mmHg: millimeters of mercury; MAP: mean arterial pressure; CPAP and BiPAP: continuous positive and bilevel positive airway pressure, respectively; SpO2: oxygen saturation; CO2: carbon dioxide; L/min: liters per minute; mmHg: millimeters of mercury; mL/kg/h: milliliters per kilogram per hour; mg/dL: milligram per deciliter; mcL: microliter; INR: international normalized ratio.
Changes in vital signs or laboratory values can trigger Best Practice Alerts (BPAs) in the electronic medical record to help identify patients with severe sepsis while in the hospital. Once a BPA is triggered, the staff report findings to the primary care provider, who may then activate the SERT through the internal paging system. Once severe sepsis criteria are met, the SERT team is activated. The majority of activations begin in the ED. The first responders to the patient are the primary RN, the SERT RN, and pharmacist. If the patient is hemodynamically unstable, a physician will also be present. The SERT RN employs bedside ultrasound and a device to assess fluid responsiveness and/or fluid tolerance74 –76; this is discussed in more detail below. The primary physician, pharmacist, and the SERT RN then discuss the plan of care, including suspected source of infection with appropriate antibiotic coverage and orders are placed to ensure that the first hour bundle is met. If a fluid bolus is ordered, the SERT nurse will continue to recheck the patient post-bolus until resuscitation goals are met. Throughout the SERT activation the RN works actively with the primary team to ensure all bundle elements are completed, and the patient receives the highest quality of sepsis care.
Concerns and considerations
Though there are benefits to having dedicated care teams, one challenge faced by the SERT is that during times of high census, it can be difficult to get timely follow-up ultrasound exams—as described below—after a fluid bolus is given, because the SERT also responds to code strokes, cardiac arrests, and rapid responses. Therefore, human resource constraints are an obvious, ubiquitous, problem. Another challenge involved in SERT success is promoting effective team performance when caring for the acutely ill. Regular feedback from the “quality assurance” arm of the SERT in addition to peer-to-peer feedback after challenging cases improves team-based care over time; others note success with simulation training for improving teamwork. 77 Finally, the methods and technologies described below do require significant capital and human investment as we have previously described. 78 While long-term, patient-centered, outcome studies using our approach and technologies are lacking, we note that a similar argument can be made for many monitoring paradigms and tools in the ED and ICU (e.g., arterial lines, end-tidal carbon dioxide, pulse oximetry) where data are used to make physiologically guided decisions without obvious mortality benefit in a wide diversity of patients. Indeed, using point of care ultrasound to guide fluid resuscitation is a nascent area of research with a small and conflicted body of evidence to date.79 –81
Integrating point of care ultrasound into our SERT
In order to reduce mortality associated with sepsis, early recognition, source control, and antimicrobial therapy are essential.12,14 Nevertheless, there is controversy regarding SSC bundle compliance and patient outcome,6,11,16,17,19,21 especially with regards to IV fluid provision. 10 Criticism of mandated IV fluid administration to septic patients ultimately reduces to the tension between “guideline-based” and “personalized” medicine. That is to say, following “check boxes” disconnects the clinician from deliberating each patient’s unique presentation, pathology, physiological reserve, and therapy. 17 On the other hand, proponents of guidelines argue that bundles ensure basic standard-of-care for each patient, reduces practice variability and cognitive load, which can be important in fast-paced clinical scenarios. 82 To reconcile discord between guideline-informed and bespoke therapy, our nursing-led SERT employs POCUS for each patient to gauge the risk–benefit profile of IV fluid administration. 23
POCUS training
Initially, SERT trainees receive basic POCUS instruction, which includes operating specific ultrasound machines, differentiating phased, linear, and convex array probes, and determining their optimal use. The details of heart and lung ultrasound training are outlined in Table 4.
Basic and advanced POCUS competencies for SERT nurses.

LVOT: left ventricular outflow tract; VExUS: venous excess ultrasound score; SERT: sepsis emergency response team; POCUS: point of care ultrasound; IVC: inferior vena cava. RV and LV are right and left ventricles; RA and LA are right and left atria. TV, MV, PV, and AV are tricuspid, mitral, pulmonic, and aortic valves, respectively.
During all sepsis activations on their shift, the trainee performs a POCUS examination with an experienced rapid response RN preceptor. All images are saved and reviewed with a physician. Each trainee reviews 10 POCUS exams and clinical assessments with the lead physician in charge of ultrasound instruction. The trainee must demonstrate aptitude in order to pass training and certify competency. Afterward, SERT RNs maintain competency by exhibiting the aforementioned POCUS skills with the lead physician instructor yearly. To illustrate the effectiveness of our training program, we conducted a prospective observational study revealing that ED physicians agreed with SERT RN POCUS assessments in 99% of cases.78,83
Gauging the benefit–risk profile for IV fluids
When our SERT responds to a patient, we perform a clinical examination supplemented by POCUS to gauge the benefit–risk profile for IV fluid. We consider IV fluid like a drug with organ-specific side-effects.84,85 We are particularly diligent about IV fluids because we see a high volume of septic patients with dialysis dependence, methamphetamine use, pulmonary hypertension, and congestive heart failure. As well, sepsis itself commonly associates with right ventricular dysfunction, which predicts mortality. 86 Given this, our approach to IV fluid is shown in Figure 2 and reduces to 3 basic questions:
(1) Does the patient have a clinical indication for IV fluid?
(2) Is the patient clinically tolerant to IV fluid?
(3) Is the patient fluid responsive?
If the answer to all three questions is “yes,” then the patient has a high benefit–risk ratio and IV fluids are encouraged. If the answer to any of these questions is “no,” then the provider is advised to reconsider fluids or give a smaller bolus volume with rapid re-evaluation (Figure 2). Accordingly, the more of these questions that are answered “no,” the lower the benefit-risk ratio for the individual patient. Brief elaboration of these concepts is considered in turn.

Overview of suggested fluid therapy pathway for our SERT.
Question 1: Does the patient have a clinical indication for IV fluid?
While this question is obvious, it is sometimes forgotten. Like any drug, there should be a clinical need prior to provision. 84 If there is no need, then the therapy should be reconsidered. How do we assess need for IV fluid? Because the heart pumps blood volume to the organs and tissues, we look for signs and symptoms that this particular function (i.e., stroke volume or cardiac output) is impaired. A good list is the “10 signs of vitality” enumerated by Funk et al. 22 Of the 10, we pay particular attention to: unexplained tachycardia (i.e., not from pain, anxiety, fever, medication effect/withdrawal, etc.), hypotension (i.e., systolic blood pressure less than 90 mmHg or MAP less than 60–65 mmHg), tachypnea, diminished consciousness, oligoanuria (i.e., 30 mL/h for 5 h or <100 mL/4h—excluding renal failure), capillary refill longer than 3 s, 87 and elevated serum lactate. Though not specific, each of these markers could indicate reduced cardiac output and, therefore, oxygen delivery to organs and tissues. If any are present, then an indication for IV fluid (i.e., to augment stroke volume and cardiac output) is present; we then turn to assessing the patient’s safety profile for IV fluid, considered below in questions 2 and 3.
Question 2: Can the patient clinically tolerate IV fluids?
Within the sphere of critical care medicine, the entity of “fluid tolerance” has recently emerged.74,76,88 Nevertheless, this concept is a time-honored question grounded in the physical exam. Very simply, a patient can “tolerate” IV fluid volume if there are no signs of venous hypertension (i.e., venous congestion) or overt fluid overload. On examination, this is displayed by sunken eyes, reduced skin turgor, lack of underarm sweat, the absence of pre-tibial edema, jugular venous pulsations below the clavicle and normal cardiac and pulmonary auscultation (e.g., absence of murmurs, rubs, gallops, and bilateral crackles). 89 We supplement the clinical exam with both basic and advanced POCUS including data from a new wireless, wearable ultrasound Doppler.90 –95 Specifically, we evaluate for ultrasound signs of venous hypertension/congestion by looking for an inferior vena cava with less than 50% collapse on inspiration (i.e., a sign that the CVP is more than 10 mmHg), 96 reduced left ventricular (LV) ejection fraction (i.e., a LV high end-diastolic volume), bilateral B-lines on lung ultrasound (i.e., an ultrasonographic sign of pulmonary edema),97,98 or internal jugular venous Doppler signals consistent with high CVP in the semirecumbent position ascertained by the wearable Doppler device. 90 Furthermore, some of the SERT team is comfortable with advanced POCUS measures of venous hypertension, including the venous excess ultrasound score (VExUS). 99 We consider a VExUS score of 2 or more abnormal.88,100 We note that the LV assessment is a qualitative one and that integrating more advanced, objective measures such as mitral annular plane systolic excursion (MAPSE), e-point septal separation (EPSS), and fractional area changes are advanced measures that could reduce operator bias. If there are no physical or ultrasound exam findings concerning for venous hypertension or fluid overload, then the patient is considered “fluid tolerant,” and we proceed to question 3.
Question 3: Is the patient fluid responsive?
Fluid (or preload) responsiveness is a broad topic that has slowly gained traction over the last 20 years in the intensive care unit, operating room, and emergency department.101 –103 The definition of fluid responsiveness is a 10%–15% increase in stroke volume or cardiac output (i.e., SV or CO) following an IV fluid bolus. 103 Measuring the change in flow from the heart (i.e., SV or CO) is critical because other measures (including blood pressure) do not adequately predict whether or not IV fluid administered to a patient actually increases the SV or CO. In other words, if you need to know if the heart will increase flow, then flow needs to be measured! To make this more concrete, consider two patients who receive an IV fluid bolus; neither increase their mean arterial pressure (MAP). Patient A has a significant increase in SV or CO; this is quite common in sepsis (e.g., 50%–60% of patients who increase cardiac output do not increase their MAP).104,105 Patient A might benefit from more fluids and this clinical state would have been missed if using only MAP to guide therapy (e.g., the provider may have decided to use vasopressors prematurely). On the other hand, patient B has no change in SV or CO with IV fluids. Many patients early in sepsis care stop having a significant increase in SV or CO with IV fluids (e.g., 20%–30%).87,106 When these patients stop receiving IV fluid (i.e., cease trying “another bolus”), they have superior clinical outcomes and save the hospital money.107 –109
To test for fluid responsiveness, the SERT nurses perform a passive leg raise (PLR) maneuver110,111 with either SV monitoring via noninvasive or minimally invasive, uncalibrated pulse contour analysis, 112 or measured change in the corrected flow time of the carotid artery (ccFT) 113 measured by the wireless, wearable Doppler ultrasound system.114 –116 The change in the ccFT is measured automatically by the wearable, continuous wave, Doppler ultrasound that has a fixed, 60° insonation angle.94,95,117 The wireless, wearable ultrasound does not generate an image; its wide (>2 cm) ultrasound beam insonates the entire carotid artery, displays a continuous Doppler spectrogram, and automatically determines the largest change in ccFT during a 2-min PLR.75,93 Furthermore, some SERT RNs can measure the change in left ventricular outflow tract velocity time integral (LVOT VTI) 118 ; however, this is often too cumbersome to perform with the PLR, so is rarely done. Both the ccFT and LVOT VTI are Doppler ultrasound surrogates for SV. The PLR is a maneuver where a patient is moved from the semirecumbent position to supine with the legs elevated. This mobilizes 250–300 mL of blood from the abdomen and legs to the heart, acting like an internal blood bolus. 110 How the heart responds to this maneuver predicts how the heart will respond to IV fluids. 119
Concerns and considerations
It is not uncommon for patients to have signs, symptoms, or ultrasound findings concerning for fluid intolerance and/or fluid unresponsiveness early in their care.87,106,120 More specifically, and as shown in Figure 2, if a patient has findings that suggest that fluid intolerance (e.g., distended IVC, pulsatile jugular vein Doppler or B-lines, on lung ultrasound), and/or if the patient does not have a significant increase in SV or surrogate (e.g., ccFT, LVOT VTI) with PLR, the primary provider still makes the final decision regarding IV fluid. We do not treat this algorithm dogmatically or dichotomously, there is lots of physiological “gray zone,” and the response need not be “stop fluids entirely” or “keep giving fluids ceaselessly.” Patients can change rapidly in the ED. Therapies such as antibiotics, antipyretics, adrenergic agents, anxiolytics, analgesics, supplemental oxygenation, or assisted ventilation can all, theoretically, mediate how the heart responds to IV fluids within minutes-to-hours. This is because the inflammatory and sympathetic milieu change early in sepsis and following treatment. Thus, we stress re-assessment when IV fluid therapy is in doubt. Additionally, while ccFT and LVOT VTI change are clinically acceptable ways to detect fluid responsiveness with PLR,113,121,122 measurement error and expertise level can affect accuracy.123 –125 Lastly, in some cases of sepsis and septic shock, patients can be so “volume down” (e.g., if there is concomitant volume loss of approximately 1.5 L or more)126,127 that a PLR does not adequately bolus the heart and the patient will appear fluid unresponsive. In this scenario, after giving some crystalloid (i.e., roughly 20 mL/kg), the patient can become fluid responsive after the stressed venous volume is replenished.
Conclusions
We think it is likely that hospitals and health systems will form RRTs specifically for sepsis, that is, sepsis emergency response teams (SERTs). We believe this because there is good, and growing, evidence that urgently dedicating personnel and equipment to patients with sepsis increases compliance with time-sensitive treatment bundles laid out by the SSC. Because bundle compliance will soon be tied to hospital reimbursement, financial pressure will favor common-sense solutions. We believe strongly that rapid recognition and treatment of sepsis is expedited by SERTs and this likely improves patient outcome. Indeed, our review of SERT literature shows that SERT implementation associated with decreased sepsis mortality, though study methodology and variability preclude definitive conclusions. Perhaps the most controversial aspect of the SSC bundle is IV fluid provision. To address this, our SERT has trained RNs to implement a novel, ultrasound-supplemented treatment algorithm with the goal of expediting personalized sepsis resuscitation. This treatment pathway is not rigidly prescriptive; when there is uncertainty about IV fluid, the algorithm leans on the provider’s clinical judgement and suggests more frequent reassessment, just like any therapy in medicine. The algorithm is founded on using the physical exam and ultrasound to weigh the benefit–risk ratio for each patient. Incorporating “fluid responsiveness” testing in this algorithm will be considered “best practice” by the European Society of Intensive care medicine (ESICM) and has been shown to improve clinically meaningful outcomes in patients with sepsis and is cost-saving. In summary, a nursing-led SERT with competency in basic and advanced POCUS is feasible, novel and expedites individualized sepsis resuscitation.
Supplemental Material
sj-docx-1-smo-10.1177_20503121241290378 – Supplemental material for A nursing-led sepsis response team guiding resuscitation with point-of-care ultrasound: A review and model for improving bundle compliance while individualizing sepsis care
Supplemental material, sj-docx-1-smo-10.1177_20503121241290378 for A nursing-led sepsis response team guiding resuscitation with point-of-care ultrasound: A review and model for improving bundle compliance while individualizing sepsis care by Jared Nunnally, So Mi Ko, Kristen Ugale, Tammy Lowe, Jacyln Bond, Jon-Emile S Kenny, Ramiz A Fargo and Korbin Haycock in SAGE Open Medicine
Footnotes
Acknowledgements
Not applicable
Author contributions
Conceptualization, TL, JB, RAF, and KH; methodology, JN, SMK, KU, and J-ESK; formal analysis, JN, SMK, KU, and J-ESK; resources, TL, JB, RAF, and KH; data curation, JN, SMK, KU, and J-ESK; writing—original draft preparation, JN, SMK, KU, and J-ESK; writing—review and editing, TL, JB, RAF, and KH; supervision and project administration, TL, JB, RAF, and KH. All authors have read and agreed to the published version of the manuscript.
Data availability statement
Data is available upon reasonable request.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: J-ESK is the co-founder and chief medical officer of Flosonics Medical, the remaining authors declare no conflicts of interest.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics approval
Not applicable.
Institutional review board statement
Not applicable.
Informed consent statement
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
