Abstract
Objectives:
The goal of this study was to define the parameters of movement of indocyanine green in the upper extremity of normal control and hand transplant recipients. The purpose was to establish a non-invasive method of determining the level of lymphatic function in hand transplant recipients. In hand transplantation (and replantation), the deep lymphatic vessels are rarely repaired, resulting in altered lymphatic connections. In most cases, the relatively rapid inosculation of superficial lymphatic networks and drainage via the venous systems results in sufficient interstitial fluid and lymph drainage of the graft to prevent edema. However, our group and others have determined that some transplant recipients demonstrate chronic edema which is associated with lymphatic stasis. In one case, a patient with chronic edema has developed chronic rejection characterized by thinning of the skin, loss of adnexal structures, and fibrosis and contracture of the hand.
Methods:
Lymphatic function was evaluated by intradermal administration of near-infrared fluorescent dye, indocyanine green, and dynamic imaging with an infrared camera system (LUNA). To date, the assessment of lymphatic drainage in the upper extremity by clearance of indocyanine green dye has been studied primarily in oncology patients with abnormal lymphatic function, making assessment of normal drainage problematic. To establish normal parameters, indocyanine green lymphatic clearance functional tests were performed in a series of normal controls, and subsequently compared with indocyanine green clearance in hand transplant recipients.
Results:
The results demonstrate varied patterns of lymphatic drainage in the hand transplant patients that partially mimic normal hand lymphatic drainage, but also share characteristics of lymphedema patients defined in other studies. The study revealed significant deceleration of the dye drainage in the allograft of a patient with suspected chronic rejection and edema of the graft. Analysis of other hand transplant recipients revealed differing levels of dye deceleration, often localized at the level of surgical anastomosis.
Conclusion:
These studies suggest intradermal injection of indocyanine green and near-infrared imaging may be a useful clinical tool to assess adequacy of lymphatic function in hand transplant recipients.
Introduction
The lymphatic system plays an important role in the initiation and resolution of inflammation, allograft rejection, and even tolerance.
1
The lymphatic system has several functions, among them filtration of bacteria, foreign materials, toxins and harmful bodies, drainage of excessive fluid from tissues, transport of fluid, protein and cells to the blood stream, augmentation of lymphocyte and antibody production,
1
and absorption and transport of fat from the intestine to the liver.
2
The functional unit of the lymphatic system is called the lymphangion, which is a contractile chamber that propels lymph fluid between two valves.3,4 The phasic contraction of collecting lymph vessels increases in rate with an increase in luminal pressure and relies on activation of voltage-dependent Ca2+ channels.
5
Thus, while lymphatic flow is affected by blood pressure and arterial and venous pulsation, flow does not rely entirely on these mechanisms. The endothelial cells that make up the walls of lymphatic vessels are not bound tightly, but instead overlap one another. Each capillary is thin-walled, relatively large and is anchored to connective tissue via filaments. The overlapping structure of the vessel walls allows them to act as a
The use of indocyanine green (ICG) imaging has helped define the current classifications of lymphedema for oncologic patients. Descriptive patterns have been proposed by Chang et al. 9 as well as Yamamoto et al. 10 Both groups describe four patterns of lymphatic flow based on linear flow versus deceleration and accumulation of ICG. These patterns of dye movement are described as linear, diffuse, stardust, or splash of the dye and reflect the severity of the lymphatic defect. There are limited reports on the objective measurement of upper limb lymphatic function in normal control subjects.11,12 Yamamoto et al. 10 used the contralateral limb in unilateral breast cancer patients as controls for dynamic ICG evaluation and demonstrated the usefulness of this technique for evaluation of upper limb edema.
Graft edema has been observed in patients receiving vascularized composite allotransplantation (VCA) 13 as well as in various animal models of VCA.14,15 Experimental models have shown that lymphatic re-anastomoses typically occur independently through a process requiring vascular endothelial growth factor (VEGF) activity. 16 The time required for this to occur in animal models varies between 5 and 14 postoperative days. 15 Mundinger et al. 14 reported that allograft swelling decreased in their non-human primate (NHP) VCA model in the time in which lymphatic connections had occurred between graft and recipient. Edema is a common finding following transplantation and may reflect a combination of venous outflow issues, rejection, and inadequate lymphatic drainage, 13 and may even also be associated with drug toxicity. 17 Cavadas et al. 13 found evidence of moderate lymphedema via scintigraphic studies in two of three hand transplant recipients studied. It has been proposed that lymphostasis may result in increased fibrosis, adipogenesis, and inflammation. This increased inflammation may give rise to subsequent graft rejection. 18
We hypothesize that significant impairment of lymphatic drainage in hand transplantation does not protect from immune activation, but may aggravate or allow the development of chronic inflammation/fibrosis and atrophy of the skin. In addition, while acute cellular rejection in the form of a rash or erythema rarely affects graft function, edema does reduce flexibility and range of motion, both critical to function in hand transplantation. Chronic edema can also put pressure on peripheral nerves and result in intraneural ischemia clinically manifested as slow progression of sensation recovery, and in the long-term paresthesias, weakness and pain in the transplanted graft. Finally, whether the edema seen in our patients is a result of impaired lymphatic function, inadequate venous drainage, or blood capillary vessel leakage is not always clear. Capillary leakage can also result in edema, and in rare cases, such as patients with Clarkson’s disease, can result in death from massive leakage of fluids and macromolecules into tissue. 19
In order to define abnormal lymphatic function present in transplant recipients, it is critical to understand upper extremity lymphatic function in normal subjects. An objective of the present study was to establish parameters of upper limb lymphatic function in normal, healthy control subjects, defined by time of ICG clearance after intradermal injection of ICG. Similar studies were also performed on hand transplant recipients with the goal of defining adequate and/or inadequate lymphatic function in hand transplant and replant patients.
Materials and methods
Patient population
After obtaining internal review board (IRB) approval, we enrolled 13 healthy subjects and 4 hand transplant patients. Exclusion factors included a known allergy to indocyanine green, iodine, or shellfish. Eight males and five females were enrolled in the normal control group. Four hand transplant recipients were enrolled in the study. Age average was 57.7 (range 42–69), and time post-transplant was 8.5 years (range 17 years to 5 months). Three of the subjects were male, and one was female. The male recipients received a unilateral hand transplant at the midforearm level (Htx01 and Htx03) or at the wrist level (Htx07), and the female recipient (Htx10) was a quadrilateral amputee who received a bilateral hand transplant at the midforearm level on the left side, and at the wrist on the right side.
Imaging system
We used an indocyanine green fluorescent lymphangiography system (LUNA system: Stryker, Kalamazoo, MI) composed of a camera unit, near-infrared laser, and a controller unit that operates the camera. The system allows visualization of anatomical structures by detecting near-infrared radiation in the tissue at a depth of approximately 20 mm from the surface.
Reagents
ICG dye (Stryker, Kalamazoo, MI) dissolved in sterile water to a stock concentration of 2.5 mg/mL concentration was diluted in sterile water to working concentrations. ICG becomes fluorescent upon binding to proteins after injection. These ICG-labeled proteins are cleared by lymphatic fluid with time. ICG maximum absorption occurs at 806 nm (between 750 and 810 nm), and its maximum fluorescence is present in plasma concentrations between 4 and 10 mg/mL. Serum clearance half-life is between 2.5 and 3.0 min.
ICG administration
After initial studies to determine the lowest concentration of ICG required for efficient visualization with the LUNA system, and after studies of various injection sites on the dorsal and ventral sides of the hand as well as the wrist, individuals were given intradermal injections into the second and fourth web spaces consisting of 10 µg of ICG diluted in 100 µL of saline per injection site. Infrared imaging with the LUNA fluorescence angiography unit was performed immediately after or during injection and video was saved for 2 min at the injection site. Subsequent videos of approximately 2 min in length were taken in the volar antebrachial area, and then again at the elbow, the axilla was viewed at the end of each elbow video. These sequences were repeated every 15 min for a minimum of 1 h. The time required for ICG to be detected at the cubital lymph node area was noted as the time to elbow (TTE).
Definition of lymphatic patterns
We assessed and analyzed lymph clearance using dynamic ICG lymphography to differentiate severity of dermal backflow (splash, stardust, and diffuse patterns) according to Yamamoto et al. 10 The patterns were described based on ICG lymphography dermal backflow screen observations. As lymphedema severity progresses, dermal backflow patterns change from splash pattern, to stardust pattern, and finally to diffuse pattern. These observations were used at first on breast cancer–related lymphedema, but we found it useful to describe dermal backflow seen in VCA upper extremity transplant patients (as shown in Figure 3).
Results
Dynamics of subcutaneous ICG injection over time in normal control subjects
In pilot studies, ICG was injected subcutaneously at various places on the back of the hand and the palm. The initial doses of dye were 250 µg of dye in 100 mL of sterile water for injection per application site. We found that injection in the second and fourth web spaces of the non-dominant hand dorsum allowed clear visualization of proximal drainage of ICG in a linear fashion from the site of injection to ventral cubital area and to the axilla. In addition, we observed that the ICG traveled in packets of dye, likely corresponding to accumulation at successive lymphangions. By decreasing the amount of dye, it was determined that 10 µg of ICG was sufficient to allow excellent visualization. Higher doses of dye were associated with excessive retention, or “blooming,” of dye at the injection site with no improvement in vessel visualization (Figure 1). Another important variable that was identified in pilot studies was the critical need for

Initial studies using 250 mg per injection of 100 mL subcutaneously into each of the second and fourth dorsal web spaces. Example of excessive dye “blooming” in hand dorsum of normal control subject 5 min (a) and 1 h (b) after injections.

ICG visualization after administration of 10 μg ICG injected with a 27-gauge needle into the second and fourth dorsal web spaces of normal controls as shown in photograph. During pilot studies to determine the best dye concentration, it was determined that an oblique intradermal injection resulted in reproducible dye movement and fewer cases of the dye being injected too deep or inadvertently into a blood vessel, as compared with more vertical subcutaneous injections.
Subject demographics.

Imaging studies of lymphatic flow in the upper extremity in normal controls and hand transplant recipients by ICG injection revealed similar patterns of dye movement as has been shown in studies of the upper extremity in treated breast cancer patients 10 and lower extremity in treated uterine or ovarian cancer patients. 20 A linear pattern (Figure 3(a)) was typical in the normal subjects as was also observed at minimum within the allograft in all hand transplant recipients. Dermal backflow patterns also described as splash (Figure 3(b)), stardust patterns (Figure 3(c)), or diffuse (Figure 3(d)) were observed in the hand transplant subjects. A splash pattern has larger bright spots with less intense, almost diffuse surrounding it, while a stardust pattern is less intense overall, but with small areas of bright intensity, similar to the night sky. A diffuse pattern appears as dim similar intensity over a given area. Using this terminology, we determined the pattern of lymphatic clearance of 10 µg ICG after intradermal injection in non-surgical, healthy subjects and four-hand transplant patients at various times after their surgeries.

Patterns observed while assessing lymphatic drainage after intradermal injection of ICG in the hand. Control and hand VCA transplant patients exhibited linear pattern drainage (a) from the injection sites, corresponding to lymphatic collector vessels. Dermal backflow patterns observed in the VCA recipients include those termed splash (b), stardust (c), and diffuse (d).
In all normal controls, ICG clearance pattern was seen in the larger linear collectors without accumulation in the superficial lymphatics that give rise to the other dye dispersal patterns. In the obtained images, linear patterns were found in all the control individuals at the hand (Figure 4(a)), forearm (Figure 4(b)), medial elbow (Figure 4(c)), and mid arm (Figure 4(d)). The ICG was visualized moving in a linear pattern between 5 and 15 min from the injection site over the dorsal side of the hand and ventral side of the wrist, and cubital appearance occurred within 30 min for most subjects. When the mean arterial pressure (MAP) of each individual at the time of assessment was compared to the TTE, an inverse correlation was found with an R2 = 0.724 for all the subjects together, and R2 = 0.672 for females and R2 = 0.845 for the male subjects (Figure 5). Table 2 shows MAP versus the ICG transit time from the injection to appear in the middle elbow (TTE). In general, the higher the arterial pressure, the faster the dye moves through the lymphatic vessels.

In all the normal controls, we found linear patterns as shown in the hand (a), forearm (b), elbow (c), and axilla (d).

Time of transit of ICG to elbow after intradermal injection is inversely correlated with subject blood pressure. The time to ICG found at the cubital area varied, with the majority of subjects showing dye at the elbow within 30 min (a). However, when analyzed based on mean arterial pressure, time to elbow showed an inverse relationship, R2 = 0.724 comparing all healthy subjects (b) or R2 = 0.672 for females and R2 = 0.845 for males (c). When placed onto graphs with control subjects (diamonds) and two of the four hand transplant recipients (triangles).
Mean arterial pressure (MAP) and ICG dye time to travel to elbow (TTE) for normal control subjects.

Lymphatic drainage in upper limb VCA transplant patients
Intradermal ICG clearance in the graft was assessed in four of our hand transplant patients. One of the four had edema in the transplanted hand at the time of assessment. In the patient with chronic edema and evidence of chronic rejection (64 years old, male, 8 years post-transplant at right midforearm level), the dermal backflow of ICG was mixture of stardust, linear, and splash patterns within the transplanted hand. The ICG was imaged in a linear pattern over the wrist but then appeared as a stardust pattern at the border between graft and native tissue (Figure 6(a)). This area over the wrist showed a linear pattern between site of injection and the graft/recipient border approximately 20 min after injection. Accumulation of ICG at the donor side of donor/recipient skin junction showed a star-like pattern which remained for the duration of the 1-h study (Figure 6(b)). This pattern suggests drainage is mainly through deep venous system that came with the graft and is connected at the time of the transplant (comitans veins) (Figure 6(a)).

Htx03, 64-year-old male patient, 8 years post-transplant done at midforearm level. Clinical picture showing skin changes that may in part be due to chronic edema (a). ICG lymphography showing a mixture of stardust, linear, and splash dermal backflow patterns 20 min after injection (b) reduction of linear patterns and emergence of stardust and splash patterns at 1 hour post injection.
Two other long-term VCA hand transplant recipients were also imaged for lymphatic drainage. Neither of these patients had signs of edema at the time of assessment but both exhibited mixed patterns of ICG dermal backflow. The second VCA recipient imaged was a 42 year old male who received a unilateral hand transplant three years prior to his participation in this study. Immediate drainage after ICG injection was linear and progressed in distinct packets within the graft moving toward the wrist. Initial movement from the injection site was strikingly similar to controls (Figure 7). Similar to the first patient, the linear pattern of flow changed to a stardust/diffuse pattern at the level of the anastomosis and donor/recipient skin interface. (Figure 7). Appearance of ICG in the cubital fossa was evident by 45 min, which was slower than control males with similar MAP (Figure 5).

ICG clearance in Htx07: 42-year-old male patient, 3 years post-transplant. Linear pattern of dye clearance (a) led changed to a stardust pattern at the interface of graft and donor skin (b), but then continued as linear pattern proximal to the anastomosis of the graft toward the cubital lymph node (c). Green arrow denotes direction of lymphatic flow from hand toward axilla.
The third VCA recipient imaged was a 55-year-old male, unilateral hand transplant, at 17 years post-transplant. The ICG movement occurred was mixed in pattern, with both splash and linear patterns seen in the graft, and stardust/diffuse near the wrist (Figure 8(a)). The linear pattern highlighting the lymphatic vessel between the hand and the cubital fossa on the ulnar side similar to normal controls crosses from graft to recipient side of the forearm (Figure 8(b)). Interestingly, there was a distinct right angle turn in the ICG highlighted vessel near the border between graft and native tissue (red arrow in Figure 8(b)), likely indicating the path of growth that particular graft lymphatic vessel made in order to establish connection with the lymphatic vasculature of the recipient. In addition to this linear clearance pattern, ICG also exhibited a stardust pattern of superficial drainage/accumulation on the dorsal side of the hand. The drainage kinetics were similar to those of the normal controls with higher blood pressure (Figure 5). Figure 9 is a graph of the MAP seen in the two hand transplant recipients who had ICG flow mapped, compared to normal controls. In general, the higher the MAP the faster the dye moves. However, note that our hand transplant patient with the highest MAP did not have the fastest ICG transit time.

ICG clearance in Htx01, 55–year-old male patient, 17 years post-transplant. The ICG movement occurred in a mixed fashion immediately from injections sites with linear and stardust pattern from both second and fourth web injection sites to the wrist (a). A linear pattern was seen from the fourth web injection site (mid left side area of (a)) that proceeded past the wrist to the forearm and from donor into native recipient tissue towards the elbow as shown in (b). The linear pattern between the hand and the cubital lymph node area, was similar to normal controls, especially on the ulnar side. The red arrow in (b) indicates a right angle turn in the lymphatic vessel near the donor/recipient border. Green arrow denotes direction of lymphatic flow from hand toward axilla.

Graphic of the mean arterial pressure (MAP) of normal controls (diamonds) and hand transplant recipients (triangles). Although N is small, hand transplant patients showed slower lymphatic clearance to elbow than did control individuals with similar blood pressures.
The fourth patient imaged is a 69-year-old female bilateral hand transplant recipient. Lymphatic drainage in the left hand was assessed at 4 weeks after transplant. ICG movement from injection site to the graft edge was immediate, linear and in packets, showing the lymphangions pump and strikingly fast. At the border of graft and native tissue, two interesting findings were observed: (1) in addition to linear drainage within the graft, clear superficial lymphatic networks were observed, which had not been observed in previous subjects, either control or hand transplant recipients; (2) clear leakage of dye into tissue where donor deep lymphatic vessels end was noted (as shown by the red arrow in Figure 10). Conversely, this leakage of dye into the tissue was not seen in the same patient in the right hand (Figure 11). The TTE was similar to that observed in control female hands, taking between 10 and 15 min to reach the cubital lymph node area. When ICG imaging was repeated at 21 weeks, the transit time was slower and the lymphatic precollectors were not as highlighted as at the fourth week. It is not clear if the reduced transit time observed at the second time point in this patient was due to reduced flow, or the inclusion of deeper venous drainage which made imaging more difficult.

ICG clearance in left-hand graft of Htx10, 69-year-old female bilateral hand transplant 1 month post-transplant. (a) Photograph image of patient left hand. (b) Mixed linear and stardust patterned IGC clearance in left hand 5 min after administration. (c) Linear drainage from injection sites to wrist, where dye accumulates in tissue at the level of anastomosis. (d) Bright pooling of ICG on recipient side of skin border (donor/recipinet skin border noted by yellow broken lines in photograph of patient elbow/forearm in (e), which is followed by dim area, just distal to the elbow, were cubital LN is easily visualized (white arrow). Green arrow denotes direction of lymphatic flow from hand toward axilla.
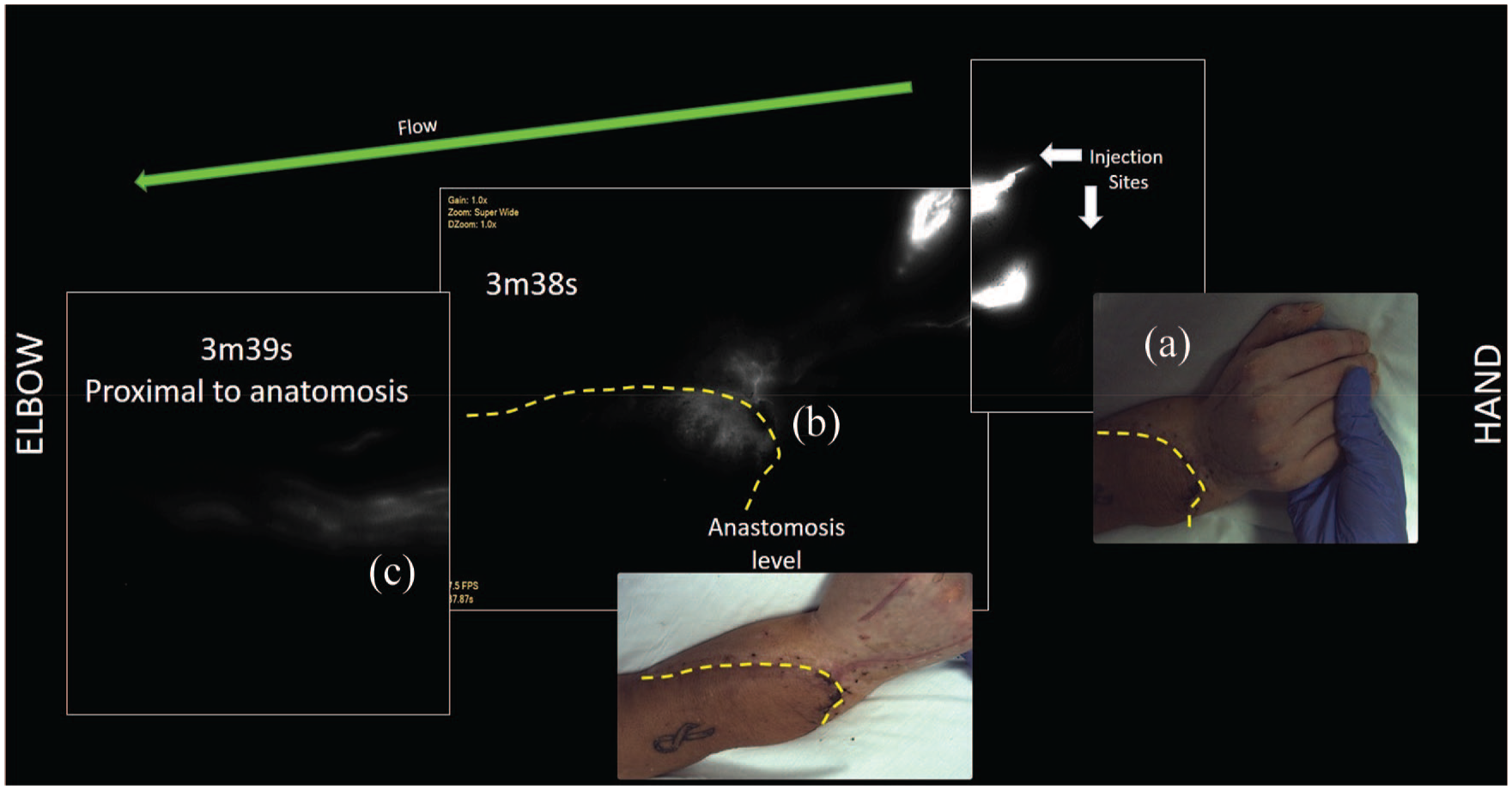
ICG clearance in right-hand graft of Htx10, 69-year-old female bilateral hand transplant. Images are of right hand at 1 month post-transplant. Clearly open and draining deep lymphatic vessels were not observed on this side. (a) Photograph of hand, (b) Stardust pattern seen before and after level of anastomosis at 3 min after injection. (c) ICG was seen near the cubital fossa very quickly (~4 min) after injection.
Discussion
As stated previously, the lymphatic system has been shown to be important in the development of systemic immune responses and has even been proposed as a target to treat inflammatory disease. 21 There is an argument that the lack of connections between graft and recipient lymphatic vessels is beneficial to graft maintenance, since initiation of any adaptive immune response is dependent on the movement of donor antigen-containing dendritic cells from the graft site to regional lymph nodes and the spleen. This movement is most effective through afferent lymphatic vessels, and in allogeneic corneal transplants, the development of anti-donor immunity is dependent on lymphatic vessels rather than blood vessels.22,23 In addition, the pharmacologic inhibition of lymphatic vessel generation can significantly prolong islet allograft survival while simultaneously inhibiting lymphangeogenesis to the site of transplant, under the kidney capsule. 24 The role of lymphatics in inflammation is an area of intense research, in some cases producing conflicting results. For example, in an experimental mouse model of inflammatory bowel disease, VEGF-C stimulation of lymphatic function has been shown to both ameliorate 25 and promote 26 intestinal inflammation.
The observation by our group and others that some degree of lymphostasis occurs in upper extremity transplantation, suggest that analysis of lymphatic function may be an important immunomonitoring assay for VCA patients. In contrast to normal control patients who show linear patterns of lymphatic drainage, we found a mixture of different patterns of dye movement, particularly around the vascular anastomosis level in hand VCA.
In hand transplantation surgery, in which the lymphatic trunks are not connected, superficial and deep stasis of the lymph will occur at least initially, and in many cases, the venous system will compensate to drain fluids from the tissues. We have objective evidence that all the patients have some degree of extravasation/deceleration around the transition between the donor and recipient site. However, we found that there is proximal clearance of the dye as early as 4 weeks after transplant. In the early post-transplant period, the dye may follow the donor lymph vessels, exit the open lymphatics (as seen in the left hand of Htx10 at 1 month (Figure 10)), and accumulates in the interstitial space. The dye may then find and later finds a proximal recipient lymphatic vessel to re-enter the system (Figure 12). We found that although all patients have shown clearance of the ICG the, question of how fast, or what patterns of clearance indicate sufficient lymphatic drainage to prevent edema is not answered in this study. In mouse models, while an absence of lymphatic vessels prevented the movement of dendritic cells and the subsequent immune response, 27 further studies demonstrated that very few vessels are needed to support normal dendritic cell movement. 28 While very few vessels may support the immune activation function of the lymphatic system, it is not clear whether similarly few vessels are sufficient to maintain fluid homeostasis.

Cross-section diagram of skin to muscle at site of VCA anastomoses and skin closure. Level of connection indicates where the donor/recipient tissue is joined. Unlike the arteries and veins, none of the lymphatic vessels are surgically re-connected. Superficial lymphatics do inosculate and establish donor-to-recipient connections through VEGF-C-dependent mechanisms. In the early post-transplant period, the dye may follow the donor lymph vessels, leave the open ended deep lymphatics outside the donor tissue, accumulate in the interstitial space, and later finds a proximal recipient lymphatic vessel, emulating a cistern.
In most of our transplant patients, and in most hand surgery patients with traumatic disruption of the deep lymphatics, post-surgical edema resolves rapidly. We know from animal model studies that the superficial lymphatic network is established within a few days across the donor/recipient skin border, and both host- and donor-derived lymphatic endothelial cells participate in the lymphatic sprouting.16,29 However, in the absence of VEGF-C stimulation of lymphoneogenesis, the generation of the larger collecting lymphatics is absent, or greatly reduced. 30 Presumably, performing lymphaticovenous anastomoses or direct lymphatic vessel anastomosis at the time of the transplant would improve large collector draining. This surgery is feasible and in selected patients has shown promise in resolution and prevention of symptoms both as a treatment and for prophylaxis of lymphedema.9,31 The most challenging aspect of this approach in upper extremity allotransplantation is that an already long and difficult operation would be extended. A more practical surgical approach is to perform as many as possible venous anastomosis from both deep and superficial vascular systems during the transplantation surgery.
A limitation of this study is the small number of study subjects. Another limitation is the depth and resolution of ICG dye by the LUNA system. If the collectors are deeper than the LUNA capacity deep of 20 mm, the lack of pattern visualization on ICG lymphographies do not necessarily indicate absence of functional collectors, or absence of lymphatic clearance via deep venous system. 7 The study would also have been strengthened by comparative standard lymphoscintigraphy studies to support or refute findings of lymphedema. However, as this test was not clinically indicated, the cost and risk (albeit small) of the radioactive tracer did not justify its use in this group of subjects. In future studies, we plan to include both intradermal and intravenous injection of ICG. The intravenous injection can be performed easily on the dorsal aspect of the hand. By comparing patterns of intradermal dye drainage with the patterns seen in the venous drain system, through the vein anastomosis which should be able to determine whether collectors are present. In addition, if the subject has edema due to leaky blood vessels as opposed to abnormal lymphatics, the clearance of dye from the vascular system may be impacted.
In summary, our studies on normal control subjects and hand transplant recipients confirm LUNA ICG lymphography is easy, safe and provides immediate feedback, and easily identifies normal intradermal clearance of ICG. The procedure can be performed in any outpatient clinic. A linear pattern of drainage was seen in 100% of normal controls between the hand and cubital lymph node, continuing on to the axilla. We have identified a consistent time parameter for lymphatic clearance, which is inversely correlated with MAP. We have observed both normal linear patterned clearance from normal controls and VCA patients, as well as splash and stardust patterns of dye deceleration within the graft tissue and at the border areas between graft and native tissues in VCA patients. Moving forward, imaging of lymphatic clearance of intradermal ICG injection can be used to determine whether graft swelling that routinely occurs with VCA rejection episodes is related to lymphatic dysfunction. Further studies are needed to determine whether similar assessments would be useful evaluating and/or treating patients with lymphostasis related to burns, cellulitis and other infections, or compartment syndrome.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Department of Defense through the Reconstructive Transplantation Research Program under Award No. (W81XWH-13-2-0057), and the Jewish Hospital and St Mary’s Healthcare Foundation.
