Abstract
Objectives:
The optimal antiretroviral therapy for patients with the M184V/I mutation is not known. The primary objective of this study was to determine the efficacy of various antiretroviral therapies in patients with HIV and the M184V/I mutation based on the number of active antiretroviral agents.
Methods:
A retrospective chart review was conducted of 100 treatment-experienced patients harboring the M184V/I mutation seen at an urban HIV clinic. Efficacy was classified as percentage of patients with viral suppression defined as HIV RNA viral load <200 copies/mL at last measurement on current antiretroviral therapy, stratified by the number of active antiretroviral agents.
Results:
The primary outcome of viral suppression occurred in 70.6% (12/17) of patients on <2 active agents, 77.2% (44/57) on 2–2.5 active agents, and 69.2% (18/26) on 3 active agents. No significant difference was found between viral suppression and patients on <2 and 2–2.5 antiretroviral agents (odds ratio = 0.71, 95% confidence interval = (0.21, 2.39), p = 0.8) or between patients on 3 and 2–2.5 active agents (odds ratio = 0.66, 95% confidence interval = (0.23, 1.88), p = 0.7). The most commonly prescribed regimen consisted of a boosted protease inhibitor with an integrase strand transfer inhibitor and two nucleoside reverse transcriptase inhibitors, one of which being lamivudine or emtricitabine.
Conclusion:
Similar rates of viral suppression were observed in patients regardless of the number of active antiretroviral agents prescribed. Regimens containing less than 3 active agents may maintain virologic suppression in patients with the M184V/I mutation. Further studies are needed to determine optimal antiretroviral therapy for patients with the M184V/I mutation.
Introduction
The human immunodeficiency virus (HIV) has a high mutation rate and therefore it is important to consider information regarding genotypic resistance when initiating or modifying antiretroviral (ARV) therapy (ART). The current Department of Health and Human Services (DHHS) guidelines for HIV-1-infected adults suggest initial ART for ARV-naïve patients comprising three, and in select patients two, fully active agents expected to have uncompromised activity based on genotypic resistance test results. 1 For treatment-experienced patients with virologic failure, past ART and resistance history must be taken into consideration when selecting new ART. 1
The nucleoside reverse transcriptase inhibitor (NRTI) mutation, M184V/I, is most commonly selected by two NRTIs, lamivudine (3TC) and emtricitabine (FTC) and confers high-level resistance to both agents.2–4 Lamivudine or FTC are recommended as a part of the NRTI backbone of all recommended ART and are included in all currently available triple-therapy single tablet regimens. 1 The M184V/I mutation also reduces susceptibilities to abacavir (ABC) and didanosine (ddI), while increasing susceptibility to zidovudine (ZDV/AZT) and tenofovir (TAF/TDF).2–4 Additionally, the M184V/I mutation occurs in the YMDD motif of reverse transcriptase (RT) causing structural changes close to the catalytic site in the enzyme which reduce RNA primer usage and decrease viral fitness through slowing viral replication. 5 Although the presence of the M184V/I mutation confers high-level resistance to both 3TC and FTC, the reduction in viral replication fitness may allow for the use of less than three fully active agents while maintaining virologic suppression.
Prescribing practices vary for patients harboring the M184V/I mutation due to its effects on viral replication and NRTI susceptibilities and based on whether patients have additional resistance mutations. Some practitioners opt to continue 3TC or FTC in the setting of M184V/I as treatment-experienced patients have been found to experience less virologic failure and preservation of CD4 count with continuation of 3TC despite the presence of resistance.6,7 The MOBIDIP study found that continuation of 3TC with a boosted protease inhibitor (bPI) after treatment failure in patients with the M184V mutation led to significantly less virologic failure compared with bPI monotherapy. 8 The DHHS guidelines suggest the following possible regimens for patients with the M184V/I mutation only and virologic failure: (a) continuation of same regimen, (b) bPI with two NRTIs, (c) integrase strand transfer inhibitor (INSTI) with two NRTIs, or (d) a bPI with an INSTI. For options that include NRTIs, it is recommended that at least one of the NRTIs should be considered an active agent. It is recommended that regimens contain at least two, and preferably three, fully active agents. 1
There has been a shift toward the use of simplified, two active agent regimens in the general population with HIV to minimize adverse events, drug–drug or drug–food interactions, and pill burden based on success in recent clinical trials.1,9–11 However, patients with major resistance, including M184V/I, are excluded from these trials. For most patients with the M184V/I mutation, the use of three active requires the use of more complex, multiple tablet regimens, especially in the setting of additional resistance mutations. For example, many patients with the M184V/I mutation are prescribed a multiple tablet regimen containing three active agents (two NNRTs, bPI, and INSTI) based on limited prospective data indicating efficacy of this regimen. 12 Therefore, there is an interest in whether regimens with <3 active agents could simplify regimens and decrease pill burden while maintaining efficacy.
The optimal ART for patients with the M184V/I mutation is not known. It is also not known whether the features of this mutation may allow for the use of less than three fully active agents while maintaining virologic suppression. The primary objective of this study was to assess virologic suppression in patients with a documented M184V/I mutation based on the number of active agents in the ARV regimen. Secondary objectives of the study included assessing efficacy of ART in maintaining CD4 count >200 cells/mm3 based on the number of active agents and characterizing prescribing practices for ART in patients with an M184V/I mutation.
Methods
Study design and patients
We conducted an observational, retrospective study of patients with HIV-1 seen at an urban HIV clinic. A convenience sample of 100 patients that were seen at the clinic between 1 September 2014 and 31 August 2017 were included in the study if they were HIV-infected, 18–89 years of age, on ART, and with at least an M184V/I mutation documented via genotypic resistance tests. Patients were excluded if there was no documented HIV RNA viral load (VL) available in the results section of the electronic medical record within the year prior to study inclusion date or while on current ART.
Data collection
Data collected from patients’ electronic medical record included patient demographic information: age, gender, and race. The HIV data collected included most recent ART and start date, ART within the past 3 months (if different from current ART), most recent VL and CD4 count on current ART, presence of VL >100,000 copies/mL at any point on current ART, CD4 nadir, duration of HIV infection, and presence of concern for non-adherence. Patients were considered to have presence of concern for non-adherence if there was documentation of concern of “non-adherence” or “non-compliance” in the clinic visit notes from the four most recent visits. All documented resistance-conferring HIV mutations, historical and present, were collected from the results section of the patients’ medical records.
Study endpoints
The primary endpoint for this study was the proportion of patients with viral suppression while on current ART, stratified by the number of active ARVs. Viral suppression was defined as VL < 200 copies/mL at the most recent measurement while on current regimen. Although viral suppression is confirmed by the lower limits of detection of used assays, the definition of virologic failure as defined in the DHHS guidelines was used. This was done to take into consideration any virologic blips that may occur during therapy that would not indicate a change in ART. A single VL value was used because confirmatory VL measurements were not always available given the retrospective nature of the data. The number of active agents was determined based on the genotypic susceptibility score (GSS). 13 The Stanford HIVdb genotypic resistance classification was used to calculate GSS. A GSS of 1 was assigned for drugs deemed susceptible or with potential low-level resistance, while a GSS of 0.5 was assigned to drugs with intermediate or low-level resistance, and a GSS of 0 was assigned to drugs with high-level resistance. Both 3TC and FTC were assigned a GSS of 0 in the presence of an M184V/I mutation. The sum of the GSS for each drug in the regimen was considered as the number of active agents. Secondary endpoints included proportion of patients with most recent CD4 count >200 cells/mm3 while on current ART stratified by the number of active ARVs and the proportion of patients on various combinations of ARVs.
Statistical analysis
Data were analyzed using descriptive statistics for demographic data. Multivariable analysis was performed to measure the association of all predictors and outcomes. For the multivariable analysis, a logistic regression model was used for outcomes with demographic characteristics. To identify potential confounders, the analysis checked for other factors associated with exposure of interest including age, gender, HIV duration in years, presence of VL >100,000 copies/mL in HIV history, CD4 nadir during treatment history, and presence of concern for non-adherence by using t-test or Wilcoxon rank-sum test for continuous variables and Fisher’s exact test for categorical variables. Factors with a statistically significant relationship on univariable analysis were included in the multivariable analysis. The odds ratios (ORs) with 95% confidence interval (CI) were calculated to measure the associations of outcomes and effects. Analyses were performed with SAS version 9.4 (SAS Institute, Cary, NC).
Results
One hundred patients with a documented M184V/I mutation were enrolled in this study. Demographic and resistance mutation data for the cohort are represented in Table 1. There were no significant differences in demographic data between virally suppressed versus not suppressed patients, except that non-adherence was more prevalent in the patients that were not suppressed (39.2% and 80.8%, p = 0.0005). Twenty-nine patients (29%) harbored only the M184V/I mutation. Thirty-one patients expressed thymidine analog mutations (TAMs) and the median number of TAMs among these patients was two. One patient had a multidrug-resistant reverse transcriptase mutation present (Q151M) which conferred resistance to all available NRTIs. The frequency of historical plasma RNA resistance mutation to at least one drug among non-nucleoside reverse transcriptase inhibitors (NNRTIs), PIs, or INSTIs was 32%, 6%, and 4%, respectively. The four patients (4%) with mutations conferring resistance to INSTIs remained susceptible to dolutegravir (DTG) and bictegravir (BIC).
Demographic data.

SD: standard deviation; VL: viral load; IQR: interquartile range; NRTI: nucleoside reverse transcriptase inhibitors; TAM: thymidine analog mutation; NNRTI: non-nucleoside reverse transcriptase inhibitors; INSTI: integrase strand transfer inhibitor; PI: protease inhibitor.
TAMs were defined as M41L, D67N, K70R, L210W, T215Y/F, and K219E/Q/R in RT. TAMs observed were M41L (n = 11), D67N (n = 14), K70R (n = 10), L210W (n = 6), T215Y/F (n = 13), and K219E/Q (n = 15).
ARV therapy for patients included in the study is represented in Table 2. There were no patients that had switched ART within the 3 months prior to inclusion. Most patients (57%) were prescribed 2–2.5 active agents. NRTI-containing regimens were used in 85% of patients. Of patients prescribed NRTI-containing regimens, 81 (95.3%) patients were prescribed 3TC or FTC. The most commonly prescribed ART (n = 27) consisted of two NRTIs (one being 3TC or FTC), a bPI and an INSTI. Within the regimen, the most commonly prescribed PI was darunavir (DRV) and the most commonly prescribed INSTI was DTG, though eight patients received elvitegravir (EVG) as a part of a combination tablet. The second most common regimen (n = 24) was two NRTIs (one being 3TC or FTC) and an INSTI. The INSTI most commonly used in this regimen was DTG. The most commonly prescribed regimen among patients harboring only the M184V/I mutation (11/29 patients, 37.9%) was two NRTIs (one of which being 3TC or FTC) and an INSTI (either DTG or EVG) with a GSS of two.
Antiretroviral regimens based on the number of active agents defined by GSS.

GSS: genotypic susceptibility score; NRTI: nucleoside reverse transcriptase inhibitor; NNRTI: non-nucleoside reverse transcriptase inhibitor; PI: protease inhibitor; INSTI: integrase strand transfer inhibitor; EI: entry inhibitor.
Viral suppression occurred in 12 patients (70.6%) on <2 active agents, 44 patients (77.2%) on 2–2.5 active agents, and 18 patients (69.2%) on 3 active agents. The most commonly prescribed ART in patients with viral suppression on <2 active agents was two NRTIs (one being 3TC or FTC) and an INSTI (most commonly DTG), for 2–2.5 active agents was two NRTIs (one being 3TC or FTC) with an INSTI (most commonly DTG), and for 3 active agents was two NRTIs (one being 3TC or FTC) with a bPI and an INSTI (most commonly DRV and DTG, respectively). No significant association was found between viral suppression and patients on <2 and 2–2.5 ARV agents (OR = 0.71, 95% CI = (0.21, 2.39), p = 0.8) and patients on 3 and 2–2.5 active agents (OR = 0.66, 95% CI (0.23, 1.88), p = 0.7), see Figure 1. There was also no significant association found between CD4 count and patients on <2 and 2–2.5 active agents (OR = 0.78, 95% CI (0.21, 2.85), p = 0.5). Patients on three active agents were less likely to have a CD4 count ⩾200 cells/mm3 compared to 2–2.5 active agents (OR = 0.24, 95% CI = (0.08, 0.66), p = 0.01). There was more non-adherence in patients with a CD4 count <200 cells/mm3 (67.9% vs 43.1%, p = 0.04); however, non-adherence appeared to be similar between patients on <2, 2–2.5, and 3 active agents. Finally, there were similar rates of viral suppression regardless of whether a regimen was NRTI-containing or NRTI-sparing, 73% and 80%, respectively (OR = 0.59, 95% CI = (0.14, 2.49), p = 0.5).
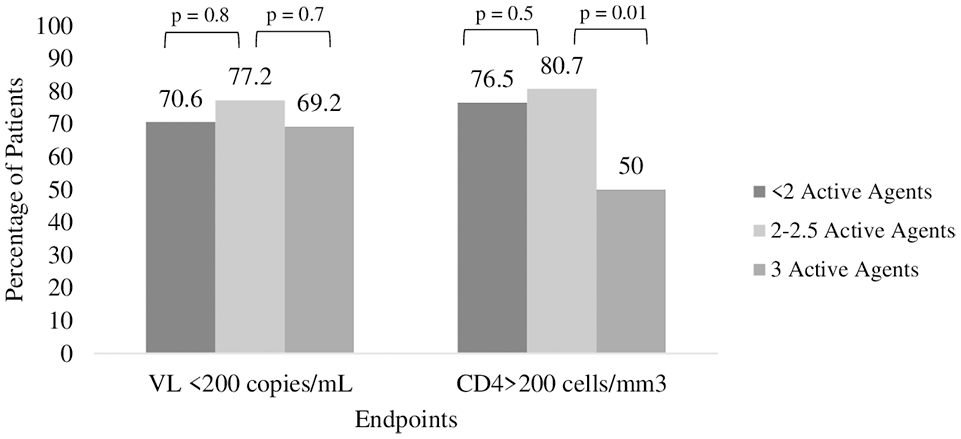
Frequency distribution of suppressed VL and CD4 count <200 across the number of active agents.
Discussion
Similar rates of viral suppression were found regardless of the number of active ARVs in this study of treatment-experienced patients with HIV and at minimum an M184V/I mutation. The most commonly prescribed regimen in this study was a bPI, an INSTI, and two NRTIs (either 3TC or FTC). This is likely reflective of the fact that the most convincing prospective data available in treatment-experienced patients, 85% with an M184V/I mutation and 26% with 3-class resistance, utilized an NRTI-containing three active agent regimens. 12 However, given that the majority of patients in our study had multiple class resistance, this commonly used regimen often contained only 2–2.5 active agents. The second most commonly prescribed regimen also contained <3 active and consisted of an INSTI and two NRTIs (either 3TC or FTC). Most patients in the study were continued on 3TC or FTC. The results of our study lend support to the growing body of evidence that patients with the M184V/I mutation may maintain viral suppression in the setting of less than three fully active agents.
The use of less than three active agents in patients with the M184V/I mutation has not been evaluated in large, prospective studies. There are several small or retrospective studies that have examined the use of two or less active agents in this population. The DOLULAM study, a single arm, prospective cohort of 27 treatment-experienced, virologically suppressed patients, 17 (63%) of which had a documented M184V mutation, found that switching to DTG and 3TC maintained viral suppression in 100% of patients at 2 years. 14 The patients in DOLULAM had excellent adherence and rigorous follow-up which differs from the 50% non-adherence in our study and may account for why we saw overall lower rates of virological suppression. Gagliardini et al. 15 retrospectively evaluated switching to dual therapy with 3TC plus either bPI or INSTI in 436 patients, 87 (20%) of which had the M184V mutation. They found that the M184V mutation was not associated with increased risk of virologic failure or treatment discontinuation at a median follow-up of 1.3 years compared with patients with at least one non-M184V mutation. The presence of the M184V mutation was, however, associated with higher probability of experiencing viral blips. Perez-Valero and colleagues prospectively evaluated switching to a single tablet regimen containing two active agents (EVG, cobicistat, FTC, and TAF) in 34 virologically suppressed patients with the M184V/I mutation. Suppression was maintained in 100% of patients at 24 weeks. 16 Of note, the patients in this trial had only the M184V/I mutation, unlike the population in our study which often had multiple resistance-conferring mutations present. Olearo et al. 17 retrospectively examined switching to a regimen containing ABC, 3TC, and DTG in treatment-experienced patients and found that the M184V mutation, present in 137 patients (8.4% of study population), was not associated with increased risk of virologic failure. Finally, Sahloff and colleagues retrospectively evaluated the use of boosted DRV plus TDF/FTC in 32 treatment-experienced patients with at least an M184V/I mutation. Viral suppression was achieved in 84% of patients. 18 These studies, in congruence with the results of our study, suggest that regimens containing <3 active agents and continuing 3TC or FTC may maintain acceptable viral suppression in the setting of an M184V/I mutation.
Nucleotide reverse transcriptase inhibitors–sparing regimens were used in 15% of the patients in our study. There are several reasons why a patient may not be prescribed an NRTI, including adverse events and resistance; however, there are potential benefits of maintaining selection pressure for the M184V/I mutation. There is little evidence regarding optimal NRTI-sparing regimens in patients with the M184V/I mutation. We were unable to detect a difference in outcomes in patients on NRTI-sparing versus NRTI-containing regimens; however, this is likely due to a small sample size.
There are limitations to be considered when interpreting the results of this study. This was a small, retrospective chart review conducted at a single site. Given the small sample size included in our study, it was not powered to detect a difference in outcomes between drug regimens. Most patients (93%) were Black or African American which may limit the generalizability of our data. There was also potential for information bias due to inconsistencies in documentation of previous genotypes, previous ART, and non-adherence. We did not have access to prescription refill data to determine adherence to ART and therefore relied on documentation of concern for non-adherence within clinic visit notes which may be subjective and may over or underestimate non-adherence. Additionally, this was a treatment-experienced population, often with multiple mutations, and a presence of concern for non-adherence in 50% of the patient population. This may explain why the rates of viral suppression in this study population (69.2%–77.2%) were lower compared with the overall viral suppression rate (85%) for patients on ART within the study HIV clinic. Although the endpoints were adjusted for non-adherence as a confounder and this is likely representative of a more “real-world” urban patient population, it is possible that this hindered our ability to detect a difference in the primary outcome. Finally, new ARV agents have been introduced since our analysis, most notably bictegravir.
Conclusion
Regimens containing 2–2.5 active agents were commonly prescribed in patients with M184V/I mutations and most patients were continued on 3TC or FTC despite the presence of resistance. Similar rates of viral suppression were observed in this study population regardless of the number of active agents prescribed. Regimens containing less than three active agents may maintain viral suppression in patients with the M184V/I mutation. Larger, prospective trials examining ART containing <3 active agents and the M184V/I mutation are needed to determine optimal ART for such patients.
Supplemental Material
Original_Table_2_2 – Supplemental material for Virologic suppression in patients with a documented M184V/I mutation based on the number of active agents in the antiretroviral regimen
Supplemental material, Original_Table_2_2 for Virologic suppression in patients with a documented M184V/I mutation based on the number of active agents in the antiretroviral regimen by Mallory T Mouradjian, Emily L Heil, Hyunuk Sueng and Neha Sheth Pandit in SAGE Open Medicine
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was waived by the University of Maryland, Baltimore (UMB) Institutional Review Board (IRB) because it was determined that the probability and magnitude of harm/discomfort anticipated in the research were not greater in and of themselves than those ordinarily encountered in daily life or during the performance of routine physical or psychological examinations/tests. Information was recorded by the investigator in such a manner that the identity of the human subjects cannot readily be ascertained directly or through identifiers linked to the subjects, the investigator did not contact the subjects, and the investigator will not re-identify subjects.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Informed consent was not sought for the present study because this was a retrospective study and with the nature of the design patients were not contacted. All data had already been collected as a part of each patient’s routine care and was stored in the patient’s electronic medical record.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
