Abstract
Objectives:
Despite many advances in medicine, not all individuals with HIV are able to achieve complete virologic suppression. This retrospective study identifies variables associated with persistent HIV viremia in an academic clinic.
Methods:
We studied 66 HIV-infected patients with a viral load of >200 copies/mL over 1 year, with controls matched 1:1 via a propensity score utilizing age at diagnosis, era of diagnosis, gender, and initial CD4 count. We collected data on multiple variables including medications, adherence, comorbidities, hospitalizations, and insurance status. Conditional logistic regression was used for unadjusted and adjusted analyses.
Results:
A total of 66 viremic cases/matched controls were included. Fewer viremic patients were on antiretroviral therapy for all 12 months (45% vs 77%; odds ratio: 0.33, p = .018) and fewer were of white race (52% vs 70%; odds ratio: 0.49, p = .053). Hospitalization (11% vs 3%; odds ratio: 10, p = .028), underinsurance (20% vs 1%; odds ratio: 5.87, p = .022), and conflicting personal beliefs about their disease (17% vs 3%; odds ratio: 5.5, p = .027) were more common in viremic patients. Psychiatric illness increased the odds of viremia in patients who had four or more visits (odds ratio: 1.63/6.64 with four/five clinic visits, respectively).
Conclusion:
Psychiatric illness is an important contributor to the presence of persistent viremia in HIV-infected patients and deserves further study.
Introduction
Recent estimates show that currently over 1 million adults in the United States are living with human immunodeficiency virus (HIV) infection. 1 Advancements in antiretroviral therapy (ART) have markedly changed the field of HIV, changing what was once a fatal disease into a manageable condition. 2 Patients are being identified more promptly due to proactive screening initiatives and are initiating ART at an earlier stage in their disease process with single-tablet drug regimens that are more potent than their predecessors.3–6 This improved management has had a significant impact on the clinical course of HIV infection and the likelihood of transmission to others. These interventions have contributed to the significant reduction in mortality in the HIV population, which is comparable to that of the general population if CD4 cell counts are maintained at 500 cells/µL or greater. 7
However, despite these advances, not all individuals are able to achieve complete virologic suppression. Many causes for persistent viremia have been identified in previous studies, including high viral load prior to treatment, as well as drug resistance and decreased absorption of medication.8,9 In addition, other factors such as medication adherence, illicit drug use, incarceration, and unstable housing have also been implicated in virologic failure. 10 Researchers have previously investigated optimization of HIV management in circumstances where the disease is more difficult to manage (i.e. prior to advances in drug development or in geographical areas with limited resources). However, there remain questions as to what factors contribute most to virologic failure in this contemporary period, with significant resources available, along with simple single-tablet regimens. Therefore, we conducted a retrospective study to identify variables associated with persistent viremia in adult patients at an academic medical center HIV clinic.
Methods
We performed a retrospective chart review of data from the University of Michigan Health System HIV/AIDS Treatment Program (HATP) clinic. The study period was from 1 June 2013 to 31 May 2014. To identify patients with viremia, we first examined active patients evaluated at the clinic during the study period with a detectable HIV viral load greater than 200 copies/mL, thus omitting those with only transient low-level viremia (“blips”). These blips were excluded because prior research has identified that patients with low-level viremia do not have a higher rate of virologic failure or treatment resistance when compared to those who achieve complete virologic suppression.11–13 We furthermore excluded those patients who were adherent to newly initiated ART during the study period and became aviremic within the next 6 months following the study period. We then identified 1:1 matched controls for each of the viremic study patients. These controls were matched via a propensity score utilizing age at diagnosis, era of diagnosis (breakdown of years into consecutive 5-year periods), gender, and initial CD4 count. The control subjects each had viral loads <40 copies/mL for the duration of the study period.
We collected data on a number of different clinical variables. These included patient demographics, treatment course, medication/clinic adherence, comorbid medical and psychiatric conditions, personal patient beliefs (situations where a physician recommended the patient initiate ART, but the patient elected not to for personal reasons), and additional social variables. Medication adherence was assessed by clinician documentation in the electronic health record and was defined as ART adherence during 75% or more of the days of the study period. Clinic attendance was assessed by review in the electronic health record of clinic appointments scheduled and attended, not attended, or canceled. Adherence was defined as clinic attendance of 75% or more of the scheduled visits during the study period. Underinsurance was defined as those who were documented in the electronic health record as lacking medical insurance coverage during the study period.
All analyses were conducted using R 3.3.1 (R Foundation for Statistical Computing, Vienna, Austria). A two-tailed p value < .05 was considered statistically significant. Conditional logistic regression was used for unadjusted and adjusted analyses, with viremia as the binary dependent variable, and this was implemented in R using the survival package, version 2. 14 The final multivariable model was built using backwards elimination via the likelihood ratio test with a threshold α < .05 for retention of predictors. Interactions among variables in the final model were tested and included if significant.
Results
There were 745 active patients who were evaluated at the University of Michigan HATP clinic during the study period. Of them, 86 patients had a detectable HIV viral load greater than 200 copies/mL, and 20 patients were adherent to newly initiated ART during the study period and became aviremic within the next 6 months following the study period, and thus were excluded. Our final study sample included 66 viremic cases (9% of the total clinic population) and 66 matched controls. Demographics are listed in Table 1. Of the viremic cases, the average age was 38 ± 11.2 years; 74% were male and 52% were of white race. The average peak viral load in the viremic group was 143,024 copies/mL (range: 213–2,135,243 copies/mL), and the average CD4 count was 281 cells/mm3 (range: 1–536 cells/mm3). The average treatment duration was 8.2 ± 5.2 months. At the study start, only 12% of the viremic patients were on a single-table regimen (STR), while 21% were on a STR at the study end. Medication and clinic adherence were lower in the viremic population than in the control population (21% vs 62% and 23% vs 65%, respectively). The number of infectious disease clinic visits were similar between viremic and control patients (2.36 vs 2.15, respectively). Substance use was more common in the viremic population (33% vs 18%), but a psychiatric comorbidity was more frequently seen in the control population (24% vs 45%). Hepatitis and opportunistic infections occurred with similar frequency in each group.
Demographics.
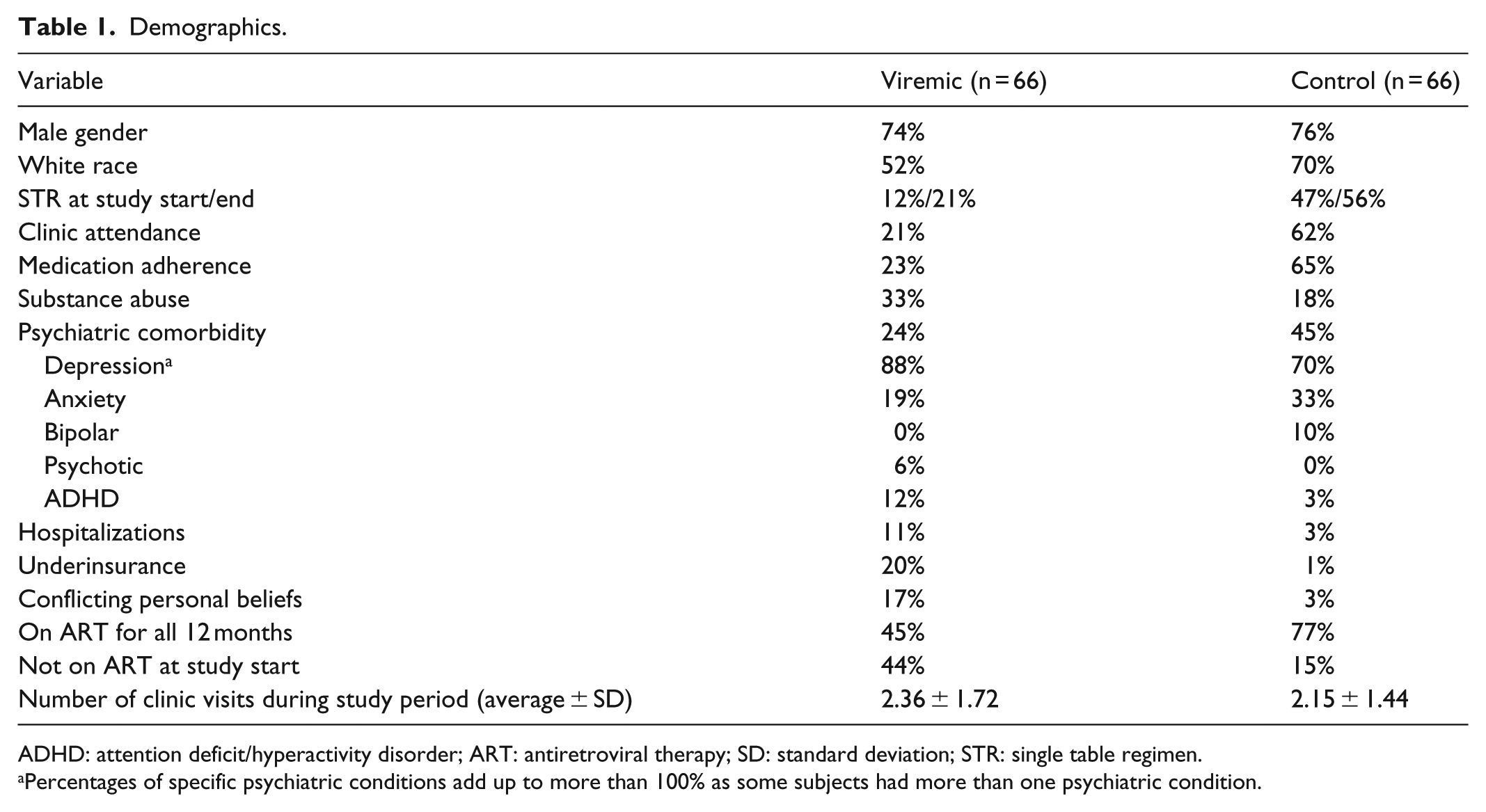
ADHD: attention deficit/hyperactivity disorder; ART: antiretroviral therapy; SD: standard deviation; STR: single table regimen.
Percentages of specific psychiatric conditions add up to more than 100% as some subjects had more than one psychiatric condition.
On unadjusted analysis (Table 2), there were fewer viremic patients on ART for all 12 months (odds ratio (OR): 0.33, 95% confidence interval (95% CI): 0.13–0.83, p = .018) and fewer were of white race (OR: 0.49, 95% CI: 0.24–1.01, p = .053). Hospitalization (OR: 10, 95% CI: 1.28–78.1, p = .028), underinsurance (OR: 5.87, 95% CI: 1.3–26.6, p = .022), and conflicting personal beliefs about their disease (OR: 5.5, 95% CI: 1.22–24.8, p = .027) were more common in the viremic population. In the multivariable analysis (Table 3), hospitalization and not being on STR at the study start was associated with viremia.
Unadjusted analysis.
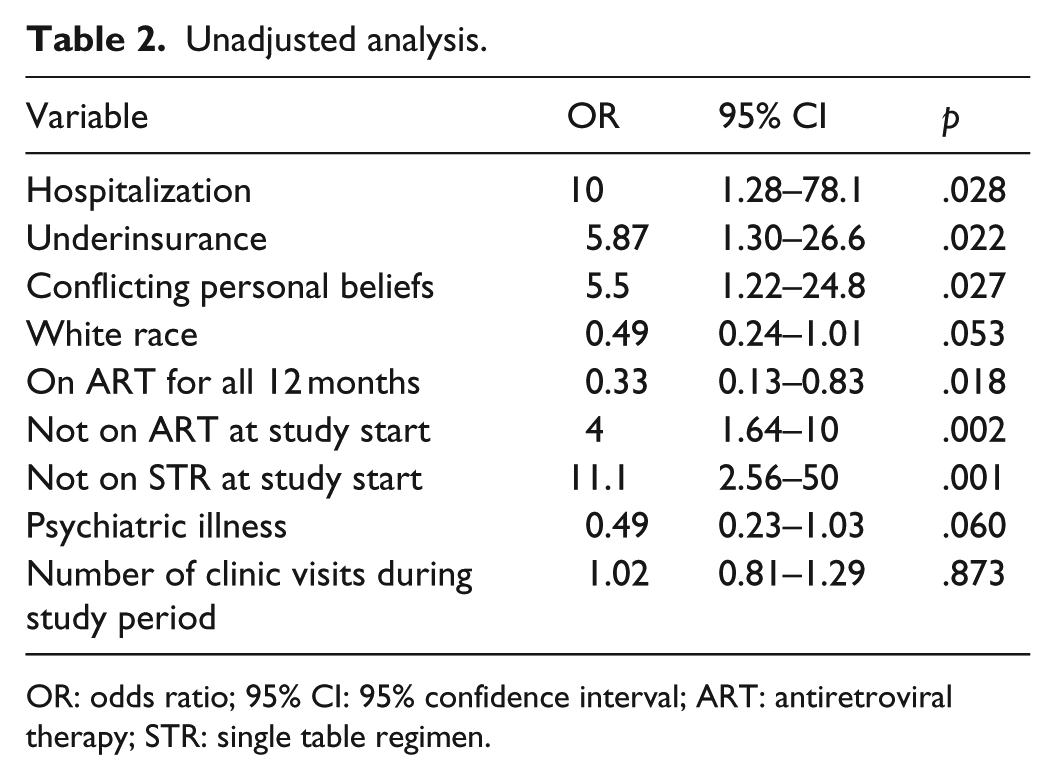
OR: odds ratio; 95% CI: 95% confidence interval; ART: antiretroviral therapy; STR: single table regimen.
Multivariable model.

OR: odds ratio; 95% CI: 95% confidence interval; SE: standard error; STR: single table regimen.
When an interaction is present, the OR cannot be calculated and significance testing cannot be interpreted for the individual terms, but only for the interaction; given the interaction between a categorical and numeric variable here, the change in OR is best presented graphically, as in Figure 1.
Notably, in the final model, a significant interaction was observed between psychiatric illness and the number of infectious disease clinic visits during the study period, complicating the analysis and the interpretation of the results (Table 3). When interaction is present, the individual variables cannot be interpreted independently. Thus, we examined how the presence of psychiatric illness is associated with viremia at various levels of clinic attendance. Though at low levels of clinic attendance (⩽3 visits), psychiatric illness appeared paradoxically protective against viremia, and this reversed to an increased odds of viremia in patients who had four or more clinic visits during the study period (p = .02; Figure 1). Specifically, an OR of 1.63 and 6.64 for viremia was seen when psychiatric illness was present among patients that also had four and five clinic visits during the study period, respectively (Figure 1). Further investigation was performed to better understand how the type of psychiatric condition associated with persistent viremia and is detailed in the “Discussion” section.

An interaction between psychiatric illness and the number of infectious disease clinic visits during the study period.
Discussion
We sought to identify the factors associated with persistent HIV viremia in adult patients at an academic medical center practice. We found possible associations of viremia with hospitalization, underinsurance, and conflicting personal beliefs about their disease/treatment on unadjusted analysis. There was a strong interaction with viremia associated with psychiatric illness and number of clinic visits.
Hospitalization can impact both the cause and the effect of viremia; however, we were unable to further delineate that relationship in this analysis. Multiple studies have demonstrated that improved ART adherence is associated with decreased risk of hospitalization.15–17 Patients with poorly controlled HIV are prone to a number of infectious and medical complications that may require hospitalization, both acquired immunodeficiency syndrome (AIDS) and non-AIDS related. 18 Furthermore, there may be other consequences of hospitalization, such as poor oral intake or newly prescribed interacting medications, which may make it difficult for patients to adhere to their ART or decrease its effectiveness.
Personal beliefs of the patients about their disease and approach to management as a barrier to treatment have also been noted in prior research. Studies have identified lower adherence rates when patients do not identify a personal need for ART. These beliefs have also been found to be difficult to change over time.19–21 Some patients do not identify a need for treatment until they begin to manifest signs or symptoms of illness, a theme common to management of chronic diseases. To overcome this obstacle, it is important to continue to foster healthy relationships between physicians and patients, provide appropriate education about the disease and management, and continue to readdress patient concerns and questions throughout the course of therapy.
The multivariable analysis revealed associations between viremia and hospitalizations, as well as not being on STR at the study start. The latter result can be explained by the assumption that being on STR at the study start confers a longer duration on targeted therapy, and thus a higher likelihood of viral suppression.
We were interested to find that psychiatric comorbidities in this sample were of a higher documented prevalence in the aviremic controls than in the viremic population, since psychiatric illness has been frequently noted in other studies to affect a significant number of HIV-infected patients. 22 A representative sample of American HIV-infected patients determined that 47% of infected subjects had a positive screen for any psychiatric disorder. 23 One Swedish cohort study also noted a higher prevalence of multiple psychiatric disorders in the HIV-infected population as compared to the non-infected population. In the male population with HIV, psychotic disorder was seen 2.9 times more frequently, bipolar disorder 3 times more frequently, and depression 3.4 times more frequently. 24 It remains possible in our study that some of the viremic patients possessed a psychiatric condition that was not yet identified, diagnosed, or documented during the time period under examination.
In the final analysis, a significant interaction was identified between psychiatric illness and number of outpatient HIV clinic visits. This was associated with decreased odds of viremia at low levels of clinic attendance, but increased odds of viremia with four or more clinic visits. Both viremic and control patients had a similar average number of clinic visits during the study period. The results of this study may indicate that as a psychiatric illness becomes more difficult to manage, it can require additional clinic visits for monitoring of multiple aspects of the patient’s complex medical care and this, in turn, increases the risk of viremia. Increased use of healthcare resources in HIV patients with mental health disorders has been documented in prior studies. 25 Furthermore, people with more psychiatric conditions may be prone to additional social issues that necessitate more frequent follow-up. The effect of improved management of psychiatric illness on viremia warrants further study, and the need for improved psychiatric management in HIV-infected patients is a topic that is supported in the literature.26–30 Appropriate and prompt management of a patient’s psychiatric comorbidities can have significant impact on patient morbidity and mortality. 31 In addition, certain antiretroviral medications, such as efavirenz, can have adverse psychiatric effects, and this must be taken into consideration when determining a suitable medication regimen for each patient.
Analysis of the breakdown of psychiatric illness subtype (anxiety, depression, bipolar disorder, psychotic disorder, and attention deficit/hyperactivity disorder) as well as psychiatric medication subtype (no medication, antidepressants, antipsychotics, benzodiazepines, and stimulants) was performed. Depression was the most common psychiatric condition in both viremic and aviremic patients (88% and 70% of the psychiatric comorbidities, respectively), and existed both by itself and in addition to the other psychiatric disorders (Table 1). However, the breakdown analysis did not show a significant association between viremia and either a particular subtype of psychiatric disorder or a medication (data not shown).
This study emphasizes that social and psychiatric factors (i.e. underinsurance, psychiatric comorbidities, and personal beliefs) play a significant role in HIV viremia. Some government-funded programs have been established to improve the health of HIV-infected patients and to provide expanded treatment to those in need. As many of these individuals do not have the appropriate social support to obtain medical care, programs such as the Ryan White Human Immunodeficiency Virus/AIDS Program (RWHAP) were developed to provide both medical and nonmedical resources to these populations in need.32–34 This allows patients without suitable financial resources and insurance coverage to receive ART treatment, reducing economic barriers. Studies have also shown that providing supplementary services, such has mental health treatment and housing assistance to help address personal and environmental obstacles to health, may improve HIV treatment outcomes. 35
It should be noted that this study does have limitations. Primarily, it consists of a small sample size of 66 patients and 66 controls, from one academic institution, over a timespan of 1 year. This potentially restricts the ability to generalize the results of this study to other centers. In addition, though the gender distribution of the patient population is consistent with Centers for Disease Control and Prevention (CDC) data of the national HIV population (with approximately three-quarters of the population male), our sample does not represent the national ethnic distribution of HIV, as our patient population is made up of a larger percentage of subjects of white race (52% vs 27% nationally).36,37 In addition, some variables (in particular, medication adherence) were determined by physician assessment in clinic documentation, which can be challenging to interpret and may be subject to underreporting.
This study provides insight into factors predictive of viremia in HIV-infected adult patients at an academic medical center, with possible associations observed between viremia and hospitalization, duration of ART, underinsurance, and conflicting personal beliefs of the patients. The effect of improved management of psychiatric illness on viremia warrants further study based on the significant interaction we detected between psychiatric illness and infectious disease clinic visits on the odds of viremia.
Footnotes
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from the Institutional Review Board of the University of Michigan Medical School (approval number/ID: HUM00093712).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was supported by grants from the Claude D. Pepper Older Americans Independence Center (grant no. AG-024824) and the Michigan Institute for Clinical and Health Research (grant no. 2UL1TR000433). The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.
Informed consent
Informed consent was not sought for this study because our IRB waived this requirement, as it was low-risk retrospective research involving chart review only.
