Abstract
Objective:
The study aimed to assess the prevalence of intestinal parasites and associated factors among people living with HIV/AIDS on antiretroviral therapy at Bule Hora General Hospital, West Guji, Oromia, Ethiopia.
Introduction:
Parasitic infection of the intestinal tract is a major source of disease in patients with HIV, particularly in the tropics, where diarrhea is a common complaint with variable severity.
Method:
An institution-based cross-sectional study was conducted from May to August 2021 on 179 systematic randomly selected HIV patients. Data on sociodemographics were collected by interviewing the study subjects using a pretested structured questionnaire. Stool specimens were collected from the study subjects and processed and examined by formol-ether concentration techniques. Data were obtained from each individual using data sheets and then entered into SPSS version 20 for analysis. The chi-square test was calculated, and a p value <0.05 was considered statistically significant.
Results:
Out of 179 study subjects, 52 (29.1%) were infected with intestinal parasites. The most frequently detected parasites were Giardia lamblia (38.5%) and Ascaris lumbricoides (25%) Entamoeba histolytica/dispar (15.4%), followed by Strongyloides stercoralis (11.5%). Factors such as the CD4+ T-cell status and source of drinking water were significantly associated with the prevalence of intestinal parasites among HIV/AIDS patients. The prevalence of intestinal parasites was highly observed among patients who scored CD4+ T cells <200 cells/mm3 90.4% (47/52) and drink ground water 73.1% (38/52).
Conclusion:
There was moderate prevalence of intestinal parasites among HIV/AIDS patients; therefore, treating unprotected water before drinking and diagnosis of early parasites for HIV/AIDS patients whose CD4+ T-cell count is less than 200 cells/mm3 should be practiced to prevent intestinal parasite infection.
Introduction
Intestinal parasite infection plays an important role in the progression of HIV infection by further distributing to the immune system. All CD4+ T-cell types, including peripheral blood macrophages, skin Langerhans cells, and brain microvillus cells, are susceptible to HIV infection. 1 Intestinal parasites induce immunological alterations that favor the progression from HIV sero conversion to AIDS. The HIV viral load was dramatically increased in people with various parasitic infections and correlated positively with a parasitic load decrease after elimination of the parasite with antiparasitic treatment. 2
Globally, approximately 3.5 billion people are infected with different intestinal parasites, and 450 million suffer from this illness. The intestinal parasitic infection prevalence is high in sub-Saharan Africa, which is similar to HIV/AIDS cases. Diarrhea causing opportunistic parasites such as Cryptosporidium parvum, Isospora belli, Giardia lamblia, Entamoeba histolytica, and helminths are common among HIV-positive persons when the CD4+ T-cell count is less than 200 cells/mm3. 3
Diarrhea, wasting, and weight loss among HIV-positive patients are remarkably high in sub-Saharan Africa due to parasitic infections associated with poverty and lack of nutrition. Opportunistic parasitic infections are common in HIV/AIDS infections. The majority (80%) of AIDS patient deaths occur due to AIDS-related infections, including intestinal parasites, rather than HIV infection itself, which usually occur later in the course of HIV infection when CD4+ T-cell count has been severely depleted, mostly below 200 cells/mm3.4–6
Ethiopia is a sub-Saharan country that is vulnerable to the high prevalence of HIV/AIDS and intestinal parasites. This is due to lower economic status and environmental hygiene. In addition, high prevalence of risk factors such as low coverage of clean water, lack of awareness, low coverage of media for teaching society about self and environmental hygiene facilities also cause high prevalence of intestinal parasites.7,8
Knowing the magnitude of intestinal parasites among HIV/AIDS patients and predictors is important to strengthen the treatment services and to develop effective preventive measures against secondary infections. However, the majority of studies done in some parts of Ethiopia on intestinal parasite among people living with HIV/AIDS identified inconsistent prevalence and predictors of parasitic infection. Moreover, epidemiological data on the intestinal parasitic infection among HIV/AIDS patients are still absent in the southern parts of Ethiopia, particularly West Guji zone. Therefore, this study was conducted to assess intestinal parasites and associated risk factors among living with HIV/AIDS who are on antiretroviral therapy (ART) at Bule Hora General Hospital.
Methods
Study design, period, and setting
A hospital-based cross-sectional study was conducted from May to August 2021 at Bule Hora General Hospital. Bule Hora is a town in southern Ethiopia located on the paved Addis Ababa–Moyale highway in the West Guji zone of the Oromia region, 471 km from central Ethiopia. It is the largest town in this zone and is mainly inhibited by Guji Oromo. It has a latitude and longitude of 5^35′N38^15′E and an altitude of 1716 meters above sea level. According to the 2007 Ethiopian Census, this zone has a total population of 27,820, of whom 14,519 are men and 13,301 are women. 9 Bule Hora General Hospital provides different health services for 1,389,821 populations, including ART and CD4+ T-cell count services for HIV patients.
Study population and selection criteria
HIV-positive patients who attended ART in the hospital during the study period were enrolled in the study. Patients who took anti-intestinal parasite drugs in the last 2 weeks were excluded from the study.
Sample size determination and sampling technique
The total sample size is calculated using the following formula: n = (Za/2)2*P (1 − P)/d2, where n is the minimum sample size needed, Z is the critical value for a given confidence interval, P is the previous study prevalence of intestinal parasites among people with HIV/AIDS, 10 d is the margin of error and 95% level of significance. Then, n = (1.96)2 × (0.176 × 0.824)/(0.05)2 = 3.8416 × 0.1450246/0.0025 = 223
The final sample size, including a 10% non-responding rate, was 179. Then, study participants were selected using convenience sampling technique.
Data collection and sample processing
The data were collected by trained BSc nurses at ART clinic. First, the patients were informed about the objectives of the study. Then, face-to-face interview was undertaken to collect data on sociodemographic and other independent variables via pretested structured questionnaire were adapted from previous literature.11–15 Then, the patients were provided the specimen containers labeled with a specific code number and applicator sticks to bring sufficient amounts of stool specimens. Stool sample consistency was determined by macroscopic observation and then examined through microscope-formed and semi-formed stools preserved by formal ether for further microscopic examinations. Macroscopically, its appearance was observed, while microscopically was used to examine the sample.
Laboratory examination
The portion of each fresh stool sample was taken and then processed to examine using the formol-ether concentration technique. In order to detect intestinal parasites, approximately 1 g or 2 mL of stool was placed using an applicator stick in a clear 10 mL conical centrifuge tube containing 7 mL of formalin saline. The resulting suspension was filtered through a sieve into another conical tube. After adding 3 mL of diethyl ether to the formalin solution, the content was centrifuged at 3200 r/min for 3 min. The supernatant was discarded and the tube was replaced in its rack. Finally, a smear was prepared from the sediment and observed under a light microscope with ×100 and ×400 magnifications. 14 Finally, blood samples collected from HIV/AIDS-infected patients from fingerstick and CD4+ T cells were counted using Pima CD4 Cartridge.
Data quality assurance
The questionnaires were pretested on 5% of the study population at Bule Hora Health Center before begin the actual data collection process to assess the clarity, understandability, and flow of each question and the time to fill the questionnaire would be assessed. The quality of normal saline and formol-ether concentration was checked by positive slides prepared from positive preserved samples. Microscopic examination of intestinal parasites as well as CD4+ T-cell count was done following manufacturer’s instruction. To avoid technical error encountered during sample collection and microscopic examination, internal quality control was performed. The data collection format of each data point was checked daily for completeness of missed or other relevant information. In addition, the formed and semi-formed stools will be preserved by formalin for further microscopic examination. The completeness of the questionnaires was checked before entering them into the software.
Statistical analysis
Data obtained from patients were encoded and then entered into SPSS version 20 for analysis. Descriptive statistics, frequency, percentage, and data were summarized using frequency tables, bar graph, and pie chart. Categorical risk factors for intestinal parasites were analyzed, and the strength of association was measured using chi-square test (χ2 test). For all analyses, a 95% confidence level and p value of <0.05 were considered statistically significant.
Ethical approval and consent to participate
Ethical approval was obtained from Bule Hora University Institutional Review Board (Ref: IRB/RC/2021). After ethical clearance, a formal letter was written to Bule Hora General Hospital. After obtaining permission, selected participants were informed, and then data collection commenced by explaining the objectives and procedures of the study to each study participant. Written informed consent was obtained from all subjects or their legally authorized representatives (in case of minor subjects/illiterate subjects) prior to study initiation. The study was conducted according to recommendations of the Declaration of Helsinki.
Confidentiality of information was kept. No references were made in oral or written reports that could link participants to research. Intestinal parasite-positive participants were reported and treated by communicating to the hospital.
Results
Sociodemographic characteristics of the study subjects
Among the study participants, 46.9% were male, and 53.1% were female. The age ranged from 4 to 74 years with a mean of 34 (±3.1) years. Concerning educational status, the majority (84.4%) of the participants were able to read and write. In the case of their occupation, most of them were merchants (21.2%), followed by (19.6%) farmers, and the residence status of the majority (57%) of study participants was in the urban area. Among respondents, most (53.6%) were married. Most of the study participants started attending ART treatment for more than a year (43.6%) (Table 1).
Sociodemographic characteristics of HIV/AIDS patients.
Stool consistency
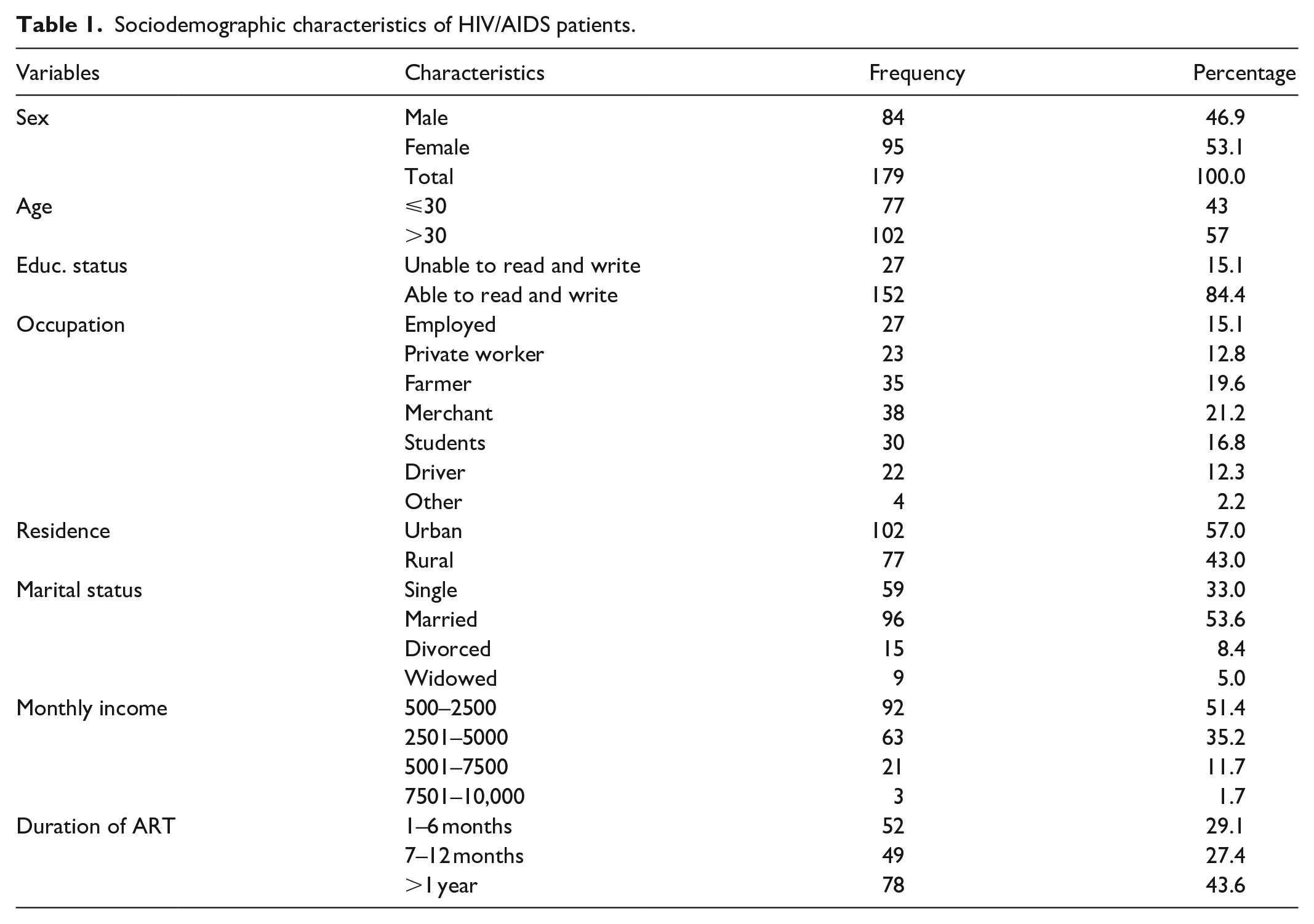
Concerning the appearance of stool, the majority of the study participants, 71 (39.7%), had formed stool, followed by loose-stool consistency, 52 (29.6%), and diarrhea, 39 (21.8%) (Figure 1).
Stool consistency of HIV/AIDS patients attend at Bule Hora General Hospital.
Prevalence of intestinal parasites
Approximately, 52 (29.1%) patients were HIV-positive for one or more intestinal parasites. The most frequently detected parasites were G. lamblia (38%), followed by Ascaris lumbricoides (25%) and E. histolytica/dispar (15.4%) (Figure 2).
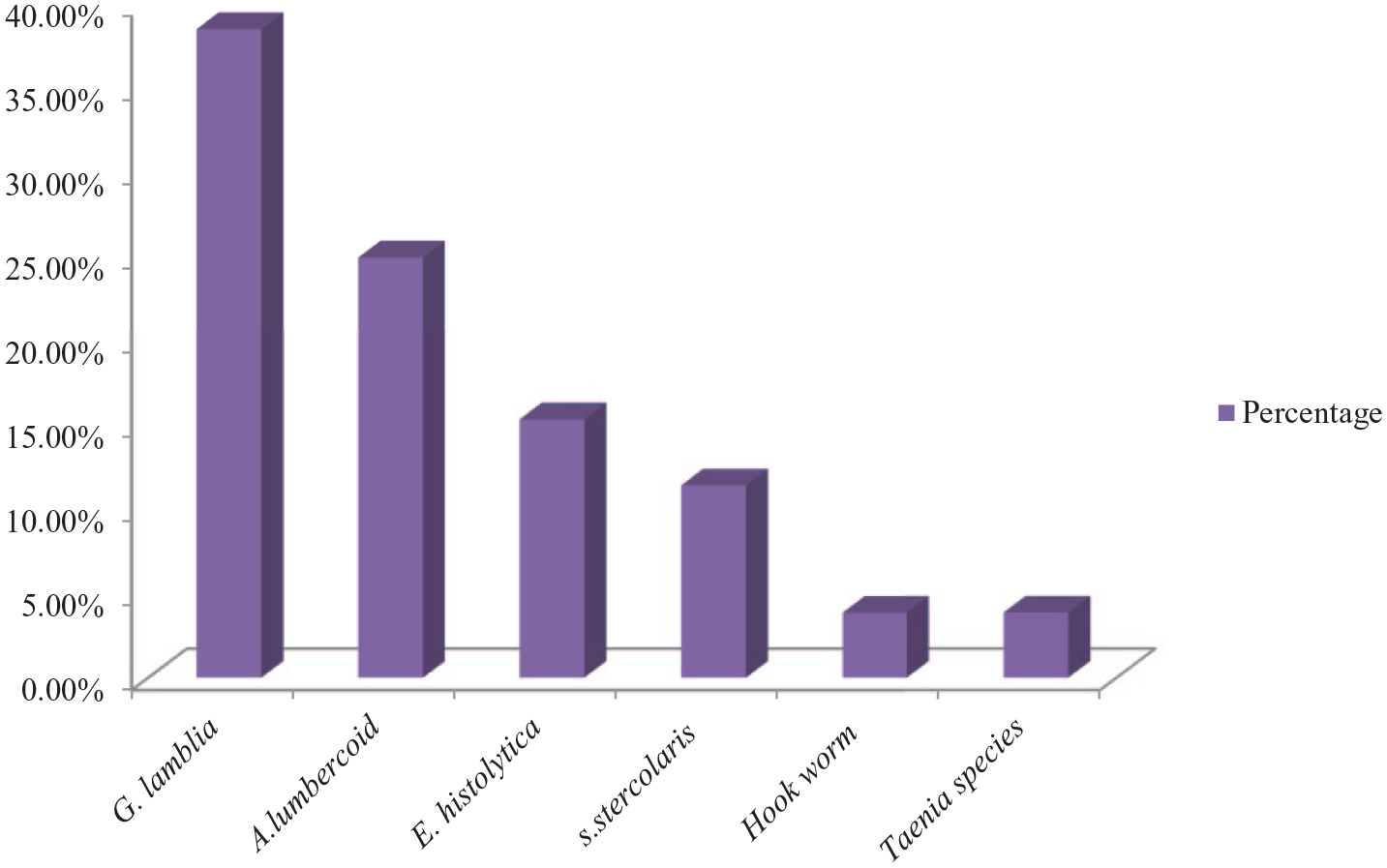
Prevalence of parasite species among HIV patients attended at Bule Hora General Hospital.
Factors associated with parasite prevalence
Chi-square test showed that the source of drinking water (χ2
Factors associated with parasite prevalence among people living with HIV/AIDS on ART attended at Bule Hora General Hospital, Oromia, Ethiopia 2021.

Statistical significance at ***p < 0.001; **p < 0.01; *p < 0.05 and χ2—chi-square.
Discussion
This study determined the prevalence of intestinal parasites among HIV-infected patient participants to assess whether a trend of occurrence was evident. The study attempted to provide information regarding HIV patients. In our study, the overall magnitude of intestinal parasites was 29.1% (52/179), which was almost equal to that in a study conducted in south India (28.9%); 16 Integrated Counselling and Testing Centre, India (27.6%); 17 Hiwot Fana, Eastern Ethiopia (28.6%) 11 and Bahir Dar (30.6%). 12 However, the findings of this study were higher than those of a study conducted in Nigeria (11.4%) 18 and Dessie Hospital (17.6%), 10 Gonder Hospital (20%), 14 and Debre Tabor General Hospital (25.3%). 13 The differences could be due to variations in sociodemographic characteristics, sample size, and time period. In addition, an implementation of relevant health intervention programs such as deworming, vaccination, and improvement in health service coverage with the active involvement of health extension workers in some places might have contributed. 13
In contrast, our finding was lower than the studies conducted in Cameroon (82.3%), 19 Southwest Ethiopia (44.8%), 20 Hawassa Teaching Hospital (55%), 21 Jimma Teaching Hospital (52.6%), 8 Gambi Bahir Dar (69%), 22 Hawassa University Hospital (35.8%), 23 and Nekemte Specialized Hospital (73.3%). 15 The possible reason for this might be the long-term data collection time, large sample size, and difference in test method. However, in those studies, female and urban dwellers were the more predominant participants of the study.
In addition, the most frequently identified intestinal parasite was G. lamblia, with 38.5%, which was similar to study conducted in Nekemte Specialized Hospital (35%) 15 However, this finding was higher than the 16% reported in the studies conducted in Adama, Afar, and Dire Dawa, Ethiopia; 3 3.8% in Jimma, Southwest Ethiopia; 8 8.1% in Hiwot Fana, Eastern Ethiopia; 11 4.3% in Bahir Dar, 12 and 7.9% in Hawassa University Hospital. 23 This may be due to climatic conditions, drinking unprotected water, and living standards of the population; therefore, it can be prevented through water treatment before drinking or use for other purposes.
In this study, the drinking ground water was significantly associated (p < 0.05) with the prevalence of intestinal parasites among HIV/AIDS patients. This finding agreed with a study conducted in Nekemte Specialized Hospital 15 and Gonder Hospital. 14 The possible explanation for this is unprotected water source favor parasite growth which can transmit through drinking water and easily infect immunocompromised HIV/AIDS patients. 15
This study revealed that the status of CD4+ T-cell count was significantly associated (p < 0.05) with the prevalence of intestinal parasites among HIV/AIDS patients. According to this study, HIV/AIDS patients with CD4+ T-cell count less than 200 /mm3 were more likely to be infected with intestinal parasites. This finding agreed with a study conducted in Cameroon, 19 Dessie Referral Hospital, Northeast Ethiopia, 10 Gonder Hospital, 14 and Hiwot Fana, Eastern Ethiopia. 11 HIV/AIDS usually diminishes CD4+ T-cell count which plays a major role in the host defense mechanism against intestinal parasitic infection, so that the individual loses the ability to defend their body and activation from such parasites may affect the progression of HIV/AIDS. 13
Limitations of this study
One of the limitations of this study was the nature of cross-sectional study design which may not show cause–effect relationship. Since chi-square test was applied for analysis-associated factors, the confounder may interfere the finding of the study. Due to small sample size used, the finding could not be generalized to determine the exact prevalence of intestinal parasites among the general population in the area.
Conclusion
In this study, there was moderate prevalence of intestinal parasites among HIV-infected patients. The risk factors significantly associated with intestine prevalence were source of drinking water and CD4+ T-cell status. The prevalence of intestinal parasites was higher among patients who scored CD4+ T cells < 200 cells/mm3 and patients those who drink ground water (73.1%). Therefore, groundwater should be treated before drinking and diagnosis of early parasites should be performed for HIV/AIDS patients whose CD4+ T-cell count is less than 200 cells/mm3 to prevent intestinal parasite infection. Large-scale longitudinal studies assessing all associated factors and continuous surveillance should be conducted to halt the discrepancies and routine examination of stool samples for intestinal parasites. Health education should be given to HIV/AIDS patients to ensure good personal hygiene and environmental sanitation.
Supplemental Material
sj-docx-1-smo-10.1177_20503121221124685 – Supplemental material for Assessment of intestinal parasites and associated factors among HIV/AIDS patients on antiretroviral therapy at Bule Hora General Hospital, West Guji, Ethiopia
Supplemental material, sj-docx-1-smo-10.1177_20503121221124685 for Assessment of intestinal parasites and associated factors among HIV/AIDS patients on antiretroviral therapy at Bule Hora General Hospital, West Guji, Ethiopia by Alqeer Aliyo and Tibeso Gemechu in SAGE Open Medicine
Footnotes
Acknowledgements
First, we would like to express our heartfelt gratitude to the Bule Hora University College of Health and Medical Science Office for their permission to conduct our research. We would also like to express our heartfelt gratitude to Henok tesfaye, Kefalew gebeyhu, and Arsema Reta for their contributions. Last but not least, for all of you who are with us during this journey, we would like to thank you.
Author contributions
Alqeer Aliyo: Conceptualization, data curation, formal analysis, funding acquisition, investigation, methodology, project administration, resources, software, supervision, validation, visualization, writing original draft, writing review and editing. Tibeso Gemechu: Conceptualization, formal analysis, investigation, methodology, software, supervision, validation, writing original draft, writing review, and editing.
Availability of data and materials
Data essential for the conclusion are included in this article. Additional data can be obtained from the corresponding author upon reasonable request.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study was obtained from *Bule Hora University Institutional Review Board (Ref: IRB/RC/2021)*.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from all subjects or their legally authorized representatives (in case of minor subjects/illiterate subjects) prior to study initiation.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

