Abstract
Objective:
We aimed to identify and represent factors associated with thrombocytopenia in intensive care unit, especially the pathogens and drugs related to severe and extremely thrombocytopenia. Then, we aim to compare the mortality of platelet transfusion and non-transfusion in patients with different degrees of thrombocytopenia.
Methods:
We identified all thrombocytopenic patients in intensive care unit by using platelet-specific values and then extracted electronic health records from our Hospital Information System. Data were statistically analyzed with t test, chi-square test, and logistic regression.
Results:
We found that infections (32.7%) were the most frequent cause associated with thrombocytopenia, followed by sepsis shock (3.93%) and blood loss (2.99%). Meanwhile, antifungals (p = 0.002) and bacterial infection (p = 0.037) were associated with severe and extremely severe thrombocytopenia. Finally, we found that the mortality of platelet transfusion and non-transfusion in patients was statistically significant for patients with platelet counts between 30 and 49/nL (χ2 = 9.719, p = 0.002).
Conclusion:
Infection and sepsis emerged as two primary factors associated with thrombocytopenia in intensive care unit. Meanwhile, antifungals and bacterial infection were associated with platelet counts less than 49/nL. Finally, platelet transfusion may be associated with reduced mortality in patients with platelet counts between 30 and 49/nL.
Introduction
Thrombocytopenia is one of the most common laboratory abnormalities in the intensive care unit (ICU) patients. 1 The reported incidence of thrombocytopenia is between 13% and 60%.2–7 It has been associated with an increased risk of blood product transfusions, bleeding, length of stay, and mortality.8–11 Thrombocytopenia can be a result of decreased platelet production, increased destruction, increased aggregation, dilution, and sequestration. 12 The cause of a low platelet count in ICU may be difficult to determine and is often multifactorial.
In clinic, thrombocytopenia caused by various infections and drugs is not uncommon, but mostly recovers with the control of the disease. Infection-related thrombocytopenia is a thrombocytopenia caused by viruses, bacteria, rickettsia, mycoplasma, fungi, and other pathogens. The main causes of thrombocytopenia are the direct destruction of platelets by pathogens, the influence of platelet production, and the increase of platelet consumption. Among the causes of thrombocytopenia, drugs are often suspected when the underlying causes of thrombocytopenia have been excluded. It has been estimated that 10%–25% of thrombocytopenia cases in critically ill patients may be caused by drugs.13,14 Drugs can induce thrombocytopenia through various mechanisms, including drug-dependent antibodies, myelosuppression, and impaired platelet production. 15 Although many studies have assessed risk factors for thrombocytopenia in critically ill patients, few have included infection and medications in their analyses.2,3,16 More importantly, these studies have included a small number of thrombocytopenia cases, of which 25 and 68 cases were evaluated.
Platelet transfusion is common, with 9%–30% of all ICU patients receiving platelets in the ICU.17,18 The benefits of platelet transfusion and the effect of platelet transfusion on mortality in patients with different degrees of thrombocytopenia are still controversial.19,20 Consequently, we wished to answer those questions to some extent.
The purpose of this study was to identify and describe factors associated with thrombocytopenia, especially the pathogens and drugs related to severe and extremely thrombocytopenia. Then, we aim to compare the mortality of platelet transfusion and non-transfusion in patients with different degrees of thrombocytopenia.
Methods
Patients
We retrospectively collected data from all patients admitted between June 2014 and November 2018 to ICU of the Aerospace Central Hospital, a tertiary hospital in Beijing. We only collected data of patients during their stay in ICU. This was an observational study. All collected data were anonymized. We identified all thrombocytopenic patients by extracting electronic health records from our Hospital Information System. Thrombocytopenia was defined as a platelet count of <150 × 109/L 21 at any time during admission. Exclusion criteria were (1) pregnancy and lactation, (2) those who had organ transplants, (3) age less than 18 years old, (4) patients with incomplete clinical data, and (5) transfer to ICU for less than 24 h. Patients with thrombocytopenia were divided into four groups according to the lowest platelet count during their stay in ICU, and classified as mild (platelet count: 100–149 × 109/L), moderate (platelet count: 50–99 × 109/L), severe (platelet count: 30–49 × 109/L), or extremely severe (platelet count: <30 × 109/L) according to standard classification. 22 The flow diagram of patient inclusion was shown in Figure 1.

Flow diagram of patient inclusion.
Data collection
The following data were collected from patient medical electronic records: gender, age, etiology, source of infection, comorbidities, microbial organism, medications, bleeding, diagnosed by the final International Classification of Diseases (ICD) code and the use of hemostatic drugs, hospitalization expenses for the ICU stay, length of stay in ICU, and ICU mortality. Associated factors with thrombocytopenia were judged independently by two of the authors (M.-k.Z., Resident and X.-x.H., Attending Physician) using a standard list of factors form. Disagreements were resolved by discussion or consensus with a third reviewer (X.-y.X., Chief Physician). Exposure to the following medication classes was evaluated because of the previous implication in thrombocytopenia and frequent use in the ICU: nonsteroidal anti-inflammatory agents (i.e. naproxen, ibuprofen, diclofenac, and indomethacin), anticonvulsants (i.e. carbamazepine, phenytoin, and valproic acid), antiplatelet agents (i.e. aspirin and clopidogrel), anticoagulant (i.e. heparin and low-molecular-weight heparin), quinolones (i.e. levofloxacin, ciprofloxacin, and moxifloxacin), H2 antihistamines (i.e. famotidine and ranitidine), penicillins (i.e. ampicillin, penicillin, and amoxicillin), cephalosporins (i.e. ceftazidime, cefazolin, and cefuroxime), and antifungals (i.e. amphotericin B and fluconazole). 23
Statistical analysis
Statistic analysis was performed using the SPSS software version 22 and GraphPad Prism version 5.01 (GraphPad Software, San Diego, CA, USA). Measurement and numeration data were statistically analyzed with t test and chi-square test, respectively. Multivariable logistic regression models were used to find out the pathogenic microorganism drugs associated with severe and extremely severe thrombocytopenia (platelet count < 49 × 109/L). In all tests, the p value < 0.05 was accepted as significant.
Results
General data
The demographics and clinical characteristics of the study population are displayed in Table 1. Of the total patients screened, 737 patients were included in the study.
Demographics and clinical features.

ICU: intensive care unit.
Associated factors with thrombocytopenia
Table 2 lists the associated factors with thrombocytopenia as judged according to the specific situation of patients by treatment teams. In 737 patients, multiple factors associated with thrombocytopenia. Infection was the most common factor (32.7%), followed by sepsis shock (3.93%) and lose blood (2.99%). Among the associated factors of the hematological diseases, the top three factors were acute leukemia, hemolytic anemia, and myelodysplastic syndromes. In 18 patients, no factor was clinically apparent associated.
The associated factors with thrombocytopenia.

MODS: multiple organ dysfunction syndrome.
Pathogens associated with thrombocytopenia
Since infection was the primary associated factor, we analyzed the pathogenic microorganism and distribution of infection sites of the infection. Pathogens were identified from clinical specimens (including blood, sputum, puncture drainage, excrement, urine) and antibody IgM or virus quantities of venous blood-associated viruses. Distribution of specific pathogens in patients with infection-associated thrombocytopenia was shown in Table 3. The logistic regression analysis of pathogens associated with severe and extremely severe thrombocytopenia was shown in Table 4. Bacterial infection was significantly associated with increased odds of platelet counts less than 49/nL (p = 0.037). Distribution of infection sites in patients with infection-related thrombocytopenia was shown in Table 5. Pulmonary infection occurred in 53.19% was the most common infection of all patients.
Distribution of pathogens in patients with infection-associated thrombocytopenia.

HIV: human immunodeficiency virus.
The multivariate logistic regression analysis of pathogenic microorganism associated with severe and extremely severe thrombocytopenia (platelet count: <49 × 109/L).
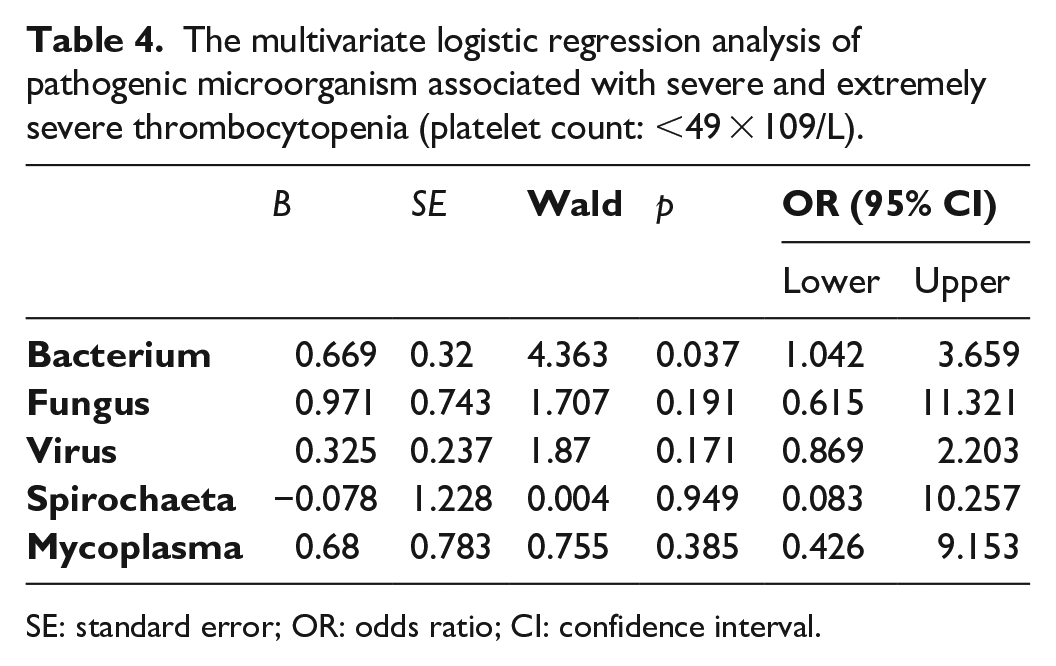
SE: standard error; OR: odds ratio; CI: confidence interval.
Distribution infection site of patients with infection-associated thrombocytopenia.

Drugs associated with severe and extremely severe thrombocytopenia
It has been estimated that 10%–25% of thrombocytopenia in critically ill patients may be caused by drugs. In the logistic regression analysis, the drug with a statistically significant correlation with severe and extremely severe thrombocytopenia (Table 6) was antifungals (p = 0.002).
Multivariate regression analysis of drugs significantly associated with severe and extremely severe thrombocytopenia (platelet count: <49 × 109/L).

SE: standard error; OR: odds ratio; CI: confidence interval.
Platelet transfusion
We compared the mortality of platelet transfusion and non-transfusion in patients with different degrees of thrombocytopenia, and the mortality between the two groups was statistically significant for patients with platelet counts between 30 and 49/nL (χ2 = 9.719, p = 0.002). See Table 7.
The mortality of platelet transfusion and non-transfusion in patients with different degrees of thrombocytopenia.

Discussion
Thrombocytopenia in critically ill patients is usually multifactorial. In our study, infection and sepsis emerged as two primary factors. It has been recognized that thrombocytopenia might be an early warning sign of sepsis. 24 The mechanism of thrombocytopenia induced by sepsis is not fully understood and may include disseminated intravascular coagulation (DIC), 25 immune-mediated platelet destruction, and hemophagocytosis. 26 Since infection was the primary associated factor, we analyzed the pathogens associated with severe and extremely severe thrombocytopenia. We found that bacterial infection was significantly associated with increased odds of severe and extremely severe thrombocytopenia. Bacteria or bacterial products may cause endothelial damage, leading to platelet adhesion and aggregation, or may bind directly to platelets leading to aggregation and accelerated clearance from circulation.27,28 There may be a possibility of immune mechanism for development of thrombocytopenia in septicemia as there is the presence of circulating immune complex in septicemic patients and reduced number of complement complex in patients with septicemic shock.29,30 This indicates that thrombocytopenia may be considered an early but nonspecific indicator of septicemia but other causes of septicemia should also be ruled out. Therefore, in ICU patients with thrombocytopenia, we should first consider the most common cause of bacterial infection and actively seek possible infection sites and pathogens, conducting corresponding treatment at the first time. In our predominant patients with hematological diseases, the contribution of thrombotic thrombocytopenic purpura, hemophagocytosis, and myeloproliferative tumor to the incidence of thrombocytopenia was modest. In some patients, as in other ICU-based studies, 6 there was no significant associated factor with thrombocytopenia. This reflects that although thrombocytopenia is a hematological system–related laboratory abnormality, it may be more likely to indicate non-hematological system diseases such as infection in patients of ICU.
Many drugs commonly used in the ICU can result in thrombocytopenia, and most patients in ICU are receiving a large number of different drugs. However, drug-induced thrombocytopenia does not seem to be important in these patients. In our research, we found that antifungals were associated with platelet counts less than 49/nL on logistic regression analysis. Due to the particularity of fungal infection, although we have obtained results with statistical difference, we still believe that thrombocytopenia associated with antifungals is that patients are severely immunosuppressed. This may be due to severe illness or hematological disease. Therefore, the infection and severity of illness could be the cause of thrombocytopenia, rather than the antifungals themselves. The low incidence of drug-induced thrombocytopenia and the high prevalence of confounding factors such as sepsis, DIC, severely immunosuppressed, or bleeding make the diagnosis of drug-induced thrombocytopenia difficult in the ICU setting. Therefore, a lower suspicion threshold is needed to prevent the underdiagnosis of drug-induced thrombocytopenia.
Since there is no fixed platelet count threshold to indicate platelet transfusion in ICU patients, so we compared the mortality of platelet transfusion and non-transfusion in ICU patients with different degrees of thrombocytopenia. The mortality in severe thrombocytopenia between the two groups was statistically significant; this suggests that platelet transfusion may be associated with reduced mortality in patients with platelet counts between 30 and 49/nL. However, previous studies suggest a platelet count from 10–20 × 109/L, with one suggesting no.31–34 It may be that these studies focus on hematology tumor patients, trauma patients, and myelosuppression patients, and may lack ICU data from the real world. This reminds us that clinical decision to transfuse platelets to an individual patient should take into account the relative risks and benefits.
This study has some limitations. First, since this was a retrospective real-world study, we could not control for all confounding factors that could have been present in these ICU patients. Second, the number of research patients was small and non-thrombocytopenic patients in the cohort. However, we believe that our results would be credible since such studies may be hard by a prospective or a national claims data analysis. Third, thrombocytopenia was divided into four groups according to the lowest platelet count, which may affect our conclusion by converting continuous variables into categorical variables. Then, we did not calculate and justify the sample size selected in this study. However, we made sure that the number of people included in the study was 10 times that of the variables in the multivariate regression analysis. Finally, the present conclusions were derived from our hospital, mainly medical ICU, and should be confirmed in other environments.
Conclusion
Among the multiple factors associated with thrombocytopenia, infection and sepsis emerged as two primary factors. Meanwhile, antifungals and bacterial infection were associated with platelet counts less than 49/nL. Finally, platelet transfusion may be associated with reduced mortality in patients with platelet counts between 30 and 49/nL.
Footnotes
Author contributions
All authors were involved in the evaluation and management of the patient, collected and analyzed data, and have read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical approval for this study waived by the Institutional Review Board Committee of the Aerospace Central Hospital due to the retrospective nature of the study. The waiver number was 20190730-ST-12.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Considering the retrospective nature of the research, the informed consent requirement was waived by the Institutional Review Board Committee of the Aerospace Central Hospital.
Trial registration
This randomized clinical trial was not registered because the retrospective nature of the study.
