Abstract
Objective:
Predictors of mortality in severe traumatic brain injuries (TBI) in pediatric patients are not well explored. We investigate traditional and novel prognostic predictors in pediatric patients with severe TBI. The primary outcome was in-hospital mortality.
Methods:
A retrospective study of children ⩽18 years old with a Glasgow Coma Scale ⩽8 was conducted at a level I pediatric trauma center. Predictors of mortality included interventions within 72 h, Pediatric Risk of Mortality (PRISM) III at 24 h, and the pediatric Sequential Organ Failure Assessment (pSOFA) score over 3 days. Novel prognostic models included a combined PRISM III and pSOFA score, a model that combined the two prognostic scores with medical interventions, and a combined medical and surgical intervention model. Categorical and continuous variables were analyzed using Fisher’s exact test and the Mann-Whitney U test, respectively. Receiver operating characteristic curves and area under the curve (AUC) were used to assess the predictive performance of prognostic scores.
Results:
There were 97 patients with severe TBI included in this study. The combined PRISM-pSOFA day 2 model was the strongest predictor of mortality (AUC = 0.973), but did not provide a statistically significant benefit over PRISM III, which served as the referent model. While the PRISM III (AUC = 0.968) had a higher predictive ability than the pSOFA-day 2 (AUC = 0.940), the difference was not statistically significant.
Conclusion:
Predicting outcomes is essential for guiding care decisions and standardizing research. The pSOFA-day 2 score offers a comparable mortality prediction to PRISM III and serves as a practical, easy alternative that can be especially helpful when the PRISM III score is unavailable or difficult to calculate due to resource limitations.
Keywords
Introduction
Outcomes in pediatric patients with severe traumatic brain injuries (TBIs) range from recovery with minimal impairment to varying degrees of neurological deficits that, in some cases, progress to brain death. 1 Such prognostic uncertainty presents challenges for healthcare professionals when addressing goals of care with families. Objective and easily implemented prognostication tools can aid bedside clinicians in risk stratification of pediatric patients with severe TBI. Describing patterns of medical interventions during the initial presentation can also aid prognostication.
While prognostic scores for TBIs have been studied extensively in the adult population, they are lacking in pediatrics.2,3 The most recognized pediatric score, the Pediatric Risk of Mortality III (PRISM III) score, can be useful in TBIs, but it is inconsistently implemented due to complexity in scoring.4,5 In addition, the PRISM III’s predictive value declines beyond the first 24 h of admission, further limiting its utility. The pediatric Sequential Organ Failure Assessment (pSOFA) score, another validated tool with good ability to discriminate risk of in-hospital mortality, has not been thoroughly explored in pediatric TBIs.6–8 The daily pSOFA can predict in-hospital mortality beyond 24 h and demonstrates better predictive value within days 2 and 4 of hospitalization. 9 Unlike the PRISM III, the pSOFA score is also easier to implement at the bedside and therefore has more practical application. The utility of prognostic tools extends to the fact that they also characterize patients beyond their neurological injuries. Assessing a broader range of physiologic and biochemical parameters helps evaluate how other organ systems contribute to the overall in-hospital mortality of TBI patients. 10 These prognostication tools can help guide physicians and families in making informed decisions regarding goals of care.
The primary aim of our study was to characterize prognostic scores among pediatric patients with severe TBI who survived compared to those who expired. We developed novel prognostic models based on medical and surgical interventions and combining the PRISM III and pSOFA scores. Alongside the PRISM III score and the daily pSOFA score, these novel scores were developed to better discriminate risk of in-hospital mortality.
Materials and methods
Study design
We performed a retrospective descriptive study of children with severe TBIs admitted to a level I trauma center pediatric intensive care unit (PICU) between July 2012 and July 2022. Our study included all patients less than 18 years of age who met criteria for severe TBI, defined by Glasgow Coma Scores (GCSs) of eight or less. Patients were excluded if their injuries were primarily due to hypoxic injuries, infectious etiologies, drug use or toxin ingestion, tumors, or mitochondrial encephalopathies. Data obtained within the first 72 h of admission to the hospital were collected from electronic health records.
Ethical approval
The study was approved by the Institutional Review Board at the University of Texas Health Science Center at Houston (IRB# HSC-MS-22-0431). This was a retrospective study; therefore, written informed consent was waived by the IRB.
Study variables
Extracted data included: demographics, interventions (medications used, procedures done, imaging performed), and prognostic scores (PRISM III score on day 1, vasoactive inotrope score (VIS) on day 1, daily pSOFA score from day 1 to 3, and Injury Severity Score (ISS) on day 1). 11 All medication and procedure data were also collected within the first 72 h of admission to the PICU. The primary outcome was in-hospital mortality, which included both cardiac and brain death. Missing variables were assumed to be within normal limits if no value was recorded during the specified period. This process was consistent with the methodology used by the PRISM III and pSOFA scoring system.5,6,8 Such an approach is commonly employed in prognostic models and allows comparability with prior studies.
Study analysis
Demographic data, interventions, and organ dysfunction scores were compared to our primary outcome. Categorical variables were reported as frequency (with percentage) and compared using chi-square tests. Continuous variables were reported as median (with interquartile range (IQR)) and compared using Mann-Whitney tests. Logistic regression models were run to assess whether the addition of multiple scores and/or other variables could provide improved mortality prognostication than provided by the scores on their own. Post-regression estimates for predicted mortality were calculated and compared to the observed mortality using the Hosmer-Lemeshow goodness-of-fit test. Receiver operating characteristic (ROC) and area under the curve (AUC) were calculated to assess the predictive ability of prognostic scores. To control potential overfitting of the ROC-AUC models, optimism-controlled bootstraps were performed with 500 replications for each prognostic tool. Each replication drew 96 patients with replacement and fit a single predictor logistic regression model on the “in-bag” sample, followed by computation of AUC on the remaining “out-of-bag (OOB)” patients (average n ≈ 36). The models guarded against edge cases where the OOB set lacked one outcome class. Statistical significance was assumed at p-value <0.05. Analysis was performed using Stata (v.17, College Station, TX, USA).
Results
Demographic
Ninety-seven patients with severe TBI were identified during the study period. The population had a slight preponderance of males (n = 59, 61%) (Table 1).
Demographic data and medical history data for all patients with traumatic brain injury (n=97).

IQR: interquartile range.
Data reported as frequency (with percentages within each characteristic) unless noted.
Clinical course in PICU and hospital
Fifteen patients (15%) experienced a cardiac arrest, with nearly all of them (n = 13, 87%) experiencing the event outside the hospital. The mortality rate in infants, children, and adolescents was 36% (n = 4), 28% (n = 18), and 27% (n = 6), respectively (p = 0.887) (Table 2). Most of these patients experienced death by neurologic criteria (n = 19, 83%), while the rest (n = 4, 17%) experienced a cardiac death within 72 h of admission. Only one patient in the cohort had a Do Not Recuscitate (DNR) status during admission, who eventually expired on day 2.
Comparison of clinical parameters within 72 h post-admission between patients who died (N = 28) and those discharged (N = 69).

TBI: traumatic brain injury; ED: emergency department.
Percentages denote relative frequencies across rows (i.e., discharge vs death) for each stratum unless otherwise specified.
Association of clinical interventions and mortality
Just over a quarter of the patients (n = 28, 29%) died during their hospitalization. Mortality was highest among patients with a TBI secondary to assault (67%) compared to those who suffered a TBI secondary to a motor vehicle collision (30%) (p = 0.001) (Table 2).
Vasopressor administration was significantly higher in non-survivors (89%) compared to those discharged (19%) (p = 0.001) (Table 3). Similarly, administration of blood products and vasopressin for diabetes insipidus was significantly higher in non-survivors (89% and 43%, respectively) compared to those discharged (28% and 4%, respectively) (p = 0.001). Comparatively, sedative use was more prevalent among patients who were discharged (90%) compared to those who died (61%) (p = 0.002). Abdominal surgery was also more common among non-survivors (29% vs 4%, p = 0.002).
Comparison of interventions within 72 h post-admission between patients who died (N = 28) and those discharged (N = 69).

Percentages denote the number of patients in each column (discharge or death) that were exposed to each intervention. The specific interventions are not mutually exclusive.
p-value of comparison of the frequency of each intervention (row) between the two patient outcomes.
Data reported as median and interquartile range (IQR).
Clinical scores in PICU
Patient data at admission were used to calculate the PRISM III, pSOFA (pSOFA-adm), GCS, ISS, and VIS scores. The PRISM III and pSOFA-adm were high on admission with median scores of 11 (IQR: 8–25) and 8 (IQR: 6–12), respectively. The median GCS score was 3 (IQR: 3–3) while the median ISS was 25 (IQR: 16–34).
Sub-stratification of the pSOFA score into individual components showed that compared to 48 hrs, most patients had severe dysfunction in the neurological (score 4 in 84% of patients) and respiratory (score 4 in 48% of patients) components on admission (Table 4). The pSOFA-day 2 (median: 8, IQR: 6–12) and pSOFA-day 3 (median: 6, IQR: 1–9) scores were calculated for all patients if alive at day 2 or day 3, respectively.
Number of patients in each score category for pSOFA components at admission (N = 97) and at 48 h post-admission (N = 89).

pSOFA: pediatric Sequential Organ Failure Assessment; pSOFA-adm: pSOFA score at admission.
Association of prognostic scores and mortality
The pSOFA, PRISM III, and the sub-stratification of the PRISM III scores at admission demonstrated that higher scores were associated with increased mortality when compared to scores in patients who were discharged (Table 5). The sub-stratification of the PRISM III included vital signs score (GCS and pupillary reflex), biochemical score (glucose, creatinine, and blood urea nitrogen), and the hematological score (white blood cell count, platelets, and prothrombin time). The median ISS was significantly higher in patients who died (median: 34; IQR: 26–43) compared to the ISS among patients who survived and were discharged (median: 21; IQR: 14–26; p < 0.001) (Table 5). The pSOFA-day 2 and pSOFA-day 3 scores also showed statistically significant associations with mortality.
Comparison of the distribution of prognostic scores and the change in pSOFA scores from admission to day 2 and day 3 between patients who died (N = 28) and those eventually discharged (N = 69).

pSOFA: pediatric Sequential Organ Failure Assessment; adm: admission; PRISM: Pediatric Risk of Mortality; IQR: interquartile range; ΔpSOFA-2: difference in pSOFA from admission to day 2; ΔpSOFA-3: difference in pSOFA from admission to day 3.
Data based on N = 89 patients that had data for day 3.
The change in pSOFA scores from admission to day 2 (ΔpSOFA-2) and admission to day 3 (ΔpSOFA-3) were also calculated (Table 5). These scores demonstrated a greater improvement (i.e., a negative change in scores) at both 48- and 72-h post-admission (p < 0.001 and p = 0.008, respectively) among patients that got discharged compared to those who died (Table 5). When the ΔpSOFA from day 1 to 2 was positive (i.e, score increased or worsened) or when the change from day 1 to 3 was ⩾ −1 (i.e., decreased by no more than one unit or increased), the change in the score was associated with death. On the contrary, if ΔpSOFA by day 3 was negative (i.e., decreased) with a magnitude larger than one, this was more likely to be associated with a patient who was discharged. These thresholds were empirically derived from the observed distribution of score changes over the time frame studied.
Comparison of prognostic scores
Multivariable logistic regression models were run to assess whether the addition of multiple scores and/or other variables could provide improved mortality prognostication than provided by the scores on their own. Although certain clinical variables (administration of vasopressors, sedatives, and/or blood products) were associated with mortality in crude univariable logistic regression, the addition of these variables into multivariable models did not yield any improvement in the ability of the PRISM III score or the pSOFA to predict mortality.
Additional multivariable models were run to assess the association between mortality and various prognostic scores (PRISM III, pSOFA- adm, pSOFA-day 2, vital signs scores, biochemical score, hematological score, and/or ISS). Although independently associated with mortality on their own, the other prognostic scores were not independently associated with mortality when analyzed in combination with the PRISM III or pSOFA-day 2 score (Table 6). Both the PRISM III and pSOFA-day 2 score provided independent prognostication, suggesting that together they may be able to provide improved prognostication of mortality past 48 h if used in combination. Although an independent predictor on its own, ISS did not act as a confounder or effect modifier on its own in models where it was included along with PRISM III and/or pSOFA-day2 (likelihood ratio test, p-values >0.05).
Independent associations of various scores with mortality based on univariable (crude) logistic regression model and three multivariable logistic regression models that included various combinations of scores.
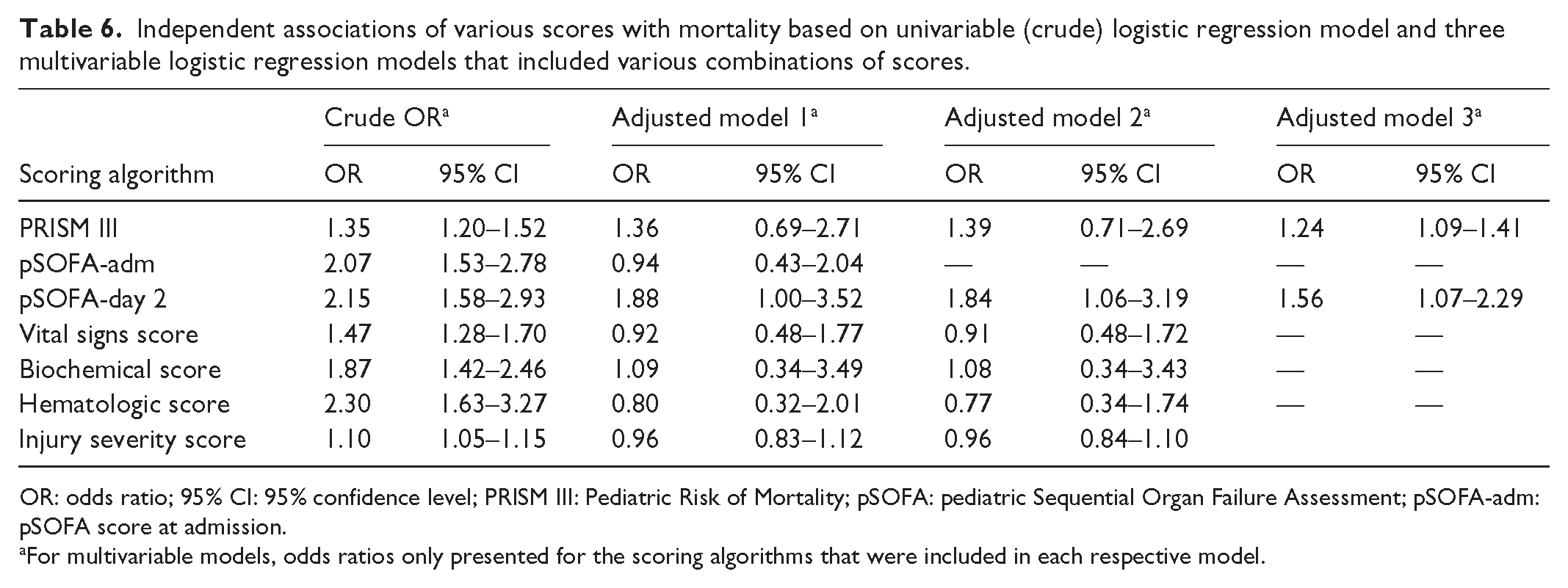
OR: odds ratio; 95% CI: 95% confidence level; PRISM III: Pediatric Risk of Mortality; pSOFA: pediatric Sequential Organ Failure Assessment; pSOFA-adm: pSOFA score at admission.
For multivariable models, odds ratios only presented for the scoring algorithms that were included in each respective model.
To assess the prognostic ability of PRISM III and pSOFA-day 2 in combination, a new scoring model was created by summing up both these scores: PRISM-pSOFAd2. The PRISM-pSOFAd2 score was analyzed as a continuous measure and as categorical variables in binary (bin-PRISM-pSOFAd2) and ternary categories (tri-PRISM-pSOFAd2). Additional risk classification models included a ternary model for PRISM III score (tri-PRISM) and a ternary model for pSOFA-day 2 score (tri-pSOFAd2). To assess the impact of variation in-patient management independent of commonly used clinical and laboratory predictors of mortality, a “med-surg” score included treatment variables (vasopressors, colloids, vasopressin, blood products, sedation, and number of surgical procedures). Lastly, a continuous risk stratification model called the “med-score” combined the scores from the tri-PRISM and tri-pSOFAd2 with a ternary classification of vasopressors and/or blood products use. Detailed descriptions of these novel scores can be found in Table 7.
AUC and 95% confidence intervals for various prognostic models to assess their ability to predict mortality.

PRISM: Pediatric Risk of Mortality; pSOFA-adm: pediatric Sequential Organ Failure Assessment admission; AUC: area under the curve from Receiver Operator Characteristic curve analysis; 95% CI: 95% confidence interval.
PRISM-pSOFAd2: continuous in nature and ranged in value from 5 to 65; bin-PRISM-pSOFAd2: binary score low (combined score <30) versus high (combined score ⩾30); tri-PRISM-pSOFAd2: ternary score classified as low (combined score <20) versus moderate (combined score 20–34) versus high (combined score ⩾ 35); Tri-PRISM: ternary model for PRISM scores (low risk: score <14; moderate risk: score 14–24; high risk: score ⩾25); Tri-pSOFAd2: ternary model for pSOFA-d2 scores (low risk: score ⩽4; moderate risk: score 5–9; high risk: score ⩾10); Med-score: continuous risk stratification model ranging from 0 through 6, that added the scores from tri-PRISM and tri-pSOFAd2 to a new ternary score classifying the administration of vasopressors and/or blood products (0 if neither, 1 if only one of them, 2 if both); Med-surg score: continuous score ranging from 0 through 9 and included information on use of vasopressors, colloids, vasopressin, blood products, sedation and the number of surgical procedures. The meds were all binary and scored as “1” if they were administered or “0” if not. The exception was “sedation,” since it was “protective” in our dataset, and coded as “1” if they did not get it (i.e., lack of sedation was a higher risk of death) and “0” if they did. The number of surgery procedures ranged from 0 (none) to 4. Unless otherwise noted within the table, all scores are calculated from the first 24 h of admission.
p-value from comparison of the AUC of each score and the PRISM III score.
The prognostic ability of all clinical, physiologic, and composite scores was compared to each other using ROC analysis. The AUC, along with its asymptotic 95% confidence intervals are presented in Table 7. The ROC curves for all the scoring methods are depicted in Figure 1. The PRISM III was our reference score. The PRISM-pSOFAd2 score demonstrated the highest discriminatory ability (AUC 0.973, 95% CI: 0.939–1.000). This new scoring method had 100% sensitivity and specificity at the cut-points of ⩽17 and ⩾40, respectively. The score had peak correct classification at a cut-point of ⩾29, where 93.75% of the cases were correctly classified with a positive likelihood ratio of 13.29. However, the PRISM-pSOFAd2 score was not statistically better than the PRISM III (AUC: 0.968; 95% CI: 0.935–1.000). The pSOFA-day 2 score on its own had a slightly lower AUC (0.940) compared to the PRISM III score, but the difference in predictive ability between the PRISM III and pSOFA-day 2 was not statistically significant. The pSOFA-day 2 also demonstrated no statistically significant difference in predictive ability compared to PRISM-pSOFAd2.
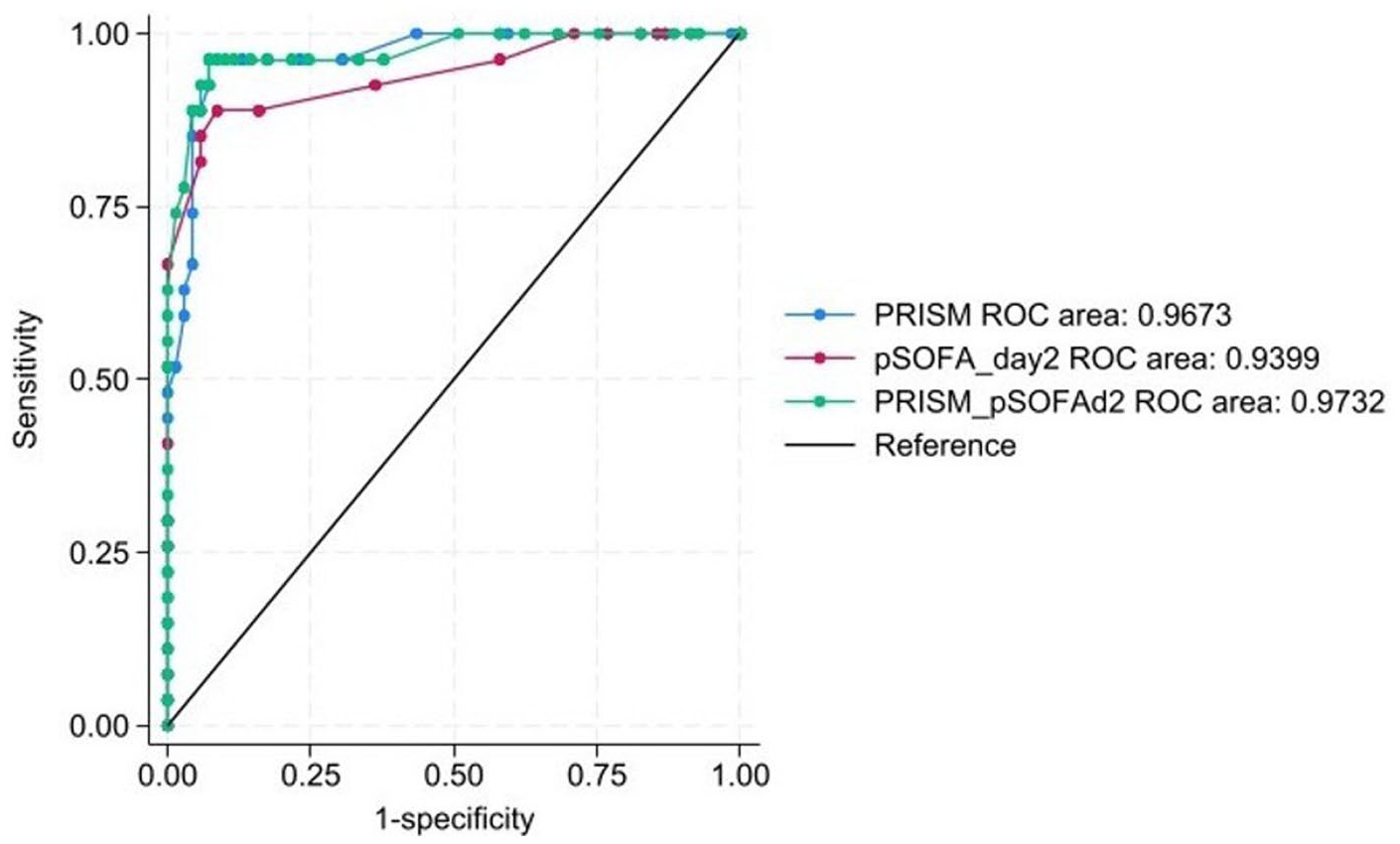
ROC curves for PRISM, pSOFA day 2, and combined PRISM–pSOFA day 2 models in predicting mortality.
Since the three main prognostic instruments of interest (PRISM III, pSOFA-day 2, and combined PRISM-pSOFAd2) yielded high AUCs and were calculated from the same dataset, optimism-controlled bootstraps were performed to assess the presence of bias due to overfitting. This analysis yielded mean optimism values of less than <0.01 (or <1%), suggesting minimal to no overfitting of the results. The 95% confidence intervals for the optimism-corrected AUCs were 0.965–0.975, 0.933–0.952, and 0.968–0.978 for PRISM III, pSOFA-day 2, and the combined PRISM-pSOFAd2 scores, respectively.
A comparison of the predictive ability of the PRISM III, pSOFA-day 2, and the combined PRISM-pSOFAd2 was also performed across clinically relevant ranges of the PRISM III and pSOFA-day 2 scores (Table 8). The predicted mortality rates from each of the three predictive algorithms were statistically not different from the observed mortality rates across all risk strata for both the PRISM III and pSOFA scores calculated on day 2.
Comparison of observed mortality versus mean predicted mortality by scoring algorithm stratified by PRISM III and pSOFA-day 2 scores.

PRISM III: Pediatric Risk of Mortality III; pSOFA: pediatric Sequential Organ Failure Assessment; 95% CI: 95% confidence interval.
Comparisons of mean predicted and observed mortality not statistically different (goodness-of-fit test p > 0.1 for all) in all PRISM III and pSOFA risk strata.
Discussion
While the PRISM III score is validated for early triage and risk stratification on admission, its reliance on 17 physiological variables makes it cumbersome at the bedside and limits its utility beyond the first 24 h. 5 In contrast, the pSOFA score is easier to apply in real time and supports serial assessment of organ dysfunction throughout the ICU stay.8,10 Despite its practicality, the pSOFA remains underutilized in clinical practice, likely due to its relatively recent validation in 2017 and the widespread familiarity with the PRISM III. 8 Our study found that serially applied pSOFA scores had excellent discrimination for in-hospital mortality comparable to more complex models, a finding not previously demonstrated in the pediatric severe TBI population. Serial measurements can inform prognosis, guide escalation or de-escalation of therapies, and provide families with objective updates on clinical trajectory.
Demographic characteristics of our patient population were consistent with pediatric studies showing a predominance of males (61%), a high mortality rate from severe TBI (29%), and assault related TBIs having a high mortality rate (67%). 12 Patients who suffered a secondary insult with cardiac arrest outside the hospital had a higher likelihood of mortality, which may be associated with either the severity of their injuries or the availability of resources at the time of, or soon after, the arrest. Age as a predictor of mortality remains controversial, with some studies demonstrating an association. Consistent with a few existing pediatric studies, our findings suggest that age was not a significant predictor of mortality.3,13–15
Although invasive ventilation, vasopressor use, colloid administration, and surgical interventions have been associated with mortality in prior TBI studies, these treatments often vary by clinician practice and were not independently predictive of mortality in our cohort.16–19 Sedatives appeared protective in our study, possibly reflecting that patients with preserved neurological function were more likely to receive sedation. However, this finding should be interpreted with caution, as it may reflect confounding by indication. Patients who were less critically ill or had different clinical trajectories may have been more likely to receive sedatives. Further research is needed to clarify the relationship between sedative use and outcomes in this population.
Neurosurgical procedures were not associated with in-hospital mortality, whereas general surgeries were, a finding likely related to surgical indications. Patients with diffuse neurological injuries or who had imaging findings concerning for herniation were often considered non-operable. Despite these observations, the med-surg score, consisting only of surgical and medical interventions, demonstrated significantly lower discriminatory ability in predicting in-hospital mortality compared to other prognostic models. These findings highlight the limited prognostic value of individual interventions.
In this study, we evaluated the change in the pSOFA (ΔpSOFA) score from day 1 to day 2 and to day 3. Our results showed a correlation between ΔpSOFA over the first 3 days and in-hospital mortality. A worsening ΔpSOFA from day 1 to 2 (defined as an increase in score) or minimal change from day 1 to 3 (defined as either no change, an increase, or a decrease of no more than one point) was associated with a higher risk of mortality. On the contrary, a substantial reduction in the ΔpSOFA score by day 3, defined as a decrease of more than one point, was more likely to be associated with a patient who was discharged. Similar trends linking the change in SOFA with mortality have been observed in adult patients. 20 Both the SOFA score and its serial changes in critically ill adult patients help quantify the degree of organ dysfunction present at ICU admission and the dysfunction that develops during the ICU stay. The relationship between ΔpSOFA and mortality in pediatric patients with severe TBIs has not been documented. Our study demonstrates how the ΔpSOFA allows prognostication beyond the day of PICU admission, supporting its potential as a dynamic monitoring tool rather than a one-time risk assessment.
We developed various predictive models of increasing complexity to predict the risk of in-hospital mortality following severe TBI. Abnormalities in laboratory parameters are frequent following TBI; however, there is a paucity of pediatric studies, and only a few adult studies have investigated their predictive value.2,13 In adult studies, glucose and coagulation parameters were shown to contribute to outcome prediction, although their effects were smaller than those of other predictors such as the pupillary reflex.2,3 Our findings were comparable; the vital signs score, which includes GCS and pupillary reflex, had a slightly lower, albeit not statistically significant, AUC compared to PRISM III. The predictive value of GCS alone was low, and it was not an independent factor in the presence of PRISM III in our multivariate analysis. This finding differs from pediatric literature that shows GCS remaining a significant predictor of mortality; however, unlike our study, that study did not include models of PRISM and pSOFA. 15 Similarly, although ISS is a known predictor of TBI mortality and was higher among patients who died, it was not an independent predictor of mortality in the presence of PRISM III. 15 ISS also fared worse than PRISM III and pSOFA-day 2 in terms of predictive ability, with a lower AUC.
Our models demonstrated that both the PRISM III and pSOFA-day 2 scores were independently associated with mortality, even when used in combination. The predictive ability of the PRISM III score alone was slightly better than the pSOFA-day 2, but the difference was not statistically significant. A combination score of the PRISM III and pSOFA-day 2 was slightly more predictive than the PRISM III alone, but again did not add a statistically significant improvement to the PRISM III model. Similarly, using different risk stratification models of the PRISM III and pSOFA scores showed no statistically better prediction of mortality. These findings suggest that the pSOFA-day 2 can serve as a practical alternative to the PRISM III in the early ICU course, especially promising in situations where the PRISM III is not calculated at admission or early on the first day. This can occur in resource-limited settings or in smaller centers where automated calculation tools are not available. In such cases, the pSOFA-day 2’s ease of calculation and reliance on fewer variables make it feasible for rapid risk stratification and ongoing monitoring.
While Matics et al. validated the pSOFA in critically ill children before applying it to a subset of septic patients, more recent studies have extended its application to various clinical settings. 8 Badke et al. demonstrated its ability to predict critical events in the PICU, while a large-scale emergency department study confirmed its predictive value for hospital mortality.6,21 Another study highlighted that descriptive scores such as the pSOFA score reflect changes in-patient condition, including responses, or lack thereof, to treatment and the course of the illness. 22 The authors note that such scores tend to offer insights that extend beyond initial baseline predictions. 22 These findings support the broader applicability of pSOFA in assessing organ dysfunction and guiding prognostication in critically ill pediatric patients, including those with TBI.
In practice, the ΔpSOFA and pSOFA could be incorporated into routine ICU workflows as part of daily multidisciplinary rounds, complementing clinical judgment with objective serial monitoring. In the first 24 h, baseline pSOFA may aid in initial triage and family counseling, whereas serial ΔpSOFA or the pSOFA-day 2 measurement could inform ongoing management decisions on adjusting therapeutic intensity and initiating discussions about prognosis. Importantly, predicted mortality from the PRISM III, pSOFA-day 2, and the combined PRISM-pSOFAd2 tracked closely with observed mortality across all clinically relevant risk strata, reinforcing the reliability of these models when applied in practice. For centers that do calculate PRISM III on admission, the pSOFA-day 2 score can still serve as a complementary measure for serial assessment, ensuring that prognostic insights extend beyond the initial 24 h of care.
The limitations of this study include its retrospective design and single-center nature, which limit the generalizability of the findings across other institutions. However, data were collected over a 10-year period during which multiple physicians staffed the unit, reflecting the typical turnover seen in large academic centers. While institutional policies or guidelines may have played a role, the variation in physician preferences over time likely mitigates the impact of any single approach. Additional limitations include occasional missing data, for which normal values were assumed in the absence of available information.5,6,8 While the assumption of missing values as normal is part of standard practice, it may underestimate abnormalities in cases where tests were not repeated despite changes in-patient status. Our focus was on physiologic and laboratory variables relevant to critical illness and organ dysfunction. The PRISM III and pSOFA scores are widely used in pediatric intensive care settings and offer a more direct assessment of physiologic derangement than ISS, which emphasizes anatomic injury. In addition, comparison of models with and without ISS demonstrated no effect modification or improvement of predictive ability by including ISS into the algorithms. No a priori sample size calculation was performed prior to data collection, which may have resulted in a suboptimal sample size. We also did not conduct a post-hoc power analysis. Statistical consensus indicates that post-hoc power analysis is mathematically redundant with the observed p-value and does not provide additional insight into the robustness of our findings. Instead, we have reported the effect sizes with 95% confidence intervals, which more directly convey the precision and uncertainty of our estimates. Due to the retrospective and exploratory nature of this project, we utilized data over as long a period as possible, where we were certain we would obtain data that would be reliable and robust without introducing any temporal effects related to changes in the management of these patients. Furthermore, a secondary analysis to calculate the minimal detectable effect (MDE) between the AUCs of PRISM III and pSOFA-d2 yielded a MDE of 0.0683, a value much larger than the observed difference of 0.0285 between the AUCs for these two scoring algorithms. This suggests that, although small, our sample size was sufficient to identify a clinically meaningful difference of at least 0.068 between the AUCs from the two main algorithms. Data were also sourced from a level I pediatric trauma center with a substantial cohort of pediatric TBI patients, allowing us to focus specifically on this population.
Conclusion
Uncertainty regarding a patient’s prognosis often complicates decision-making and care delivery. Identifying a prognostic tool that can be applied serially beyond the first day of admission could guide goals of care discussions and reduce prognostic uncertainty for families. Physicians can provide objective data and support informed decision-making about a patient’s clinical status, especially as caregivers navigate challenging decisions. Furthermore, the ability to better predict mortality using a scoring system can aid in-patient stratification and help standardize interventions across institutions. While outcome risk can be influenced by clinical practice, our models demonstrated that by eliminating variance due to physician practices, our objective measures had excellent predictive power. Our findings of the ΔpSOFA score, along with the pSOFA-day 2, as strong predictors of in-hospital mortality offer valuable insights for both clinicians and families. The simplicity and bedside applicability of the pSOFA score enhance clinicians’ ability to communicate prognostic changes throughout the patient’s clinical course.
Footnotes
Author contributions
Jefferson T. Bedell: Conceptualization, Methodology, Formal analysis, Investigation, Data curation, Writing—Original Draft, Writing—Review & Editing, Visualization.
S. Shahrukh Hashmi: Validation, Formal analysis, Data curation, Writing—Review & Editing, Visualization.
Thao L. Nguyen: Conceptualization, Methodology, Writing—Review & Editing.
Preethi Kannan: Writing—Review & Editing, Visualization.
Chinyere O’Connor: Conceptualization, Methodology, Validation, Formal analysis, Data curation, Writing—Review & Editing, Visualization, Supervision, Project administration.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
