Abstract
Background:
The selection of safe drugs for pregnant women in developing countries, such as Ethiopia, where there are limited options of drugs would be challenging. Hence, the aim of this review was to determine the extent of prescribed drugs use and their potential to cause fetal harm among pregnant women in Ethiopia based on the United States Food and Drug Administration risk category.
Methods:
Relevant studies were identified through systematic searches conducted in PubMed, HINARI, Google Scholar and Researchgate. Data on study characteristics and outcomes were extracted using the format developed in Microsoft Excel. The primary measure was pooled prevalence of prescription drugs use during pregnancy. The I2 index was used to assess heterogeneity among studies. The presence of publication bias across studies was evaluated using funnel plot. A random effects model was used to estimate the pooled prevalence.
Results:
A total of nine studies published between 2013 and 2019 were included. The pooled prevalence of prescription drugs during pregnancy, excluding minerals and vitamins, was 45.9 (95%CI: 29.3, 62.5)%. The pooled prevalence of prescription drug use, including minerals and vitamins, was 86.9 (95%CI: 81.2, 92.6)%. The pooled proportion of medications used based on the United States Food and Drug Administration risk category was 56.1 (95%CI: 43.0, 68.4)%, 29.0 (95%CI: 27.9, 30.1)%, 12.1 (95%CI: 7.9, 18.1)%, 4.1 (95%CI: 3.6, 4.6)%, and 2.5 (95%CI: 1.8, 3.6)% for the United States Food and Drug Administration fetal risk category “A,” “B,” “C,” “D,” and “X,” respectively.
Conclusion:
The use of prescription drugs during pregnancy, excluding supplements, in Ethiopia was high. Drugs with evidence of fetal harm were widely used. Hence, health care providers should select relatively safe drugs. Stakeholders should ensure safe prescribing practice for pregnant women through developing guidelines and updating professionals on the fetal risk status of commonly prescribed drugs.
Introduction
The use of drugs during pregnancy is a concern due to associated risk for the fetus. 1 Since it is difficult to determine the effects of drugs on pregnant women and their fetus through clinical trial due to maternal and fetal safety concern and ethical considerations, the use of nearly all drugs is not recommended during pregnancy.2–5Although avoiding pharmacological treatment, particularly prescription drugs in pregnancy, is an ideal approach, often this is not possible because some women enter pregnancy with medical conditions that require ongoing and episodic treatment, while others develop pregnancy-attributed medical cases during the gestational period.5,6
Medication used during pregnancy can be teratogenic. According to the United States Food and Drug Administration (US-FDA), the term
The extent of fetal damage by drugs used during pregnancy is dependent on many factors, including fetus’s stage of development. 5 The gestational period is classified into three trimesters each comprising 3 months. The risk of teratogenicity is high if the drug is used in the first trimester as this period involves major organ formation. 11 Drugs reaching the fetus during this stage may cause a miscarriage, birth defect, and sometimes defect which is noticeable only later in life. 12 Second trimester onward, the fetal development is primarily maturation and growth. Exposure to drugs during this period often not leads to major congenital malformations but growth inhibition and malfunction of already formed organs. 13
In some pregnancy, the use of prescription drugs is highly beneficial for both mother and fetus. Mothers may enter into pregnancy with chronic disease, such as epilepsy, diabetes mellitus, asthma, hypertension, migraine, and severe depression, or develop disease during pregnancy which is not only dangerous for mother but also for the fetus. 5 Other mothers may develop transit diseases, such as infections during their preganancy. 1 In both cases, the health care providers are supposed to select relatively safe drugs and limit the number of drugs used at a time and period of exposure to drugs. 13
Although the type and extent of drugs used varies from region to region, evidence shows that large numbers of pregnant women take drugs. 14 For example, a systematic review conducted in developed countries indicated 27%–93% consumption of prescription drugs, excluding vitamins and minerals. The authors attributed the wide variation seen in prescription drug use across countries to lack of consistence among studies conducted in terms of study methods and reporting.15,16 Similarly, studies from developing countries reported high prescription drugs use during pregnancy.17–19
It is important to consider the type of medications used during pregnancy. Generally, medications are categorized based on the harm they can pose on the fetus. Consequently, there have been efforts to categorize drugs according to their potential risk on fetus to guide the users. Although the US-FDA pregnancy risk category has often criticized for oversimplification, it is most widely used risk classification up until recently. In response to critics, the FDA issued a new guideline in 2015, Pregnancy and Lactation Labeling Rule. This guideline presents the summary of relevant clinical information on the drugs in way which is suitable for decision-making.20,21
According to the FDA risk category, the drugs in category “A” are considered as safe, whereas for drugs categorized under “B” and “C” class, there is lack of evidence on the risk to human fetus or positive evidence is reported in animal studies. Category “D” and “X” is for drugs with clear risk of fetal abnormality, and “X” is absolutely contraindicated, whereas “D” can be used in certain situation where the disease carries higher risk for both mother and fetus, such as epilepsy. 22 Supplements, such as folic acid and vitamins, are safe to use during this period. However, often drugs that have clearly established teratogenic effect are used during pregnancy. 23 Medications, such as antihistamine, NSAIDs, antiasthmatics, sedatives, antihypertensives, antiepileptics, and antibiotics, are commonly used.12,24
The selection of safe drug for pregnant women in developing countries, such as Ethiopia, where there are limited options of drugs would be challenging. Thus, identifying the extent of prescription drugs use during pregnancy and their fetal risk categories would help health care providers and policymakers in their action toward achieving appropriate drug use in this population. Therefore, this review was conducted to determine the extent of prescribed drugs use among the pregnant women and describe the pregnancy risk level of medications prescribed during pregnancy according to the US-FDA pregnancy risk classification. This review is expected to encourage judicious use of prescription drugs, positive approach, and awareness toward the drug use for improving optimal maternal and child health.
Methods
Review protocol
The protocol for the study was developed before the commencement of the review and it is available on reasonable request from the principal author. The Preferred Reporting Items for Systematic Review and Meta-Analysis (PRISMA) checklist was used for conducting and reporting this review. In addition, we used PRISMA flow diagram to depict the process of identification, screening for eligibility, and final inclusion. 25
Eligibility criteria
Original research articles evaluating the extent of exposure to prescription drugs, excluding supplements (vitamins and minerals), during pregnancy were included in the study. There were no restrictions on publication year, but only studies that were written in English and conducted in Ethiopia were considered for inclusion. Studies reporting utilization rate of drugs other than prescription drugs, such as self-medication, illicit drugs, and traditional medicines, were excluded. In addition, studies with unrelated or insufficient or missing outcome measures were excluded.
Data source and searching strategy
The search was conducted in September 2019 and checked for a newly published article up until the review process was finished on PubMed and HINARI using the following keywords and indexing terms: “prescription drug,” “pregnancy,” and “ Ethiopia.” Advanced Google Scholar and Researchgate search was also conducted to identify other relevant published and unpublished works, including dissertations, institutional repositories, and organizational manuals, among others. Boolean operators (AND, OR) and truncation were used when appropriate to increase the number of relevant findings. In addition, we searched reference lists from retrieved articles to identify further relevant studies.
Study selection
The records retrieved from different databases were exported to EndNote version 9. The original articles identified using the search strategy were subject to screening by two independent reviewers, Y.A. and A.T., after removing duplicate files. The title, abstract, and full text of each article were screened for eligibility based on the established criteria. Any disagreement among the reviewers was resolved by A.N.M. after thorough discussion.
Data extraction and outcome measurement
Data extraction format was developed in Microsoft Excel. The study characteristics, such as study setting, study design, year of publication, study subjects, sample size, sampling technique, and data source, were extracted. The primary outcome measure was the percentage of pregnant women who used prescription drugs, excluding vitamins and minerals. Furthermore, we identified the percentage of pregnant women who used prescription drugs, including vitamins and minerals, mean number of drugs, frequently used therapeutic class of drugs, percentage of drugs prescribed by the US-FDA risk classification category, and the percentage of pregnant women used at least one prescription drugs by trimester.
Study quality assessment
The quality of the studies which fulfilled the inclusion criteria was checked prior to data extraction using the Joanna Briggs Institute Prevalence Critical Appraisal Tool for prevalence studies. 26 The instrument has 10 criteria with “Yes,” “No,” “unclear,” and “not applicable” options. The mean score of the three authors who were taken for decision and studies with less than 50% score was excluded.
Data processing and statistical analysis
The extracted data were exported from Microsoft Excel to OpenMeta[Analyst] software for analyses of outcome measures and sub-grouping. Pooled prevalence of outcomes was calculated assuming DerSimonian–Laird random effects model at 95% confidence level (CI). The significance of heterogeneity of the studies was assessed using I2 statistics based on I2 percent variation across studies. The presence of publication bias was assessed by using funnel plots. A statistical test with a
Results
Study selection
To assess the prevalence of prescription drug use during pregnancy in Ethiopia, our search identified 93 records of which 51 were duplicate titles. After evaluating the articles based on the title and abstract, 27 articles were excluded of which 13 articles were totally unrelated to our review outcomes, 8 reported self-medication practice, 3 reported herbal medicine use, 2 reported supplement use, and 1 reported the use of single class of drugs. In total, 15 articles were considered relevant and were subjected for quality assessment. Finally, 9 articles were considered to be of good quality and included in the systematic review and meta-analysis (Figure 1).

PRISMA flow chart of literature search and study inclusion criteria.
Summary of study characteristics
Table 1 shows the characteristics of nine studies included in this systematic review. Regarding the study area, five studies were conducted in northern part of Ethiopia,27–31 two were conducted in central part, Addis Ababa,18,32 one in eastern Ethiopia, Harar, 33 and one in western Ethiopia, Jimma. 34 Most of the studies were conducted in antenatal care clinic (ANC) of hospitals,27,29,34 hospitals and health centers, 30 and health centers alone.31,32 There were also studies which included pregnant mother attending other wards, such as psychiatry, ambulatory, internal, and obstetrics and gynecology ward.30,34 All studies were published 2009 onward and conducted using cross-sectional study design through either chart review27,29,32,33 or chart review and patient interview.28,18,30,31,34 The sample size in studies included ranged from 263 29 to 1268. 18 Approximately, all studies used either simple random or systematic sampling techniques to select the study participants except one study which used convenient sampling. 34 The prevalence of prescription drug use among pregnant women, including vitamins and minerals, ranged from 71.30% 18 to 97%, 27 whereas vitamins and minerals excluded prevalence ranged from 11.12% 33 to 70.40%. 28 The mean number of drug used varied from 1.1 30 to 2.4. 28 The risk of bias was assessed using Joanna Briggs Institute Prevalence Critical Appraisal Tool for prevalence studies. Majority of articles included scored more than 75%.
Characteristics of studies conducted on prescription drugs use among pregnant women in Ethiopia.

Psychiatry, ambulatory clinic, and internal medicine ward. ANC: antenatal care; MCH: maternal and child health; CS: cross-sectional; OBG: obstetrics and gynecology; PD: prescription drug.
Outcome measures
Prevalence of prescription drug use
The pooled prevalence of prescription drugs used during pregnancy, excluding supplements, that is, iron products and vitamins, was 45.9 (95%CI: 29.3, 62.5)%. There was significant heterogeneity across studies as it is indicated by I2 value of 99.45%,

Forest plot depicting pooled analysis of nine studies reporting prescription drug use during pregnancy, excluding vitamins and minerals in Ethiopia.

Forest plot depicting pooled analysis nine studies reporting prescription drug use during pregnancy, including vitamins and minerals in Ethiopia.
Prescription drugs use by the US-FDA category and trimester
Table 2 shows the percentage of drugs prescribed according to the US-FDA risk category and percentage of women used prescription drugs in each trimester. All studies reported the fetal risk category of drugs used according to the US-FDA risk category. The use of potentially harmful drugs was reported in all studies. However, only three studies reported the use of drugs from the US-FDA risk category “X.” The drugs reported from this class were warfarin, statins, and estradiol valerate.27,18,34 However, only four studies reported the extent of use of prescription drugs by trimester .27,28,18,32,34 There were wide variation in the percentage of women used drugs per each trimester ranging from 2.25% 27 to 68.75% 32 in the first trimester, 14.74% 34 to 70.42% 18 in the second trimester, and 29.79% 34 to 73.25% 27 in the third trimester.
Prescription drug use by the US-FDA risk category and trimester among pregnant women in Ethiopia.
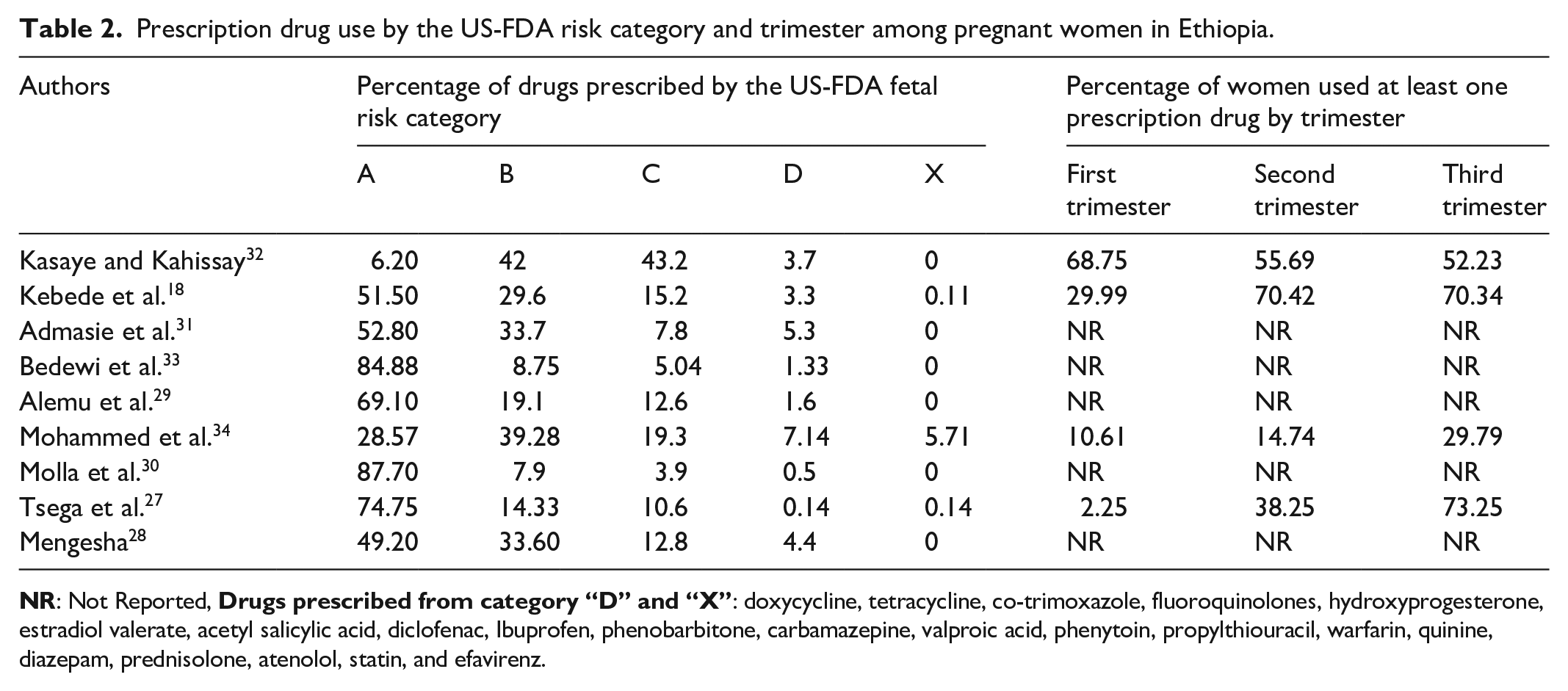
The pooled percentage of medications used based on the US-FDA risk category was 56.1 (95%CI: 43.0, 68.4)%, 29.0 (95%CI: 27.9, 30.1)%, 12.1 (95%CI: 7.9, 18.1)%, 4.1 (95%CI: 3.6, 4.6)%, and 2.5 (95%CI: 1.8, 3.6)% for category “A,” “B,” “C,” “D,” and “X,” respectively. Although, only four studies reported percentage of women prescribed with drugs per trimester, we estimated pooled proportion. Accordingly, 17.6 (95%CI: 5.7, 43.0)%, 43.1 (95%CI: 20.9, 68.4)%, and 57.2 (95%CI: 37.8, 74.6)% of women used prescription drugs in the first, second, and third trimester, respectively.
Commonly used class of drugs
All studies included in this review reported commonly used class of drugs among pregnant women. However, there was no consistence across studies both on therapeutic categorization and common class of drug used. The most commonly reported classes of drugs were antianemic, anti-infective, analgesics, gastrointestinal drugs, including antiemetic and antacid (see Table 3). Antianemic preparations were most frequently used ranging from 46.8% 18 to 89.1% 30 followed by anti-infective from 7.8% 30 to 42.5%, 34 and analgesics from 0.99% 27 to 40.1%. 34
Commonly used class of drugs among pregnant women in Ethiopia.

NR: Not Reported.
Publication bias
Publication bias was evaluated using funnel plot of standard error by logit event rate. The plot showed a slight asymmetry, indicating that publication bias may exist (Figure 4).

Funnel plot assessed for publication bias in the studies conducted on prescription drugs use during in Ethiopia.
Discussion
The use of medications during pregnancy is common practice. While it is challenging to avoid the use of drugs during pregnancy, it is possible to indicate and encourage the use of medications with good safety profile. Pregnant mothers get exposed to drugs through different source; prescription drugs, over-the-counter medication, and herbal medicine. The aim of this review was to determine the extent of prescribed drugs use and their potential for fetal risk among pregnant women in Ethiopia.
In this review, the pooled prevalence of prescription drugs use during pregnancy excluding minerals and vitamins was 45.9 (95%CI: 29.3, 62.5)%. The prevalence was less than half of the prevalence recorded when vitamins and minerals were included implying high rate supplement consumption among pregnant women, 86.9 (95%CI: 81.2, 92.6)%. The commonly prescribed supplements were iron products and vitamins, particularly folic acid separately or in combination with iron preparations. This practice is in line with World Health Organization recommendations. 35 Hence, health care providers should encourage the use of other supplements additionally to iron and folic acid as there are evidences supporting the added benefit of the use of multiple micronutrients in developing countries. 36
Although the figure for prevalence of prescription drugs use during pregnancy excluding supplement as is still high, it might be underestimated. This can be attributed for different reasons. First, some of the studies included were based on the chart review; hence, they might have missed unrecorded medications. Second, because mothers who had miscarriage and termination of pregnancy are usually not included in such studies, which could be due to drugs with potential for teratogenicity, there might be underestimation of the use of drugs with potential for fetal risk.37,38
To guide appropriate selection of drugs, the potential for fetal risk of each drug is evaluated and different risk categorization approach has been used, such as the US-FDA, Australian Drug Evaluation Committee, and Swedish Catalog of Approved Drugs. In Ethiopian health care settings, the US-FDA category is applied to rate the fetal risk associated with each drugs. In this systematic review, the pooled percentage of medications used based on the US-FDA risk category was found 56.1 (95%CI: 43.0, 68.4)%, 29.0 (95%CI: 27.9, 30.1)%, 12.1 (95%CI: 7.9, 18.1)%, 4.1 (95%CI: 3.6, 4.6)%, and 2.5 (95%CI: 1.8, 3.6)% for category “A,” “B,” “C,” “D,” and “X”, respectively. It is clear that more than third quarter of drugs were prescribed from category “A” and “B” which is relatively safe group of drugs. However, the presence of drugs from “X” and “D” category is worrying since it carries high risk of teratogenicity, though the use of category “D” is justifiable when the benefit overweight the risk. 22 Drugs, such as co-trimoxazole, fluoroquinolones, diclofenac, Ibuprofen, phenobarbitone, carbamazepine, valproic acid, phenytoin, propylthiouracil, warfarin, diazepam, prednisolone, atenolol, and statins were commonly prescribed drugs from category “D” and “X.” Not only drugs from category “D” and “X” are dangerous, one cannot rule out the risk for category “C.” 34
Although there are few studies reported the use of unknown FDA status drugs in current review, it is believed that drugs without unknown FDA status are highly used. This is because a drug can stay in the market without having assigned FDA risk status for 27 years on average. 39 Hence, health care providers should try to collect evidences regarding the safety of drug from different sources. Moreover, providers should be aware of new labeling system proposed by the FDA since 2015 which is hoped to provide better gudiance. 20 The policymaker should adopt and ensure implementations of such kind of guidelines.
In this study, the pooled prevalence of prescription drugs use among women in first, second, and third trimester was 17.6 (95%CI: 5.7, 43.0)%, 43.1 (95%CI: 20.9, 68.4)%, and 57.2 (95%CI: 37.8, 74.6)%, respectively, showing increased use of drugs in later period of pregnancy. This might be due to delayed initiation of ANC follow-up in this setting leading to unrecorded prescription drug use in the early pregnancy. 40 However, physicians might become more relaxed in prescribing in the later stage of pregnancy. 41 Regardless of the cause of such practice, it should be discouraged as teratogenic drugs uses in the later stage of pregnancy can lead to functional disability of child if not organ malformation.42,43 For example, NSAIDs used in the late pregnancy can lead to premature closure of the fetal ductus arteriosus and oligohydramnios. 11
Limitations
Although this review can be source of information for health care providers and policymakers, it is important to notice that the quality of research included lacks methodological rigorousness. First, considerable inconsistence in measuring outcome variables, medication classification, and reporting was observed across reviewed studies. Second, the studies included might have suffered from underreporting for variety of reasons. For example, the majority of the studies were conducted among pregnant mothers attending ANC which might not be representative of the whole population since this population is under supervision of health care providers and more rational drug use is expected. Similarly, high dependence on the secondary data was observed. In addition, there were no efforts to track drugs use history of mothers who ended up aborting or had miscarriage leading to possible underestimation of the use of potentially teratogenic drugs. It should be noted that the pooled prevalence of percentage of drugs use per each trimester was calculated only from four studies. Moreover, we were not able to calculate the pooled proportion for commonly used class of drugs and pooled mean number of drugs due to incomplete and inconsistent reporting.
Conclusion
The use of prescription drugs during pregnancy, excluding supplements, in Ethiopia was high. The use of supplements, particularly iron and folic acid, was highly prevalent and should be encouraged. Drugs with potential for fetal risk were widely used and there was high tendency to use prescription drugs in the later state of pregnancy. Hence, health care providers should select relatively safe drugs during pregnancy. Stakeholders should ensure safe prescribing practice for pregnant women through developing guidelines and updating professionals on the risk status of commonly used drugs. Finally, we recommend standardize approach in conducting researches on drugs use during pregnancy.
Footnotes
Acknowledgements
The authors thank the researchers who responded promptly when they were contacted for additional information on their respective publications.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
