Abstract
Background:
The frequent occurrence of bacteria-associated diarrhea together with increased antimicrobial resistance poses a significant public health challenge worldwide.
Objectives:
The aim of this study was to assess the prevalence, antimicrobial susceptibility pattern, and associated factors of
Methods:
A cross-sectional study was conducted among 232 patients with diarrhea at public health facilities in Adama, Ethiopia, from February 2017 to March 2017. Data were collected using a structured questionnaire. Stool samples were examined for
Results:
The prevalence of
Conclusion:
Introduction
Diarrheal diseases are one of the leading causes of deaths worldwide, with an estimated 1.4 million deaths in 2010. 1 The morbidity and mortality are high in developing countries where living standards, access to safe and adequate clean water supply, and proper sewage disposal system are often limited.2,3 Diarrhea is defined as the passage of three or more loose or watery stools in the 24-h period within the 2-week period. 4 Its epidemiology is aggravated by the lack of access to clean and safe drinking water, poor sanitary disposal of human waste, lack of washing hands, poor housing conditions, cohabitation with domestic animals, and lack of access to adequate and affordable health care.5,6
Potential enteric bacterial pathogens that cause life-threatening diarrheal diseases across the world include
The increase in drug resistance in
There are pocket studies on diarrhea from different regions of Ethiopia. However, most of them were restricted either to clinical data, specific age group, or common bacterial strains with or without associated factors and antimicrobial sensitivity tests.6,20–22 This study investigated the prevalence, antimicrobial susceptibility pattern, and associated factors of
Methods
Study settings, design, and period
The facility-based cross-sectional study was conducted at public health facilities in Adama, Ethiopia, from February 2017 to March 2017. Adama, which is found at 99 km southeast of Addis Ababa, Ethiopia, has 10 public health facilities. Approximately 5000 patients with diarrhea visited these health facilities every year. 23
Study population and exclusion criteria
Patients who reported three or more diarrhea episodes within the last 24 h were enrolled in the study. Newborn, inpatients and those who had persistence diarrhea and taken antimicrobial treatment 2 weeks prior to and at the time of data collection were excluded from the study.
Sample size and sampling technique
The sample size was calculated using a single population proportion formula considering the prevalence of 17.4% culture-confirmed bacteria-associated diarrhea, 20 95% confidence interval (CI), and a 5% margin of error. After adding 10% non-response rate, the final sample size was 243. Out of the total (10) public health facilities, six (Adama Hospital Medical College (AHMC), Adama Health Center (AHC), Biftu Health Center (BHC), Denbela Health Center (DHC), Hawas Health Center (HHC), and San Francisco Health Center (SHC)) were selected using a simple random sampling technique (lottery method). Proportional allocation of the sample size was made for each health facility based on their average size of patients with diarrhea (Figure 1). The study participants were enrolled consecutively until the intended sample size fulfilled.
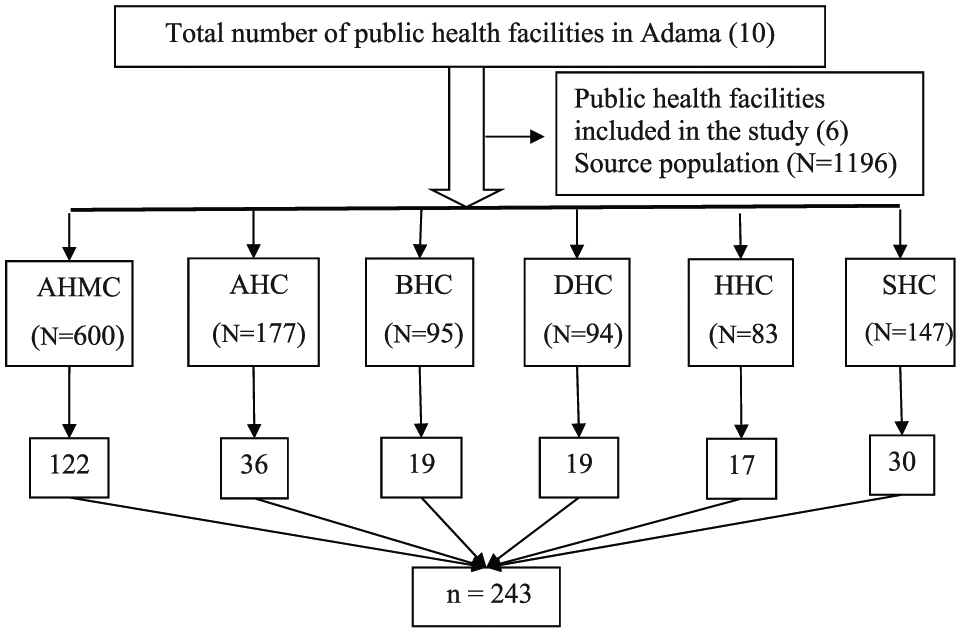
Schematic presentation of sample size allocation.
Data collection and laboratory procedures
Data were collected using a pre-tested structured questionnaire adapted from the World Health Organization (WHO) core questions and different kinds of literature that were designed to explore factors related to diarrhea.6,19,24,25 The adapted questionnaire was contextualized to the local situation and to the study objectives. The questionnaire comprised sociodemographic characteristics (such as sex, age, residence, level of education, and occupational status) and associated risk factors (feeding of raw/uncooked food, washing hands before the meal and after using the toilet, the frequency of washing hands, contact with domestic animals among other related factors). Participants were instructed to collect ~1 g of fresh stool (or 1 mL if loose) in a sterile screw-capped tube containing 9 mL buffered peptone water (Oxoid, Basingstoke, England). The stool specimens were transported in cold box to the Oromia Public Health Research, Capacity Building and Quality Assurance Laboratory, Adama, Ethiopia for further processing and microbiological analysis.
In the laboratory, the isolation and characterization of
Antimicrobial susceptibility testing
Each isolated strain was tested in vitro for antimicrobial susceptibility according to a modified Kirby Bauer disk diffusion method as described by the Clinical and Laboratory Standards Institute (CLSI). 28 The commonly prescribed antimicrobials such as ampicillin (10 µg), amoxicillin/clavulanate (20/10 µg), cefoxitin (30 µg), ciprofloxacin (5 µg), chloramphenicol (30 µg), tetracycline (30 µg), and nalidixic acid (30 µg) (Oxoid, Ltd., UK) were used to screen the susceptibility of the isolates. Three to five pure colonies of bacteria were picked and suspended in sterile normal saline (0.85% NaCl) until the suspension became equivalent to 0.5 McFarland turbidity standard. A bacterial suspension was placed at the center of Mueller Hinton agar plate (Oxoid, Ltd., UK) supplemented with 5% sheep blood and evenly spread using a sterile cotton-tipped applicator. After drying for 3–5 min, antimicrobial disks were placed and incubated aerobically at 37°C. After overnight incubation, the diameter of the zone of inhibition was measured using a digital caliper and interpreted as sensitive (S), intermediate (I), or resistance (R) based on the CLSI interpretive criteria. 28
Operational definitions
Acute bacterial diarrhea is defined as the passage of three or more loose or watery stools in a 24-h period before the study and the isolation of at least one
Antimicrobial resistance is the complete insensitivity of the isolates for which they are sensitive before. 28
Data quality control
The questionnaire was initially prepared in English and translated into local languages (
Data analysis
Data were checked for completeness, coded, and entered into the EpiData software (version 3.1,
Results
Participant characteristics
Of the total (243), 232 patients with diarrhea were enrolled in this study, making a response rate of 95.5%. The majority of them were females (57.3%). The age of the study participants ranges from 1 to 70 years with a mean age of 22.3 years (±16.5 standard deviation). Most of the study participants were urban dweller (89.2%) and had a primary level of education (43.1%) (Table 1).
Sociodemographic characteristics of patients with diarrhea at public health facilities in Adama, Ethiopia, 2017.

Prevalence of acute bacterial diarrhea
Out of the 232 stool specimens investigated, 42
Antimicrobial susceptibility pattern of
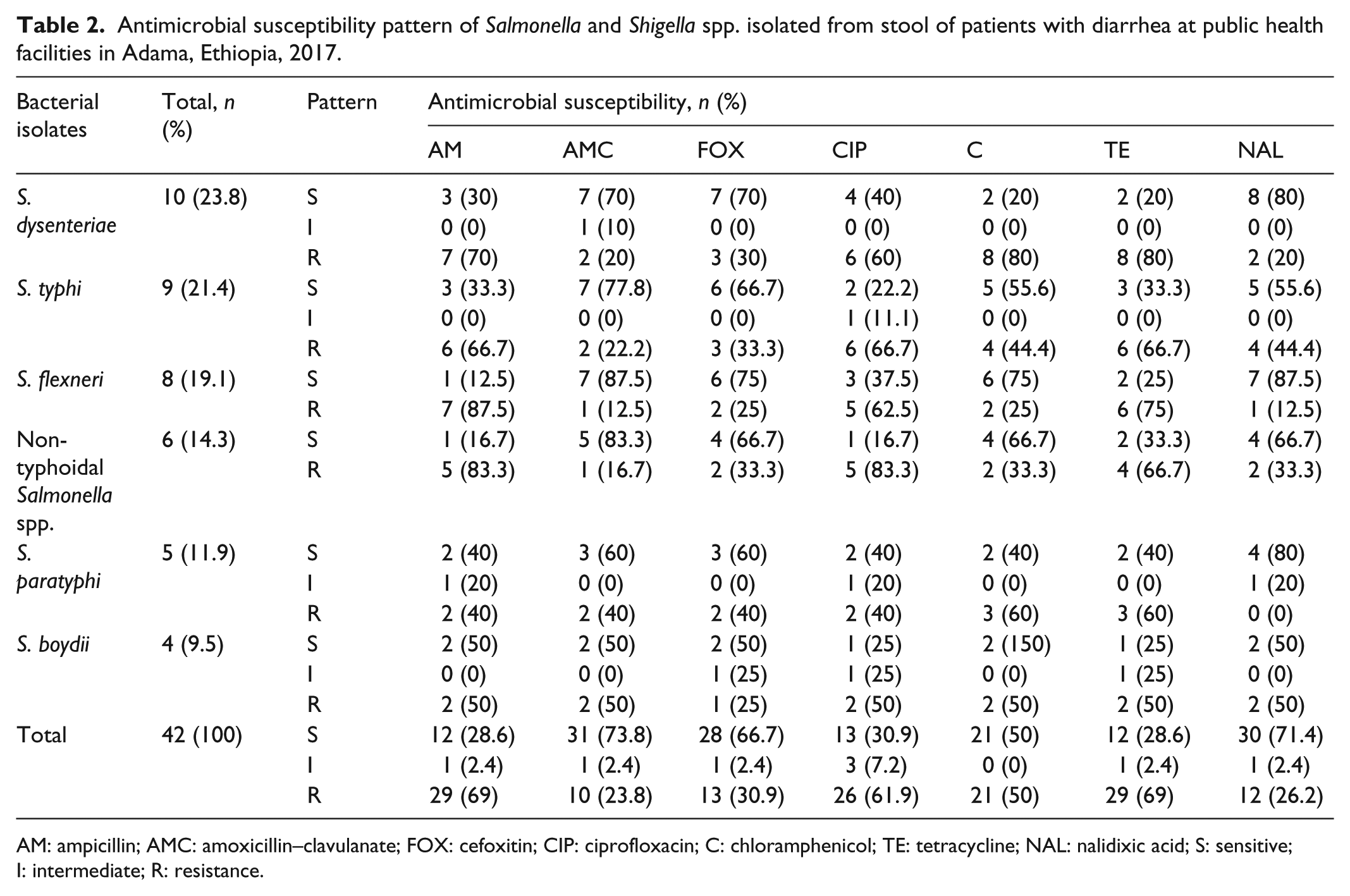
AM: ampicillin; AMC: amoxicillin–clavulanate; FOX: cefoxitin; CIP: ciprofloxacin; C: chloramphenicol; TE: tetracycline; NAL: nalidixic acid; S: sensitive; I: intermediate; R: resistance.
Antimicrobial susceptibility pattern of the isolates
High rates of resistance against multiple antimicrobials were observed in most of the isolates. The isolates showed 69% resistance to each of ampicillin and tetracycline, and 61.9% to ciprofloxacin. The most resistant isolates from
Factors associated with acute diarrhea
Patients in the age range of 11–20 years had almost five times higher odds of getting acute bacterial diarrhea than those in the age of less than 11 years (adjusted odds ratio (AOR): 4.61, 95% CI: 2.48, 7.34). The odds of having acute bacterial diarrhea were about fourfold higher among patients who feed raw vegetables compared with their counterpart (AOR: 3.67, 95% CI: 1.32, 8.59). The odds of being infected with acute bacterial diarrhea were almost three times higher among patients who did not wash their hands before a meal with soap compared to those who did (AOR: 2.68, 95% CI: 1.96, 7.48). The odds of acute bacterial diarrhea were about threefold among patients who did not wash their hands after using the toilet compared to those who did (AOR: 3.25, 95% CI: 1.43, 7.36) (Table 3).
Factors associated with

OR: odds ratio; CI: confidence interval.
Statistically significant association at a
Statistically significant association at a
Discussion
Bacteria-associated diarrheal diseases are a major public health problem in developing countries where illiteracy, poverty, overcrowding, poor sanitation, and unsafe drinking water supply are common.1–3 In this study, the prevalence of
Acute diarrhea caused by
The ecological repartition of the four pathogenic
Antimicrobial treatment can reduce the symptoms of a disease, decrease the number of carriers, and prevent the spread of the infection. However, in resource-limited countries, clinicians are enforced to clinically diagnose and prescribe broad-spectrum antimicrobials empirically that led drug-resistant bacterial strains to emerge.
17
Infections caused by these bacteria could lead to a high cost of treatment, prolonged hospital stays, and an increase in mortality with its concomitant loss in manpower and societies.24,31 In this study,
In this study, patients aged 11–20 years had higher odds of getting acute bacterial diarrhea. This may be due to their substandard personal hygiene and higher exposure to unavailability of safe drinking water and lack of washing hands. The higher odds of acute bacterial diarrhea among patients who did not wash their hands with soap before a meal and after using the toilet were in accordance with the findings of the study conducted in Mekelle, Ethiopia. 6 Another factor that was found to increase the odds of acquiring acute bacterial diarrhea was the feeding of raw vegetables. This was supported by a large variety of evidence that demonstrated as the feeding of contaminated vegetables predisposes consumers to infectious diarrhea.36,37 The occurrence of acute diarrhea could be decreased by interventions aimed to increase awareness in the identified associated factors.
This study has some limitations. The likelihood of underestimation of the prevalence of
Conclusion
In the present study,
Supplemental Material
Questionnaire – Supplemental material for Salmonella and Shigella among patients with diarrhea at public health facilities in Adama, Ethiopia: Prevalence, antimicrobial susceptibility pattern, and associated factors
Supplemental material, Questionnaire for Salmonella and Shigella among patients with diarrhea at public health facilities in Adama, Ethiopia: Prevalence, antimicrobial susceptibility pattern, and associated factors by Bedada Teshome, Zelalem Teklemariam, Desalegn Admassu Ayana, Dadi Marami and Nega Asaminew in SAGE Open Medicine
Footnotes
Acknowledgements
The authors would like to thank Haramaya University for providing an ethical clearance, Oromia Public Health Research Capacity Building and Quality Assurance Laboratory for material support, and the study participants for their valuable information.
Author contributions
B.T. conceived the study and participated in data collection and laboratory analysis. B.T., Z.T., D.A.A., and D.M. have participated in study design, proposal development, interpretation, and an initial and final write-up of the manuscript. N.A. participated in proposal development, sample analysis, and manuscript write-up. All authors read and approved the final manuscript.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical approval
Ethical clearance was obtained from the Institutional Health Research Ethics Review Committee of the College of Health and Medical Sciences, Haramaya University. Permission to conduct the study was also secured from respective health facilities. Ethical approval for this study was obtained from the Institutional Health Research Ethics Review Committee of the College of Health and Medical Sciences, Haramaya University (approval number: IHRERC 065/2017).
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was materially supported by the Oromia Public Health Research, Capacity Building and Quality Assurance Laboratory, Adama, Ethiopia. The supporter had no role in study design, data collection, and analysis, decision to publish, or preparation of the manuscript.
Informed consent
Data were collected after informed, voluntary, written, and signed consent was obtained from the study participants aged ⩾18 years and assent for those <18 years of age from the child’s parent or legally authorized representative guardian before the commencement of the study. The confidentiality of the information was strictly maintained.
Supplemental material
The data used to support the findings of this study are available from the corresponding author upon request.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
