Abstract
Background/objectives:
The results of phase 2 and 3 clinical trials, which justify decisions regarding marketing approval for new drugs, are used for comparison of drugs in the post-marketing phase. A number of meta-analyses of approved antidiabetics have been performed, but the heterogeneity of trials has not been fully examined. The aim of this study was to explore factors that may influence baseline HbA1c in trial samples and treatment outcomes (i.e. HbA1c reductions and effect sizes), with the goal of providing unbiased and fair retrospective comparisons between different antidiabetics.
Method:
We conducted three meta-regression analyses using 78 randomized or non-randomized comparative phase 2 or 3 trials of 24 approved antidiabetics in Japan, conducted from 1987 to 2012.
Results:
Baseline HbA1c of each arm was higher in phase 2 trials, trials with a greater number of subjects, trials with a lower proportion of male subjects, trials of combination therapy, or trials with longer subject disease duration. Entry criteria were different among drug classes and caused variations in baseline HbA1c. HbA1c reductions were larger in non-randomized trials, trials with a shorter treatment period, or trials with a lower proportion of male subjects. Effect sizes were larger in phase 2 trials, or trials of combination therapy. Larger effect sizes were observed in drugs with later market entry for alpha-glucosidase inhibitors and glinides.
Conclusion:
Baseline HbA1c, an important characteristic of subjects enrolled in trials of antidiabetics, differed significantly across trials. Differences in features of study subjects were caused by explicit stipulations in eligibility criteria of HbA1c and also by other conditions (e.g. trial design, regulatory guidance, treatment guideline) and/or interventions of investigators and pharmaceutical companies that were specific to drugs and trials. Healthcare professionals should carefully consider these heterogeneities in trials used for marketing approval review when making a retrospective comparison to select the best treatment option for patients.
Keywords
Introduction
The number of individuals diagnosed with diabetes mellitus (DM) or at high risk for DM has increased tremendously, to over 100 million in the United States in 2015 and about 20 million in Japan in 2016.1,2 DM is a major cause of death and is an established risk factor for coronary heart disease and ischemic stroke. 3 In addition to diet and exercise regimens, optimal glycemic control using antidiabetic drugs is considered a key element in the treatment of DM. Several oral antidiabetics have been introduced since 1960s, with alleged therapeutic advantages over older drugs. However, several studies suggest difficulties in comparing efficacy between old and new drugs in a “fair” manner, because patient backgrounds, clinical trial regulations, and treatment guidelines have changed over decades.4,5 Trial design and characteristics of study subjects were shown to have a significant impact on the results of pivotal trials of recently approved drugs. 6 Antihypertensive drugs, for example, showed time trends in baseline blood pressures (BP) and treatment effects. 7 The observed trends, particularly those in baseline BP, seem to reflect historical changes in the target population in the past decades and indicate that heterogeneity in trial results should be considered in evaluating treatment effects of different drugs.
It is inevitable that pivotal phase 3 trials conducted in different settings entail a certain level of heterogeneity. These trials are used as the rationale for regulatory decisions regarding new drug approvals and eventually exploited in post-marketing for multiple purposes such as comparative analyses of treatment including economic evaluation, advertisements, and the rationale for treatment guidelines and/or algorithms.5,8 Irrespective of their critical roles in approval and post-marketing activities, however, the levels and causes of heterogeneity among the trials have not been fully investigated.
We aimed to explore factors that might influence baseline HbA1c, changes in HbA1c and treatment effect sizes (ESs). It is of importance to investigate all three dependent variables because baseline HbA1c is commonly included as a covariate in the primary analysis for pivotal studies and those variables are closely related to each other. We analyzed whether baseline and clinical outcomes of phase 2 or 3 clinical trials (i.e. changes in HbA1c and treatment ESs) used for marketing approval decisions were associated with characteristics of drugs, designs of trials, characteristics of enrolled subjects, or trial start year. Our study presented candidate factors to which attention should be paid in comparing drug efficacy and also provided a general description of features associated with successful comparative trials.
Methods
We gathered data on phase 2 and 3 trials conducted in Japan for all antidiabetics available on the market. A total of 26 drugs were identified. For two, trial data were unavailable in the public domain. Five multi-regional trials conducted for marketing approval in Japan were included. We used data from a Japanese population in those studies to eliminate any ethnic differences in the results.
The trial data were obtained from common technical documents (CTDs) on the Pharmaceutical and Medical Devices Agency website (http://www.info.pmda.go.jp/), “interview forms,” which provide healthcare professionals with comprehensive, specific, and practical guide for proper use of drugs, and publications on trials (Table S1). Baseline HbA1c and changes in HbA1c in each treatment arm were collected as explanatory and/or objective variables. National Glycohemoglobin Standardization Program (NGSP) values were used for our analyses; Japanese Diabetes Society values found in CTDs, interview forms and publications, were converted to NGSP values. 9 We conducted regression analysis using all the arms, as in previous studies.7,10 Studies were characterized by trial design, the entry criteria for HbA1c, mean age, disease duration, proportion of male subjects, and drug class.
We analyzed changes in HbA1c and ESs for each drug-placebo pair. Both are important outcomes used for various post-marketing comparisons, but the two have somewhat different practical implications. Changes in HbA1c are a direct measure of response to a drug; they reflect changes of a trial arm; they commonly appear in treatment guidelines and/or package insert of the drug. ESs, which are calculated based on changes in HbA1c and standard deviations, are a measure of how much a test drug is effective over a comparator/placebo in a comparative trial; they reflect differences between trial arms; they do not usually appear in guidelines or package inserts in their own, but in the form of various statistical measures including
Statistical analysis
We applied random-effects meta-regression models to account for both within-trial variances of patient backgrounds and treatment effects (weighted by the sample size of each trial), and the residual between-trial heterogeneity. The objective variable (

We categorized entry criteria based on lower and upper limitations of HbA1c: without criteria for a lower limitation, 7.0% or less and higher than 7.0%; without criteria for an upper limitation, 10.0% or less and higher than 10.0%.
A rank correlation test was performed to investigate correlations between ES of drugs and the order of market entry for alpha-glucosidase inhibitors (aGI), glinides, dipeptidyl peptidase-4 (DPP-4) inhibitors, and sodium glucose co-transporter 2 (SGLT2) inhibitors.
The significance level was set at
Results
For 24 antidiabetics approved from 1961 to 2015 in Japan, 78 comparative phase 2 or 3 trials, conducted from 1987 to 2012, with 176 treatment arms were used for the analyses (Figure S1). Explanatory variables and ES for HbA1c in 78 clinical trials were summarized in Table 1. Baseline HbA1c for an arm ranged from 6.91% to 10.78%, and the average baseline was 8.20%. Changes in HbA1c in response to active drug treatment ranged from −1.63% to 0.16%, and the average change was −0.74%. There were significant differences in baseline HbA1c (including placebo,
Summary statistics of explanatory variables and effect sizes for HbA1c in 78 clinical trials.
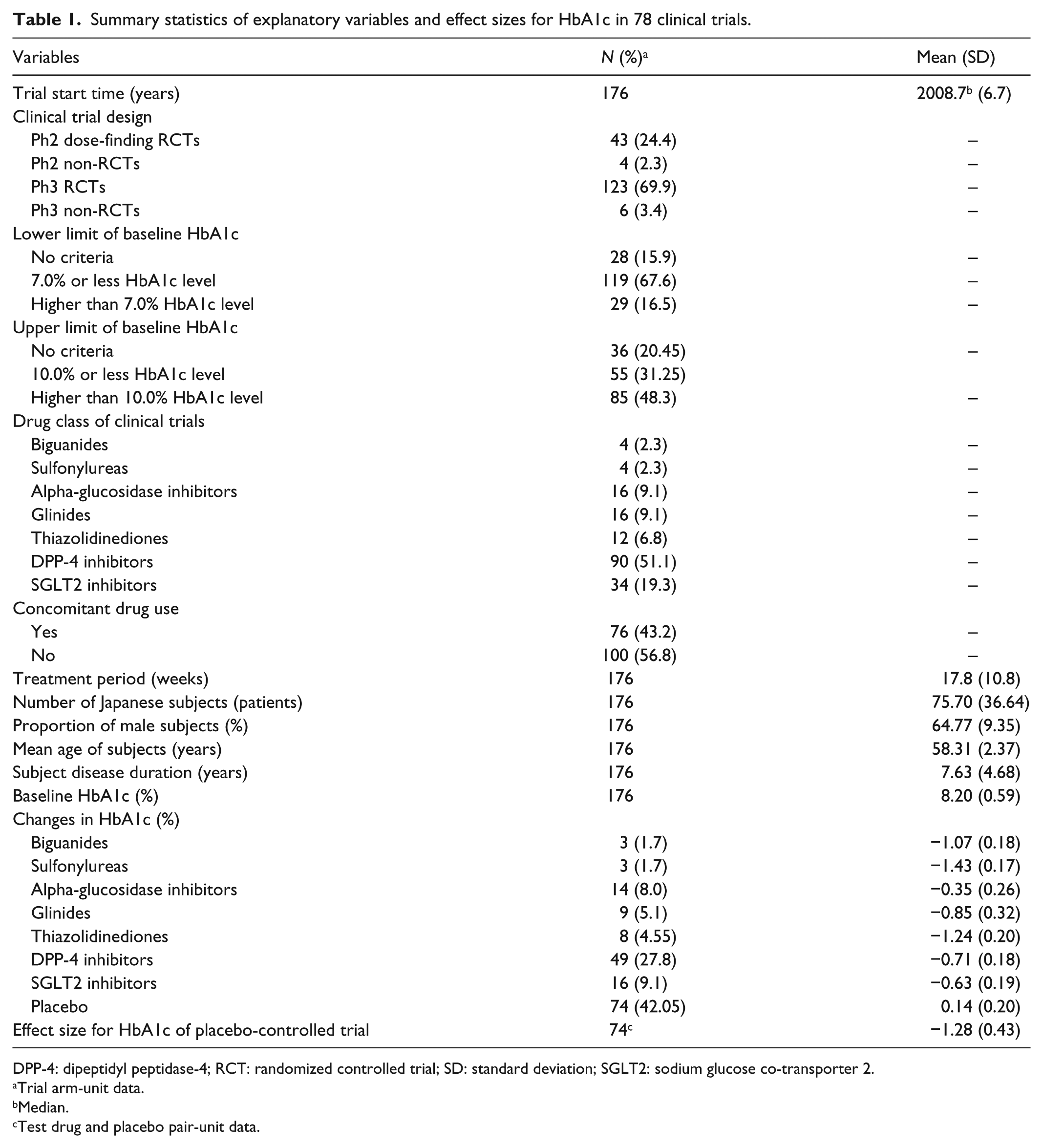
DPP-4: dipeptidyl peptidase-4; RCT: randomized controlled trial; SD: standard deviation; SGLT2: sodium glucose co-transporter 2.
Trial arm-unit data.
Median.
Test drug and placebo pair-unit data.
Baseline HbA1c
The results of regression analysis regarding baseline HbA1c are summarized in Table 2. Phase 2 dose-finding randomized controlled trials (RCTs) showed higher baseline compared to phase 3 RCTs (Coefficient: −0.2492 in Model 1 and −0.1872 in Model 1′). Baseline was higher in trials of combination therapy than in trials of monotherapy (0.2614 in Model 1 and 0.1846 in Model 1′) or in trials including a larger number of Japanese subjects (0.0015 in Model 1 and 0.0016 in Model 1′). Subjects’ disease duration was positively associated with baseline when we did or did not adjust for variables representing drug class (0.0149 in Model 1 and 0.0073 in Model 1′), while proportion of male subjects was negatively associated with baseline when we did not adjust for drug class (−0.0130 in Model 1). Trials without entry criteria for a lower limit of baseline HbA1c showed higher baselines compared to trials with 7.0% or less HbA1c level and higher than 7.0% HbA1c level entry criteria (−0.8353 and −0.3900 in Model 1). Trials of thiazolidinediones and sulfonylureas showed higher baselines (0.3636 and 1.6685 in Model 1′), and trials of biguanides, glinides, DPP-4 inhibitors, and SGLT2 inhibitors showed lower baselines compared to trials of aGI (−0.4339, −0.5603, −0.3460, and −0.3718 in Model 1′). The heterogeneity (
Meta-regression analysis of baseline HbA1c.

DPP-4: dipeptidyl peptidase-4; RCT: randomized controlled trial; SE: standard error; SGLT2: sodium glucose co-transporter 2.
The objective variable was baseline HbA1c (%).
Treatment effects of drugs (HbA1c changes and ESs)
Absolute changes in HbA1c after drug treatment were associated with some variables related to study design and subject profile (Table 3). Phase 2 and phase 3 non-RCTs showed larger HbA1c reduction compared to phase 2 dose-finding RCTs (Coefficient: −0.5852 and −0.3516 in Model 2). Treatment period and proportion of male subjects were positively associated with HbA1c changes (i.e. smaller reduction; 0.0075 and 0.0039 in Model 2).
Meta-regression analysis of changes in HbA1c.

DPP-4: dipeptidyl peptidase-4; RCT: randomized controlled trial; SE: standard error; SGLT2: sodium glucose co-transporter 2.
The objective variable was changes in HbA1c (%).
The ES of placebo-controlled trials were also related to some variables (Table 4). Phase 2 dose-finding RCTs showed larger ES compared to phase 3 RCTs (Coefficient: 0.2280 in Model 3). Concomitant drug use including insulin combination therapy in three trials was negatively associated with ES (i.e. larger treatment effect in combination therapy than monotherapy; −0.2223 in Model 3). Trials of sulfonylureas and thiazolidinediones showed larger treatment effects compared to trials of aGIs (–1.3726 and −0.9084 in Model 3).
Meta-regression analysis of effect sizes for HbA1c.

DPP-4: dipeptidyl peptidase-4; RCT: randomized controlled trial; SE: standard error; SGLT2: sodium glucose co-transporter 2.
The objective variable was effect size for HbA1c.
Placebo-controlled trials were used for this analysis.
Test drug × placebo.
Neither baseline HbA1c nor subject entry criteria were associated with HbA1c changes or ES. Trial start time indicating the time trend variable did not show statistical significance in any regression analyses, although it was negatively correlated with baseline HbA1c and ES, and positively correlated with absolute changes (data not shown). The heterogeneity (
Rank correlations of ESs
Rank correlations of ES by the order of market entry were tested for certain drug class where the number of drugs was three or more. Significant negative rank correlations were shown in aGIs and glinides (i.e. larger ES for later market entry; Kendall’s tau-b = −0.539,
Discussion
We explored how the clinical trial results of antidiabetics were associated with trial backgrounds and subjects’ characteristics. We focused on phase 2 and 3 trials that were submitted for regulatory approval and commonly used as clinical evidence of efficacy in marketing activities. Our analysis clarified that characteristics of study subjects were determined by stipulations in the protocol and other conditions specific to drugs and trials and that trial results were associated with such characteristics of enrolled subjects and trial design components. The associations between trial results and demographics of enrolled subjects or trial design components suggest that the “observed” treatment ES does not solely depend on the efficacy of a drug per se, but also on other factors including study conditions that can be determined by sponsors or investigators, study subjects actually enrolled, clinical trial regulations, and disease treatment guidelines at the time of a trial reflecting public health needs regarding the underlying DM population, as reported in previous studies for DM and also for antihypertensives.7,12
We investigated whether study subjects enrolled in clinical trials have changed over time. The results of this analysis indicated that the mean baseline HbA1c of a study arm was higher and showed considerably more variation in the 1990s than in the 2000s (without adjustment for other variables). This implies that study subjects were different between studies at least in baseline HbA1c, one of the most important patient characteristics in DM. Baseline was significantly associated with entry criteria in relation to HbA1c level and study design, as explained later, and a time trend was not observed in regression analysis (Table 2). These results suggest that study subjects were selected based on eligibility criteria in each protocol, which seem to reflect pertinent diagnosis guidelines, therapeutic interventions, and regulations at the time of a trial.
In this regard, it is also of interest to examine the extent to which subjects in clinical trials were similar to real-world patients. According to a cross-sectional study and the Japan National Diabetes Surveys, mean HbA1c increased from 1997 to 2002 and decreased from 2002 to 2011 in the general population of patients with type 2 DM.13,14 Observed changes in average HbA1c levels in the trials in our study did not contradict the overall trend in patients, suggesting that the study results may be extrapolatable for a specific time, but not for all periods.
Baseline HbA1c in phase 3 non-RCTs/RCTs was statistically lower than that in phase 2 RCTs (Table 2), but baseline HbA1c was unexpectedly not associated with HbA1c reduction or ES (Tables 3 and 4). Results of phase 3 RCTs were somewhat different from those in phase 2 and/or non-RCTs; HbA1c reductions were smaller in RCTs than those in non-RCTs (Table 3), and ES were smaller in phase 3 RCTs than those in phase 2 RCTs (Table 4). Smaller effectiveness outcomes in phase 3 might be explained in part by “regression to the mean,” because our data reflect only successful development pathways. However, when considering the clear differences in baseline between phase 2 and 3, as well as in outcomes between RCTs and non-RCTs, we surmise that study subjects differed somewhat between different phases and objectives. A possible explanation is that patients were selected more carefully in phase 3 RCTs, probably with the aim of creating homogeneous samples with small variances, rather than samples highly responsive to test drugs.
Baseline was higher in subjects enrolled in trials of combination therapy, and ES were also larger in such trials, even when baseline level was adjusted (Tables 2 and 4).14,15 These results are consistent with previous findings that combination therapies showed larger effects in reducing the HbA1c level than did monotherapy for patients with severe conditions in whom monotherapy was unsuccessful. 16 Trials with longer treatment periods demonstrated less absolute changes in HbA1c (Table 3), which is in line with previous studies reporting a progressive deterioration of diabetes control and a cumulative incidence of failure of monotherapy with long-term antidiabetic treatment.17,18
Demographic features of subjects were associated with baseline HbA1c and treatment effects in complicated ways. Proportion of male subjects was negatively associated with baseline (Model 1 in Table 2), but the association disappeared when the drug class variables were included (Model 1′ in Table 2). In terms of treatment effects, HbA1c reductions were smaller in trials with a higher male proportion, but ES were not (Tables 3 and 4). A model without baseline yielded similar results (data not shown). These results indicate that sex ratio may have some impact on HbA1c reductions, but the size of the possible impact appears minimal when trial backgrounds are adjusted. Previous studies report that mean HbA1c value at study enrollment was significantly higher in Japanese women with type 2 diabetes than in men,13,19,20 and our findings without adjustment were consistent with these results. In the entire Japanese population (i.e. healthy adults included), baseline was higher for men than for women. 2 It has been reported that women with type 2 diabetes are less likely to reach the goals for recommended HbA1c levels compared to men.21–25 Female patients with higher baseline need larger HbA1c reductions, but adverse events and/or intrinsic hormonal circumstances sometimes prevent achievement of therapeutic goals. 26
Mean age of subjects was not associated with baseline or treatment effects. Disease duration of subjects, one of the severity measures for DM, showed a positive association with HbA1c baseline, but did not show an association with treatment effects (Tables 2 and 3). Other studies also reported that disease duration of subjects was associated with HbA1c baseline but not with treatment effects.12,27
Our results indicated that different patient entry criteria led to different study samples in terms of baseline HbA1c. Pre-specified lower limits of HbA1c level in entry criteria seemed to enhance the enrollment of patients with poor glycemic control and lead to lower baseline levels in study samples, as was intended (Model 1 in Table 2). However, the association disappeared when the drug class variables were added (Model 1′ in Table 2). This probably reflects the fact that there are differences (at least) in entry criteria among trial protocols of different drug classes. In contrast, upper limits in entry criteria were not associated with observed baselines. The sponsor and investigators choose appropriate inclusion/exclusion criteria to achieve their study objective, but their choices have been constrained inevitably by the pertinent diagnostic and therapeutic guidelines.5,28 Sponsors and investigators should also follow domestic and international drug development guidelines, for trials to be submitted for New Drug Application (NDA) purposes. 4 Although all trials in our analyses were used to obtain regulatory approval in general type 2 diabetes populations, the entry criteria were not consistent because applicable guidelines were not the same during the period. Prior to 2000, few trials had entry criteria for HbA1c level, while almost all trials had some entry criteria for HbA1c after 2000. These historical changes were inevitable as a response to changes in the requirements for clinical development. However, in comparison of trial results, it is essential to pay attention to what criteria were applied to specific trials, because such factors could lead to heterogeneity in sample populations.
Average baseline HbA1c in each arm was not related to treatment effects in our meta-regression models. The correlation between average baseline and HbA1c reduction was weak (
Differences in treatment effects among drug classes were clearly observed in this analysis. These differences reflect not only a drug’s effectiveness per se, but all other factors that could affect how clinical trials are designed and implemented. Drug class was significantly associated with baseline HbA1c level even when critical entry criteria (i.e. upper and lower limits of HbA1c) and some patient backgrounds were controlled (Table 2). The observed imbalance in baseline occurred before the administration of drugs and was not attributed to the effectiveness of drugs, which apparently implies heterogeneous study subjects among trials with respect to baseline, at least. Heterogeneity in study samples may reflect the competitive environment of trials. We found significant rank correlations of ES by the order of market entry for aGIs and glinides (Kendall’s tau-b = −0.539,
One implication of this study is that treatment outcomes depend on the intention, implementation, and environment of trials in DM, as observed in other therapeutic fields. Trial design components that were declared in the protocol prior to recruitment, including eligibility criteria, treatment period, and the usage of concomitant medications (i.e. monotherapy or combination therapy), would affect results. In addition, we confirmed that trial results might be influenced by patient demographics in each trial. Investigators and pharmaceutical companies can also intervene in the actual process of subject enrollment to some extent. For example, they can enroll patients for whom preferable outcomes would be expected as long as eligibility criteria are satisfied and an apparent imbalance in patient characteristics does not occur. Heterogeneity in clinical trials, irrespective of whether it is intended, is a long-standing issue related to the extent to which trial results can be extrapolated. The possible impact of heterogeneity should be carefully considered when trial results are retrospectively compared.
This study had limitations as follows: our findings are based solely on the results of phase 2 and 3 trials of approved antidiabetics and cannot automatically be extrapolated to antidiabetic trials in general. Results for unapproved drugs are not considered in this research. Our regression analysis was for exploratory purposes with only a small number of variables, and it would thus be inappropriate to infer any definitive causal links. We collected only data presented in publications; undisclosed information may affect our conclusion, for instance, the concomitant medications for complications such as statins and thiazides which may have effect on HbA1c level during the trial.31,32
Conclusion
Baseline HbA1c level of study subjects was significantly different among trials of antidiabetic drugs, which reflected the fact that eligibility criteria had been changing over time. Changes in HbA1c were related to proportion of male subjects enrolled and treatment period. Study sample characteristics, trial status in clinical development, and trial design features were associated with observed treatment effects. Our results suggested that differences among study subjects were caused by explicit stipulations in eligibility criteria as well as by other conditions and/or interventions that were specific to drugs. Such differences directly or indirectly affected trial results and eventually statements on efficacy in guidelines, package inserts, and/or advertising materials. Impact of heterogeneities in trials should be considered in the retrospective evaluation of therapeutic outcomes, especially when medical professionals compare treatment effects of different antidiabetic drugs and select the best one for patients.
Supplemental Material
fig_1_(1) – Supplemental material for Exploratory analysis of comparative clinical trials used for marketing approval in patients with type 2 diabetes in Japan
Supplemental material, fig_1_(1) for Exploratory analysis of comparative clinical trials used for marketing approval in patients with type 2 diabetes in Japan by Reina Kaneko and Shunsuke Ono in SAGE Open Medicine
Supplemental Material
Manuscript_ID_SOM-AOP-18-0383_Table_S1 – Supplemental material for Exploratory analysis of comparative clinical trials used for marketing approval in patients with type 2 diabetes in Japan
Supplemental material, Manuscript_ID_SOM-AOP-18-0383_Table_S1 for Exploratory analysis of comparative clinical trials used for marketing approval in patients with type 2 diabetes in Japan by Reina Kaneko and Shunsuke Ono in SAGE Open Medicine
Footnotes
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: R.K. is an employee of Daiichi Sankyo Co., Ltd, Tokyo, Japan. S.O. declared no potential conflict of interest in preparing this article.
Ethical approval
Ethical approval was not sought for this study because it is not a research involving human participants.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by a Japanese government-based grant-in-aid from the Ministry of Education, Culture, Sports, Science and Technology, Tokyo, Japan (Grant KAKENHI: 26460215).
Informed consent
Informed consent was not sought for this study because it is not a research involving human participants.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
