Abstract
Objectives:
To evaluate outcomes of colorectal screening using sodium picosulfate and magnesium citrate compared with other prescription bowel-preparation agents. Primary endpoints were rates of procedure-associated hospitalizations, diagnosis at hospitalization, and rates of early repeat screenings.
Methods:
This retrospective cohort study identified patients using the Truven Health Analytics MarketScan databases, which contain fully adjudicated, de-identified, medical- and prescription-drug claims, as well as demographic and enrollment information for individuals with commercial, Medicaid, and Medicare supplemental insurance coverage. Patients who had a colonoscopy or sigmoidoscopy over a 3-year period were identified using International Classification of Diseases Clinical Modification procedure codes, recorded on claims from physicians and facilities. First, screening colonoscopy was identified for each patient, and the study was limited to those patients who could be observed for ≥6 months before and 3 months after the screening procedure. Total number of hospitalizations and rates of early repeat screenings were evaluated for all patients who received sodium picosulfate and magnesium citrate and compared with those who received other bowel-preparation agents. Individual prescription medications that could affect the outcome of the cleansing agent were identified; further evaluations were made to establish whether patients had comorbid conditions, such as chronic kidney disease, cardiovascular disease, or psychiatric illness. Statistical methods included descriptive statistics, two-tailed t-tests, and multivariate logistic regression.
Results:
A total of 566,628 procedures were identified in the MarketScan databases and included in the study. Sodium picosulfate and magnesium citrate performed well in terms of safety outcomes, with no hospitalizations due to diagnosis of hyponatremia, dehydration, or other fluid disorders in the 10 days after procedure. Early repeat rates among sodium picosulfate and magnesium citrate patients were comparable with rates observed for all other cleansing agents.
Conclusion:
Outcomes of colorectal screening using sodium picosulfate and magnesium citrate were not significantly different compared with other prescription bowel-preparation agents.
Keywords
Introduction
Colorectal cancer (CRC) is currently the fourth most common cancer in the United States.1–3 The incidence is higher in men, and compared with many other forms of cancer, CRC has a relatively high mortality rate with a 5-year survival rate estimated at 65.1% (Supplementary Figure 1).1–3 While the median age at CRC diagnosis is 68 years with a disproportionate gender distribution, regular screening of at-risk patients improves the odds of detecting CRC at an early stage. 4 Colonoscopy is the most sensitive method for the detection of CRC and adenomatous polyps, which also allows the removal of precancerous lesions to prevent further development and is thus the gold standard for CRC screening.5,6
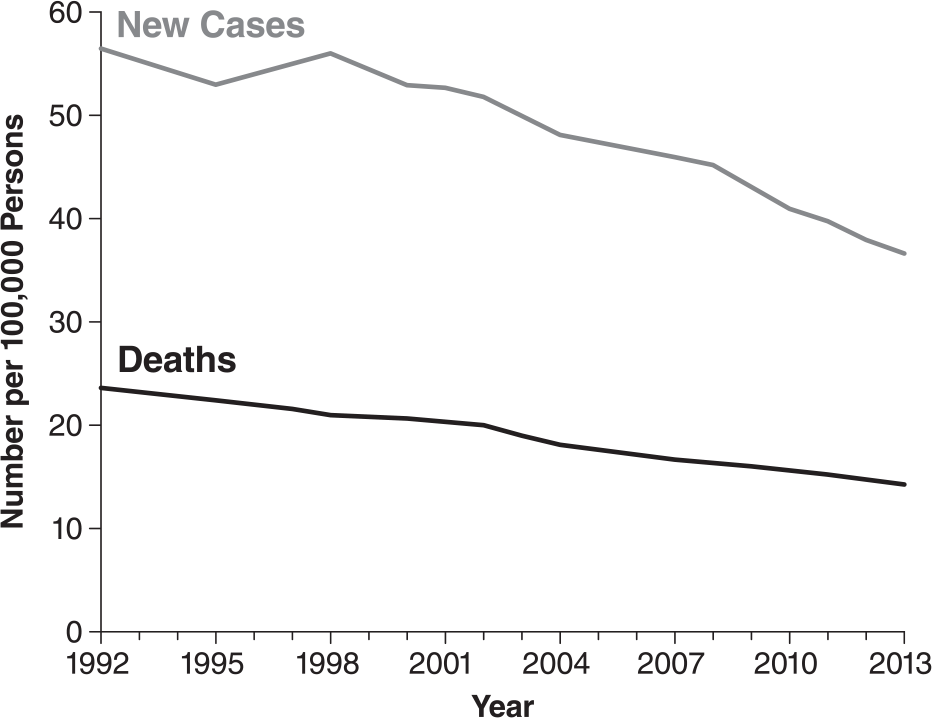
Colorectal cancer incidence and number of deaths in the United States from 1992 to 2013.
Adherence to colonoscopy screening guidelines with adequate bowel preparation that allows full visibility of the colon—defined as the ability to identify lesions >5 mm7–9—is associated with decreased CRC incidence and mortality rates.10,11 Adequate bowel preparation is critical not only for safe and effective colonoscopy screening,12,13 but is also associated with a significant decrease in the adenoma miss rates, early repeat screenings, hospitalizations, and other high-cost events.13–17 While outcomes have a strong association with successful preparation, a recent retrospective analysis assessing degree of bowel cleansing in both inpatients and outpatients undergoing colonoscopy found that inadequate cleansing was recorded in 11.2% of the patients. 18 No significant difference was observed between inpatients and outpatients.
An adequate bowel preparation involves the use of a cleansing agent prescribed prior to the procedure, such as high-volume (HV) bowel-preparation products (i.e. 4-L polyethylene glycol (PEG) solutions), low-volume (LV) products (e.g. 2-L PEG solutions), or other over-the-counter (OTC) products (e.g. magnesium citrate).
Adverse events (AEs) following ingestion of bowel preparations are uncommon, but can be serious. 19 Regarding the safety of colonoscopy and bowel preparation, a recent study found the rate of AEs to be low even under severe conditions, although the majority of these patients would not have been coded for a screening procedure. 20 HV products have been associated with AEs, such as nausea, vomiting, bloating, and cramps, and typically require patients to consume up to 4 L of a solution with an often unpalatable salty taste. As a result, patients may be less tolerant of HV agents and at greater risk for inadequate preparation. LV products are reportedly easier for the patient to consume and are generally well tolerated with similar or better cleansing quality compared with HV agents.12,21,22 Documented AEs with LV preparations include dehydration, hyponatremia, electrolyte imbalances, and in rare cases, kidney damage. 23
Prepopik® (Ferring Pharmaceuticals Inc., Parsippany, NJ, USA) is a LV sodium picosulfate and magnesium citrate (P/MC) bowel-preparation agent approved for colon cleansing prior to colonoscopy or sigmoidoscopy in adults. 24 For each bowel preparation, the patient is required to consume the drug dissolved in 5 oz of water at two different times, followed by 40 or 24 oz, respectively, of a clear liquid of the patient’s choice, to be consumed within 5 h. 24 In previous clinical trials, bowel cleansing using P/MC preparations have been demonstrated to be both safe and effective with the potential to increase patient adherence to colorectal screening guidelines.25–27
The rationale for this study was based on a misconception of efficacy concerning CRC procedures driven by the use of various bowel preparation scales in clinical trials. The objectives were to evaluate real-world clinical outcomes of CRC screening by comparing P/MC with different prescription bowel-preparation agents, including the incidence of early repeat-screening events and hospitalization rates, and assessment of the evidence in a retrospective analysis.
Methods
Study design
Using employer-based health insurance claims filed between 1 January 2012 and 30 June 2014, this retrospective cohort study evaluated outcomes of colorectal screening in patients using P/MC compared with patients who used other cleansing agents. The patients were identified using the Truven Health Analytics MarketScan databases (now part of IBM Watson Health), which contain fully adjudicated, de-identified, medical- and prescription-drug claims, as well as demographic and enrollment information for individuals with commercial, Medicaid, or Medicare supplemental insurance coverage. Colonoscopies and sigmoidoscopies were identified using the International Classification of Diseases, 9th revision, Clinical Modification (ICD-9-CM) procedure codes, recorded on claims from physicians and facilities (Supplementary Table 1); the claims data for this study were filed before the transition to ICD-10 on 1 October 2015. Because physicians performing these procedures can file claims separately from the facilities where procedures are performed, we grouped all claims occurring within 1 day of each other and considered them as one event. All patient data were tracked for 9 months, with patients required to have had no colonoscopy or sigmoidoscopy in the 6 months prior to the initial screening and repeat screenings defined as secondary screenings that occurred ≤3 months after the initial screening.
Codes used to identify colonoscopies and sigmoidoscopies.
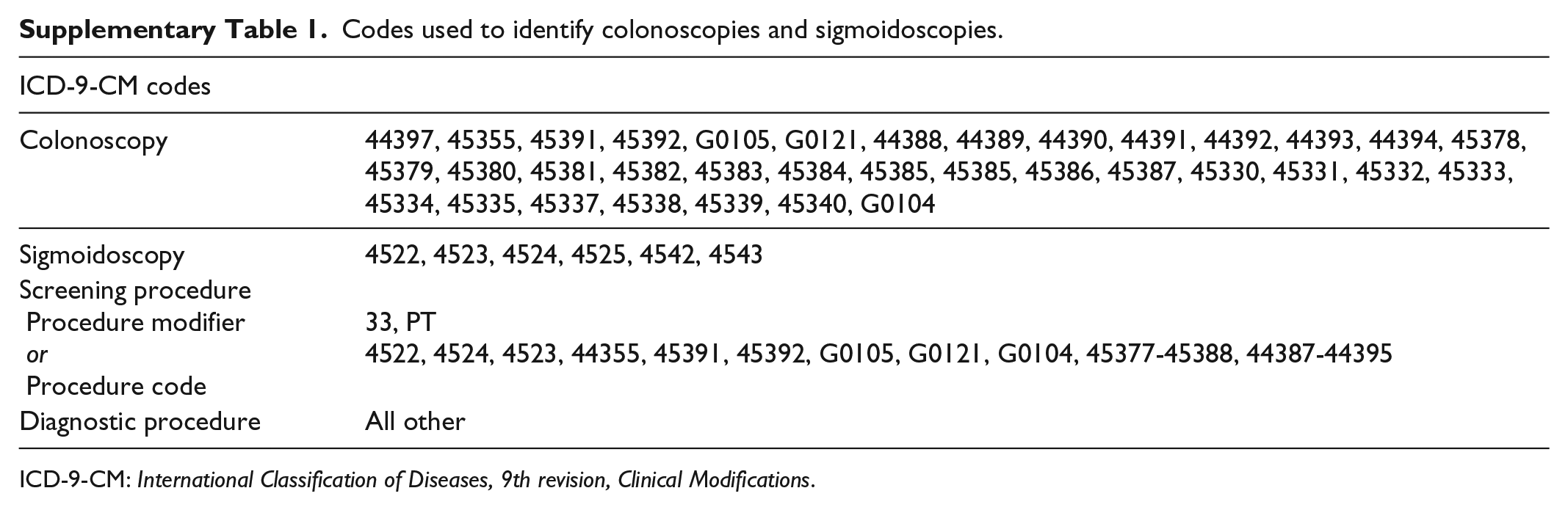
ICD-9-CM: International Classification of Diseases, 9th revision, Clinical Modifications.
The bowel-preparation agent used with each procedure was identified through pharmacy records of the closest filled prescription within 90 days prior to the colonoscopy or sigmoidoscopy. All solution-based cleansing agents were classified as either HV (≥4-L solution) or LV (<4-L solution), based on their approved labeling and directions for use (Table 1). Tablet-based agents (OsmoPrep® and Visicol®, Salix Pharmaceuticals, Inc., Raleigh, NC, USA) were categorized as “other bowel preparations,” and OTC products were not included in this analysis.
Classification of bowel preparations.

HV: high-volume; LV: low-volume.
Ferring Pharmaceuticals Inc., Parsippany, NJ.
Pharmascience Inc., Montreal, QC, Canada.
Salix Pharmaceuticals, Inc., Raleigh, NC.
Braintree Laboratories, Inc., Braintree, MA.
GAVIS Pharmaceuticals, Somerset, NJ.
Wallace Pharmaceuticals Inc., Somerset, NJ.
Inclusion/exclusion criteria
The purpose of this study was to focus on average-risk patients. Key inclusion criteria were age ≥18 years, at least one identified claim for a colonoscopy or sigmoidoscopy within the study period, at least one prescription for a cleansing agent filled within 90 days before the procedure, and the availability of medical and pharmacological data for each patient (≥6 months prior and ≥3 months after the procedure). Data with no identifiable prescription for a cleansing agent (e.g. use of OTC products or physician samples) prior to the procedure were excluded from the analysis. Patient health status was estimated using the Charlson Comorbidity Index (CCI), 28 which is widely used as a measure of patient health status that summarizes comorbidities of patients based on the diagnosis codes found in administrative claims data, with higher scores indicative of poorer health.
To minimize potentially confounding comorbidities, patients determined to be at high risk for CRC based on medical claims were excluded. These high-risk patients were identified through the colonoscopy procedure code G0105 and a selection of ICD-9 codes (Supplementary Table 2).29,30
ICD-9 codes used to indicate high-risk patients. 30

ICD-9: International Classification of Diseases, 9th revision.
Study measures
Patient demographics (i.e. age, sex, geographic region, and risk status) and cleansing agent used for each procedure were collected and used to stratify the results. Primary endpoints were rates of procedure-associated hospitalizations, diagnosis at hospitalization (i.e. CRC vs non-CRC), and rates of early repeat screenings. Hospitalizations were defined as any hospital admission that occurred within 10 days of a procedure. Fluid levels are a particular concern for LV agents, and non-CRC diagnoses were defined as those directly indicative of product safety that could have been associated with the procedure (i.e. diverticulitis, hyponatremia, dehydration, and other fluid or electrolyte disorders), based on principal discharge diagnosis code (Supplementary Table 3). The total number of hospitalizations and the proportion of non-CRC hospitalizations were compared between patients who received P/MC and those who used other LV products, HV products, or other bowel-preparation agents.
Codes used to identify hospitalizations for selected conditions.

ICD-9: International Classification of Diseases, 9th revision.
On the recommendation from the US Multi-Society Task Force (MSTF) of CRC, patients with CRC should undergo a colonoscopy within 3–6 months after surgery (for a duration of 2–3 years). For patients who have undergone curative resection of either CRC or rectal cancer, the MSTF recommends surveillance colonoscopy 1 year after surgery. 31 In this study, early repeat screenings were defined as repeat colonoscopies or sigmoidoscopies scheduled within 90 days of a previous procedure. The rates of early repeat screenings were evaluated for all patients who received P/MC and compared with those who received other LV products, HV products, or other prescription bowel-preparation agents. Individual prescription medications that could affect the outcome of the cleansing agent were identified, including those that affect renal function (e.g. loop and thiazide diuretics) and those associated with hypokalemia or hyponatremia (e.g. cardiac glycosides and corticosteroids). Further evaluations were made to establish whether patients had comorbid conditions, such as chronic kidney disease, cardiovascular disease, or psychiatric illness.
Statistical methods
Patient demographics, patient health status, and presence of repeat procedures and associated hospitalizations were summarized using descriptive statistics, whereas significance of subgroup differences was evaluated using chi-square test. Multivariate logistic regression was used to evaluate the association between each bowel-preparation group and outcome, and the likelihood of an early repeat screening or hospitalization, with adjustment for age, sex, geography, and CCI score. These analyses were conducted to evaluate whether the rates of early repeat screenings were significantly different for P/MC compared with other agents.
Results
Patient population
Out of 1,329,751 screenings of de-identified patients in the MarketScan databases, a total of 566,628 procedures were deemed eligible to be included in the study (Table 2). A majority of these procedures were performed using LV preparations defined as “other LV” agents (69.0%), followed by HV agents (21.9%), P/MC (5.9%), and other agents (3.2%). Mean age for all patients was 56.4 years (standard deviation (SD), 10.9), with a slightly higher proportion of women than men (53.5% vs 46.5%, respectively). The patient population who used P/MC was slightly younger than average (mean age, 55.1 years; SD, 10.6; p < 0.0001), with a larger proportion of female patients (58.2%; p < 0.0001).
Procedures and patient characteristics.

P/MC: sodium picosulfate and magnesium citrate; LV: low-volume; HV: high-volume; SD: standard deviation; CCI: Charlson Comorbidity Index.
LV agents, except P/MC.
Mean CCI scores for the total study population were low (0.41; SD, 0.97), indicating that patients were healthy, with a relatively low overall comorbidity burden (Table 2). P/MC patients were healthier, with a lower comorbidity burden and lower mean CCI scores (0.36; SD, 0.89), compared with all others (0.41; SD, 0.97; p < 0.0001). Patients who received P/MC were generally prescribed cardiovascular medications at lower rates compared with the mean overall rate among all patient groups, including angiotensin-converting enzyme inhibitors (14% vs 16.3%; p < 0.0001), loop diuretics (2% vs 3.1%; p < 0.0001), beta blockers (13% vs 14.8%; p < 0.0001), calcium channel blockers (9% vs 10.5%; p < 0.0001), and statins (27% vs 27.8%; p < 0.0001). Other classes of medication, including psychiatric medications, were prescribed at similar rates across patients using all bowel-preparation agents.
Efficacy/safety outcomes
Hospitalizations
Out of the 566,628 procedures in the study, a total of 3433 (0.6%) were associated with a hospitalization within 10 days of screening, of which 505 cases (14.7%) were for diagnoses of CRC, and the remaining 2928 cases (85.3%) were admissions for any cause other than CRC (Table 3). The rates of non-CRC hospitalization per 1000 screens were 3.78 for P/MC, 5.01 for other LV, and 6.13 for HV preparations.
Procedures and hospitalizations within 10 days of screening.

P/MC: sodium picosulfate and magnesium citrate; LV: low-volume; HV: high-volume; CRC: colorectal cancer.
LV agents, except P/MC.
Percentage of number of procedures by agent.
Percentage of total hospitalizations by agent.
Percentage of non-CRC-related total hospitalizations by agent.
While non-CRC admissions potentially related to the use of a cleansing agent (i.e. hyponatremia, dehydration, or other fluid/electrolyte disorders) were generally infrequent in the 10 days following the procedures (n = 23/2928 total non-CRC hospitalizations; 0.8%), the majority of these patients used other LV agents (n = 20/23), whereas no admissions were observed for P/MC patients. All other non-CRC admissions were for diagnoses unrelated to CRC screening.
While patients who used P/MC were hospitalized to a lesser degree in the 10 days following a procedure compared with all bowel-cleansing agents (4.6 vs 6.1 per 1000 screenings, respectively), a larger proportion of P/MC patients were hospitalized with a diagnosis of CRC compared with all agents (n = 27/154 (17.5%) vs n = 505/3433 (14.7%) hospitalizations, respectively).
Early repeat screenings
A total of 4359 (0.8%) screenings were identified as early repeat events, defined as any repeat screening occurring within 90 days of a previous procedure (no code was implemented), with the greatest early repeat rate associated with HV preparations (0.9%; Table 4).
Early repeat-screening events.
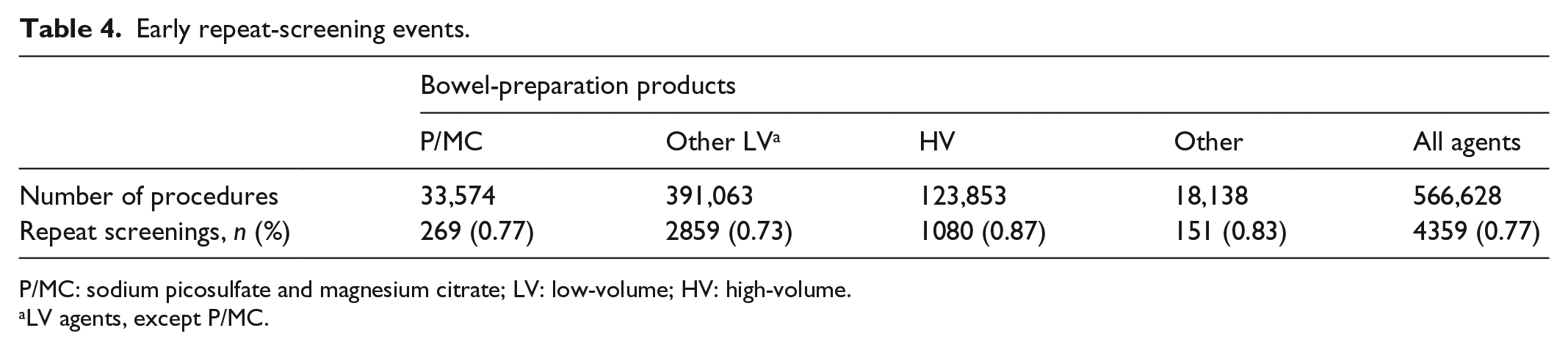
P/MC: sodium picosulfate and magnesium citrate; LV: low-volume; HV: high-volume.
LV agents, except P/MC.
Adjusted analyses
Adjusted analyses, controlling for age, sex, geographic location, and health status, showed that all bowel-cleansing agents compared favorably with each other. While estimates on repeat-procedure rates for patients who used P/MC were not significantly different compared with other LV products (odds ratio (OR), 1.103; 95% confidence interval (CI), 0.973–1.251; p = 0.1266), repeat-procedure rates for P/MC and other LV agents were significantly lower when compared with HV bowel-preparation products (OR, 0.858; 95% CI, 0.799–0.921; p < 0.0001; Table 5). In these analyses, the only factors significantly associated with higher rates of early repeat screenings were older age (all estimates, p < 0.03) and poor health status (all estimates, p < 0.0001). In adjusted analyses that compared P/MC with other LV agents and controlled for age, sex, and health status, no significant differences were detected in CRC hospitalizations within 30 days of screening agents (Table 5).
Odds ratio estimates.

OR: odds ratio; CI: confidence interval; P/MC: sodium picosulfate and magnesium citrate; HV: high-volume; LV: low-volume; CCI: Charlson Comorbidity Index; CRC: colorectal cancer.
LV agents, except P/MC.
Discussion
In this retrospective study of real-world clinical outcomes associated with colonoscopy and sigmoidoscopy procedures, we found that P/MC compared similarly with other commonly used LV/HV agents and standard-of-care bowel-cleansing preparations. P/MC performed well in terms of safety outcomes, with no hospitalizations due to a diagnosis of hyponatremia, dehydration, or other fluid disorders in the 10 days after a procedure, and the early repeat rate among P/MC patients was comparable with the rate observed for all other bowel preparations.
Previous studies have suggested that the timing of the preparation could influence the quality of the cleansing and the outcome of the procedure.22,32 According to these studies, a shorter preparation-to-colonoscopy interval may be more favorable than a longer interval, which leads to more efficacious bowel cleansing. The outcome associated with a shorter preparation-to-colonoscopy interval22,32 further suggests that preparations that are easier to consume may have an inherent advantage.
We found that patients using P/MC had fewer concomitant medications, which could be indicative of general health status, accompanied by a lower rate of hospitalization, a higher comparative proportion of CRC diagnoses, and no significant differences in post-screen CRC-hospitalization rates. The results could be associated with explicit patient characteristics, such as the relative younger age and higher proportion of female patients within this population.
While we have established a large sample size with a rigorous identification of measures and outcomes, a major limitation of the study is that we used data that reflect experiences of relatively younger individuals (mean age, 56.4 years) with insurance and as such, may not be generalizable to all patients who undergo colorectal screening. The short follow-up window should also be considered a limitation, as rates of repeat screenings and CRC diagnoses may be more accurate when observed over an extended period. However, it remains unclear how hospitalizations that occur ≥3 months after a screening should reflect on the outcome and success of a procedure. It may also be considered a limitation that canceled or rescheduled procedures would not show up in claims data, as the claims only reflect services rendered. Neither did this analysis include OTC agents, which are the majority of cleansing agents used prior to colonoscopy. It is also a significant limitation that split-dose preparations (standard of care) cannot be evaluated in cross-group comparisons using claims-based analysis. However, it is likely that the repeat screen rates constitute a mixture of split/non-split dose patients. Another limitation of the claims database is that it is not possible to extrapolate which patient populations were given which products or identify how those products were administered. However, given the large number of patients screened, these factors should be similar among the different agents, and would be factored into the number of patients returning for a repeat screen, despite being given the most appropriate bowel preparation. Finally, the use of claims data may have masked the influence of individual factors on the outcome of the bowel preparations and the subsequent screening procedures. 11
While the statistics from the National Cancer Institute have demonstrated a seemingly steady decline of CRC incidence and CRC mortality between 1992 and 2013 among those aged ≤50 years (Supplementary Figure 1), overall CRC incidence and death rates have increased by almost 2% per year within the same time frame.2,3 This observation is an indication of an ongoing and unmet need for adequate bowel preparations and more efficient and timely screening procedures. As the median age of the American population is steadily increasing, 33 it has been estimated that costs associated with CRC and the future economic burden to the Medicare program and its beneficiaries will be substantial, further highlighting the importance of adequate colorectal screening preparations. 34
Conclusion
The findings presented herein may assist physicians and policymakers in modifying standard-of-care screenings to reduce the negative outcomes associated with inadequate preparations. P/MC and LV products are well tolerated with similar cleansing quality and AEs comparable with those of HV products. Future research may provide further guidance on how the proper use of P/MC, LV, and HV agents, or other bowel-cleansing agents, may increase rates of adequate preparation and subsequent successful screening procedures, as well as reduce rates of hospitalization and early repeat events, ultimately enhancing overall patient care.
Footnotes
Acknowledgements
Editorial support was provided by The Curry Rockefeller Group, LLC, Tarrytown, NY, USA.
Declaration of conflicting interests
L.E.Y. and D.N.D. are employees of Ferring Pharmaceuticals Inc. N.C.S., P.L.C., and A.S. are employees of Precision Health Economics.
Ethical approval
Ethical approval was not sought for this study because this study is based on claims data from Truven Health Analytics MarketScan databases.
Funding
This work was supported by the Ferring Pharmaceuticals Inc.
Informed consent
Informed consent was not sought for this study because this study is based on claims data from Truven Health Analytics MarketScan databases.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
