Abstract
Dermal filler and botulinum toxin injections are among the most performed non-invasive procedures for rejuvenation and contouring, internationally. Although most dermal fillers and approved botulinum toxins are well tolerated with a high safety profile, adverse events and/or complications can happen. Injection-related sequelae and side effects are usually rare, mild, transient, and self-limiting in nature. However, devastating and life-changing complications, such as intravascular occlusion, skin necrosis, permanent visual impairment, and stroke, have been reported. Risk reduction measures include an in-depth knowledge of anatomy, physiology, and pharmacology, correct patient selection, correct identification of clinical indications, and, fundamentally, the importance of appropriate skill and training. In this paper, some of the complications are discussed and advice on how to minimise adverse events and complications from these injectables is provided.
Prim Dent J. 2023;12(3):65-72
Learning Objectives
To review the use of terminology in terms of sequelae, complications, adverse events, and side effects in relation to the injection of hyaluronic acid and botulinum toxins
To understand and recognise adverse events and complications of hyaluronic acid and botulinum toxin injections for facial areas
To advise on preventive measures to avoid adverse events and complications following hyaluronic acid and botulinum toxin injections
Notes
Complications and adverse events (AEs) are used interchangeably in the literature. Please see the terminology section (Table 1).
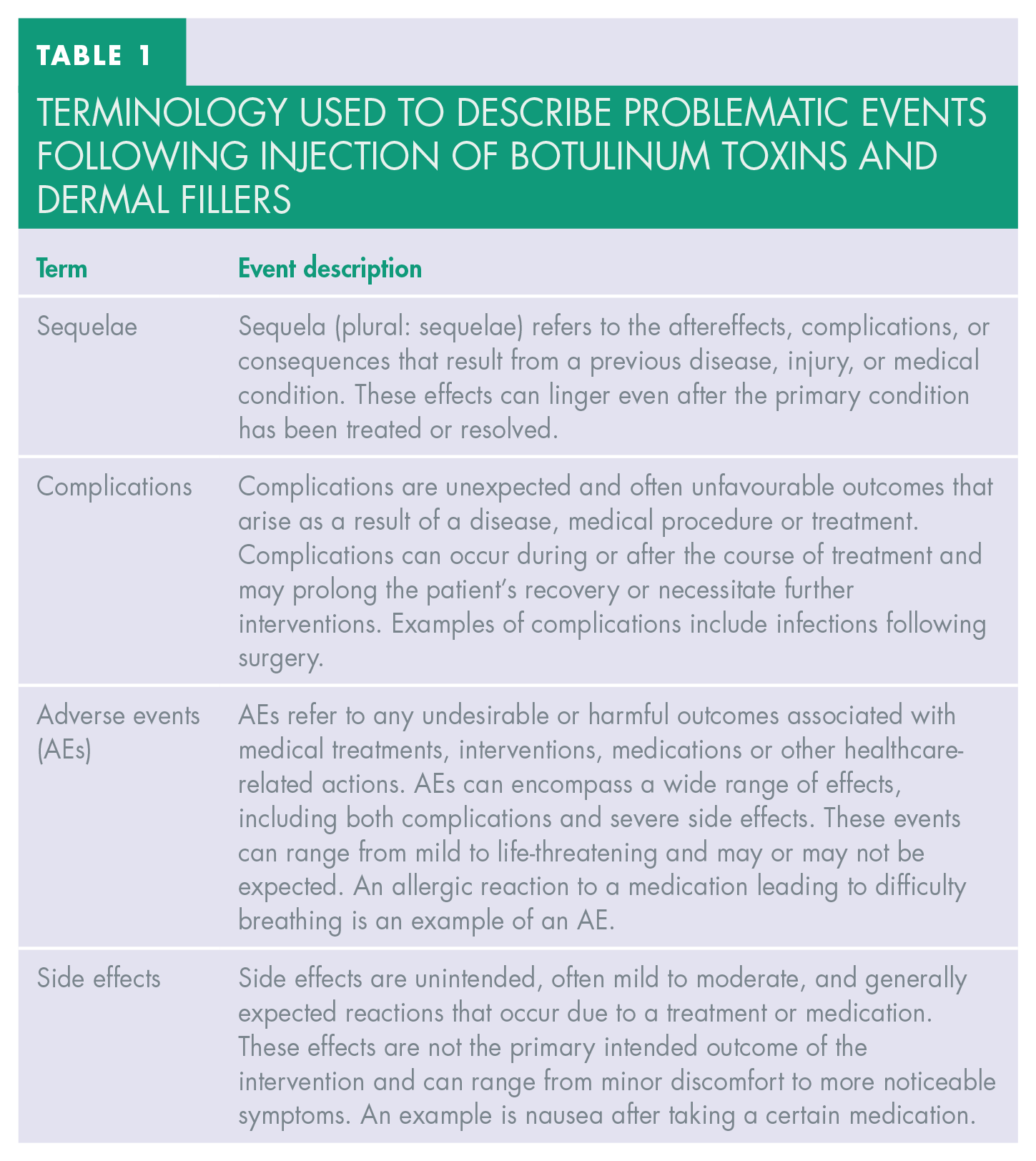
Terminology used to describe problematic events following injection of botulinum toxins and dermal fillers
Botulinum toxins, neuromodulators, neurotoxins are used interchangeably in the literature.
Botulinum toxin type A (BoNT-A) is produced by the bacterium Clostridium botulinum and is available in several different formulations, including but not limited to:
OnabotulinumtoxinA (Botox, Botox Cosmetic)
AbobotulinumtoxinA (Dysport)
IncobotulinumtoxinA (Xeomin)
PrabotulinumtoxinA (Jeuveau)
Each of these products has specific indications, dosage, and administration practices. Therefore, practitioners should indeed familiarise themselves with the specifics of the product they are using. It is important to note that these products are not interchangeable. Their units of biological activity are different, their characteristics are different and may also have different safety profiles. The individual products should be used according to their approved indications and dosing recommendations.
Introduction
The demand for non-surgical and minimally invasive procedures for rejuvenation and prevention of ageing signs is increasing exponentially worldwide.1-3 This is not limited to new technologies, devices or injectables but also encompasses pharmaceuticals, supposed “youth pills,” nutraceutical supplements, hormones, and stem cells.4,5 Anti-ageing interventions in aesthetic medicine partly fall into anti-ageing medicine, a discipline of medical science and applied medicine that is still developing. Anti-ageing interventions attempt to treat the foundations of ageing, prevent ageing, and alleviate any age-related disease to extend healthy and youthful life. 5 Aesthetic medicine focuses on anti-ageing and rejuvenating procedures.
The use of botulinum toxin injections and dermal filling agents has been reported as the most requested and delivered non-surgical cosmetic treatment worldwide. 6 Botulinum toxin was the most popular non-surgical cosmetic procedure globally in 2020. According to the statistics published by the American Society of Plastic Surgeons, hyaluronic acid (HA) fillers were the second most popular non-surgical cosmetic procedures. This has been consistent for many years. 6
The safety profile and tolerability of BoNT-A and HA dermal fillers have been proven through extensive clinical experience internationally.7-9 However, AEs have become more noted in recent years, and expected to increase. 10 This can be due to a combination of various factors, including:
lack of regulation and licensing in many countries. This means people with minimal or no formal training are providing injections of botulinum toxins and dermal filling agents. Unfortunately, this includes non-healthcare professionals who lack a basic understanding of anatomy, physiology, pharmacology, procedures, complications prevention, and general patient management;
lack of structured, quality-assured training;
the introduction of many new products to the market, some of which do not have supporting scientific evidence or licensing;
increased population demand for such procedures
Current literature shows that the overall rate of AEs is comparable among BoNT-A products, with individual AEs being somewhat dependent on the facial area treated with post-injection sequelae (see Table 1) and headache being the most reported.7,8,10-13
There are many filling agents on the market, including fat, bio-stimulatory agents, temporary, semi-permanent, and permanent agents. These can be categorised as fast biodegradable fillers and slow, late, or minimally biodegradable fillers. HA dermal fillers are the most popular and commonly used fillers, and due to the limited scope of this paper, our focus will be on HA dermal fillers.
When administered with thorough knowledge of anatomy and correct injection techniques, HA fillers are generally associated with a low incidence of complications.14-18 However, AEs can occur even in the most experienced hands, ranging from self-limiting to serious issues requiring prompt recognition and management.18-20
Immediate sequelae and AEs associated with HA dermal fillers include pain, oedema, erythema, bruising and haematoma, and paraesthesia. Palpable or visible lumps are mostly technique related. Serious complications are rare and include delayed onset nodules and/or vascular compromise/occlusion that can lead to tissue necrosis, stroke, and permanent visual impairment.19,21-30 These can be prevented and minimised with comprehensive knowledge of the relevant anatomy, pharmacology, and physiology, together with the use of a correct injection technique.
Fundamentals of prevention of adverse events (AEs)
AEs with BoNT-A and HA dermal fillers can generally be categorised into three groups:31,32
patient-related
product-related
technique-/procedure-related lack of knowledge insufficient experience
Botulinum toxin A (BoNT-A)
BoNT-A formulations have demonstrated outstanding safety and tolerability profiles across various cosmetic and therapeutic uses, based on published research, clinical experience, retrospective reviews, and meta-analyses.7,8,33,34 Different formulations available in the market are chemically and pharmacologically unique with different manufacturing processes, and hence their doses are not interchangeable. Furthermore, their dose-response curves are not parallel.35,36 This is an important consideration. AEs are mainly due to technique-related factors, inaccurate assessment of the target muscles (facial assessment), lack of knowledge of facial anatomy and lack of understanding of product characteristics, including “excessive action of the drug,” or “diffusion to nearby unwanted targets”.25,31,32 Examples of these include: 7
blepharoptosis (eyelid ptosis)
brow ptosis
eye sensory disorders
lip asymmetries
imbalances of the lower face
Patient-related factors include assessing the patient’s overall well-being and health, taking a comprehensive medical history, checking for body dysmorphic disorder (BDD), and checking for hypersensitivity type I or IV to botulinum toxins or any ingredient in the formulations used. Pregnancy, breastfeeding, infection at the injection site, neuromuscular disorders are other examples of patient-related factors. 10 Clinicians should assess for contraindications, warnings, or precautions for each individual, including medical problems, medications, allergies, recent or planned medical, dental, and other cosmetic procedures, and previous cosmetic treatments (date and modality of the procedure). It is beyond the scope of this paper to discuss all medical problems and medications, and these are dependent on the type of injectable being provided. A short list of medical problems and medications that contradict injection of BoNT-A can be seen below:31,32,37,38
allergic reaction or hypersensitivity to botulinum toxin or any of the ingredients in the formulation
infection at the proposed injection site
immunocompromised patients
neurological disorders such as amyotrophic lateral sclerosis (ALS), myasthenia gravis, or Lambert-Eaton syndrome
pregnancy and breastfeeding
concurrent use of certain antibiotics, such as aminoglycosides, that can increase the effects of the toxin
Recommendations and prevention strategies:10,12,34
an individualised treatment plan should be utilised, as textbook or recommended injection points in the literature do not apply to all individuals. Comprehensive examination should be carried out for each person and guidelines should be used to draw a treatment plan that is most suitable according to the individual’s anatomy, desires and expectations and the planned outcome
various formulations are not interchangeable, and the clinician should be familiar with the characteristics of the product they use
analysis of target muscles and muscles in the proximity, and their interaction for various facial expressions and functions is paramount
the lowest optimal dose should be administered and the number of booster injections reduced/eliminated
non-essential over-the-counter drugs that might affect coagulation and platelet activity can be discontinued for one week before the procedure to reduce the potential risk of bleeding and bruising. These include some herbal supplements. Many herbal products possess antiplatelet and/or anticoagulant properties. Contemporary literature suggests that many herbal supplements known to interfere with blood clotting predominantly work by inhibiting platelet activation and aggregation.39-42 Furthermore, use of some of these supplements interfere with the activity of various prescribed medication.43,44 Some examples include ginkgo biloba, garlic, Asian ginseng, St. John’s wort, and saw palmetto
cleanse the skin of dirt and make-up with a cleanser
antisepsis of the area using chlorohexidine, iodine, or hypochlorous acid based disinfectant
after antisepsis, adhere to aseptic non-touch technique (ANTT) and only touch the injection site with a sterile disposable needle
provide patients with verbal and written aftercare instructions, including information for contacting the clinic in case of emergencies
Dermal fillers
The injection of HA dermal fillers is similar to placing an implant into various layers and units of the face. Understanding this point highlights the importance of the clinicians’ knowledge of anatomy, product characteristics and expertise (injection technique) in reducing the risk of complications, enhancing outcomes, and managing AEs. Although HA dermal fillers have limited longevity and are biodegradable, the tissue longevity of these products is substantially longer than advertised by pharmaceutical companies.45-47 Various types of HA dermal fillers have different properties and vary in terms of cross-linking and cross-linking type, gel particle size, and concentration of cross-linked and free HA. Several types of dermal fillers (distinct from HA fillers) exist with unique chemical and biophysical properties and safety profiles which necessitate careful examination by clinicians. For example, calcium hydroxylapatite (CaHA) fillers are biocompatible and biodegradable, known for their collagen-stimulating properties. 48 Poly-L-lactic acid (PLLA) fillers, composed of biodegradable synthetic polymers, typically yield gradual results and can influence collagen synthesis. 49 Polycaprolactone (PCL) fillers are a type of biodegradable, collagen-stimulating dermal filler. 50 Polymethylmethacrylate (PMMA) microspheres (non-biodegradable and providing a permanent implant effect) present a different safety profile which requires careful evaluation. 51 However, a thorough and detailed discussion of each filler type is beyond the scope of this paper.
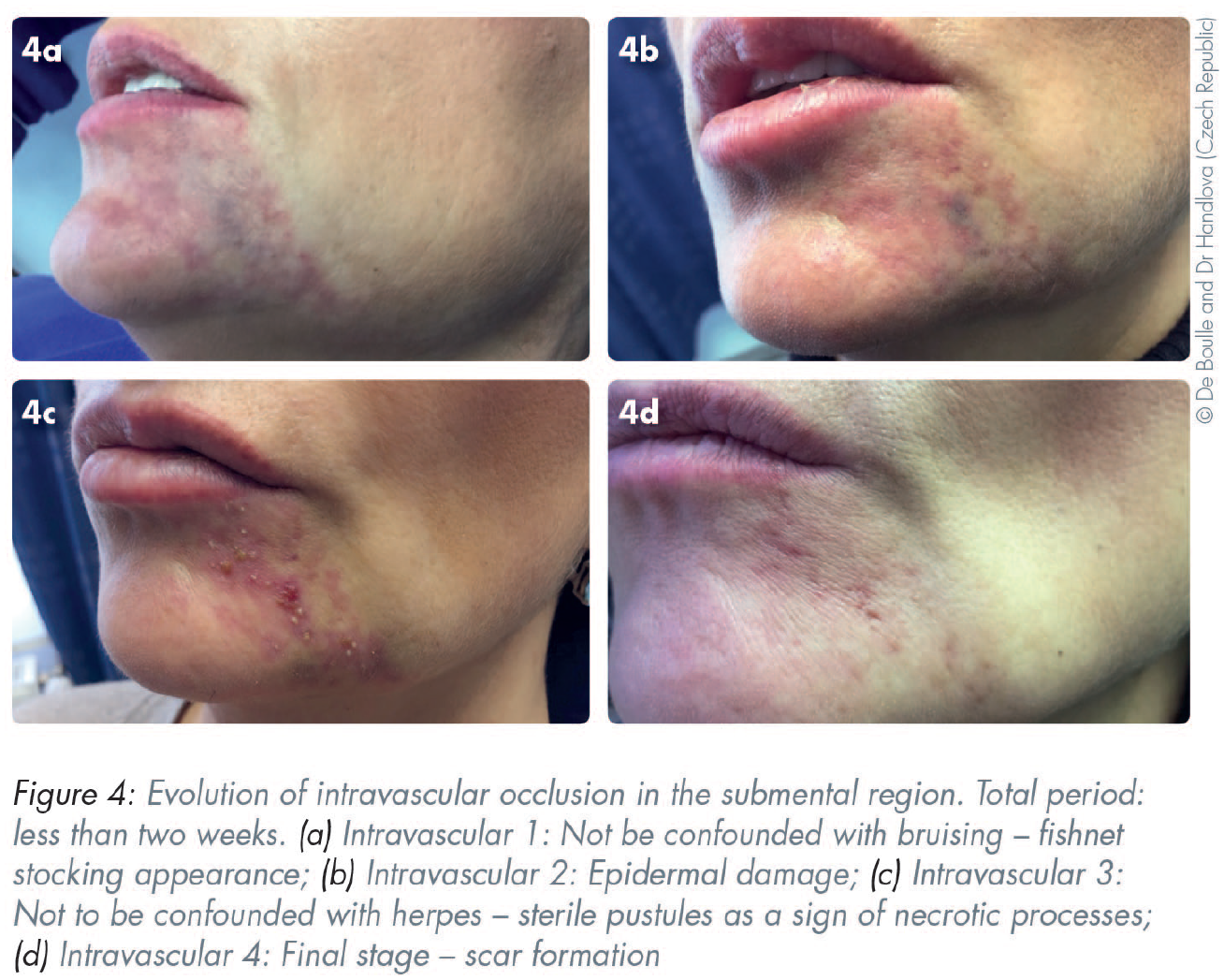
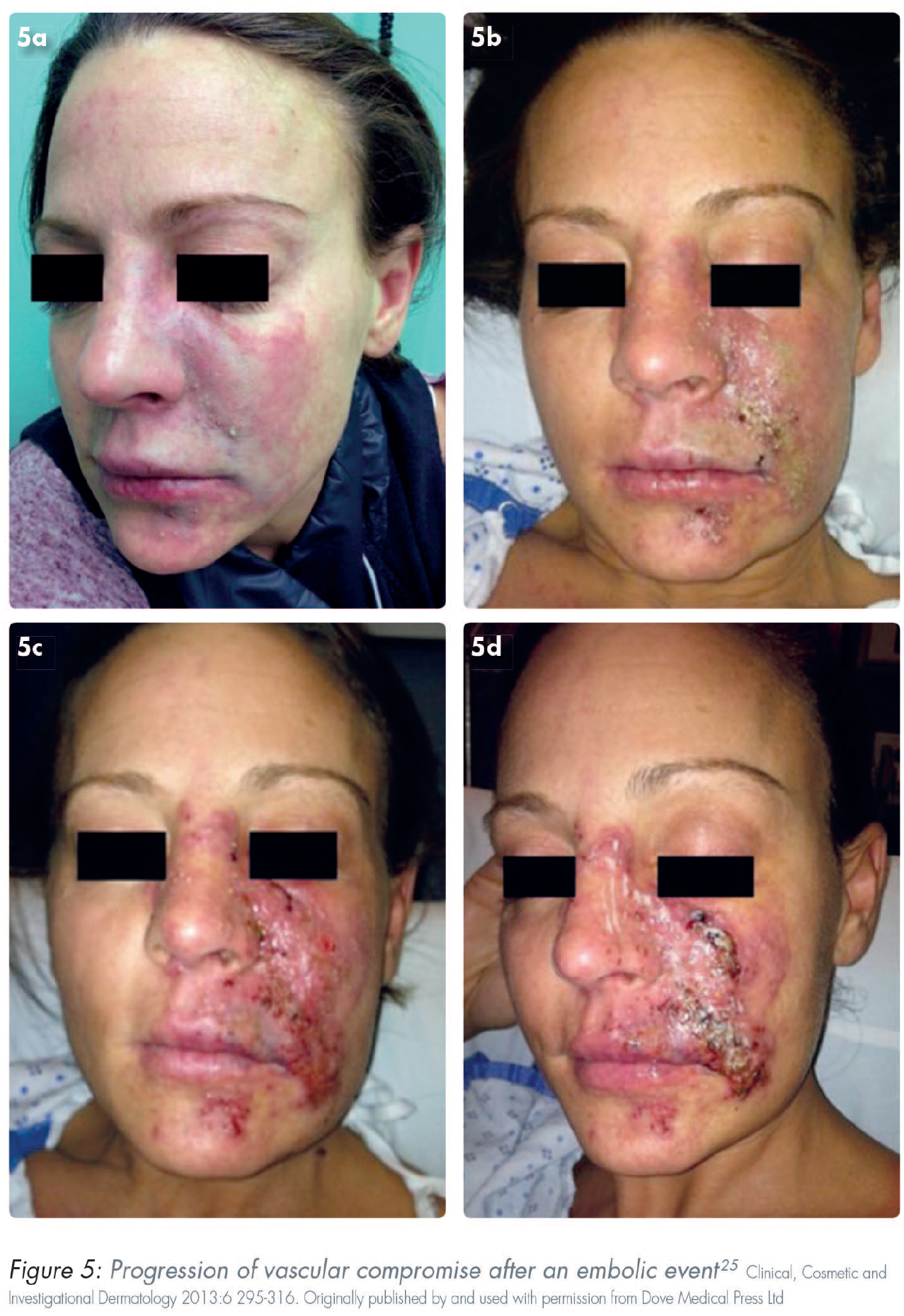
HA dermal fillers have a good overall safety profile.19,52-54 Mild to severe reactions might occur immediately after injection or may be delayed. Common sequelae include localised and transient responses. Examples of these include erythema, bruising, and pain. Nodules, vascular occlusion, and visual complications are less common but have more severe consequences (Figures 1–5). Delayed-onset adverse reactions include cyclic or persistent oedema, erythema, late-onset inflammation, and nodules. Late-onset nodules have been reported and could occur due to a variety of factors, including hypersensitivity, foreign body reaction, injection placement, infection, sterile abscess, or biofilm development (Table 2).18,25,55-58
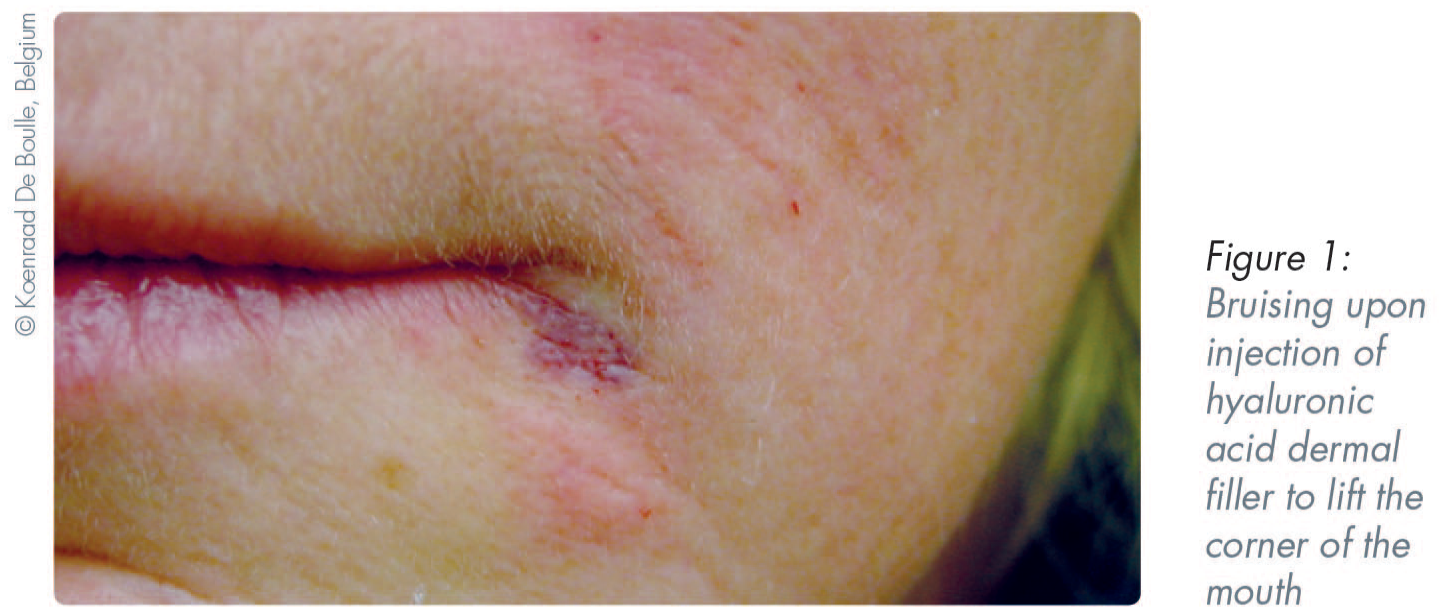
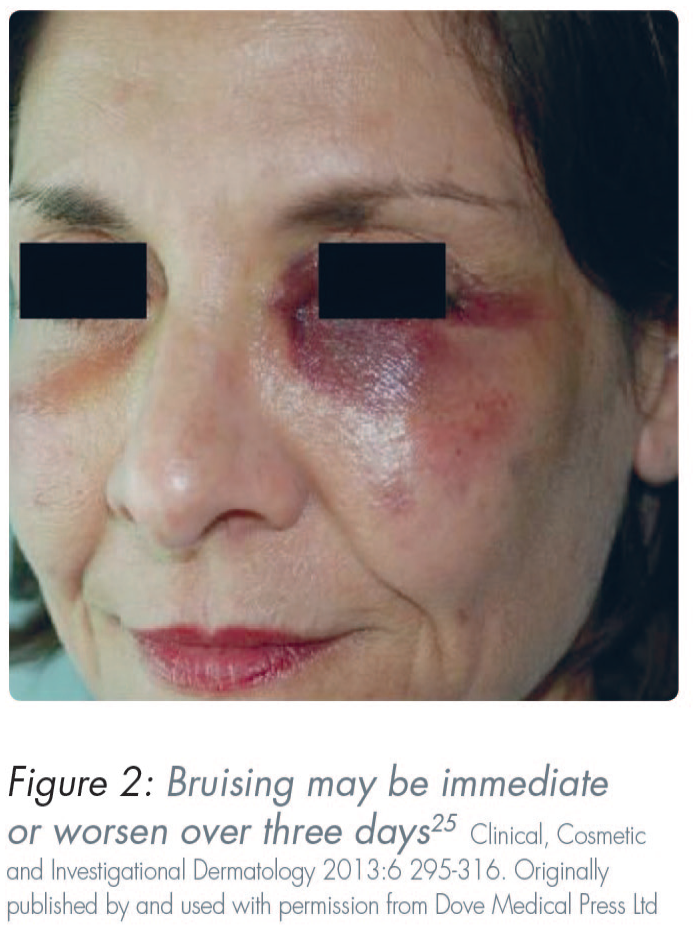
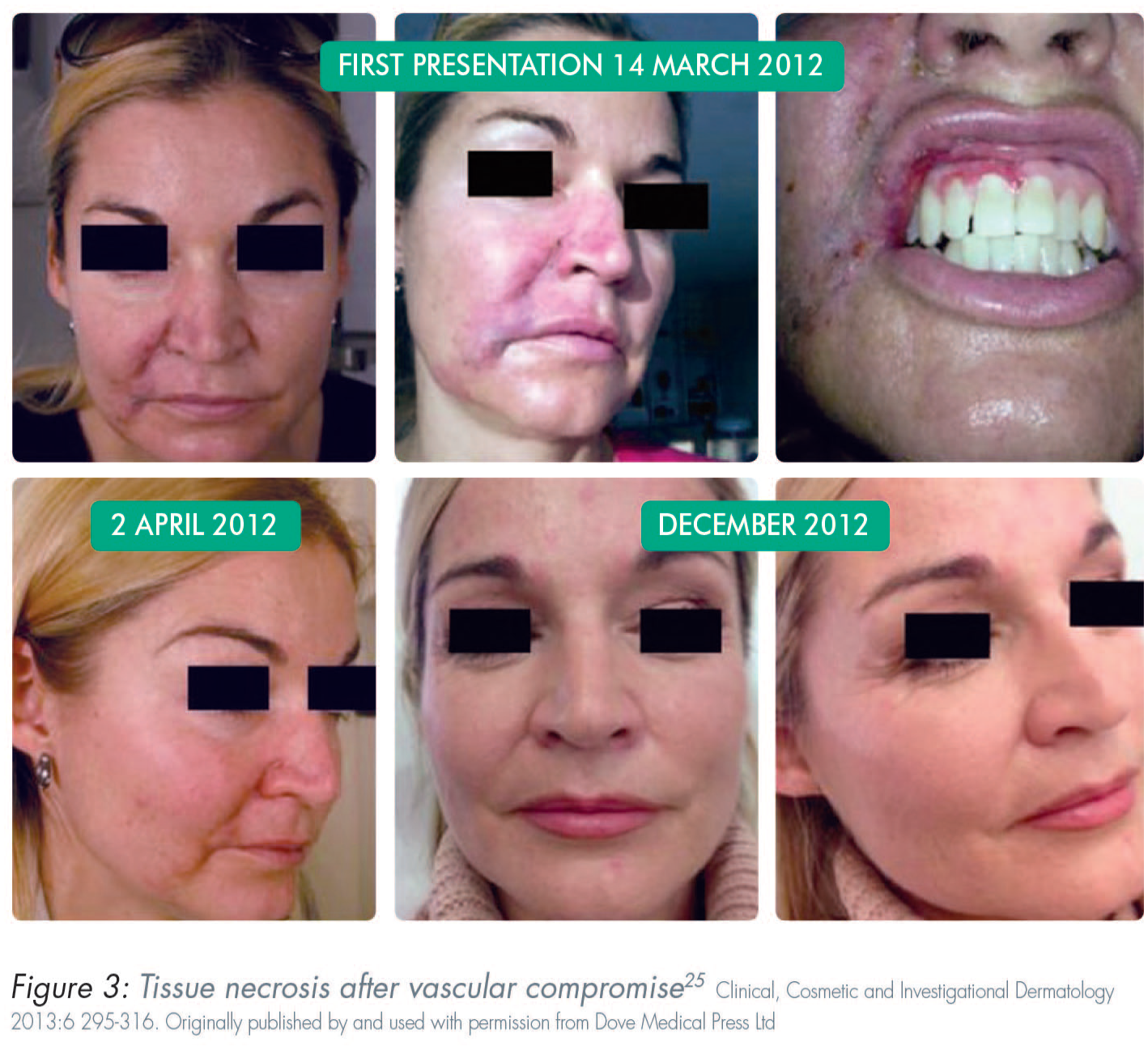
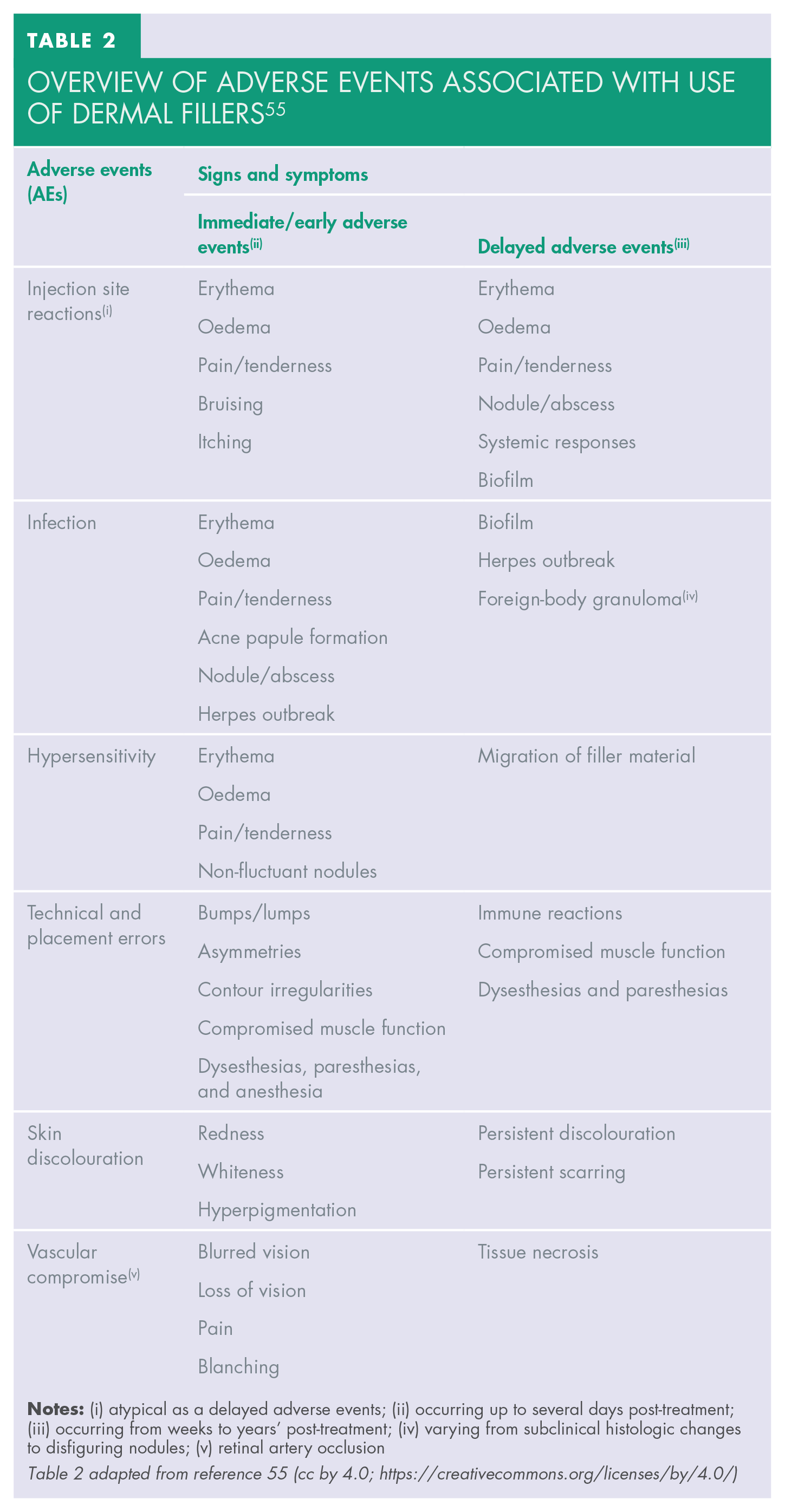
Overview of adverse events associated with use of dermal fillers 56
Careful evaluation should be carried out before providing any injectable cosmetic procedures. Recent medical problems or existing and chronic conditions, allergies (including hypersensitivity to HA or gram-positive bacterial protein, lidocaine or amide-type local anaesthetics) and mental well-being should be screened. It should be kept in mind that patients are not always forthcoming with their medical and cosmetic history, affecting diagnosis and proper management of reactions. Risk factors include:10,59
history of prior complications with soft-tissue fillers
multiple prior filler treatments
poor personal hygiene
poor oral hygiene
immunocompromised state
chronic or recurrent skin conditions
current herpes labialis
uncontrolled type 2 diabetes mellitus
history of previous surgery or permanent implants/fillers in the target area
Treatment for unwell patients (e.g. those with fever, cold or influenza, or overly tired, overly stressed) should be delayed. 10 Clinicians should appropriately assess the risk:benefit ratio according to patients’ medical conditions, such as controlled autoimmune disease with sound healing.
All risks (minor, major, common, and rare) associated with dermal filler injections should be included in informed consent forms including all possible visual complications (e.g. ocular pain, double vision, or blindness in one or both eyes). Vascular occlusion may occur up to 72 hours following treatment; it is therefore essential that clinicians are always available for review, or a plan has been made for a colleague who can be available and have access to hyaluronidase. Hyaluronidase is an enzyme that can degrade HA, a major component of the extracellular matrix in the human body. In the context of dermal fillers, hyaluronidase is often used to dissolve HA-based fillers in the case of complications or if the patient is unsatisfied with the results of the filler injection. 60 Consent for use of hyaluronidase and the reversal of HA fillers should be obtained. 10
A list of formulated recommendations and prevention strategies to help the aesthetic practitioner are highlighted here:10,12,34
Patient consultation and the importance of understanding the patient’s all underlying systemic and local disease, allergies and hypersensitivities, cosmetic history and any planned medical, cosmetic, or dental procedures should not be undermined.
Practitioners should remain vigilant for patients with unrealistic expectations of aesthetic procedures, as this can indicate potential dissatisfaction with treatment outcomes and may require additional patient education or psychological evaluation.
It’s imperative that aesthetic practitioners are well-versed in recognising symptoms of BDD, a psychiatric condition characterised by excessive preoccupation with perceived flaws in appearance. Patients demonstrating characteristic signs of BDD warrant referral for appropriate mental health evaluation and counselling. Insufficient familiarity with BDD can lead to unnecessary procedures on individuals whose dissatisfaction with their appearance is primarily psychological. Therefore, if aesthetic practitioners lack the necessary knowledge and skills to identify these patients, it’s strongly recommended that further training is undertaken. Many postgraduate courses in aesthetic medicine include components that provide practitioners with these crucial identification skills.61,62
All risks, including vascular compromise, permanent visual impairment, and stroke, should be included in the consent forms and patients educated.
Hyaluronidase and AE management protocols must be available before carrying out the procedure.
Aseptic technique.
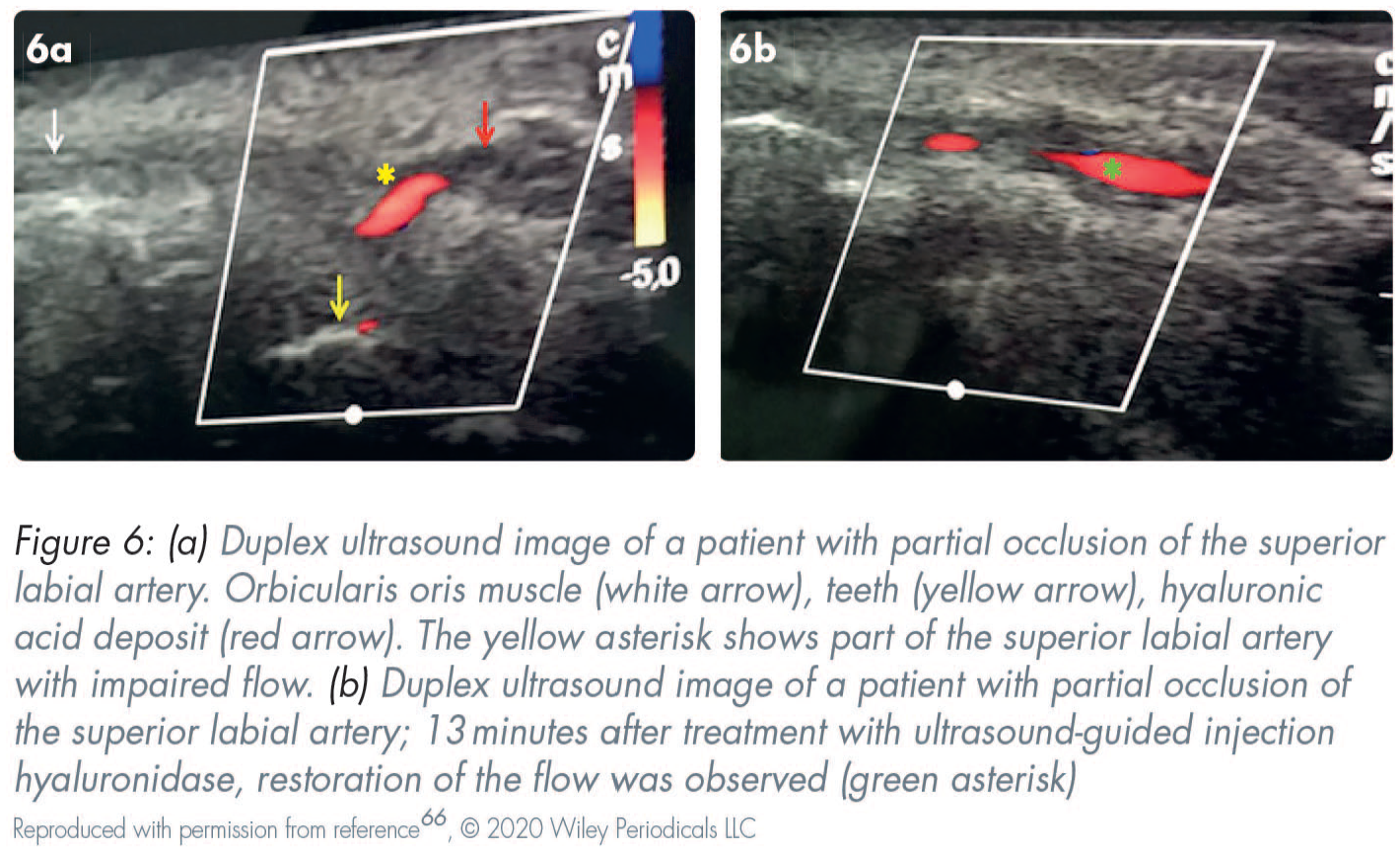
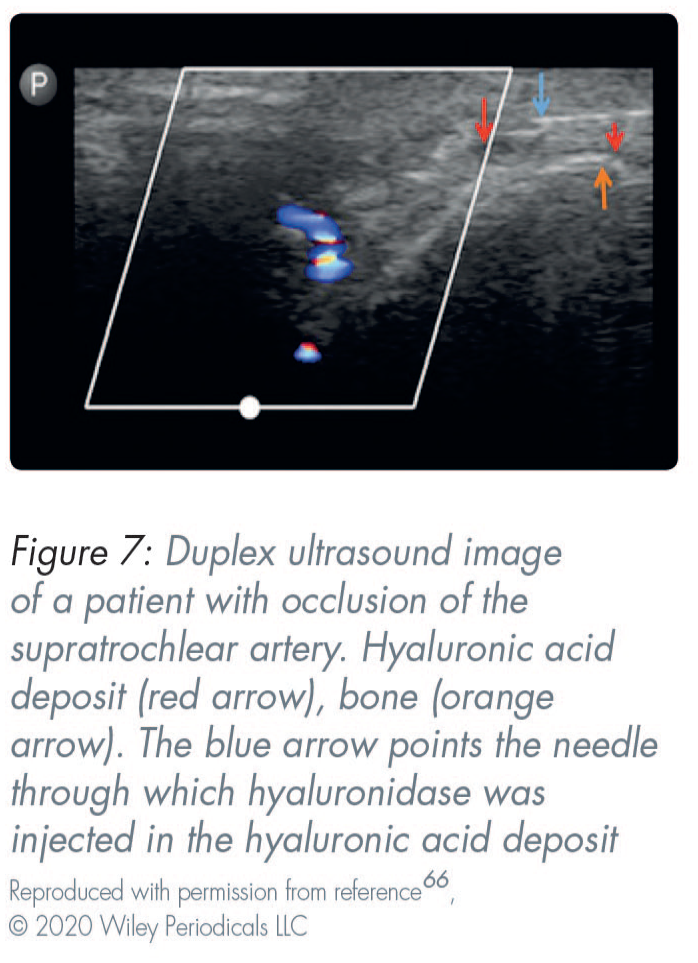
High-frequency ultrasound scanning can be used for assessment and during treatment provision to minimise complications (Figures 6 and 7). This is a non-invasive, convenient, and rapid technique. 63 Ultrasonography is a valuable and non-invasive tool for identifying the site, quantity, real-time diagnosis of the type of implant/filler that has been placed/injected, and for the study of complications.64,65 Before injections, duplex ultrasound is useful for assessing the individual anatomy and mapping the vascular structures. This helps reduce the chances of intravascular injections. 66
Recommendations10,31,32,67
Attend formal structured training courses rather than one-day training sessions.
Keep up-to-date with publications and current recommendations.
Attend complications management workshops on an annual (or more regular) basis.
Be willing to learn from more experienced colleagues and ask questions when in doubt.
Practice in a clinical environment.
Take time during consultations and during patient counselling.
Patient education plays an important role. Ensure patients are well-informed regarding their treatment procedure, products used, and all possible complications (common and rare), post-care instructions, and be “alert” regarding possible late-onset/AEs.
Cosmetic treatments are medical procedures and should be treated with the same respect, clinical judgement, and patient care.
Assess your patients for possible underlying psychological issues such as BDD. By improving your communication skills, you will feel more comfortable conversing with your patients, refusing treatments, and making the required referrals.
Keep AE protocols in your practice and readily available (similar to CPR training instructions and manuals).
Team training can help manage complications and deal with anxious patients in the case of AEs.
Do not underestimate patient-related factors.
Purchase your products from reputable resources.
Have the tools available in your clinic and be prepared to treat AEs confidently.
Remember that prevention is critical.
Conclusions
Injectables such as BoNT-A and HA filling agents that are produced through comprehensive research and development have a well-established safety profile. AEs and complications are infrequent, and most are of mild-to-moderate severity, transient and self-limiting. However, in some rare cases, severe and life-changing AEs (e.g. intravascular injection, necrosis, and visual impairment) can occur. These outcomes need to be diagnosed and managed promptly. The majority of other complications are related to the techniques and delivery systems employed. As a result, clinicians must be completely aware of all potential consequences, be prepared to utilise established preventative techniques and provide appropriate remedial treatment. For optimal outcomes, a detailed knowledge of anatomy and accurate injection techniques are essential.
