Abstract
Objective
To investigate if the addition of bedaquiline and clofazimine to a treatment regimen for multidrug-resistant tuberculosis (MDR-TB) could improve patient outcomes.
Methods
A prospective, randomized, controlled study was conducted in patients with MDR-TB. Treatment was for 18 months. Patients in the experimental group received bedaquiline and clofazimine in addition to their regular treatment regimen whereas patients in the control group did not.
Results
68 patients with MDR-TB were randomised to treatment, 34 to each group. At the end of treatment, cure rates were statistically significantly greater for the experimental group compared with the control group (82% vs. 56%). There was no difference between groups in the number of severe adverse events (3[9%]) in both groups and none were skin-related.
Conclusions
The addition of bedaquiline and clofazimine to the treatment regimen significantly improves outcomes for patients with MDR-TB. Clinicians should be aware of the clinical benefits of this addition but be mindful of contraindications and adverse effects.
Introduction
Multidrug-resistant tuberculosis (MDR-TB) refers to patients infected with Mycobacterium tuberculosis whose drug susceptibility testing in vitro confirms resistance to isoniazid and rifampin. 1 According to the World Health Organisation (WHO), in 2021 there were 450,000 new cases of rifampin-resistant tuberculosis (RR-TB) reported. 2 MDR-TB has become a serious public health problem and a serious challenge for TB control.3–5 Long treatment courses, high treatment costs, low cure rate, and adverse reactions to second-line drugs are just some of the problems perplexing researchers working in the treatment of MDR-TB.6–8 These difficulties combined with the strong infectivity of TB, has made the control of MDR-TB an important problem to be solved.9–10
The discovery of bedaquiline, has given new hope to patients with MDR-TB. 11 Bedaquiline is a diarylquinoline that disrupts the activity of the mycobacterial adenosine triphosphate (ATP) synthase. 12 The drug binds to the oligomer subunit C of ATP synthetase 13 and affects the activity of the ATP synthetase proton pump leading to obstruction of ATP synthesis.14,15 Therefore, it prevents the ATP energy supply of M. tuberculosis and has both bacteriostatic and bactericidal effects. 15 Although there are several reports from various countries on the efficacy of bedaquiline in the treatment of MDR-TB, there are few studies that have evaluated bedaquiline combined with clofazimine in MDR-TB.16–18 The purpose of this present study was to investigate the efficacy of the addition of bedaquiline and clofazimine to a treatment regimen for MDR-TB.
Methods
Study population
Consecutive patients who began treatment for MDR-TB at Wuhan Jinyintan Hospital from January 2019 to December 2019 were eligible for this prospective, randomized, controlled study. Eligible patients were ≥18 years of age, had no symptoms or signs of pregnancy, and no serious or uncontrolled heart, liver, kidney, or psychiatric disease. In addition, they had confirmed MDR-TB by phenotypic or genotypic drug susceptibility testing. Phenotypic drug susceptibility testing was conducted using the proportional agar method on Lowenstein-Jensen medium, while genotypic drug susceptibility was assessed using the GenoType MTBDRplus assay. Exclusions were as follows: extensive drug resistant-TB (i.e., XDR-TB [resistant to fluoroquinolones AND second-line injectable drugs in addition to MDR-TB]); pre-XDR-TB (resistant to any fluoroquinolone OR second-line injectable drug in addition to MDR-TB);1,19,20 infected with nontuberculous mycobacteria; severe comorbidity; presence of human immunodeficiency virus (HIV); acquired immunodeficiency syndrome (AIDS).
The reporting of this study conforms to the CONSORT statements. 21 The study was approved by the Medical Ethics Committee of Wuhan Jinyintan Hospital and registered with the Chinese Clinical Trial Registry (ChiCTR 1800020391). Written informed consent was obtained from each patient and patient data were anonymized prior to analysis.
Study procedures
Randomization was conducted using a computer generated random-number table. When the researchers determined that a patient was eligible, basic information was transmitted by telephone to central randomization system staff who assigned the patient to a group based on a random-number table and relayed this information to the researcher. Neither the researchers nor the patients were aware of the assigned treatment.
Patients were randomized to an experimental or control group for 18 months. Patients in the experimental group received bedaquiline, levofloxacin, linezolid, cycloserine and clofazimine for six months. (Figure 1). For the next 12 months they received the same treatment regimen without bedaquiline. Patients in the control group received bedaquiline, levofloxacin, linezolid, cycloserine and protionamide for six months. Ethambutol was used instead of protionamide in cases of drug sensitivity or intolerance. For the next 12 months they received the same treatment regimen without bedaquiline.

All patients received 18 months of daily treatment. Patients in the experimental group received bedaquiline (Bdq); levofloxacin (Lfx); linezolid (Lzd); cycloserine (Cs); clofazimine (Cfz) for the first six months. For the next 12 months they received: Lfx; Lzd; Cs; Cfz. Patients in the control group received: Bdq; Lfx; Lzd; Cs; protionamide (Pto) for 6 months. For the next 12 months they received: Lfx; Lzd; Cs; Pto. Ethambutol (EMB) was used instead of Pto in cases of drug sensitivity or intolerance.
For bedaquiline, patients received 400 mg qd for two weeks, followed by 200 mg qd three times weekly for 22 weeks. For levofloxacin, patients ≤45 kg received 750 mg qd and patients >45 kg received 1000 mg qd. For linezolid, patients received 600 mg qd. For cycloserine, patients ≤45 kg received 250 mg bid and patients >45 kg received 250 mg in the morning and 500 mg in the evening. For clofazimine, patients received 100 mg qd. For protionamide, patients ≤45 kg received 300 mg bid and patients >45 kg received 400 mg bid. For ethambutol, patients ≤45 kg received 750 mg qd and patients >45 kg received 1000 mg qd. In addition, vitamin B6 was added to all treatment regimens to prevent any neurotoxicity.
Patients’ signs, symptoms and adverse events were assessed monthly. Pre-treatment and then at monthly intervals the following tests were obtained: routine blood and urine; liver and kidney function; electrolyte levels; thyroid function; electrocardiogram (ECG); sputum smear microscopy; sputum TB culture; TB drug sensitivity; visual acuity; colour vision. Chest computed tomography (CT) scans were taken pre-treatment and then every three months during treatment. Changes in CT lesions were defined as absorption, invariance or deterioration. Absorption was further classified as, ‘effective’ or ‘ineffective’ (i.e., unchanged/worsened).
For TB sputum culture, at least two consecutive negative samples, ≥30 days apart, was considered a negative result. The time taken for the first sputum sample to be recorded as negative was taken as ‘negative time’. Treatment outcomes for patients with MDR-TB were defined according to WHO guidelines:
22
They were as follows:
Cure: the patient completed the course of treatment without evidence of treatment failure and had ≥3 consecutive negative sputum cultures at least 30 days apart after the intensive phase. Treatment completed: the patient completed the course of treatment without evidence of treatment failure, and there was no evidence of negative sputum cultures in ≥3 consecutive sputum cultures at least 30 days apart after the intensive phase. Treatment failed: patients had to stop treatment or permanently change their regimen (more than two drugs) for the following reasons: (a) at the end of the intensive phase, TB sputum culture was positive; (b) TB sputum culture became negative and then returned to positive in the continuation phase; (c) evidence of resistance to fluoroquinolones or second-line injectable drugs; (d) severe adverse drug reaction. Death: The patient died from any reason during the course of treatment. Lost to follow-up: the patient stopped treatment for two months or more for any cause.
These outcomes were subsequently classified as either favourable (i.e., cure) or adverse (i.e., treatment failure, death, or lost to follow-up).
Statistical analyses
Statistical analysis was performed using SPSS software (version 22.0 for Windows®; SPSS Inc., Chicago, IL, USA). A P-value <0.05 was considered to indicate statistical significance.
Data were described as mean ± standard deviation (SD). The student’s t-test and Wilcoxon U test were used for normally and non-normally distributed variables, respectively. Counting data were expressed as percentages and compared using χ2 test. According to sample size calculations, if the probability of making type I error was ≤5%, the probability of making type II error was ≤10%, and the two groups were required to have the same number of cases; at least 24 patients were required in each group. However, 34 patients were enrolled in each group and so an effective sample size was achieved.
Results
In total, 68 patients with MDR-TB were randomised to treatment, 34 to the experimental group and 34 to the control group (Figure 2). One patient was lost to follow-up in each group and so 33 patients in each group completed the 18-month study. There were no statistically significant differences between groups in demographic or clinical characteristics (Table 1). Across both groups, 51 (75%) patients had previously received anti-TB treatment, with a median treatment duration of 19 months (range 2–120 months). Drug resistance characteristics of both groups are shown in Table 2. Most patients had shown resistance to two or three TB drugs. At the end of treatment, the rates of sputum TB cultures turning negative in the experimental and control groups were 82% (28/34) and 56% (19/34), respectively (Table 3). This difference was statistically significant (χ2 test, 5.58; P < 0.025). At the end of treatment, the rates of ‘effective’ absorption of pulmonary lesions as assessed on CT scans were 82% (28/34) and 56% (19/34), in the experimental and control groups, respectively (Table 4). The difference between groups was statistically significant (χ2 test, 5.58; P < 0.025).

CONSORT Flow diagram. 21
Demographic and Clinical Characteristics.

Data are expressed as mean ± standard deviation or n (%).
ns, not statistically significant; TB, tuberculosis.
Drug resistance characteristics of the two groups.

Data are expressed as n (%).
ns, not statistically significant.
Negative sputum tuberculosis cultures at each timepoint.

Data are expressed as n (%).
Number of patients with ‘effective’ absorption of lung lesions as assessed by CT scan*.
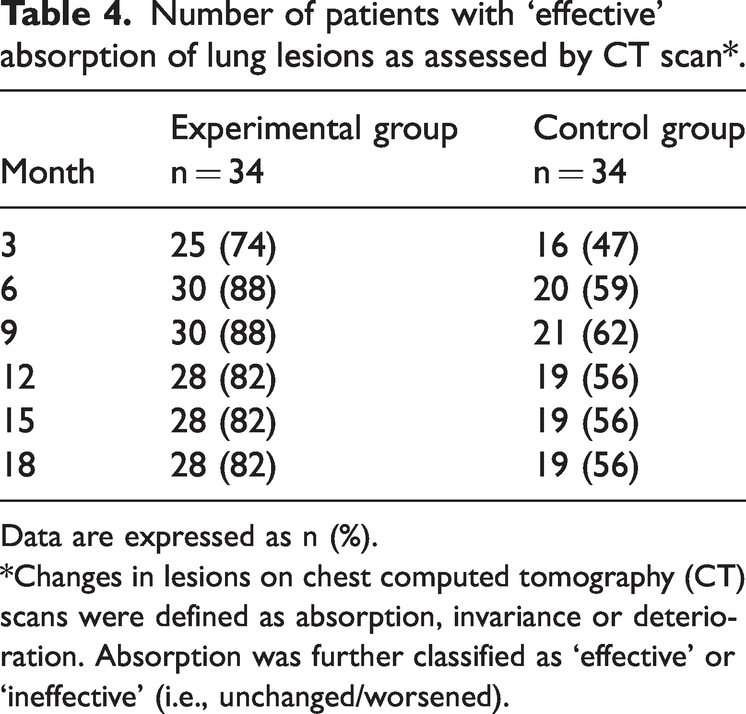
Data are expressed as n (%).
*Changes in lesions on chest computed tomography (CT) scans were defined as absorption, invariance or deterioration. Absorption was further classified as ‘effective’ or ‘ineffective’ (i.e., unchanged/worsened).
At the end of treatment, cure rates (i.e., favourable rates) were 82% (28/34) and 56% (19/34) in the experimental and control groups, respectively (Table 5). The difference between groups was statistically significant (χ2 = 5.58, P < 0.025). In addition, there were statistically significantly less adverse outcomes in the experimental group compared with the control group (6 [18%] vs 15 [44%]; χ2 = 5.58, P < 0.025). There was no difference between groups in the number of severe adverse events in both groups (i.e., 3 [9%]; Table 6).
Treatment outcomes. 22

Data are expressed as n (%). −, not assessed.
*Outcomes were classified as either favourable (i.e., cure) or adverse (i.e., treatment failure, death, and lost to follow-up).
α3, severe adverse events; 2, continuous positive sputum samples.
β3 severe adverse events; 10, continuous positive sputum samples.
Severe Adverse Events.

Data are expressed as n (%).
ECG, electrocardiogram.
Discussion
China is a global ‘hotspot’ for MDR-TB, with an estimated incidence of 110,000 cases per annum. 23 Moreover, as shown in this study, a significant proportion of patients have no history of TB treatment. This suggests that many patients acquire MDR-TB infection through human-to-human transmission. 24 This discovery emphasizes the importance of rapid diagnosis, effective treatment and source control of MDR-TB.
Compared with sensitive TB, MDR-TB is associated with a high treatment cost and poor treatment effect, which has had a serious negative impact on families and society. 25 In this study, we found that the cure rate of patients with MDR-TB was significantly improved by the addition of bedaquiline and clofazimine to their treatment regimen (i.e., 82% vs. 56%). In addition, we found a significant difference between groups in ‘effective’ absorption of pulmonary lesions (i.e., 82% vs. 56%). Interestingly, our observed cure rate for the experimental group (i.e., 82%) was significantly higher than that reported by the WHO in its 2022 Global TB Report (i.e., 60%). 2 However, it was lower than the 90% cure rate found by investigators using a combination of bedaquiline, pretomanid, and linezolid in patients with XDR-TB. 26
While the precise mechanism of action of clofazimine in the treatment of MDR-TB is unclear, the drug is known to activate the activity of phospholipase A2 and cause accumulation of lysophosphatidylcholine, and so inhibit the growth of M. tuberculosis.27,28 In addition, clofazimine accelerates the production of hydrogen peroxide and enhances the bactericidal effect of macrophages.29,30 Clofazimine also inhibits bacterial replication and transcription by binding to the guanine base of M. tuberculosis DNA. Inhibition of DNA-dependent RNA polymerase, inhibits RNA synthesis which in turn inhibits protein synthesis. 31 Importantly, clofazimine is a moderate to potent cytochrome P450 isoenzyme 3A4 (CYP3A4) inhibitor.32–34 Furthermore, clofazimine has been shown to have good antibacterial activity against slow-multiplying M. tuberculosis.35,36 These slow-multiplying bacteria are responsible for biofilm and granuloma formation; therefore, the inhibitory effect of clofazimine may facilitate the contact between bedaquiline and M. tuberculosis and so enhance the anti-tuberculosis activity of bedaquiline. This hypothesis may explain the results from this present study that showed superior efficacy of the experimental group compared with the control group. Nevertheless, clofazimine has been reported to cause skin colour and skin ichthyosis changes which may influence compliance with treatment. Although, these changes have been found to decrease following drug withdrawal they may take 1–2 years to clear up completely. 37
In this present study, there was no difference between groups in the number of serious adverse events and none were skin-related. Furthermore, we found that 82% patients in the experimental group had negative TB culture after six months and this rate remained constant for the next 12 months. This finding suggests that a short course of this new treatment regimen may be effective which will undoubtedly reduce exposure and the possibility of potential side effects.
The study had several limitations. For example, the number of patients was relatively small, which may have led to sample bias. In addition, patients with HIV/AIDS were not included in this study which may have limited the applicability of our results. Prospective, randomized, controlled studies with larger sample sizes and the inclusion of different patient groups are required to confirm our conclusions.
In summary, we have shown that the addition of bedaquiline and clofazimine to a MDR-TB drug regimen improves patient outcomes. Clinicians should be aware of the clinical benefits of adding bedaquiline and clofazimine to treatment regimens for patients with MDR-TB but be mindful of contraindications and carefully monitor patients for signs of adverse effects.
Footnotes
Acknowledgements
We wish to thank the patients and their families. In addition, we thank staff and the management team at Wuhan Jinyintan Hospital.
Declaration of conflicting interests
The authors declare that there are no conflicts of interest.
Funding
The authors disclose receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by Medical Research Projects of the Health and Family Planning Commission of Wuhan Municipality (Project number: WZ17B09).
