Abstract
Chronic pulmonary aspergillosis (CPA) often arises in the setting of structural lung damage and is associated with chronic inflammation, which increases the risk of venous thromboembolic disease (VTE), including pulmonary embolism (PE). However, the clinical context and pathophysiological mechanisms underlying CPA/VTE remain poorly defined. This study aims to highlight the clinical challenges through a case series and systematic review of existing literature regarding this rare co-occurrence. We present three complex cases from Kiruddu National Referral Hospital in Uganda: two males, one female, aged 44–80 years, with post-tuberculosis lung disease. All three were diagnosed with Aspergillus IgG-positive CPA and confirmed PE (two acute, one with chronic thromboembolic pulmonary hypertension (CTEPH)). One presented with concurrent active tuberculosis (TB) relapse and severe immunosuppression (CD4 T-cells < 200 cells/mm3), and two had HIV. All patients received itraconazole and anticoagulation with rivaroxaban. Two patients survived and are stable on therapy, while one patient died due to septic shock. A systematic search of PubMed, Embase, Scopus, and Web of Science yielded seven studies reporting nine patients with coexisting CPA and VTE/PE. Underlying conditions most frequently included prior TB (n = 3) and chronic obstructive pulmonary disease (n = 3). PE was chronic in three patients (including one with CTEPH), with CPA subtypes ranging from simple aspergilloma to subacute invasive aspergillosis and angioinvasive aspergillosis. Proposed mechanisms included endothelial damage due to Aspergillus angioinvasion and creation of post-PE lung cavities/infarcts suitable for Aspergillus colonization. Anticoagulation was used in six cases, but was complicated by hemoptysis in three. Two-thirds of patients improved with antifungal therapy, while three deaths were reported. CPA may coexist with PE/DVT due to fungal angioinvasion, chronic inflammation, or immobilization from advanced lung disease. Management requires individualized balancing of antifungal and anticoagulation therapy, with heightened vigilance for bleeding.
Plain language summary
Chronic pulmonary aspergillosis (CPA) is a long-term lung infection caused by Aspergillus fungus, usually affecting people whose lungs have been damaged by conditions such as tuberculosis (TB). Because CPA causes ongoing inflammation, it may increase the risk of blood clots in the lungs and legs—known as venous thromboembolism (VTE), including pulmonary embolism (PE). However, this combination is uncommon and not well understood. In this study, we describe three patients from Kiruddu National Referral Hospital in Uganda who had both CPA and PE. The patients were between 44 and 80 years old, all with previous TB-related lung damage. All tested positive for Aspergillus IgG, confirming CPA, and also had PE—two acute cases and one long-standing case that had progressed to chronic thromboembolic pulmonary hypertension (CTEPH). Two of the three patients were living with HIV, and one had a TB relapse with severe immune suppression. All three received antifungal treatment (itraconazole) and blood-thinning medication (rivaroxaban). Two patients recovered well, while one died from septic shock. We also reviewed published studies on this rare combination. Across seven studies, only nine similar cases were found worldwide. The most common underlying lung problems were previous TB and chronic obstructive pulmonary disease (COPD). Some patients had chronic PE, and others had different forms of CPA—from simple cavities to more invasive fungal disease. Researchers suggested two possible explanations: 1. Aspergillus may directly damage blood vessels, increasing clot risk. 2. Lung cavities or infarcts caused by PE may create environments where Aspergillus can grow. Treating both conditions together is challenging. Blood thinners can cause bleeding, especially coughing up blood, which occurred in several cases. Still, most patients improved with antifungal treatment. Overall, CPA and PE can occur together and require careful, individualized management
Background
Chronic pulmonary aspergillosis (CPA), a spectrum of chronic lung infections caused primarily by the colonization of Aspergillus species, typically arises in individuals with preexisting structural lung damage such as post-tuberculosis lung disease (PTBLD), chronic obstructive pulmonary disease (COPD), and bronchiectasis.1,2 It is characterized by a progressive and often debilitating course involving chronic cough, hemoptysis, progressive respiratory decline, and death.3,4
Pulmonary embolism (PE) and other forms of venous thromboembolism (VTE) remain a significant cause of morbidity and mortality in hospitalized and chronically ill patients. 5 PE is a thrombotic disorder governed by Virchow’s triad—endothelial injury, hypercoagulability, and circulatory stasis. 6 Chronic inflammatory and infectious states, such as active tuberculosis (TB) and deep-seated fungal infections, are well-established risk factors for hypercoagulability, inducing a pro-thrombotic state through cytokine release and direct endothelial damage.7–10
Given that CPA frequently occurs in patients with preexisting conditions such as PTBLD, COPD, and bronchiectasis—all of which are established risk factors for PE11–13—a theoretical mechanism links CPA directly to venous VTE, particularly PE. However, clinical reports detailing the concurrent diagnosis, management, and proposed pathophysiology of CPA and PE/VTE remain scarce, often limited to single case reports.14,15 The inherent complexity of managing these dual conditions—specifically balancing the need for therapeutic anticoagulation against the high risk of catastrophic hemoptysis from CPA lesions—presents a significant clinical challenge. 16
We aim to bridge this knowledge gap by presenting a unique case series of three patients from a high-TB burdened setting who presented with CPA and concurrent PE (Table 1), and by systematically synthesizing the available literature on this rare co-occurrence. We seek to characterize the clinical features, comorbidities, and outcomes of this co-occurrence, providing vital insights for improved patient management.
Summary of cases with chronic pulmonary aspergillosis and PE/VTE at Kiruddu National Referral Hospital.
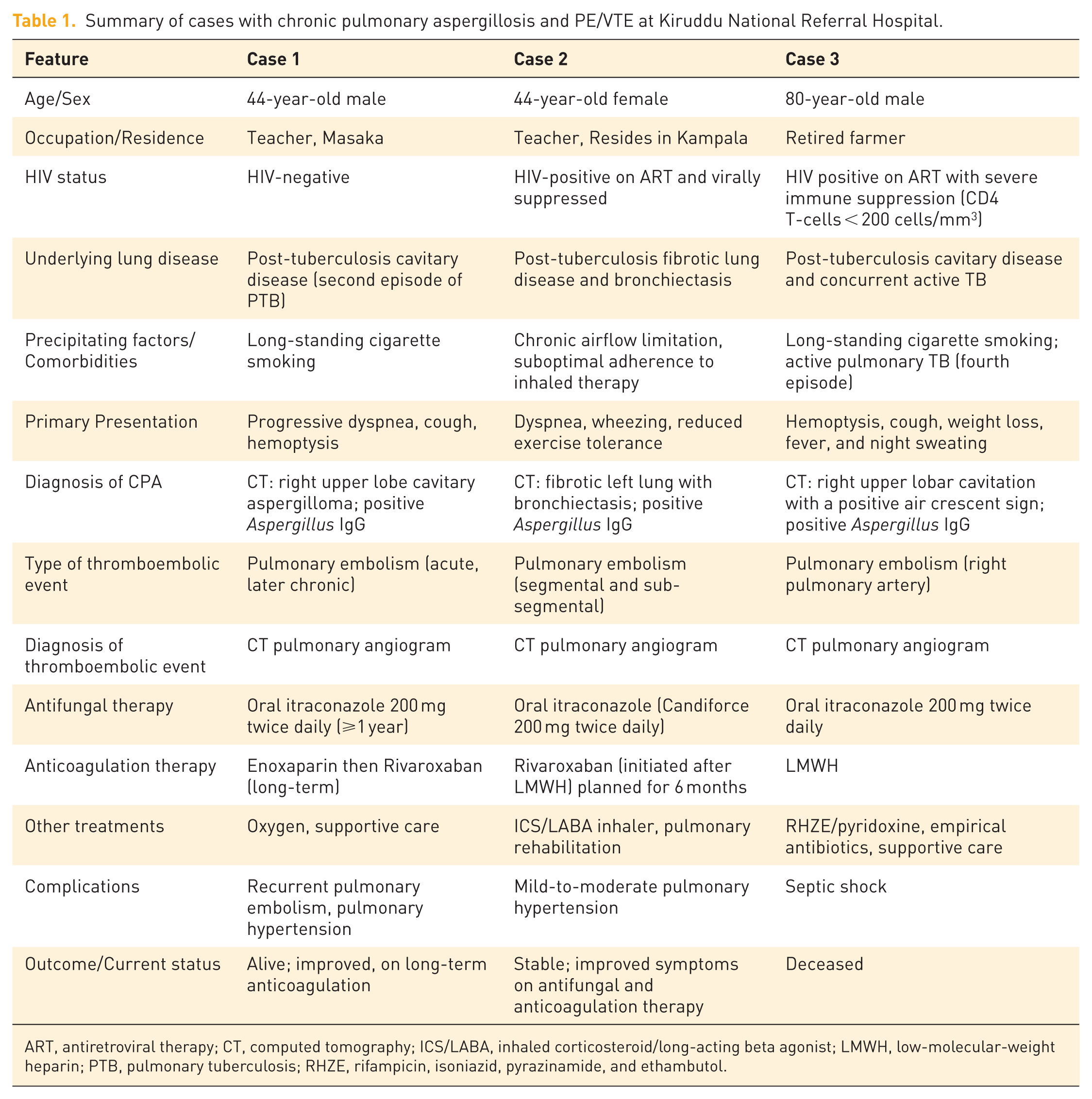
ART, antiretroviral therapy; CT, computed tomography; ICS/LABA, inhaled corticosteroid/long-acting beta agonist; LMWH, low-molecular-weight heparin; PTB, pulmonary tuberculosis; RHZE, rifampicin, isoniazid, pyrazinamide, and ethambutol.
Case presentations
The study was conducted and reported in accordance with the CARE statement (Supplemental Appendix 1). 17
Case 1: CPA complicated by PE in a school teacher
A 44-year-old male, a teacher from Masaka in rural Uganda, who was being treated for a second episode of pulmonary TB diagnosed by chest radiograph, was referred to Kiruddu National Referral Hospital in Kampala with progressive difficulty in breathing, cough, and hemoptysis. He reported a long-standing history of cigarette smoking and had previously been diagnosed with pulmonary TB 4 years earlier by GeneXpert, for which he completed treatment with documented microbiological clearance.
On evaluation, he was dyspneic and mildly hypoxic (SPO2—91% on room air), with reduced breath sounds over the right hemithorax. Chest computed tomography (CT) demonstrated thick-walled cavities with internal soft-tissue densities and pericavitary inflammation, consistent with aspergillosis. A working diagnosis of PTBLD with cavitating aspergillosis was made, and the patient was commenced on oral itraconazole 200 mg twice daily, planned for 1 year.
After 11 months of antifungal therapy, he was readmitted with worsening dyspnea, cough, and chest pain, although hemoptysis had resolved.
On examination, he was dyspneic, had grade IV digital clubbing, blood pressure 141/98 mmHg, pulse 98 bpm, and oxygen saturation 94% on 5 L/min via nasal prongs. A pulmonary CT angiogram (Figure 1) showed features of PE involving the right pulmonary artery. A diagnosis of PE complicating CPA was made, and he was initiated on subcutaneous enoxaparin, later transitioned to oral rivaroxaban. He improved clinically and was discharged after 2 weeks.

Computed tomography images showing thick-walled cavities with an internal soft-tissue density in the right apical segment, as shown in Panel a and b; axial and coronal planes of the mediastinal window. Panel c and d are axial contrasted CT images showing a filling defect in the right pulmonary artery; pulmonary embolism.
Approximately 8 months later, the patient was readmitted with acute shortness of breath and right-sided chest pain. He was alert but tachypneic (respiratory rate 45 breaths/min) with oxygen saturation 100% on 15 L/min of supplemental oxygen, pulse 99 bpm, and blood pressure 117/91 mmHg. Physical examination revealed inward movement of the lower chest wall during inspiration and persistent finger clubbing. A new pulmonary CT angiogram showed no residual cavitary lesions but revealed extensive right pulmonary artery emboli. A diagnosis of chronic thromboembolism with pulmonary hypertension (CTEPH) (following a transthoracic cardiac echocardiogram) was made, and long-term anticoagulation was continued. At the time of this report, he remains an outpatient at Kiruddu National Referral Hospital.
Case 2: CPA and PE in an HIV-positive woman
A 44-year-old woman, on antiretroviral therapy (ART) for 19 years and virally suppressed (current regimen was Tenofovir/Lamivudine/Dolutegravir), presented to Kiruddu National Referral Hospital with progressive difficulty in breathing and wheezing that worsened on exposure to dust, cold air, smoke, or perfumes. She had a history of three prior episodes of pulmonary tuberculosis (in 2002, 2003, and 2004) that had been reported as cured.
A CT scan performed in 2021 had shown loss of left lung volume with fibrosis, lobar atelectasis, and cystic bronchiectasis (images not available). Since then, she had been managed for chronic airflow limitation (diagnosed clinically from a peripheral facility), intermittently using long-acting muscarinic antagonists (LAMA) and short-acting β-agonists (SABA) but with sub-optimal adherence.
She re-presented in June 2025 with worsening dyspnea and reduced exercise tolerance. On examination, she was a well-nourished female, with blood pressure 102/62 mmHg, pulse 88 bpm, weight 59 kg, and oxygen saturation was 92% at room air. Respiratory examination revealed reduced breath sounds on the left and a loud P2 on cardiac auscultation. An echocardiogram done 2 days prior showed features of mild-to-moderate pulmonary hypertension.
A CT pulmonary angiogram performed on this visit (Figure 2) demonstrated multiple filling defects in the right distal lobar, segmental, and sub-segmental pulmonary arteries—consistent with pulmonary embolism. In addition, there were pleural calcific granulomas, pleural thickening, cystic bronchiectasis, and volume loss with fibrotic changes in the left hemithorax, in keeping with post-tuberculous fibrotic disease.

Computed tomography of the chest and pulmonary angiogram showing cystic fibrosis, volume loss, and fibrotic changes in the left lower lobe with bilateral multifocal ground glass opacification in a “Mosaic pattern (panel a and b). Multiple filling defects in the right distal lobar, segmental, and sub-segmental pulmonary arteries—consistent with pulmonary embolism (Panel c and d–red arrow).
She also had a positive Aspergillus IgG serology, confirming CPA in a background of post-TB lung damage and HIV infection. The patient was started on oral itraconazole (Candiforce 200 mg twice daily) for CPA and therapeutic anticoagulation with rivaroxaban. She also received an inhaled corticosteroid/long-acting β-agonist (ICS/LABA) combination for airway obstruction. However, this was not confirmed due to the relative contraindication to spirometry posed by PE. The choice to use rivaroxaban despite significant interaction with itraconazole was because the patient could not afford INR monitoring if warfarin were to be used, and the long distance they needed to travel to the hospital precluded the use of low-molecular-weight heparin. We monitored her telephonically for any bleeding events.
At review in July 2025, she reported improved breathing and exercise tolerance, with only occasional nocturnal wheeze. Her vital signs were stable (BP 118/70 mmHg, pulse 74 bpm, weight 57 kg). Rivaroxaban was continued at 20 mg once daily, alongside ICS/LABA twice daily and itraconazole.
By October 2025, she again reported symptomatic improvement. Examination remained unremarkable, and she was encouraged to continue medication and begin pulmonary rehabilitation exercises. At this point, she is clinically stable on rivaroxaban, itraconazole, and ICS/LABA therapy.
By January 2026, she had completed the rivaroxaban dose and had not reported any major bleeding event.
Case 3: Chronic pulmonary aspergillosis in a patient with tuberculosis, post-tuberculosis lung disease complicated with pulmonary embolism
An 80-year-old male with HIV on ART (Abacavir/ lamivudine and Dolutegravir) for the past 8 years was admitted for a 2-month history of cough with a past history of hemoptysis but with current general body weakness, reduced appetite, weight loss, vomiting, fever, and night sweats. He had a long-standing history of cigarette smoking and had been treated for pulmonary TB thrice, with the latest episode 2 years prior to admission, for which he reported a cure.
On evaluation, he was wasted, afebrile, alert with blood pressure of 113/56 mmHg and pulse rate of 120 beats per minute. He was tachypneic on respiratory examination with a respiratory rate of 26 breaths per minute and had crepitations in the right supramammary and mammary zones. Initial investigations showed a CD4+ T-cell count of <200 cells/µL, a positive TB urine lipoarabinomannan, numerous pus cells with bacteruria on urine dipstick, and a round opacity in the right upper lung zone on chest radiograph. He was started on broad antimicrobial empirical therapy and anti-TB therapy (oral rifampicin, isoniazid, ethambutol, and pyrazinamide).
Abdominopelvic ultrasound revealed prostate enlargement with bladder wall thickening and echoes in urine. A chest CT was done (Figure 3), which showed fibro-cavitating lung disease characterized by right upper lobar cavities, with the largest containing an intra-cavitary mass with surrounding cystic bronchiectasis and fibrotic changes consistent with an aspergilloma in the background of post-TB lung disease. There were also filling defects in the right pulmonary artery, consistent with right pulmonary artery embolism. Further, bilateral apical lung reticulo-nodular opacities and bilateral mild pleural effusion were observed, which were consistent with superimposed active pulmonary TB.

Panel a and b are axial CT images, mediastinal window showing a filling defect in the right main pulmonary artery, shown by the red arrows. Panel c is an axial CT image mediastinal window showing a soft-tissue density within a cavity in the right apical segment. Panel c is an axial CT scan image, lung window demonstrating the “air crescent sign” in Panel d; the aspergilloma is shown by the green arrows.
He also tested positive for Aspergillus IgG, further supporting the diagnosis of CPA. He was then started on low-molecular-weight heparin, oral itraconazole 200 mg twice daily planned for a year, and tamsulosin/dutasteride combination.
By day five of admission, he developed septic shock, all resuscitation efforts were futile, and the patient died.
Materials and methods for systematic review
Study design
This was a systematic review following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA 2020) guidelines. 18 The study protocol was developed in advance according to a structured Population, Intervention, Comparator, and Outcomes (PICO) framework. The review sought to synthesize all published evidence describing the coexistence or association between CPA and PE or other forms of VTE. A narrative synthesis approach was employed, given the rarity of the condition and the expected heterogeneity of the available literature.
Information sources
A comprehensive and systematic literature search was conducted on September 6, 2025, using four major electronic databases: PubMed/MEDLINE, Embase, Scopus, and the Web of Science Core Collection. These databases were selected for their extensive biomedical coverage and minimal overlap. In addition to the primary database searches, Google Scholar (first 200 results) and OpenGrey were used to identify potentially relevant gray literature, including conference proceedings, theses, and institutional reports. The reference lists of all included studies and relevant reviews were manually screened to capture additional articles missed in the electronic searches. No date restrictions were applied, but only studies published in English were considered eligible.
Search strategy
The search strategy was developed in consultation with an experienced medical librarian and adapted for each database’s indexing system. Controlled vocabulary (Medical Subject Headings [MeSH] for PubMed and Emtree terms for Embase) was combined with free-text keywords to maximize sensitivity. Each search string combined terms describing chronic pulmonary aspergillosis and pulmonary embolism or venous thromboembolism. A representative PubMed strategy was as follows: (“chronic pulmonary aspergillosis”[MeSH Terms] OR “aspergilloma”[All Fields] OR “chronic cavitary pulmonary aspergillosis”[All Fields] OR “Aspergillus”[MeSH Terms] AND (“lung”[All Fields] OR “pulmonary”[All Fields])) AND (“pulmonary embolism”[MeSH Terms] OR “venous thromboembolism”[All Fields] OR “deep vein thrombosis”[All Fields] OR “pulmonary artery thrombosis”[All Fields]).
Search strings were adapted for each database syntax, and complete queries are available in the supplementary material (relitsearch.zip). The search results were exported to EndNote X9 for de-duplication and subsequently imported into Covidence systematic review software for title/abstract screening, full-text assessment, and data management. Additional manual searches of the reference lists of the selected studies were performed to identify additional potential studies.
Eligibility criteria
Studies were considered eligible if they met the following criteria:
Reported human cases of CPA (including simple aspergilloma, chronic cavitary pulmonary aspergillosis, or subacute invasive aspergillosis) coexisting with pulmonary embolism or other venous thromboembolic events;
Included sufficient diagnostic, clinical, and management information to confirm both CPA and PE/VTE;
Were published as full-text articles in peer-reviewed journals.
Eligible study designs encompassed case reports, case series, observational studies, and clinical trials. Exclusion criteria included studies focusing solely on acute invasive aspergillosis, animal or experimental models, abstract-only publications lacking extractable data, and non-English reports.
Study selection
Two reviewers (IP and JMK) independently screened all titles and abstracts using Covidence software. Any study deemed potentially relevant by either reviewer proceeded to full-text review. Full-text articles were independently assessed for eligibility against predefined inclusion and exclusion criteria. Discrepancies were resolved through discussion. The selection process was documented using a PRISMA 2020 flow diagram (Figure 4), summarizing the number of records identified, screened, excluded, and included at each stage.

PRISMA flow diagram.
Data extraction and management
A structured data extraction form was designed and piloted prior to data collection. For each included study, the following information was extracted: Study details (author, year, country, design, sample size); patient demographics and comorbidities ( tuberculosis, COPD, diabetes, malignancy, HIV); CPA subtype and diagnostic criteria (imaging, microbiological culture, serology, histopathology); diagnostic modality for thromboembolism (CT pulmonary angiography, Doppler ultrasound, echocardiography, or autopsy); proposed pathophysiological mechanisms linking CPA and PE/VTE; therapeutic interventions (antifungal therapy, anticoagulation, surgical procedures); and clinical outcomes (recovery, recurrence, complications, or death).
Two reviewers independently (IP and JKM) extracted data, and inconsistencies were resolved by consensus. When clarification was needed, original articles were re-examined to ensure accuracy and completeness.
Quality assessment
The methodological quality of each included study was independently assessed by two reviewers using the Joanna Briggs Institute (JBI) Critical Appraisal Checklists. Case reports were evaluated using the 8-item checklist assessing clarity of patient presentation, diagnostic methods, intervention description, and outcomes. Case series were assessed with the 10-item checklist, which additionally considers participant inclusion, site reporting, and statistical analysis. Scores were summarized descriptively, and studies were categorized as high, moderate, or low quality depending on the proportion of affirmative responses. All included reports achieved high-quality ratings, ensuring reliability in the synthesized conclusions.
Data synthesis and analysis
Given the rarity and heterogeneity of CPA–PE co-occurrence, meta-analysis was not feasible. Instead, a narrative synthesis was employed. Findings were organized thematically across domains of epidemiology, comorbidities, diagnostic features, pathophysiological mechanisms, management approaches, and outcomes.
When available, the relationship between CPA subtype, anticoagulation use, and mortality was explored qualitatively to highlight patterns relevant to clinical management.
Reporting and transparency
All stages of the review—from database searching to data synthesis—were conducted according to the PRISMA 2020 framework. A detailed PRISMA flow diagram illustrates the screening and inclusion process, and all search strings, extracted data tables, and quality assessments are provided for transparency and reproducibility.
Results
Study selection
The initial database search yielded 1216 records: Web of Science (n = 495), Scopus (n = 353), Embase (n = 201), and PubMed (n = 167). After removal of 281 duplicates (275 through Covidence and six manually), 935 unique articles remained for screening.
Following title and abstract review, 918 were excluded for irrelevance to CPA or thromboembolic disease. Seventeen full-text articles were assessed for eligibility; 10 were excluded due to wrong outcomes (n = 3), unrelated populations (n = 1), air embolism (n = 1), lack of full text (n = 2), or being conference abstracts (n = 3). Ultimately, seven studies (five case reports and two case series) involving nine patients met the inclusion criteria and were analyzed (Figure 4).
Characteristics of included studies
The seven included studies were published between 1985 and 2024 and originated from Asia, Europe, and Africa. Five were single-patient case reports, and two were case series, together describing nine individuals with concurrent or sequential CPA and PE or VTE. Table 2 shows the studies included.
Summary of studies included in the systematic review.

BAE, bronchial artery embolization; CCPA, chronic cavitary pulmonary aspergillosis; CML, chronic myeloid leukemia; COP, cryptogenic organizing pneumonia; COPD, chronic obstructive pulmonary disease; CPA, chronic pulmonary aspergillosis; CT/CTPA, computed tomography/CT pulmonary angiography; CTEPH, chronic thromboembolic pulmonary hypertension; DKA, diabetic ketoacidosis; DVT, deep vein thrombosis; GM test, galactomannan antigen test; IPA, invasive pulmonary aspergillosis; IVS, interventricular septum; LCB, lactophenol cotton blue; LLL, left lower lobe; MPA, main pulmonary artery; PE, pulmonary embolism; SAIA, subacute invasive aspergillosis; TB, tuberculosis; VTE, venous thromboembolism; yo, year-old.
Demographics and comorbidities
Patients ranged in age from 33 to 79 years; seven were male. Common comorbidities included COPD (n = 3), previous TB (n = 3), diabetes mellitus or diabetic ketoacidosis (n = 1), and hematologic malignancy under chemotherapy (n = 1). One patient had cryptogenic organizing pneumonia, and another had no reported comorbidities.
CPA presentation and diagnosis
The spectrum of CPA subtypes comprised simple aspergilloma (n = 3), chronic cavitary pulmonary aspergillosis (CCPA) (n = 3), subacute invasive aspergillosis (SAIA) (n = 1), and angioinvasive aspergillosis (n = 1).
Diagnosis was established using combinations of: radiology (cavitating or fibrotic lesions on CT or radiograph); Microbiology or culture for Aspergillus spp.; Serologic testing for Aspergillus IgG or galactomannan, and/or Histopathology or surgical specimens in resected cases.
Radiologic features most frequently described were thick-walled cavities with soft-tissue densities, the air crescent sign, or mycetoma-like lesions.
PE/VTE diagnosis
All cases confirmed thromboembolic events through CT pulmonary angiography (CTPA), Doppler ultrasonography, echocardiography, or autopsy.
PE was acute in most patients (n = 5), chronic in two, and combined with chronic thromboembolic pulmonary hypertension (CTEPH) in one.
Management approaches
All patients received antifungal therapy, most commonly itraconazole or voriconazole, with amphotericin B or posaconazole used in selected refractory cases. Anticoagulation was administered in six patients, employing warfarin, rivaroxaban, heparin, or low-molecular-weight heparin (LMWH). However, treatment was frequently complicated by massive hemoptysis or drug–drug interactions between azoles and anticoagulants. In the report by Liu et al., 16 two patients discontinued warfarin due to hemoptysis, while in Tunney et al., 21 bronchial artery embolization was required for bleeding control. Two cases (including Kirshenbaum et al. 15 ) withheld anticoagulation entirely because of high hemorrhagic risk or thrombocytopenia. Surgical intervention, primarily lobectomy or resection, was reported in older series such as Regnard et al., 19 while more recent cases favored medical management. Duration of antifungal therapy ranged from 4 months to 1 year, depending on disease severity and tolerance.
Clinical outcomes
Of the nine patients, six (67%) achieved clinical or radiologic improvement, while three deaths (33%) were reported—two from massive hemoptysis and one from respiratory failure secondary to angioinvasive disease. No recurrence of thromboembolic events was described after appropriate antifungal and anticoagulation therapy. Overall, outcomes were favorable when both CPA and thromboembolic complications were recognized early and comanaged.
Quality assessment
All included studies scored highly on the JBI Critical Appraisal Checklists (Tables 3 and 4). Each of the five case reports fulfilled all eight quality criteria (8/8), demonstrating clear timelines, diagnostic accuracy, treatment details, and take-home messages. The two case series achieved 8/10 and 9/10. Overall, the methodological quality of available evidence was rated as high, though limited by small numbers and heterogeneity in diagnostic and therapeutic approaches.
Quality assessment for case reports included in the review.
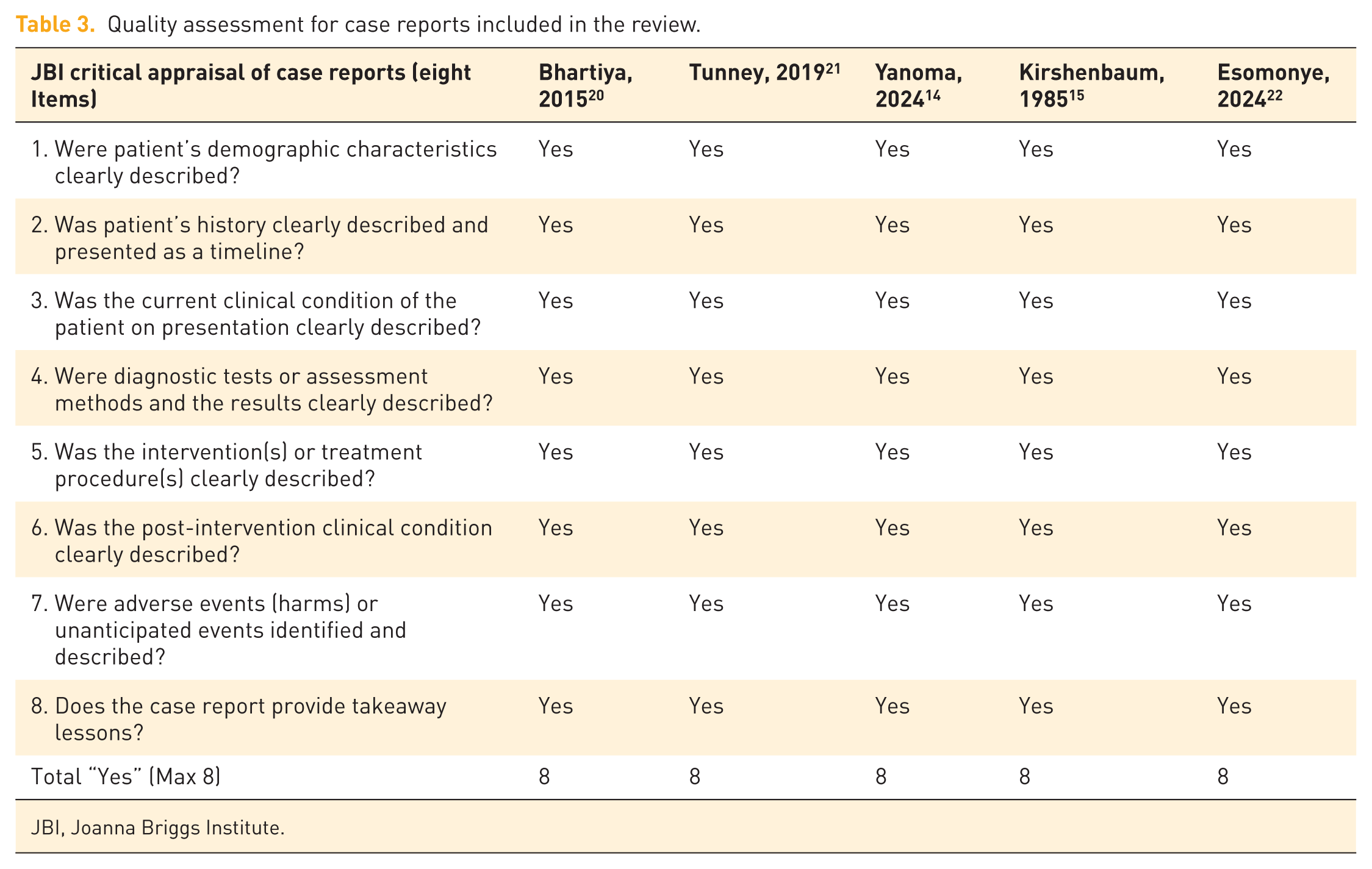
JBI, Joanna Briggs Institute.
Quality assessment for case series included in the review.

JBI, Joanna Briggs Institute.
Discussion
This study represents, to our knowledge, the first combined report describing cases of CPA complicated by PE/VTE, including cases from Africa. Across three patients managed at Kiruddu National Referral Hospital, CPA developed in the setting of PTBLD. All three patients subsequently developed thromboembolic disease confirmed by imaging. The systematic review identified only nine published cases of patients globally with coexistent CPA and PE/VTE, underscoring the rarity of this presentation. Nevertheless, the convergence of findings between our case series and prior literature highlights common risk profiles—middle-aged male adults with underlying cavitary or fibrotic lung disease, chronic hypoxia, and inflammation. Most cases were male because the underlying lung diseases (COPD and TB) are commoner in males.
The co-occurrence of CPA and VTE, particularly PE, represents a severe and challenging clinical conundrum. The synthesis of our findings with the seven prior published studies strongly suggests that this is not a coincidental finding but rather a distinct, clinically relevant association driven by specific pathophysiological mechanisms. The systematic review highlighted three proposed principal mechanisms linking CPA to PE/VTE, which were directly observable in our case presentations. First, CPA, as a chronic fungal infection, drives sustained systemic inflammation that induces endothelial damage and activates the coagulation cascade. 23 The underlying conditions in our cases (PTBLD and HIV) are themselves prothrombotic, creating a cumulative risk that predisposes patients to VTE. 24 Second, the most definitive link, exemplified in severe cases in the literature, 15 involves Aspegillus hyphae directly invading the pulmonary vasculature, which, even in the less aggressive but prolonged nature of chronic cavitary CPA or SAIA, can induce sufficient local endothelial damage to trigger in-situ thrombosis.25,26 Direct pulmonary vascular invasion of fungi presenting as pulmonary embolism has been observed in mucormycosis as well. 27 The structural destruction of lung tissue creates an abnormal hemodynamic and vascular environment conducive to thrombus formation. Finally, the systematic review explicitly confirmed cases where PE/pulmonary infarction preceded the CPA diagnosis. The resulting necrotic lung tissue creates a microenvironment ideal for colonization by inhaled Aspergillus conidia, a sequence that suggests a feedback loop of vascular damage and fungal proliferation.
The consistent finding of severe PHT in two of our cases—one presenting as CTEPH-like disease—underscores the severe prognostic implications of this co-occurrence. This necessitates a clear diagnostic imperative: PE/VTE should be actively excluded in CPA patients presenting with worsening dyspnea, new-onset chest pain, or rapid progression of PHT, even if the primary lung pathology is dominant. Obviously, the concurrent lung pathology and PE make it difficult to differentiate PHT secondary to parenchymal disease (Group 3) from PHT secondary to chronic thromboembolism (Group 4). 28 Workup with echocardiogram and CTPA is essential for diagnosis and risk stratification, particularly in settings like ours where right heart catheterization is not easily accessible. Unfortunately, VTE screening scores and biomarkers such as the Well’s score and D-dimers have not been validated in CPA and show poor diagnostic accuracy in people with established chronic lung diseases or CTEPH.29,30 This calls for specific VTE screening scores and biomarkers specific to CPA to increase the pre-test probability of CTPA.
Diagnosing CPA/PE co-occurrence is challenging because symptoms overlap—dyspnea, pleuritic pain, and cough are nonspecific. Radiological findings may also be confounded, as pulmonary infarcts and fungal cavities can mimic each other.31–33
High-resolution CT, pulmonary CTA, and serological testing for Aspergillus IgG are therefore essential for accurate differentiation. In resource-limited settings such as Uganda, access to CT pulmonary angiography and fungal serology remains limited, which may lead to underdiagnosis.
The intersection of CPA and PE also creates a critical anticoagulation dilemma. CPA is a major risk factor for massive, life-threatening hemoptysis, and anticoagulation dramatically increases this hemorrhagic risk.34,35 Our review noted three instances of anticoagulation cessation or complication due to bleeding. This dilemma necessitates highly individualized patient risk stratification and aggressive comanagement, potentially involving therapeutic drug monitoring for antifungals (which interact with many anticoagulants) and considering local interventions like bronchial artery embolization if hemoptysis risk is deemed too high. 36 Clinical prediction scores to assess the risk of bleeding in people with CPA and PE/VTE on anticoagulation are also needed.
The final implication of our study is related to the impact of underlying disease: while the predominance of PTBLD in all our cases and the literature (n = 3) reflects TB’s role as a major cause of structural lung damage globally, the inclusion of COPD, bronchiectasia, and cryptogenic organizing pneumonia from the systematic review confirms that any cause of chronic, severe structural lung disease predisposes the patient to this catastrophic triple pathology. Future studies should evaluate coagulation abnormalities and inflammatory biomarkers in CPA populations to clarify the mechanistic link and identify subgroups at risk for thromboembolism.
The rarity of CPA–PE coexistence limits generalizability. The case series, though detailed, involved only three patients from a single center. While this systematic review provides a valuable synthesis, it is limited by the rarity of the reported co-occurrence. The vast majority of the synthesized evidence consists of case reports/series (n = 7), which naturally precludes quantitative meta-analysis and introduces potential publication bias toward unusual or successful treatment outcomes. Future prospective cohort studies are needed to determine the true incidence and prevalence of PE/VTE in large CPA cohorts. Another potential limitation is that we included only English publications. This could have limited the scope of the studies.
Conclusion
CPA may coexist with PE or DVT due to fungal angioinvasion, chronic inflammation, or immobilization from advanced lung disease. Management requires individualized balancing of antifungal and anticoagulation therapy, with heightened vigilance for bleeding. Clinicians managing CPA patients should maintain a high index of suspicion for PE/VTE, particularly in those with worsening dyspnea and hemoptysis.
Supplemental Material
sj-docx-1-tai-10.1177_20499361261431900 – Supplemental material for Chronic pulmonary aspergillosis and pulmonary embolism/venous thromboembolism: a case series and systematic review of the literature
Supplemental material, sj-docx-1-tai-10.1177_20499361261431900 for Chronic pulmonary aspergillosis and pulmonary embolism/venous thromboembolism: a case series and systematic review of the literature by Joseph Baruch Baluku, Peter Isagara, Ivaan Pitua, Jeremiah Mutinye Kwesiga, Judith Mutesi, Joanitah Nalunjogi, Ahmed Ddungu, Ronald Olum and Felix Bongomin in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
