Abstract
Background:
Human cystic echinococcosis frequently involves the lungs, and colonization of a residual cavity with Aspergillus spores can lead to chronic pulmonary aspergillosis (CPA).
Objectives:
We pooled and characterized cases of CPA co-existing with pulmonary cystic echinococcosis (PCE) through a systematic review of globally published data.
Design:
Systematic review.
Data sources and methods:
Primary literature was identified through searches of various electronic databases, including CINAHL, Ovid MEDLINE, MEDLINE (PubMed), EMBASE, Google Scholar, the Cochrane Database of Systematic Reviews, and African Journal Online. The search encompassed articles from inception to February 2024, using medical subject heading search terms “chronic pulmonary aspergillosis” OR “Aspergillus” OR “aspergilloma” OR “aspergillosis” AND “hydatid disease” OR “hydatidosis” OR “echinococcosis” OR “Echinococcus.” Two reviewers independently assessed titles, abstracts, and full texts for eligibility using the Rayyan web-based software. The eligible studies comprised original observational research, single case reports, and case series that reported on CPA in patients with PCE based on established criteria (microbiological and/or radiological diagnosis of PCE) without language or geographic restrictions. Cochrane Risk of Bias Tools (ROB2 and ROBINS-I) were used to assess study quality and risk of bias, and the quality of the evidence was rated using the Grading of Recommendations, Assessment, Development, and Evaluations tool. The extracted data were summarized using descriptive statistics.
Results:
A total of 148 studies were initially identified from a literature search, and after the screening, 52 articles were selected for full-text review. We included 41 studies published from 1996 to 2023 that met the eligibility criteria and involved 3035 participants. The studies comprised case reports (75.6%, n = 31), case series (12.2%, n = 5), retrospective cohort studies (7.3%, n = 3), and one (2.4%) each of prospective cohort and cross-sectional study, respectively. More than two-thirds were conducted in India (36.6%, n = 15), followed by Turkey (14.6%, n = 6) and Iran (12.2%, n = 5). Among the 72 CPA cases, 50% were male, and 38.9% were female, with a median age of 38 years. Common comorbidities included pulmonary tuberculosis (n = 5) and diabetes mellitus (n = 3). The most frequent symptoms were cough (n = 32), hemoptysis (n = 21), and fever (n = 16). Radiological examinations were conducted in 74.4% of hydatid disease cases, while histological examinations were performed in 82.1% of aspergillosis cases. Cavitary lesions were noted in 21.4% of radiological findings. Treatment involved both surgical and medical management for 43.1% of patients, with antifungals (50%) and anthelminthic drugs (50%) commonly used. Surgical treatments included cystectomy (57.9%) and lobectomy (55.3%). All patients with reported outcomes were cured.
Conclusion:
CPA complicates PCE and has both similar clinical and radiological features. Diagnostic approaches primarily include radiology and histology, while treatment often involves a combination of surgical and medical management, with antifungals and anthelminthic drugs commonly used. Despite the complexity of CPA, patients generally achieve favorable outcomes with appropriate treatment. Further research is needed to optimize diagnostic and therapeutic strategies for this condition.
Trial registration:
PROSPERO (CRD42024510441).
Plain language summary
This study looks at how two lung diseases, cystic echinococcosis and chronic pulmonary aspergillosis (CPA), can happen together. Cystic echinococcosis is caused by a parasite and can lead to cysts in the lungs. Sometimes, these cysts can become infected with a type of fungus called Aspergillus, leading to CPA. This infection can cause symptoms like coughing, coughing up blood, and fever, and can make it harder for people to breathe. To understand how often this happens and what treatments work best, we reviewed 41 studies from around the world that included 3,035 patients. Most of the studies came from India, Turkey, and Iran. In total, 72 people with cystic echinococcosis also had CPA. These patients were mostly middle-aged, and half of them were men. Many also had other lung diseases like tuberculosis. Doctors used imaging scans and tissue samples to diagnose both conditions. Treatment often involved a combination of surgery and medicine. The most common surgeries were to remove cysts or parts of the lung, while medications included antifungal drugs and treatments to kill the parasite. The good news is that all patients who received proper treatment recovered. This research shows that while CPA can complicate lung disease caused by parasites, it can be effectively managed if diagnosed early and treated properly. More research is needed to improve ways to find and treat these overlapping lung infections.
Introduction
Echinococcosis is a rare parasitic zoonosis caused by the larval stages of taeniid cestodes within the genus Echinococcus. Echinococcosis is a neglected tropical disease that occurs in two main forms, cystic echinococcosis (also known as hydatiduria) and alveolar echinococcosis, caused by the tapeworms Echinococcus granulosus and E. multilocularis, respectively.1–3 The World Health Organization (WHO) estimates that more than 1 million people are affected with echinococcosis at any one time, with human cystic echinococcosis (HCE) being the most common form globally. 4 The incidence rates for HCE can reach more than 50 per 100,000 person-years, and prevalence levels as high as 5%–10% may occur in parts of Argentina, Peru, East Africa, Central Asia, and China. 4 A recent analysis of its global burden suggests that the number of HCE cases had increased from 134,980 in 1990 to 207,368 in 2019. 5
Primary HCE typically involves a solitary cyst; however, multiple cysts or multiple organ involvement have also been reported, with the liver being the most common site for echinococcal cysts in pastoral strains (>65%), followed by the lungs (25%).6,7 Patients with pulmonary cystic echinococcosis (PCE) remain asymptomatic for years until the hydatid cysts grow to a significant size, triggering symptoms such as chronic cough, chest pain, shortness of breath, and eventually hemoptysis, which often results from rupture of large intraparenchymal or pleural cysts.4,7
Overall mortality in HCE ranges from 2% to 5% with adequate treatment.2,8 However, in patients with PCE, superinfections or co-infections are common and contribute significantly to overall mortality. 9 Co-existent infections occur in up to about 30% of patients initially presenting with PCE, with a mortality rate of about 6%. 9 Saprophytic colonization of cystic or residual cavitary lesions with Aspergillus spores following PCE or surgical interventions is a rare complication.10–16 This results in chronic pulmonary aspergillosis (CPA)—a progressive parenchymal disease that often occurs in patients with existing structural lung diseases, such as pulmonary tuberculosis. 17 From recent estimates, the annual incidence of CPA is about 1.8 million cases, with 340,000 (18.5%) deaths. 18
A large retrospective cohort study from Turkey of 100 patients with PCE found CPA in 2% of cases. 10 Similarly, in a systematic review of 1247 cases of CPA from Africa, 19 (1.5%) had CPA as a complication of PCE. 19 Thus, PCE is an underappreciated underlying disease that leads to the development of CPA, warranting a detailed review. Moreover, PCE and CPA have overlapping symptoms; therefore, underdiagnosis is common. In this systematic review, we aimed to pool and characterize cases of CPA complicating PCE by systematically reviewing globally published data to address these gaps, with the overarching goal of reducing morbidity from this seemingly neglected disease.
Methods and materials
Study design
We conducted a systematic review of literature following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines. 20 The PRISMA checklist is provided as Supplemental File 1.
Inclusion criteria
Patient/Population (P)
Individuals with CPA and pulmonary echinococcosis.
Exposure (E)
Aspergillus and cystic lung lesions.
Outcome (O)
Clinical outcomes, including treatment types, vital statistics, and clinical manifestations.
Study design (S)
Case reports, case series, cross-sectional studies, cohort and retrospective studies, randomized clinical trials.
Data sources
Electronic databases searched included Medline (through PubMed), Scopus, Embase, Google Scholar, Web of Science, and African Journal Online (AJOL).
Search strategy
A qualified medical librarian (BM) conducted a detailed database search using the keywords and medical subject headings (MeSH) “chronic pulmonary aspergillosis” OR “Aspergillus” OR “aspergilloma” OR “aspergillosis” AND “hydatid disease” OR “hydatidosis” OR “echinococcosis” OR “Echinococcus”. No language restriction or year of publication was applied.
Inclusion criteria
We included single case reports, case series, and cohort studies, including cases of CPA in patients with microbiologically or radiologically confirmed PCE of all ages and sexes across the world. There were no language restrictions. CPA was defined as a spectrum of pulmonary disorders caused by Aspergillus species, characterized by one or more pulmonary cavities, with or without a fungal ball, associated with chronic respiratory symptoms lasting 3 months or more (e.g., cough, hemoptysis, fatigue, or weight loss), and confirmed by radiological findings and/or serological evidence of Aspergillus infection. PCE was defined as a parasitic infection of the lungs caused by the larval stage of Echinococcus species, presenting radiologically as one or more pulmonary cysts, which may be asymptomatic or associated with respiratory symptoms such as cough, chest pain, hemoptysis, or dyspnea, and confirmed by imaging and/or serological tests for echinococcal antibodies. Studies were eligible for inclusion if they explicitly reported diagnostic criteria or case definitions meeting these operational definitions.
Exclusion criteria
We excluded review articles and extrapulmonary cases.
Selection process
Two sets of independent reviewers (RO and PB, and AA and FB) conducted the initial screening of titles and abstracts, focusing on studies related to CPA and pulmonary echinococcosis co-infection. Full-text assessment was performed for selected articles. Discrepancies in inclusion/exclusion decisions were resolved through discussion or consultation with a third reviewer (FB or AA). Non-English abstracts were translated using Google Translate or DeepL.
Data extraction
A standardized data extraction form was developed by FB and AA and pilot-tested by PB and RO. Extracted data encompassed study characteristics, country, sample size, modes of diagnosis of CPA and echinococcosis, clinical manifestation, treatment for both diseases, including medical and surgical, and clinical outcomes. Data extraction was independently conducted by five reviewers (RO, PB, AA, BEE, and FB), and any discrepancies were discussed with and resolved by agreement of at least three authors.
Data analysis
We conducted a narrative synthesis using Microsoft Excel, summarizing key information on CPA diagnosis, echinococcosis diagnosis, clinical manifestation, treatment, and treatment outcomes using frequencies and percentages. No meta-analysis or subgroup analyses were conducted.
Ethical considerations
As the review involves the analysis of publicly available, previously published studies, ethical approval is not applicable.
Results
Study characteristics
A total of 148 studies were found in the initial literature search, and an additional four via citation searching. After removing three duplicates and screening the title and abstract, 97 studies were excluded, leaving 51 articles for full-text screening. Ten were excluded because they were reviews or editorials without primary case data (n = 4), reported duplicate patient populations (n = 1), or lacked sufficient clinical detail to confirm CPA complicating PCE (n = 5). Eventually, 41 studies conducted between 1996 and 2023 involving 3035 participants were included (Figure 1). The studies comprised case reports (75.6%, n = 31), case series (12.2%, n = 5), retrospective cohort studies (7.3%, n = 3), and one (2.4%) each of prospective cohort and cross-sectional study, respectively. More than two-thirds were conducted in India (36.6%, n = 15), followed by Turkey (14.6%, n = 6) and Iran (12.2%, n = 5), Figure 2.
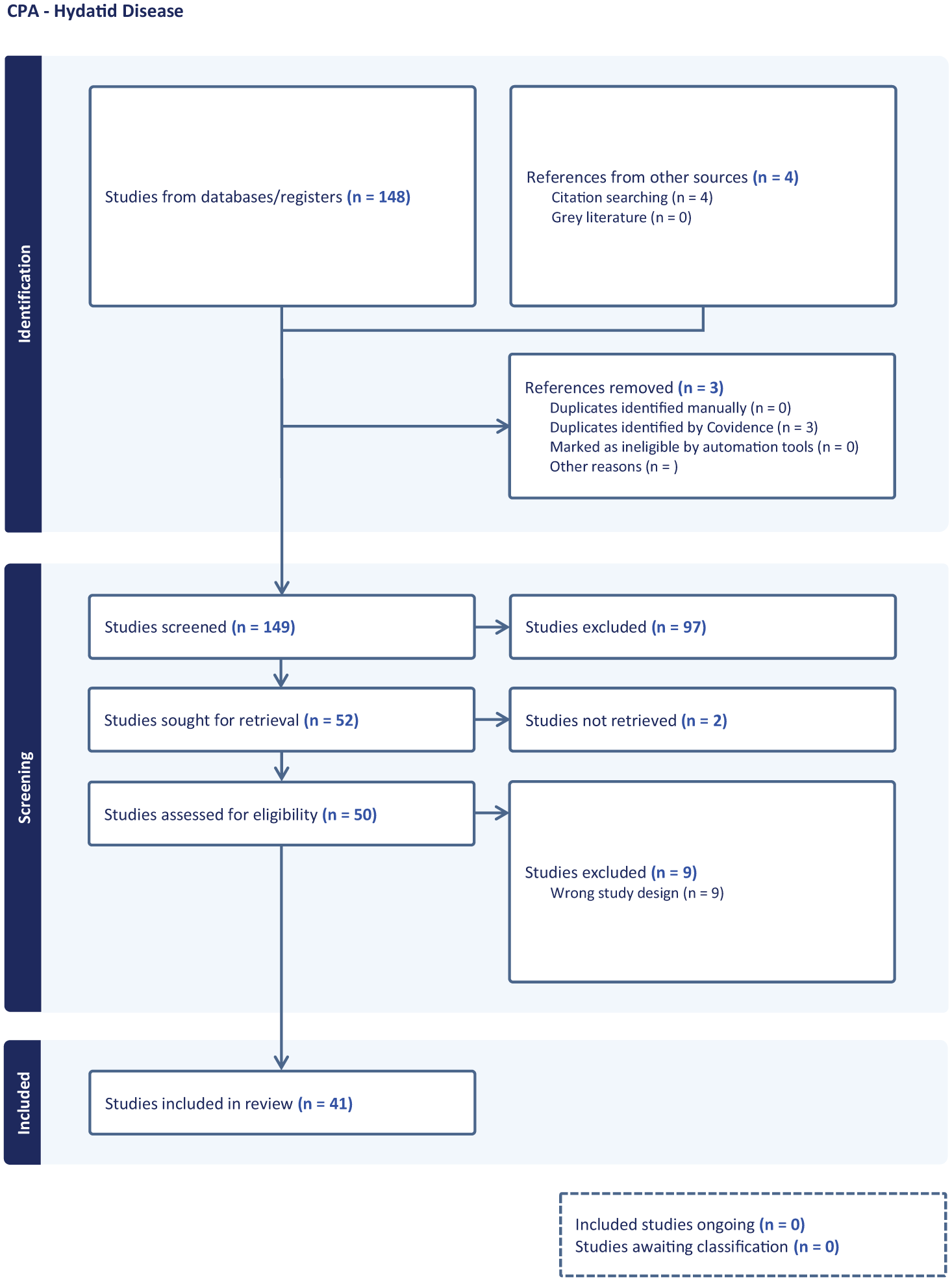
A PRISMA flow diagram showing the selection of articles for the systematic review.

World map showing cases of co-infections with hydatid cysts and aspergillosis.
Characteristics of the patients
There were 74 cases of CPA as a complication of PCE reported in the included studies (
Characteristics of included studies and the study participants.

DM, diabetes mellitus; F, Female; M, Male; PTB, pulmonary tuberculosis.
Clinical presentation
Cough (n = 34; 45.9%) with hemoptysis (n = 23; 31.1%), fever (n = 17; 23.0%), and chest pain (n = 14; 18.9%) were the most frequent symptoms. The duration of symptoms at diagnosis ranged from 2 weeks to 3 years. The most frequently reported clinical sign was decreased breath sounds, reported in nine studies (Table 2).
Clinical presentation.

Diagnostic evaluation
Regarding hydatid disease, chest imaging was the most common investigation performed in 31 studies (73.8%), followed by histology (64.3%, n = 27), serology (14.3%, n = 6), and microbiology (4.8%, n = 2). For aspergillosis, histology was most often performed (81.0%, n = 34), followed by radiology (28.6%, n = 12), microbiology (19.0%, n = 8), and serology (4.8%, n = 2; Figure 3).
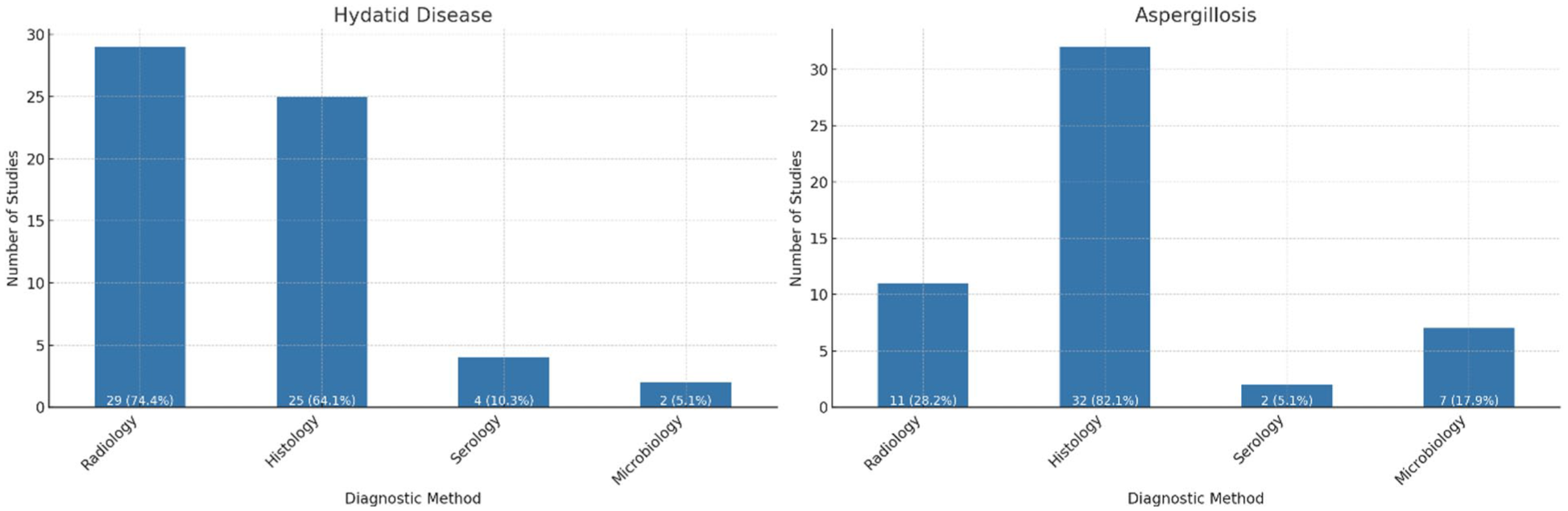
Bar charts showing the diagnostic methods used for echinococcosis and aspergillosis.
Microbiology and serology
In 17 studies reporting microbiological findings, sputum microscopy revealed Echinococcus granulosus in only one study, 40 while positive indirect hemagglutination was used to identify it in one study. 51 For the majority (88.2%, n = 15), sputum microscopy and culture did not reveal any significant findings related to E. granulosus. Aspergillus spp. was also identified in sputum cultures in only seven studies.12,30,35,40,44,47,52 None of the studies reported positive acid-fast bacilli findings or Mycobacterium tuberculosis.
Serological findings were reported for eight cases in seven studies. One case showed an intermediate-strength purified protein derivative (PPD) skin test with 15 mm induration. 12 Hydatid serology, using a specific anti-Echinococcus IgG enzyme-linked immunosorbent assay, was positive in three studies.12,40,52,53 Repeat Aspergillus serology in one of these cases was positive, with ImmunoCAP showing 195 mgA/L and a significantly raised serum IgE of 1503 kIU/L (reference range: 0–100). 40 Aspergillus-specific IgE was positive in a different case, with normal Aspergillus galactomannan levels. 31 Positive serological tests for Aspergillus and indirect hemagglutination for E. granulosus were also reported in another patient. 43 Additionally, one case confirmed the presence of Aspergillus flavus species by molecular assay. 30 However, elevated Aspergillus IgE levels are not diagnostic for CPA.
Histology
Histological findings from 34 studies involving 59 participants revealed a range of characteristics associated with co-infection with Echinococcus and aspergillosis. In 21 studies (61.8%) of the 34 with histological findings, cyst walls were infiltrated by fungal hyphae.11,13,16,21,24,26,27,30,31,33–37,41–44,47,52 Other common observations included cavities16,32,37,39,40,44,50,51 containing aspergilloma16,29,32,37,42,50,51 and chronic organized pneumonia.16,41,49,51 Polymorphs and histiocytes with fragments of Echinococcus and Aspergillus were sometimes found in smears.15,41 Seven studies showed laminated membranes of hydatid cysts,11,13,21,35,37,40,43 with Echinococcus hooklets in four studies,15,35,40,47 and septate fungal hyphae in nine studies,11,13,27,31,35,37,53 sometimes confirmed by periodic acid-Schiff11,13,27,31,35,37,53 and Grocott’s methenamine silver staining. 35 Chronic inflammatory changes in the surrounding lung parenchyma were frequently observed,28,32,42,47,53 and necrotic lung tissue was common.31,38,40 The presence of mixed inflammatory cells,21,26,30,31,37 fibroblastic proliferation,31,51 and vascular infiltration, 31 in response to fungal invasion, was also a notable finding.
Radiological findings
Radiological findings from the 36 studies with available results showed that 14 out of 58 cases (24.1%) had cavitary lesions with air-fluid levels (n = 6, 10.3%). Chest X-rays revealed oval or round densities in 10 cases (17.9%), while thoracic CT scans detailed cavitating lesions, pneumonic infiltration, and residual cavities with collapsed membranes in 15 cases (26.8%). Air crescent signs suggestive of aspergilloma were noted in 8 cases (13.8%), and fine curvilinear membranes indicating hydatid cysts were present in 2 cases (3.4%).35,49 Additional findings included thick-walled cavities near the hilum in 3 cases (5.2%),28,30,47 cystic lesions in 9 cases (15.5%),11,27–30,34–36,52 and significant consolidation in 11 cases (19.0%).16,30,32,35,38,40,48 Complex cysts often ruptured and contained air or mass-like opacities,25,26 and necrotic lesions showing intense enhancement were also reported.31,41
Bronchoscopy
Bronchoscopy findings were present in seven studies involving 21 patients co-infected with hydatid cysts and aspergillosis.10,12,26,32,47,48,53 In one case, fresh blood and necrotic material were observed coming from the superior segment of the right lower lobe. 47 Inflammation and secretions were noted in the left lung during bronchoscopic examination, with no fistula found in another case.12,32 One case reported no abnormalities. 32 In another study, a separate tissue cut section exhibited a fibrous wall containing necrotic material and dichotomous branching septate fungal hyphae admixed with neutrophilic infiltration. 26 During bronchoscopy, hemorrhagic lesions were found in the upper right lobe, and Serratia was identified in one case. 10 Findings in multiple cases included a white glistening membrane resembling tender coconut, pathognomonic for cystic hydatidosis (n = 9, 42.9%), and mucoid secretions (n = 3, 14.3%). 48
Management and clinical outcomes
Thirty-three patients (44.6%) received both surgical and medical management, while one patient received medical management only 25 and seven received surgical management only,13,16,27,35,43,46,49 Table 3. Of the 32 cases that received at least one form of medical management, 18 (52.9%) received anthelminthic drugs (albendazole), 16 (50.0%) received antifungals, and 7 (21.9%) received antibiotics. Only 7 (21.9%) patients received both antifungals and anthelminthic drugs. Itraconazole (n = 12) and voriconazole (n = 2) were the only antifungals used. Of the 40 cases (54.1%) who received surgical treatment, cystectomy (57.5%, n = 23), lobectomy (55.0%, n = 22), and capitonnage (27.5%, n = 11) were the most performed procedures (Figure 4). Abscess and hemorrhage were reported as postoperative complications in one patient each.
Management and clinical outcomes.

BAE, bronchial artery embolization.

Procedures performed in the management of chronic pulmonary aspergillosis complicating pulmonary cystic echinococcosis.
Discussion
This systematic review reports the co-existence of 74 cases of CPA as a complication of PCE. Although PCE is globally distributed, reports of cases with Aspergillus co-infection were confined to a limited number of countries, including India and Pakistan in Asia; Iran, Tunisia, and Morocco in the Middle East and North Africa; Spain and Turkey in Europe; and Peru in South America. The higher case burdens observed in these countries could be due to better case reporting, improved diagnostic capabilities, and the high endemicity of echinococcosis and TB. The review did not reveal any predilection for the female sex or immune compromise as sometimes asserted by some authors. 11 In most studies, aspergillosis was diagnosed incidentally by histology, suggesting that clinical suspicion was often low.
The patchy distribution of CPA and PCE co-infection is not surprising. Cystic echinococcosis is a neglected tropical disease that receives little funding for research and treatment relative to its burden of disease. It is, therefore, underdiagnosed. To illustrate, the disease is considered endemic in sub-Saharan Africa, but only scarce data, if any, exist for most countries. 54 The rarity of reports of CPA and PCE co-infection may, therefore, in part be attributed to the neglected status of hydatid disease.
PCE is typically asymptomatic in the early stages. As cysts become larger or more complex, symptoms may develop and vary depending on size, location, compression of nearby structures, and the condition of the cystic structures. 11 From our review, cough, fever, hemoptysis, and chest pain—symptoms that can occur in independent cases of both PCE and CPA—were the commonest presentations. Thus, the coexistence of Aspergillus in hydatid cysts is prone to being overlooked, and diagnosis can only be made by deliberate investigations, including imaging and serology.
The diagnosis of PCE is usually based on radiology. 27 On imaging, PCE is associated with a variety of signs, depending on whether the cysts are complicated or not. Intact cysts present as sharply defined round to oval homogenous opacities of variable size, typically 1–20 cm in diameter, on plain chest radiographs. 55 Partially evacuated cysts give the waterlily or Camelote sign in about 10%–15% of PCE cases. The signet ring sign, seen when a bleb of air dissects into the wall of the cyst, giving it the shape of a ring, is indicative of impending cyst rupture. 55 In contrast, ruptured cysts produce a host of signs, such as the air crescent or meniscus sign, inverse crescent sign, Cumbo sign (onion peel sign), rising sun sign, and air bubble sign. 55 The air bubble sign on a CT scan, which has high sensitivity and specificity, signifies a ruptured, infected hydatid cyst, differentiating it from abscesses or tumors.
For CPA, chest radiography is the initial screening imaging modality and can demonstrate cavities, bronchiectasis, soft tissue within the cavity, nodules, and volume loss/lung fibrosis. 56 High-resolution chest computed tomography is the imaging modality of choice for better delineation of cavities, cavity wall invasion, intracavitary soft tissue, pleural thickening, and lymphadenopathy. 56 In simple aspergillomas, chest radiograph shows an air-filled cavity with intracavitary soft tissue, usually located in the upper lobes. At the same time, high-resolution CT of the thorax typically depicts a well-defined soft-tissue mass within a lung cavity, with a characteristic Monod sign (air surrounding the aspergilloma in a pre-existing cavity).
In the diagnosis of hydatid disease, serological testing typically serves a supportive role. In addition to the assay format, the sensitivity of serological assays depends on the location, number, integrity, and viability of the cyst. 55 The seropositivity rate is higher in hepatic cysts than in pulmonary cysts. 55 In PCE, intact pulmonary cysts often give a falsely negative serological test, while ruptured cysts increase the likelihood of seropositivity. This is because the sequestration of antigens within intact cysts minimizes their ability to stimulate antibody production by the host’s immune system. On the other hand, serological tests for CPA have become a mainstay of diagnosis, as elevated Aspergillus-specific IgG antibodies are found in over 90% of patients with CPA. 57 However, our review indicates that serology for aspergillosis was rarely performed, possibly due to its non-availability or a low index of suspicion. Microbiological cultures of respiratory specimens can also be used for diagnosis, but they are less sensitive. The recovery rate of Aspergillus spp. is significantly higher for high-volume cultures than for conventional cultures. 58
PCE requires a combined surgical and medical therapy, with a PAIR (puncture, aspiration, injection, and re-aspiration) approach; medical therapy, typically with albendazole or mebendazole, is the standard of care. 59 Cystic echinococcosis generally has a good prognosis, with cure rates as high as 100% and very low recurrence rates.59,60 On the other hand, medical therapy is the mainstay of managing CPA. Oral itraconazole or voriconazole for 6–12 months or lifelong is the mainstay of therapy for most symptomatic patients and is associated with a significant improvement in quality of life.61,62 Mortality in treated cases of CPA is about 20% at 1 year and about 50% at 5 years.63–65 In CPA, surgical treatment is indicated for patients with Aspergillus nodules and for those with simple aspergilloma with good lung function test results. 66 Surgery is curative; however, a few patients relapse.67,68 In Africa, the surgical management of patients had a mortality rate of less than 10%. 69 Video-assisted thoracoscopic surgery is an emerging surgical option associated with lower post-operative complications, recurrence, and post-procedural sepsis. 70
This review provides a comprehensive analysis of CPA as a complication of PCE and a new insight into the expanding risk factor for CPA. CPA diagnosis is becoming more feasible in low-income countries with the introduction of the Aspergillus IgG–IgM lateral flow assay. 71 Demonstration of a possible occurrence of CPA-PCE -PTB is a significant proportion in this review and warrants further investigations into the need for enhanced clinical suspicion for CPA in PCE, PTB, or co-infected individuals. An important limitation of this systematic review is that we included case reports and case series, given their underreporting in the literature. This limits the statistical inferences we can make, and the descriptions are prone to selection bias.
Limitations and future directions
This systematic review has several limitations that should be acknowledged. First, most included studies were case reports and small case series, which are inherently subject to publication and reporting bias, as unusual or severe presentations are more likely to be published than typical cases. Second, there was marked heterogeneity in the diagnostic approaches used across studies, with limited application of standardized criteria for both chronic pulmonary aspergillosis and PCE. This variation limits the comparability of cases and may have led to under- or over-diagnosis in some reports. Third, important clinical details, such as comorbidities, treatment regimens, and outcomes, were inconsistently reported, further limiting the ability to draw robust conclusions. Finally, the geographic clustering of studies in a few endemic regions suggests that the true global burden of CPA complicating PCE remains underestimated, and findings from this review may not be generalizable to all settings.
Future studies should address these gaps through prospective cohort studies conducted in endemic regions, which would provide more reliable estimates of CPA incidence, clinical outcomes, and risk factors complicating PCE. The development and validation of standardized diagnostic scoring systems could also enhance comparability across studies and improve early recognition of this co-infection. In addition, integrating advanced molecular and omics-based approaches for pathogen identification holds promise for clarifying host–pathogen interactions and uncovering novel diagnostic and therapeutic targets.
Conclusion
PCE can predispose both immunocompromised and immunocompetent individuals to the development of CPA. Diagnostic approaches primarily include radiology and histology, while treatment often involves a combination of surgical and medical management, with antifungals and anthelminthic drugs being commonly used. Despite the complexity of CPA, patients generally achieve favorable outcomes with appropriate treatment. Although rare, greater awareness and understanding of this co-infection will improve diagnosis and management, ultimately enhancing patient care in areas endemic for echinococcosis. Proactive steps should be taken to detect Aspergillus co-infection using computed tomography scans and serological testing. Where these are negative, follow-up of persons managed for PCE may be warranted to detect the development of CPA promptly. Further research is needed to optimize diagnostic and therapeutic strategies for this condition.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251404362 – Supplemental material for Chronic pulmonary aspergillosis complicating pulmonary cystic echinococcosis: a global systematic review
Supplemental material, sj-docx-1-tai-10.1177_20499361251404362 for Chronic pulmonary aspergillosis complicating pulmonary cystic echinococcosis: a global systematic review by Felix Bongomin, Aayush Adhikari, Ronald Olum, Pratik Baral, Iriagbonse Iyabo Osaigbovo, Winnie Kibone, Bassey E. Ekeng, Linda Atulinda, Bethan Morgan, Shailendra Katwal, Norman van Rhijn and David W. Denning in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
Felix Bongomin is supported by the Carigest MA Conny Maeva Charitable Foundation as part of the “Chronic Pulmonary Aspergillosis: Optimization of Therapy, Immunogenetic Screening, and Diagnosis in Uganda [CPA_OPTIONS_Uganda]” as a PhD studentship at the University of Manchester, United Kingdom. CARIGEST SA did not play any role in the design, implementation, and analysis of the study.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
