Abstract
Background:
Malaria and soil-transmitted helminth (STH) infections often co-occur in Nigerian children under 5 years of age, affecting haematological functions. However, local data on their combined effect are limited.
Objectives:
To determine the prevalence and impact of malaria, STH and malaria–STH co-infection on haematological parameters in children under five in the Delta South Senatorial District in Nigeria.
Design:
Hospital-based cross-sectional study.
Methods:
A total of 269 children under 5 years of age attending selected health centres were recruited across the Delta South Senatorial District. Malaria was diagnosed by microscopy, STH by stool analysis and haematological parameters were measured using automated techniques. Associations between infection status, demographics, clinical characteristics and haematological indices were examined with descriptive statistics, T-test, ANOVA and correlation analyses.
Results:
Co-infection was most common in toddlers aged 1–2 years (40.9%) and was more frequent in males. Normal haemoglobin genotype (Hb AA) predominated (57.3%), followed by sickle cell trait (Hb AS) (36.3%) and sickle cell disease (Hb SS) (6.3%). Children with Hb SS had lower haemoglobin, packed cell volume (PCV) and red blood cell (RBC) counts, but higher white blood cell levels (p < 0.01). Children with malaria or STH, especially those with co-infection, showed a macrocytic hypochromic pattern with increased mean corpuscular volume and reduced mean corpuscular haemoglobin concentration (p < 0.001), despite only mild, non-significant reductions in haemoglobin and RBC counts. Parasitaemia varied with age (p = 0.033) but showed weak correlations with haemoglobin, RBC and PCV, suggesting that anaemia was influenced more by host factors than by parasite burden. Hb AS children had higher parasite loads but better red cell indices; Hb SS cases showed the greatest haematological derangement, particularly when coinfected.
Conclusion:
Malaria and STH infections remain a major cause of haematological abnormalities in children under five. Early red cell alterations, especially macrocytosis and hypochromia, preceded obvious anaemia and were more strongly influenced by host factors than by parasite density, underscoring the need for integrated control and careful haematological assessment in endemic settings.
Plain language summary
Malaria and intestinal parasitic infections often occur together in Nigerian children under-5 years and affects haematology. Yet, little is known about their combined effects, especially when children have different blood characteristics. This study looked at 269 children under-5 years from health centres in Delta State, Nigeria. Malaria was tested using blood samples, while helminthes were checked through stool tests. Several blood measurements were taken to understand the impact of these infections. Co-infection with both malaria and helminthes was most common in toddlers aged 1–2 years (40.9%). Boys generally had higher infection rates than girls. The most common blood genotype was Hb AA (57.3%), followed by Hb AS (36.3%) and Hb SS (6.3%). Children with Hb SS had the poorest blood results, with much lower haemoglobin, PCV, and red blood cell counts, but higher white blood cell levels. Co-infected children were more likely to have larger red blood cells (macrocytosis), while overall falls in haemoglobin and RBC counts were modest and not statistically significant. Malaria parasite density was higher in boys than in girls and peaked among newborns and five-year-olds. Children with anaemia carried higher parasite loads and developed more frequent fevers, particularly when they were infected with malaria alone. Hypochromia (pale red cells) and anaemia were common in co-infected children with Hb SS and Hb AS, showing that genetic background influences disease impact. Strong links were found among Hb, PCV, and RBC, while parasite density showed weak relationships with blood indicators. These findings show that malaria and helminthes infections together worsen blood problems in children, particularly in those with sickle cell traits. Targeted strategies combining malaria and helminthes control, and focused care for children with sickle cells, are needed to reduce sickness in affected communities.
Background
Multiparasitism is the concomitant existence of different infections of parasitic origin in a single host. This phenomenon remains a major public health concern, especially in the less economically viable areas.1–5 This condition is common in low-income communities and vulnerable populations, such as young children under 5 years of age.6,7 These children are especially vulnerable due to their developing immune systems and high exposure rates. Under-nutrition and inadequate hygiene further increase susceptibility, affecting growth, well-being and haematological stability. Co-infections with malaria and helminths can lead to compounded pathological effects, including alterations in haematological parameters, which are critical for diagnosing and monitoring these diseases.8–10
Malaria and soil-transmitted helminth infections often co-exist in many regions where they are considered endemic, and this phenomenon has largely been observed among children. There have been studies that have shown a large overlap between the two infections due to shared environmental and socioeconomic factors.11,12 Malaria and helminth co-infections have also been associated with differences in clinical and haematological manifestations compared to single infections.11,12
Malaria remains one of the most significant parasitic infections worldwide and is a leading cause of death due to Plasmodium parasites. 7 Despite decades of global control efforts, malaria has stubbornly resisted elimination and continues to pose a serious public health challenge, especially in Africa. According to the World Health Organization, malaria remained a worldwide public health concern in 2023, with an estimated 249 million cases and over 600,000 deaths worldwide, the majority occurring in sub-Saharan Africa and disproportionately affecting children under 5 years of age. 7 Notably, within this region, 76% of these malaria deaths were among children under 5 years of age.7,13 Beyond its devastating health impacts, malaria also exacerbates poverty in affected populations, perpetuating a cycle of poor health and economic hardship.7,13
In Nigeria and across sub-Saharan Africa, the burden of malaria is worsened by widespread poverty, poor access to healthcare and a lack of adequate public health infrastructure, making it even harder to combat the disease effectively. 14 Malaria, caused by various species of Plasmodium, primarily affects the blood, leading to haemolysis and severe complications such as anaemia, thrombocytopenia and abnormalities in white blood cell (WBC) counts. 15 Anaemia is often the most noticeable haematological manifestation, caused by the destruction of infected red blood cells (RBCs) and the suppression of new blood cell production due to inflammatory reactions in the body.15,16 Beyond anaemia, malaria infections can also lead to immune-related haematological changes, including leukopenia or leucocytosis and thrombocytopenia, reflecting the body’s systemic response to infection and associated tissue damage. 15 These challenges underscore why malaria remains a major public health concern in Nigeria, highlighting the need for comprehensive approaches that combine medical interventions with improvements in healthcare infrastructure and equitable access to care.
Helminth infections, caused by intestinal parasites like Ascaris lumbricoides, Trichuris trichiura and hookworms, are notorious for causing chronic blood loss and iron-deficiency anaemia due to intestinal blood loss and poor nutrient absorption.17,18 Soil-transmitted helminth (STH) infections, caused by parasitic worms such as A. lumbricoides (roundworm), T. trichiura (whipworm) and A. duodenale N. americanus (hookworms), are highly prevalent in regions where access to clean water, adequate sanitation and good hygiene (WASH) remains limited. 19 In 2023, about 1.5 billion people globally, that is, 24% of the world’s population, were affected by STH infections, with sub-Saharan Africa bearing the highest burden. 20 Young children are particularly vulnerable, with over 260 million cases documented among preschool-aged children. 20 The overlapping distribution of malaria and STH infections often leads to dual burdens, further compounding health disparities and exacerbating morbidity in affected communities.21,22 For instance, haemolysis caused by malaria may intensify helminth-induced anaemia, while helminth-mediated immune modulation could alter the severity of malaria infections.23,24 One of the most worrying combinations is malaria, concurrently with STHs, together affecting over one-third of the world’s population.20,21,25,26
Despite the high burden of malaria and STH infections globally and within Nigeria, research on the combined haematological effects in children remains limited in the Nigerian context, whereas several studies have explored this subject globally.22,26,27 Furthermore, little is known about how haemoglobin genotypes such as Hb AA, AS and SS modify haematological responses during single or co-infection, despite their clinical relevance in West African populations. Emerging evidence suggests that these genotypes may influence distinct red cell and inflammatory patterns that remain poorly described in young children. This is particularly critical, as Nigeria is highly endemic for both diseases, with its already strained healthcare resources. Such efforts are essential to address the dual burden of these infections and improve the health and well-being of children in Nigeria’s most vulnerable communities.26,27 This study investigates the individual and combined effects of malaria and STH infections on haematological parameters in children under five in Delta State, Nigeria.
Materials and methods
Study design and population
This study employed a descriptive, hospital-based cross-sectional survey. The study was carried out from May to December 2022 and from May to December 2023 to coincide with both rainy and dry seasons and account for seasonal variations in parasitic infections.14,28 The study population consisted of 269 children under 5 years of age residing within the study area and attending selected hospitals and health centres. These healthcare facilities were purposively sampled to represent each of the eight (8) local government areas in the Delta South senatorial district, in accordance with the WHO Service Availability and Readiness Assessment (SARA) guidelines. 29 Children were eligible if they were aged 0–59 months, had lived in the study area for at least 6 months and had parental or guardian consent. Exclusion criteria included a recent blood transfusion (within 3 months) or refusal of consent. Only children of parents or legal guardians who signed informed consent were included in the study. The purpose and benefit of the project were explained to them, including the process for blood and faecal sample collections following recruitment. Demographic data such as age and sex were collected from consenting individuals prior to enrolment.
Study area
The study was conducted in the Delta South Senatorial District in the southern part of Delta State, Nigeria, bordered by the Atlantic Ocean to the south. 30 The district comprises eight (8) local government areas 31 and includes riverine and coastal communities as well as mainland settlements characterised by extensive networks of creeks and rivers.32,33 The approximate geographic coordinates of the district lie between latitude 4.50°N and 5.70°N and longitude 5.00°E and 6.30°E.
The district forms part of the Niger Delta, where oil exploration co-exists with residential communities and contributes to varying degrees of urbanisation and population mobility. In combination with environmental conditions, these factors may indirectly influence living standards and access to healthcare in some communities.32–34 The area has a humid-tropical climate with a long wet season (April–October) and a shorter dry season influenced by Harmattan winds (November–March).30,32 Annual rainfall averages 2000–3500 mm; mean temperatures range between 25°C and 33°C and relative humidity is 70%–90% during rainy months.30,32
The geographical terrain can limit access to healthcare,32–34 and together with poverty and poor sanitation, contributes to the persistence of endemic diseases such as malaria and soil-transmitted helminths. 6 Primary healthcare centres and their catchment communities were therefore purposively selected to obtain a representative district-level sample, in line with WHO SARA recommendations. 29
Sample size determination
Sample size was estimated using the standard single-population proportion formula for cross-sectional studies, assuming a 95% confidence level and a 5% margin of error. 35 An expected prevalence of 22%, based on national estimates from the Nigeria Malaria Indicator Survey, was used in the calculation, yielding a minimum sample size of approximately 269 participants. 36 The study was hospital-based, and enrolment was determined by outpatient attendance and eligibility during the study period. A total of 269 children under 5 years of age were recruited for the study, which is comparable to sample sizes reported in similar hospital-based paediatric malaria and helminth studies in Nigeria and other African endemic settings.37–39 This sample size was considered adequate for estimating infection prevalence and detecting moderate differences in key haematological indices, although statistical power was limited for smaller subgroups, such as children with the Hb SS genotype.
Sample collection
Blood Samples
Approximately 2–3 mL of venous blood was drawn from each child via venipuncture using a sterile disposable syringe. The blood was then transferred into ethylenediamine-tetra-acetate (K2-EDTA) tubes for subsequent laboratory analysis.40–42 About 1 ml was used for malaria microscopy and parasite density estimation, and the remaining was used for haematological parameters.
Stool samples
Fresh stool samples were collected separately from each participant using sterile universal containers. Each sample was properly labelled and transported to the laboratory within 4 h, consistent with WHO recommendations for Kato-Katz examination.43–45
Clinical evaluation
Body temperature was measured axillary using a calibrated digital clinical thermometer for all participating children under 5 years of age. In alignment with WHO guidelines, a body temperature reading of ⩾37.5°C was used to define febrile status.42,46 This threshold is particularly relevant in malaria- and helminth-endemic regions, where fever often serves as a key clinical indicator of underlying parasitic infections. All clinical assessments and biological sample collections, including venous blood and stool sampling, were performed by trained healthcare personnel. Prior to clinical evaluation, demographic characteristics, such as sex and age, were obtained from the participants’ caregivers or parents.
Laboratory procedures
Malaria parasite microscopy and quantification
Thick and thin smears of blood were prepared from each sample, stained with Giemsa and examined microscopically using an oil-immersion objective lens to confirm the presence of malaria parasites and to quantify the level of parasitaemia.43,44,46 Parasites were quantified by counting against a fixed number of white blood cells and calculated as:
Two microscopists read each slide independently, with discrepancies resolved by a third reader.
Haematological analysis
Complete blood counts (CBC) were performed within 4 h of sample collection using the automated Sysmex KX-21N haematology analyzer, following the manufacturer’s standard operating procedures. The analyzer measured a range of haematological parameters, including haemoglobin (Hb), haematocrit, WBC counts, RBC count, platelet count and red cell indices—mean corpuscular volume (MCV), mean corpuscular haemoglobin (MCH) and mean corpuscular haemoglobin concentration (MCHC). Anaemia morphology was classified using red cell indices (MCV and MCHC) derived from the complete blood count, rather than peripheral blood smear examination. Daily internal quality control checks were performed, whereas calibration followed the manufacturer’s scheduled maintenance procedures. 42 Result interpretation was based on paediatric reference values and the World Health Organization (WHO) thresholds for anaemia.40–43 The haematological indices analysed were selected as core markers of anaemia, red cell morphology and infection-related inflammatory response in malaria- and helminth-endemic settings. Haemoglobin genotype was determined using standard haemoglobin electrophoresis on cellulose acetate at alkaline pH, following routine laboratory protocols. 42 All procedures adhered to the methodological standards described by Dacie and Lewis. 40
Faecal examination
Stool specimens were analysed using the Kato-Katz thick-smear method (41.7 mg template) according to WHO guidelines to detect and count STH eggs.43–45 Slide readings allowed species-level identification of helminth ova based on their distinct morphological features.43–45
All laboratory analyses were performed by trained laboratory scientists and technicians with experience in malaria microscopy, haematology and parasitology.
Statistical analysis
Descriptive statistics were used to summarise the sociodemographic, clinical and haematological characteristics of the study population. Continuous variables (Hb, PCV, RBC, WBC, platelet count, MCV, MCH and malaria parasite density) were reported using mean values, standard deviations (SDs) and standard error of the mean (SEM), whereas categorical variables were reported using frequency and percentage. A t-test was used to compare the means of two independent groups, while analysis of variance (ANOVA) was employed for multiple comparisons among three or more groups. In cases where post hoc comparisons were required following statistically significant differences, Tukey’s Honest significant difference was employed (p ⩽ 0.05) in partitioning the means. Furthermore, we conducted a Pearson correlation analysis to assess relationships between selected haematological indices, demographics and clinical parameters by malaria status and by Malaria and STH co-infection status. Stacked bar charts were used to show the distribution of variables by gender, age groups, genotypes and co-infection status. As the research was exploratory, the results were interpreted with care, especially when they showed weak associations. All analyses were two-tailed with statistical significance set at p ⩽ 0.05 and were performed using SAS JMP Statistical Discovery™ version 17.0 (SAS Institute, Cary, NC, USA).
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) guidelines.
Ethical considerations
Ethical approval for this study was obtained from the Delta State Ministry of Health Ethics Committee. Written informed consent was obtained from parents or legal guardians of all participating children prior to enrolment. Full ethical approval details and consent statements are provided in the Declarations section.
Results
Characteristics of participants
Figure 1 presents a stacked bar chart illustrating the proportion of male and female participants who were co-infected with malaria and STH, stratified by age group. Among co-infected children, toddlers aged 1–2 years accounted for the highest proportion (40.9%), followed by those aged 3–4 years (31.6%) and infants aged 3–11 months (14.9%). The lowest proportions were observed among children aged 5 years (8.5%) and newborns aged 0–2 months (4.1%). Across all age groups, males consistently exhibited a higher prevalence of co-infections than females, with the greatest sex disparity among toddlers and preschoolers.

Gender of children co-infected with malaria and soil-transmitted helminths by age group.
Among participants co-infected with malaria and STH, the majority had the Hb AA genotype (57.3%), followed by Hb AS (36.3%), while only a small proportion had Hb SS (6.3%; Figure 2). Across all genotypes, males were more represented than females. Specifically, 33.0% of the co-infected children were males with Hb AA, compared to 24.3% females; 22.1% were males with Hb AS, compared to 14.2% females; and 3.7% were males with Hb SS, compared to 2.6% females.
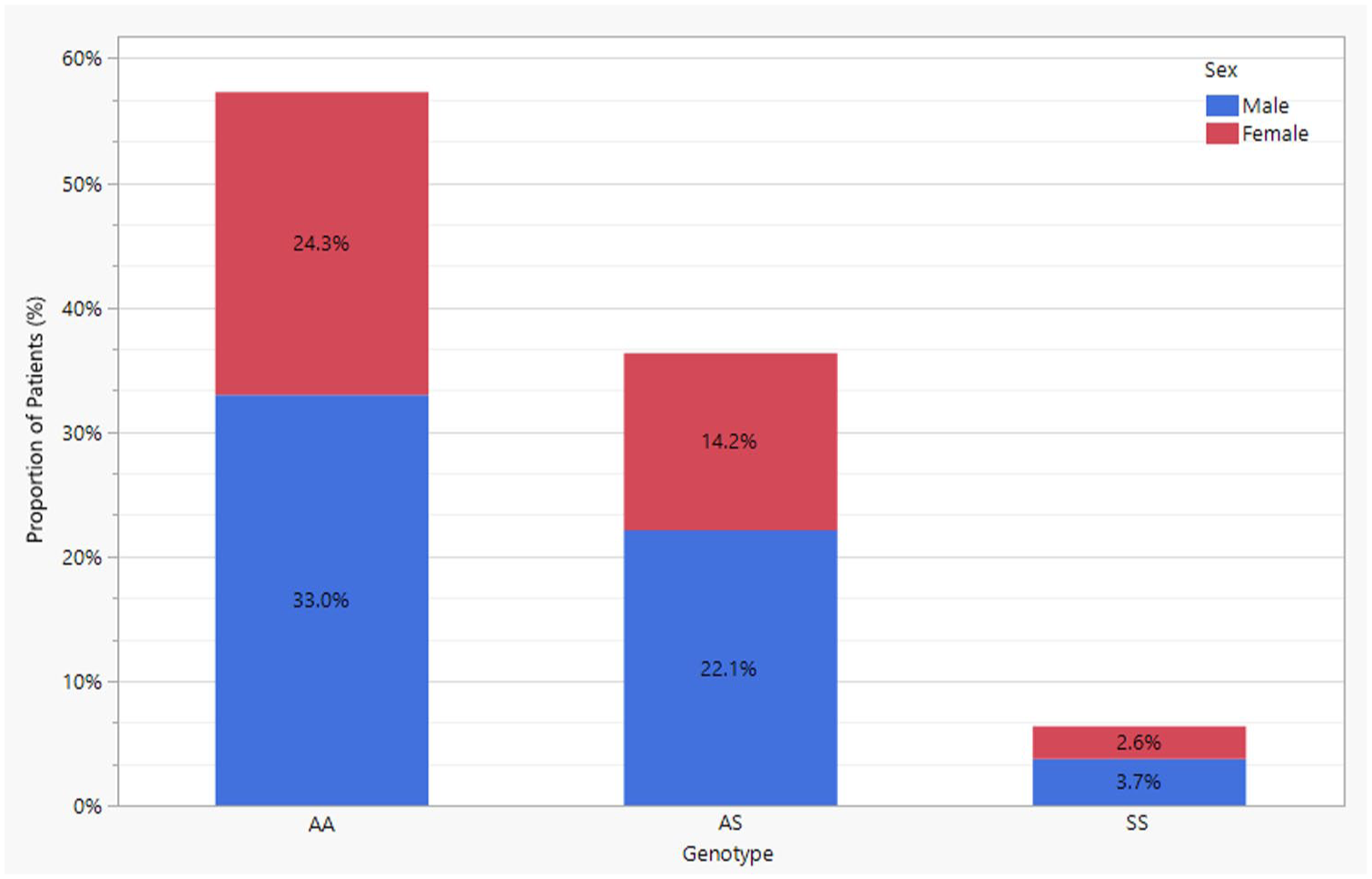
Gender of children co-infected with malaria and soil-transmitted helminths by haemoglobin genotype.
Haematological profile and infection status
Table 1 presents the haematological profile of the participants. Overall, participants exhibited mild normocytic anaemia, characterised by a mean PCV of 34.5 ± 0.4%, a mean HB of 11.3 ± 0.1 g/dL and an RBC count of 3.75 ± 0.04 × 1012/L. No statistically significant differences in haematological indices were observed across sex or age groups (p > 0.05). However, haemoglobin genotypes had a marked and statistically significant impact on haematological parameters. Children with homozygous sickle-cell disease (Hb SS genotype) demonstrated significantly lower PCV (29.4 ± 1.3%), HB (9.7 ± 0.5 g/dL) and RBC counts (3.22 ± 0.15 × 1012/L), along with significantly elevated WBC counts compared to children with Hb AA or Hb AS genotypes (p < 0.01).
Haematological parameters in malaria and soil-transmitted helminth co-infected children living in endemic areas in Delta State, Nigeria, by demographic and clinical characteristics.
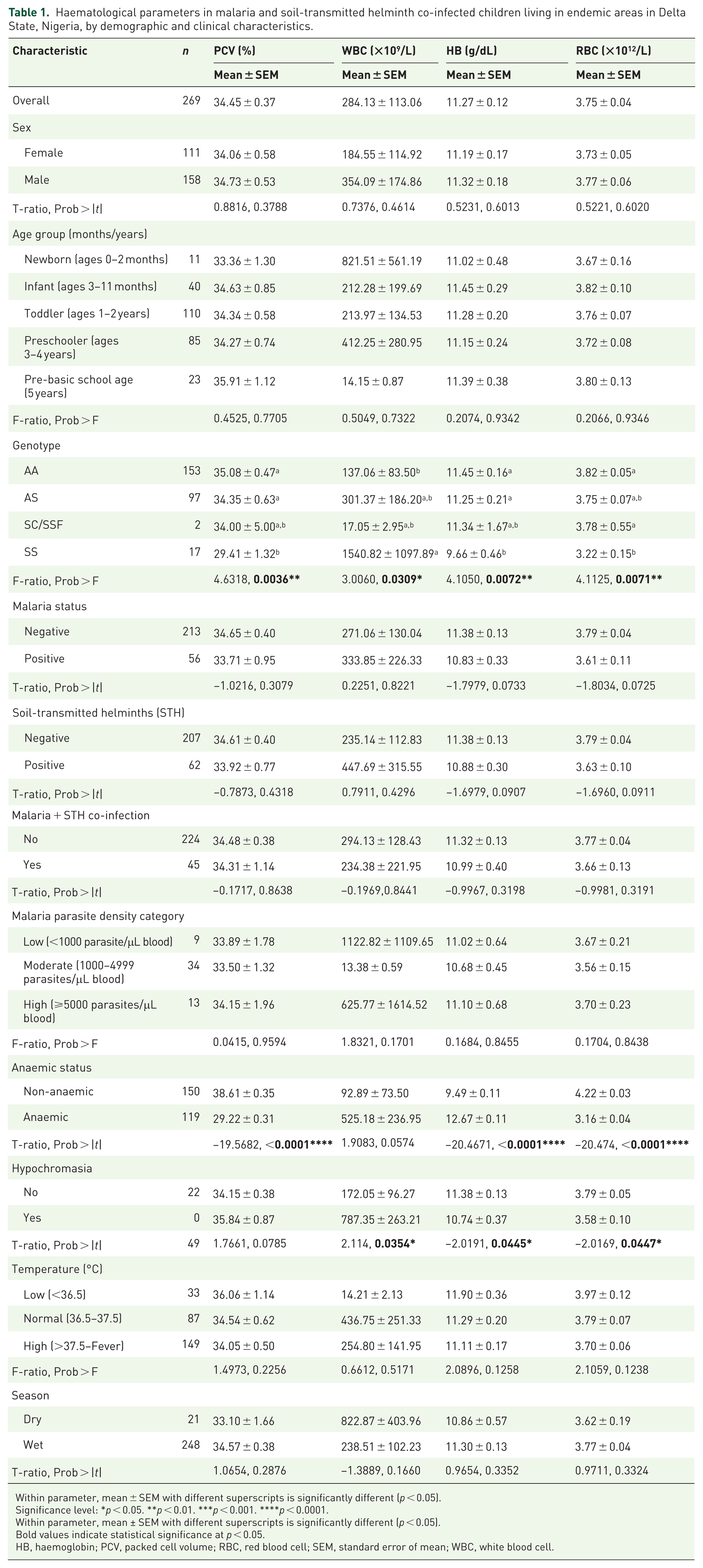
Within parameter, mean ± SEM with different superscripts is significantly different (p < 0.05).
Significance level: *p < 0.05. **p < 0.01. ***p < 0.001. ****p < 0.0001.
Within parameter, mean ± SEM with different superscripts is significantly different (p < 0.05).
Bold values indicate statistical significance at p < 0.05.
HB, haemoglobin; PCV, packed cell volume; RBC, red blood cell; SEM, standard error of mean; WBC, white blood cell.

Within parameter, mean ± SEM with different superscripts is significantly different (p < 0.05).
Significance level: *p < 0.05. **p < 0.01. ***p < 0.001. ****p < 0.0001.
MCV, mean corpuscular volume; MCHC, mean corpuscular haemoglobin concentration; SEM, standard error of mean.
When stratified by infection status, children with either malaria or STH alone exhibited modest reductions in Hb levels (approximately 0.5 g/dL) and RBC counts (around 0.15 × 1012/L). However, these decreases were not statistically significant (p = 0.07–0.09). Similarly, children co-infected with both malaria and STHs did not show any additional decline in these parameters compared to those with single infections. Notably, children who were positive for malaria or STHs showed a significant increase in MCV and a decrease in MCHC (p < 0.001), indicative of a macrocytic-hypochromic pattern. This abnormal red cell morphology appeared even when mean Hb and RBC values remained near normal, suggesting early functional iron deficiency or erythropoietic stress. Although platelet counts were slightly lower in malaria-positive cases, the difference was not statistically significant (p > 0.05).
Malaria parasite density across demographic and clinical subgroups
Table 2 illustrates malaria parasite density by demographic and clinical characteristics among the participants. Among the 56 malaria-positive children, the mean parasite density was 8290.96 ± 2092.94 parasites/µL. Parasite density was higher in males (10,579 ± 3208 parasites/µL) than in females (4755 ± 1796 parasites/µL), although the difference was not statistically significant (p = 0.1764). Age had a notable effect, with the youngest (<2 months) and oldest subgroups (5-year-olds) carrying higher parasite densities (approximately 10,387 and 41,326 parasites/µL, respectively). The lowest parasite density was observed among toddlers (1–2 years; approximately 5062 parasites/µL), with these differences reaching statistical significance (p = 0.0331). Other factors, such as genotype, co-infection, anaemia status, hypochromasia, fever and seasonality, were not significantly (p > 0.05) associated with parasite densities. Seasonal differences in PCV, HB, RBC and parasite density were minimal and not statistically significant (p > 0.05). However, WBC counts tended to be higher during the dry season. Rates of malaria, STH and co-infections were similar regardless of the seasons.
Malaria parasite density by demographic and clinical characteristics in children living in malaria endemic areas in Delta State, Nigeria.
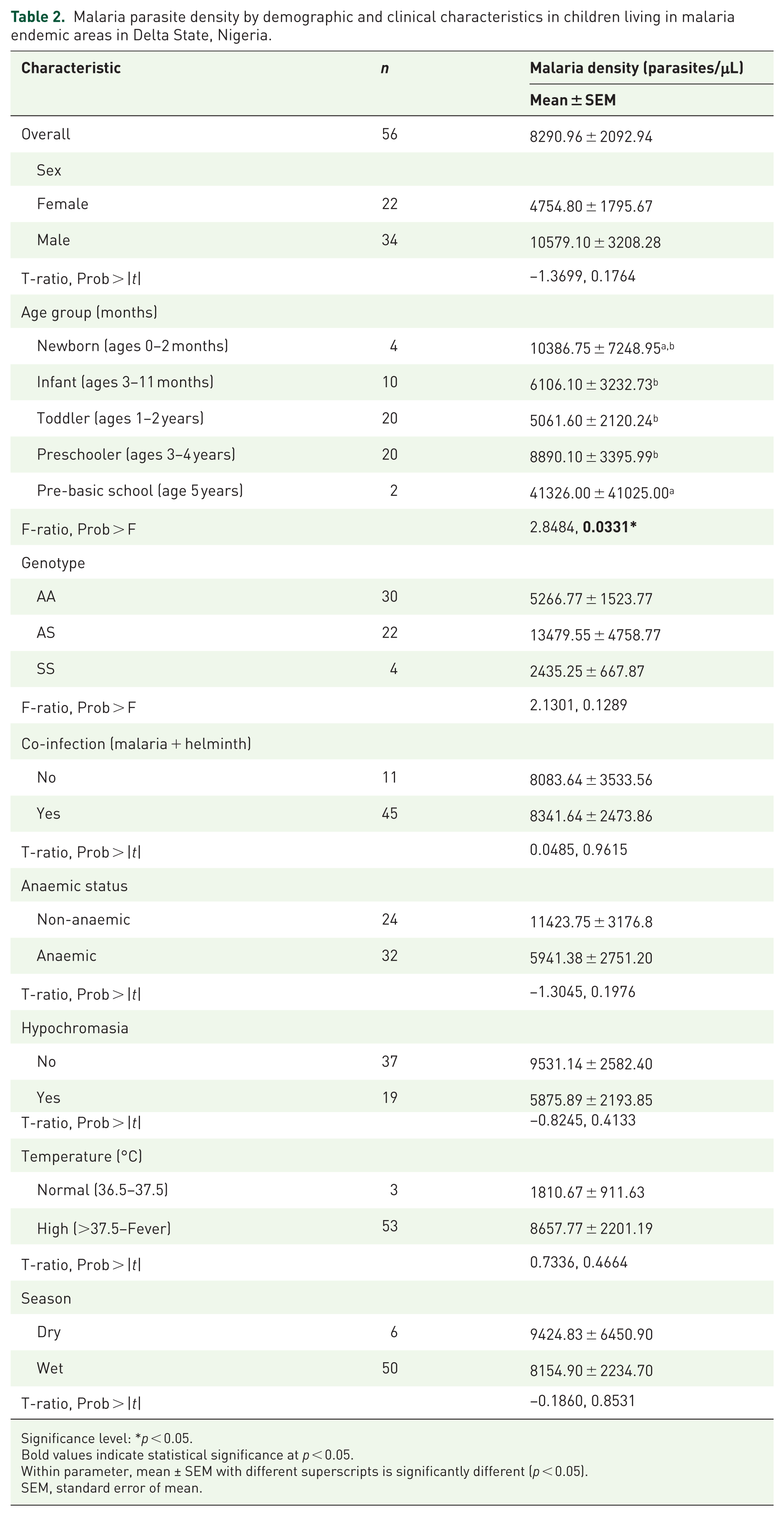
Significance level: *p < 0.05.
Bold values indicate statistical significance at p < 0.05.
Within parameter, mean ± SEM with different superscripts is significantly different (p < 0.05).
SEM, standard error of mean.
Relationship between anaemia status, malaria parasite and temperature
Figure 3 depicts the relationship between anaemia status, malaria parasite density and body temperature among children under 5 years old who were infected with malaria, with or without STH co-infection. Overall, parasite density ranged from approximately −10,000 to 80,000 parasites/µL, with the majority of children clustering around temperatures between 37.5°C and 39.5°C. Among children with malaria only (no STH co-infection), both anaemic and non-anaemic individuals exhibited elevated parasite densities, with anaemic children showing a broader temperature range and higher parasite burdens, especially at temperatures above 39°C. By contrast, children with malaria–STH co-infection showed more concentrated clustering of parasite density and temperature, particularly between 37.5°C and 39°C and at lower parasite densities (around 10,000–20,000 parasites/µL). Anaemic children tended to have higher malaria parasite densities and greater temperature variability (37.5°C–44.3°C) than their non-anaemic counterparts, suggesting an interaction between anaemia and the febrile response to malaria infection. These patterns indicate that co-infection and anaemia status may jointly influence both parasitaemia and febrile presentation among infected children.

Relationship between anaemia status, malaria parasite and temperature of children co-infected with malaria and soil-transmitted helminths.
Impact of malaria–STH co-infection on anaemia and hypochromia stratified by haemoglobin genotype
Figure 4 displays the distribution of anaemia status among the participants, stratified by haemoglobin genotype and co-infection status with malaria and soil-transmitted helminths (STH). In children without co-infection, the highest proportions were observed among those with the AA genotype, with 36.0% non-anaemic and 22.1% anaemic. Among AS genotype children, 20.7% were non-anaemic, and 14.9% were anaemic, while only 5.4% of SS genotype children were anaemic, with no non-anaemic cases observed. Among children with malaria–STH co-infection, the AA genotype group again showed the highest proportions, with 28.9% non-anaemic and 24.4% anaemic. In the AS group, 15.6% were non-anaemic and 24.4% anaemic. For the SS genotype, 2.2% were non-anaemic, and 4.4% were anaemic. Across all genotypes, anaemia was more prevalent among co-infected children, particularly in those with AS and SS genotypes, highlighting a potential synergistic effect of co-infection and genotype on anaemia burden.

Anaemia status in children co-infected with malaria and soil-transmitted helminths by haemoglobin genotype.
Figure 5 presents the distribution of hypochromia among children under 5 years of age stratified by haemoglobin genotype and malaria + STH co-infection status. In the absence of co-infection, hypochromia was most common among children with the AA genotype, with 9.5% exhibiting hypochromia and 48.6% without. In the AS genotype group, 4.1% had hypochromia and 31.5% did not, whereas in the SS genotype group, only 5.4% were non-hypochromic, with no hypochromic cases recorded. In children with malaria + STH co-infection, the prevalence of hypochromia increased markedly across all genotypes. Among those with the AA genotype, 22.2% had hypochromia and 31.1% did not. For the AS genotype, 15.6% were hypochromic, and 24.4% were non-hypochromic. In the SS group, 6.7% were non-hypochromic, and no cases of hypochromia were reported. Overall, the findings indicate that hypochromia is more prevalent among children with malaria + STH co-infection, particularly in those with AA and AS genotypes, suggesting an additive effect of co-infection on red blood cell abnormalities.

Hypochromia status in children co-infected with malaria and soil-transmitted helminths by haemoglobin genotype.
Correlation of haematological parameters with demographic and clinical factors
Table 3 presents a detailed pairwise correlation analysis of haematological and clinical parameters stratified by malaria and malaria–STH co-infection status. Hb, PCV and RBC count showed very strong positive correlations across all groups (r > 0.97, p < 0.0001), confirming their physiological interdependence in reflecting anaemia status. RBC and Hb were nearly perfectly correlated (r = 1.00) in all subgroups. Weak to moderate negative correlations between WBC and RBC/Hb/PCV were observed in malaria-negative and co-infection-negative groups (p ≈ 0.05), suggesting inflammatory responses may be inversely related to red cell indices in uninfected or singly infected children.
Pairwise correlation analysis of haematological parameters by malaria and malaria-soil transmitted helminth co-infections among children in an endemic environment.

Significance level: *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001.
HB, haemoglobin; MCHC, mean corpuscular haemoglobin concentration; MCV, mean corpuscular volume; PCV, packed cell volume; RBC, red blood cell; WBC, white blood cell.
MCV was negatively correlated with Hb and RBC in malaria-positive and co-infection-positive children (r = –0.46 to −0.52, p < 0.001), indicating a possible shift toward macrocytosis and altered red cell morphology during infection. MCHC and MCV demonstrated a strong inverse relationship in all groups (r = −1.00, p < 0.0001), a known haematological pattern reflecting compensatory changes in red cell composition. Platelet counts were negatively correlated with age in all groups, significantly so in malaria-negative (r = –0.23, p = 0.0006) and co-infection-negative children (r = –0.21, p = 0.0013), suggesting an age-related decline. Platelet counts were also significantly correlated with MCV and MCHC in some groups, indicating an interaction between thrombopoiesis and erythropoietic stress. Malaria and STH infections were associated with subtle shifts toward macrocytic anaemia patterns (increased MCV, reduced MCHC) and age-related platelet reductions.
Malaria density showed no statistically significant correlations with any haematological variable, though weak trends were noted with MCV and WBC in some groups (p > 0.05), indicating that parasite load may not directly influence red cell indices in this cohort. Higher body temperature was weak but significantly associated with lower PCV, Hb and RBC in malaria-negative and co-infection-negative groups (p < 0.05), consistent with fever-related suppression of erythropoiesis. However, malaria parasite density showed limited correlation with most haematological markers, suggesting that infection status rather than burden might drive the observed hematologic changes in endemic settings. Age was positively correlated with MCV and negatively with MCHC in malaria-negative and co-infection-negative children (p < 0.0001), consistent with normal hematologic maturation.
Discussion
In this hospital-based study of 269 preschool children, the most frequent finding was mild normocytic anaemia. Mean PCV was 34%, mean haemoglobin concentration was 11 g/dL and red cell indices were normal in size but toward the lower end of the normal concentration range. Similar findings have been reported in healthy children in Ibadan, Nigeria 47 and in Kilifi, Kenya, 48 where low-level malaria transmission and modest nutritional deficiencies were the prevailing conditions. Haematological findings, as seen in Table 1, showed no meaningful differences by sex or age, consistent with an earlier school-based study from southwest Nigeria. 49
Haemoglobin genotype significantly influenced both co-infection prevalence and clinical severity in this cohort. Approximately half of all co-infections of malaria–STH were from children with Hb AS and exhibited higher malaria parasite densities. This observation aligns with the established understanding that while Hb AS provides partial protection against severe malaria, it does not fully reduce parasite density, thereby allowing higher parasite loads in certain individuals.50,51 Despite these elevated parasite densities, Hb AS children maintained better-preserved haemoglobin levels and PCV compared with Hb AA children, suggesting a protective modulation against malaria-associated anaemia.48,50,52,53
Similar findings have been reported in East50,54 and West Africa,53,55 where Hb AS children experienced reduced clinical severity and haematological disturbances despite measurable parasitaemia.55,56 The male predominance among Hb AS co-infected children in our study may further suggest that behavioural or environmental factors, such as increased outdoor activity or exposure to vector-prone environments, amplify infection risks and interact with genetic predisposition. Similar findings from Kilifi, Kenya 48 and rural Ghana 56 have shown that Hb AS carriers tend to exhibit milder haematological disturbances, supporting the notion of a genotype-mediated immune-buffering effect.
On the other hand, fewer children with homozygous sickle cell genotype (Hb SS) presented with elevated clinical features, with the highest frequency of severe anaemia (82.4%). These children had significantly reduced PCV, reduced Hb concentration, and raised leukocyte levels, characteristics of chronic haemolysis, sustained bone marrow activity and persistent inflammation. Similar haematological profiles have been documented among children with sickle cell disease in both Ghana 56 and Nigeria. 57
Malaria and STH as a single infection lowered haemoglobin by about 0.5 g dL-¹ and the RBC count by about 0.5 g dL-¹, but these reductions were insignificant. However, a striking observation in this study was the presence of macrocytosis (increased red cell size/MCV) and hypochromia (reduced haemoglobin concentration/MCHC) in both the single and co-infection groups. These characteristics are commonly associated with reticulocytosis following haemolytic episodes and with functional iron deficiency driven by chronic inflammation. 58 This subtle macrocytic–hypochromic pattern is an important physiological signal and may represent early erythropoietic stress that precedes overt anaemia. Platelet levels were lower in the Plasmodium falciparum-positive children, indicating the mild thrombocytopenia of uncomplicated falciparum malaria. This mild thrombocytopenia is likely due to increased platelet destruction or removal during infection. 59
The mean parasite density of the malaria-positive preschool-age children in this study was just below 9000 µl, within the moderate range commonly reported across African malaria-endemic settings. Similar community-based studies from Kenya and Uganda have documented mean parasite counts of 5000–15,000 parasites/µL in asymptomatic or mildly symptomatic children under five, particularly during transmission peaks.60,61
With respect to age, the group with the highest parasite density was neonates, and the least infected were toddlers. Maternal antibodies protect most infants for the first few months, but those who do become infected tend to present high densities because their own humoral response is premature. 60 By the time children reach toddlerhood, repeated low-level infections have broadened their pool of strain-specific antibodies, which helps keep parasite counts low. As they are near school age, however, greater outdoor activity increases exposure and as our figures show, average densities climb again. Similar age-related patterns have been observed in community-based studies conducted in Kilifi on the Kenyan coast, 48 and in rural villages in The Gambia, 53 where infection rates and immune responses also varied significantly with age.
Notably, in this study, there is a statistically insignificant association between haemoglobin genotype and malaria parasite density, helminth coinfection, anaemia, hypochromasia, fever or seasonal variations. Several larger-scale studies also reported weak or no significant associations between parasite density and these clinical or host variables after adjusting for age. In Ibadan, Nigeria, parasitaemia explained less than 10% of the variation in haemoglobin after controlling for genotype and nutrition, thereby suggesting parasite density does not fully capture the complex nature of anaemia risk. 62 Another Tanzanian investigation found that recovery from malaria-induced anaemia was more strongly correlated with inflammatory markers than with initial parasite density, suggesting that inflammation, rather than parasite density, mediates anaemic responses. 54 Together, these findings suggest that inflammatory and host-related factors may play a more prominent role than parasite burden alone in shaping haematological outcomes in endemic settings, although high parasite densities may still exacerbate disease severity in susceptible individuals.63,64 Similar clinical correlations were also previously documented in a hospital-based study in Enugu, Nigeria 64 and in a Tanzania community study, 54 testifying to the understanding that increased parasite density tends to exacerbate disease severity. Furthermore, the significant genotype-dependent variation in anaemia (Figure 4) clearly demonstrates the genetic determinants of parasite co-infection severity. Children with the Hb SS genotype exhibited the highest prevalence of severe anaemia and hypochromasia, consistent with chronic haemolysis, sustained erythropoietic stress and heightened inflammatory responses likely triggered by prolonged parasitic exposure.57,65
Despite more than 90% of samples being collected during the wet season, no significant seasonal variation was observed in PCV, HB, RBC or malaria parasite density. These findings align with prior work from both Zambia 66 and Tanzania, 67 where seasonal peaks in malaria transmission had minimal impact on haematological indices, especially in areas with widespread use of insecticide-treated nets (ITNs) with coverage rates exceeding 50%. Interestingly, there was a modest rise in total WBC count during the dry season, a trend that presumably indicates an increased prevalence of non-malarial febrile illnesses, such as respiratory or enteric infections, as malaria transmission eases. A similar seasonal pattern has been reported in northern Ghana, where clinical presentations during the dry season tend to shift from malaria toward bacterial and viral infections. 68 The stability of red cell indices and infection rates across seasons suggests that widespread implementation of vector-control measures may be attenuating the typical seasonal fluctuations in malaria burden. 67 The relative stability of red cell indices across seasons may reflect the moderating effects of sustained vector control interventions in the study area. 56
This study observed weak, statistically non-significant associations between malaria parasite density and haematological parameters or body temperature, mirroring findings from community-based studies in Tanzania 54 and Mali, 55 which similarly reported weak correlations. These observations highlight the complexity underlying malaria pathology, suggesting that clinical outcomes such as fever and anaemia depend on multiple factors beyond parasite density alone, including host immunity, nutritional status, genetic predispositions and co-infections.69,70 The clear association between increased parasite density, elevated body temperature and anaemia severity illustrated in Figure 3 aligns with well-established malaria pathophysiology. 54 High parasite densities (>5000 parasites/µL) were consistently accompanied by elevated fevers, indicating that parasite burden directly contributes to inflammatory responses and systemic clinical manifestations. 71
Study strengths and limitations
This study provides important findings, but several limitations should be considered. Due to its cross-sectional design, the study cannot determine causal relationships between infection status, blood parameters and genotype. The majority of samples were collected during the rainy season (May to December), which limits the ability to fully evaluate seasonal trends in infection rates and haematological responses.
The small number of children with the Hb SS genotype may have limited the detection of more subtle genotype-specific effects, although the abnormalities observed were clinically consistent with sickle-cell physiology. Furthermore, several potential confounding variables were not evaluated, including environmental exposures, iron levels, nutritional status and inflammation markers such as C-reactive protein. Differential leukocyte subsets were not examined in this analysis, as the study focused on total WBC count alongside red cell indices central to the study objectives. These factors might have influenced haematological outcomes and could not be accounted for in this analysis. In addition, some background information was obtained through self-reported questionnaires, which may have introduced recall bias. By focusing on children who visited healthcare facilities, the study may have missed cases in those who did not seek care, possibly underestimating the true burden of infection in the community.
A further methodological limitation relates to statistical analysis. Although appropriate parametric tests (t-tests, ANOVA, Pearson correlations) were used, formal non-parametric alternatives were not applied. Visual inspection suggested approximate normality, and sample size supported parametric testing; however, this remains a limitation when interpreting subgroup comparisons.
Despite these limitations, this study has several strengths. It focuses on children under five, a population highly vulnerable to both malaria and STHs, yet often underrepresented in co-infection research. The inclusion of haemoglobin genotype data provides a crucial genetic perspective when interpreting the outcomes. A more thorough understanding of infection severity and clinical relevance was made possible by the combination of genotypic analysis, haematological evaluations and microscopy.
Importantly, the study identified subtle but clinically meaningful red cell abnormalities, particularly the early macrocytic–hypochromic pattern, even when haemoglobin levels remained near normal. This rare finding underscores early erythropoietic stress that may precede overt anaemia and is easily missed by routine Hb-based assessments. The findings’ dependability and possible applicability in other endemic areas are reinforced by the comparatively large sample size and the use of appropriate statistical techniques in the analysis. This study provides valuable insights into targeted intervention design by identifying key factors, such as age, sex and genotype. In addition to addressing a local data gap, the region-specific focus on the Delta South Senatorial District may help in developing health programs tailored to the needs of communities facing comparable issues.
Conclusion
This study provides robust evidence that among children under five in Delta South, Nigeria, STH infections and malaria continue to represent a significant health burden. Both single and co-infections were common, particularly among male children, reflecting the persistent interaction of biological and environmental risk factors in this endemic setting. Although anaemia was generally mild, distinct haematological changes, such as macrocytosis, hypochromasia and elevated WBC counts in vulnerable subgroups, including children with the Hb SS genotype, indicate ongoing erythropoietic stress and inflammation that could worsen during peak transmission seasons or in the absence of timely intervention.
Importantly, these abnormalities were observed even with modest haemoglobin reductions, underscoring the value of red cell indices in identifying early haematological compromise. Genotype-specific vulnerabilities were particularly evident, as children with the Hb SS genotype experienced the most profound anaemia and hypochromasia. These findings support the importance of targeted screening, early risk identification and proactive clinical management. The weak association between parasite density and anaemia severity also suggests that immune and genetic factors may influence clinical outcomes more significantly than parasite burden alone.
From a public health perspective, these results reinforce the need for integrated interventions. Combining seasonal malaria chemoprevention, regular deworming and better nutritional support, such as iron and folate supplementation, is likely to reduce illness in young children, especially in settings where these infections are common. Early haematological assessment may aid timely risk identification before overt anaemia develops, particularly in high-risk children. Locally adapted, evidence-based and multidimensional control interventions remain the most important factor in maximising the health and developmental prospects of children living in endemic settings.
Footnotes
Acknowledgements
The authors sincerely thank the management and staff of the participating health facilities in the Delta South Senatorial District, Nigeria, for their cooperation during data collection. We are also grateful to the parents and guardians who consented to their children’s participation in this study. Technical support provided by Emmanuel Olu-Ero during laboratory procedures and sample analysis is gratefully acknowledged.

