Abstract
Background:
Febrile neutropenia (FN) is the most common and serious adverse event of chemotherapy for solid and hematological neoplasms, with infection as a major complication. FN occurs in 10%–50% of patients with solid tumors and over 80% with hematological malignancies, with mortality rates up to 11%.
Objective:
To characterize bloodstream pathogens in post-chemotherapy FN through a systematic review and meta-analysis of studies published from 2013 to February 13, 2024.
Design:
Systematic review and meta-analysis.
Data sources and methods:
PubMed, Web of Science, Scopus, and Embase were searched using MeSH, Emtree, and keywords. Risk of bias was assessed using the JBI checklist. Random-effects proportions meta-analysis was performed.
Results:
Twenty-two studies (n = 23,319) reported 8665 positive blood cultures: 59% Gram-negative (95% CI: 46.8–67.5), 39.7% Gram-positive (95% CI: 31.3–48.2), and 2.5% fungi (95% CI: 0.9–4.1). Random-effects meta-analysis showed high heterogeneity (I2 = 98.92%, p < 0.01). Meta-regression by sample size, economic development, and risk of bias did not explain this variability.
Conclusion:
Gram-negative pathogens slightly predominate over Gram-positives in bloodstream infections among post-chemotherapy FN patients.
Trial registration:
PROSPERO (CRD42023472191).
Plain language summary
The study highlights the significant prevalence of pathogens in the bloodstream of patients experiencing febrile neutropenia (FN) after chemotherapy. It identified a total of 21,485 patients from 23 studies, revealing that 46.1% of positive blood cultures were Gram-negative bacteria, while Gram-positive bacteria accounted for 45.6%. Additionally, fungi represented only 1.9% of the pathogens. The systematic review and meta-analysis showed considerable heterogeneity among the studies, indicated by an I² of 98.92%. Despite evaluating various factors such as sample size, economic development, and risk of bias, none significantly influenced this heterogeneity. In conclusion, the findings suggest that Gram-negative bacteria are slightly more prevalent than Gram-positives in cases of FN following chemotherapy, underscoring the need for targeted antimicrobial strategies in this vulnerable patient population.
Introduction
Cancer remains a leading global cause of death, with nearly 10 million deaths and 20 million new cases in 2020. 1 Chemotherapy is central to treating both solid and hematologic malignancies.2,3 Febrile neutropenia (FN) is one of the most common and severe adverse effects of chemotherapy. FN is defined as a single oral temperature measurement of ⩾38.3°C (101°F) or ⩾38.0°C (100.4°F) sustained for more than 1 h, in patients with an absolute neutrophil count (ANC) of <500 cells/μL or expected to decrease to <500 cells/μL within 48 h.4,5 FN often necessitates hospitalization and broad-spectrum antibiotics,4,5 and is associated with increased mortality, healthcare costs, and delays or modifications in cancer treatment.4,5
FN occurs in 10%–50% of patients with solid tumors and over 80% of those with hematologic malignancies receiving chemotherapy. 6 It significantly increases the risk of serious infections, particularly bloodstream infections (BSIs), which contribute substantially to morbidity and mortality in this population. Patients with FN have an estimated 15% higher risk of death compared to those without FN. 7
The epidemiology of BSIs in FN patients has shifted over time. 9 While Gram-positive organisms predominated in the late 20th century, the past two decades have seen a resurgence of Gram-negative pathogens, likely driven by evolving chemotherapy regimens, antimicrobial use, and invasive procedures such as central venous catheterization.
Despite advances in FN management and prophylactic strategies, current data on BSI pathogens in post-chemotherapy FN remain fragmented, varying across regions and time periods. No clear consensus exists on the predominant microbial etiology, complicating efforts to standardize empiric therapy and prevention protocols. Discrepancies in trends—such as the resurgence of Gram-negatives—further hinder the development of universal treatment guidelines.
This study aims to systematically review and synthesize evidence on the distribution and characteristics of the pathogens causing BSI in adult patients with post-chemotherapy FN. Understanding these trends is crucial to inform empirical treatment, guide prophylaxis, and ultimately reduce FN-related morbidity, mortality, and healthcare burden in cancer patients.
Materials and methods
This study adheres to the Preferred Statement Reporting Items for Systematic Reviews and Meta-Analysis (PRISMA 2020) 10 and was previously registered with PROSPERO (ID number: CRD42023472191).
Study design
Systematic Review and meta-analysis were conducted and reported in accordance with the PRISMA guidelines. 10
Eligibility criteria
Primary studies on bacteremia in post-chemotherapy neutropenic patients, aged 18 years, published between 2013 and February 13, 2024, without language restrictions, were included. Exclusion criteria comprised studies involving COVID-19, pediatric or pregnant populations, non-chemotherapy-related febrile neutropenia, as well as review articles, letters to the editor, case reports, and conference abstracts.
Sources of information
A comprehensive search was conducted in PubMed, Web of Science, Scopus, and Embase using MeSH and Emtree terms (e.g., Neoplasms, Hematologic Neoplasms, Bloodstream Infection, and Prevalence), as well as free-text terms (e.g., cancer), combined using Boolean operators (AND, OR). The search strategy was tailored to each database (see Supplemental Material).
Eligibility assessment
Eligibility assessment was conducted independently and blinded by two reviewer pairs (FC-PG and FG-HA). Discrepancies were resolved by consensus. Article selection was finalized on February 13, 2024.
Data extraction
Data was extracted using a standardized Excel spreadsheet (Microsoft Excel, version 15.24; 2016). Two authors (FC and PG) extracted data from the included studies, and three others (FG, YYL, and HA) verified the data. Disagreements between reviewers were resolved by consensus.
Variables
The primary outcome was the prevalence of bloodstream infections. Secondary variables included tumor type, bacteria classification (Gram-positive or Gram-negative), specific pathogens, antimicrobial resistance, infection management, and the occurrence of sepsis or septic shock. Additional extracted data included study design, year of publication, country, sample size, FN diagnostic criteria, study period, number of centers, and funding source. Definitions of febrile neutropenia used by each author can be seen in Tables 1 and 2.
Characteristics of the studies included in the systematic review.

ANC, Absolute Neutrophil Count; N/R, Not Report.
Characteristics of included studies on febrile neutropenia treatment, infection management, and isolated pathogens.
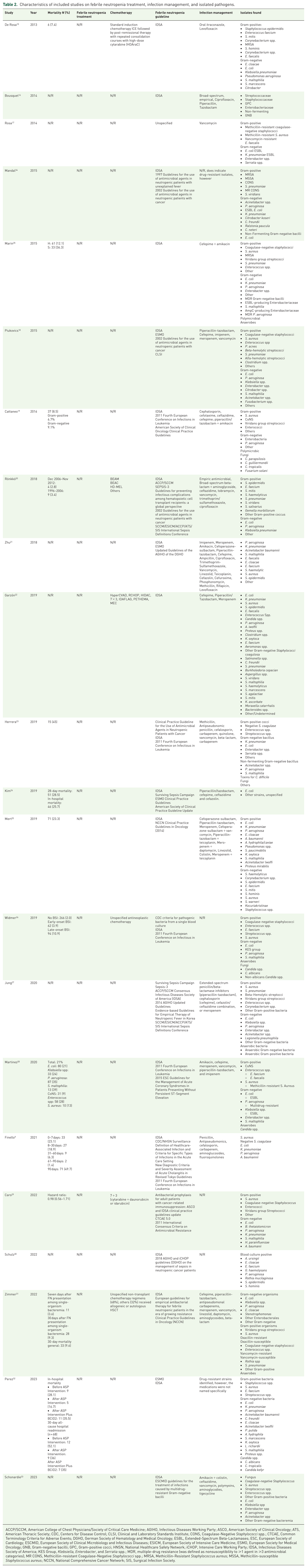
ACCP/SCCM, American College of Chest Physicians/Society of Critical Care Medicine; AGIHO, Infectious Diseases Working Party; ASCO, American Society of Clinical Oncology; ATS, American Thoracic Society; CDC, Centers for Disease Control; CLSI, Clinical and Laboratory Standards Institute; CONS, Coagulase-Negative Staphylococci spp.; CTCAE, Common Terminology Criteria for Adverse Events; DGHO, German Society of Hematology and Medical Oncology; ESBL, Extended-Spectrum Beta-Lactamases; ESC, European Society of Cardiology; ESCMID, European Society of Clinical Microbiology and Infectious Diseases; ESICM, European Society of Intensive Care Medicine; ESMO, European Society for Medical Oncology; GNB, Gram-negative bacilli; GPC, Gram-positive cocci; HNSN, National Healthcare Safety Network; iCHOP, Intensive Care Working Party; IDSA, Infectioius Diseases Society of America; KES Group, Klebsiella, Enterobacter, and Serratia spp.; MDR, multiple-drug resistance (was defined as nonsusceptibility to ⩾1 agent in ⩾3 antimicrobial categories); MR CONS, Methicillin-resistant Coagulase-Negative Staphylococci spp.; MRSA, Methicillin-Resistant Staphylococcus aureus; MSSA, Methicillin-susceptible Staphylococcus aureus; NCCN, National Comprehensive Cancer Network; SIS, Surgical Infection Society.
Study risk of bias assessment
The risk of bias was assessed independently by two reviewers (AF-C, PG) through the Joanna Briggs Institute (JBI) Critical Appraisal Checklist for studies reporting prevalence data.11–13 This tool assesses nine domains where final scores were determined based on the percentage of affirmative responses. Studies were classified as high risk of bias (score of 49% or less, leading to exclusion of the study), moderate risk (score ranging from 50% to 69%), or low risk (score above 70%). Discrepancies were resolved by consensus.
Risk of bias summaries were generated using the ROBVIS R package v.0.3.0.900. 14
Data synthesis
Descriptive statistics (percentages, frequencies, and means) were used to summarize the data. A random-effects meta-analysis was performed using the Freeman-Tukey reverse transformation model. Studies with a high risk of bias were excluded. Heterogeneity was assessed using Cochran’s Q test (significance set at p < 0.05) and the I2 statistic. Univariate meta-regression was performed using the covariates: risk of bias, sample size, and Country Development Status.
Sensitivity analyses included subgroup analysis assuming I2 = 10%, leave-one-out meta-analysis, and standard error adjustments using the Sidik-Jonkman and truncated Knapp-Hartung methods. All analyses were performed using Stata 18 statistical software (StataCorp LLC, College Station, TX, USA).
Reporting bias assessment
To assess small-study effects, funnel plots were generated, and Egger’s test was performed for studies reporting Gram-negative, Gram-positive, and fungal infections. A p < 0.05 was considered statistically significant. Trim-and-fill analysis was performed to determine if imputed studies affected the effect size.
Assessment of certainty
It was not considered.
Ethics
Secondary evidence does not require the authorization of an Institutional Review Board; thus, ethics approval was not considered.
Results
A total of 7630 articles were identified across the databases. After removing duplicates and screening titles and abstracts, 266 articles were retained for full-text review. Of these, only 22 studies8,9,15–23,24–34 met the inclusion criteria and were the basis of the qualitative and quantitative analysis (Figure 1).

PRISMA flow of selection and exclusion of the information.
Characteristics of studies
A total of 22 studies were included for the assessment.8,9,15–23,24–34 Table 1 describes the author, year of publication, type of study, country, study period, patient population, number of centers, and funding status.
Based on publication year, 14 (63.6%)15–23,24–26,34,35 studies were published before 2020 and 8 (36.4%)9,27–33 2020 and 2023. Geographically, eight studies (36.4%) were from Europe,8,15,16,18–20,27,29 6 (27.3%) from South America,9,17,22,23,32,33 5 (22.7%) from Asia,21,24–25,27,34 equivalent to 22.7%; 2 (9.1%) from North America30,32 and 1 (4.5%) from Oceania. 31 Tables 1 and 2 describe the characteristics of patients with chemotherapy-induced FN with positive blood culture.
Regarding study design, 12 studies (54.5%) were retrospective,9,15,16,18,21–23,24,25,27,29,33 8 (36.4%) were prospective,8,17,19,20,26,28,30,34 1 (4.5%) was a prospective-retrospective comparison, and 1 (4.5%) was a quasi-experimental pre-post study. 20 Most studies (81.8%, n = 18) were single center,15–18,20–23,24,25,27,29,30,32–35 while 5 (22.7%) were multicenter9,19,26,28,31 (22.7%; Table 1).
Febrile neutropenia definitions were based on guidelines and definitions explicitly referenced by the authors. Four studies (18.2%)16,22,35 used the Infectious Diseases Society of America (IDSA) definition alone. Sixteen studies (72.7%)9,18–21,23,24–28,30–34 referenced multiple sources additional to the IDSA guidelines: the 1997 Guidelines for the Use of Antimicrobial Agents in Neutropenic Patients with Unexplained Fever, the 2002 Guidelines for the Use of Antimicrobial Agents in Neutropenic Patients with Cancer, the European Society for Medical Oncology (ESMO), the fourth European Conference on Infections in Leukemia (2011), the American Society of Clinical Oncology (ASCO) Clinical Practice Guidelines, and guidelines for preventing infectious complications in hematopoietic cell transplant recipients. One study (4.5%) 29 did not use the IDSA guidelines, and one (4.5%) 17 did not specify a guideline. Definitions are detailed in Table 1.
Patients with septic shock were reported in three studies (13.6%).8,20,28 Of these, one study (4.5%) 20 used the American College of Chest Physicians/Society of Critical Care Medicine (ACCP/SCCM) definition. The remaining two studies (9.1%)8,28 did not cite a specific sepsis guideline; one article 8 offered a self-defined criterion, and the other 28 provided no definition. Table 2 describes Characteristics of Included Studies on Febrile Neutropenia Treatment, Infection Management, and Table 3 reports the classification of pathogens isolated from positive blood cultures.
Characteristics of patients with chemotherapy-induced FN with positive blood cultures.

APACHE, Acute Physiology and Chronic Health Evaluation; CCI, Charlson Comorbidity Index; ECOG, Eastern Cooperative Oncology Group; HIV, human immunodeficiency virus; HP, positive blood culture; ICU, Intensive Care Unit; IQR, Interquartile Range; MASCC, Risk identification index in neoplastic patients with febrile neutropenia; MDR, Multidrug Resistant; N/R, Not reported; PAA, Antimicrobial stewardship program; PITT, Bacteremia score; SD, Standard Deviation; SOFA, Sepsis-related organ failure assessment.
Risk of bias
In the risk of bias assessment, two studies (9.1%) presented moderate risk, and twenty studies (90.9%) were classified as low risk (see Supplemental Materials).
Characteristics of patients with post-chemotherapy febrile neutropenia with microorganism identification in blood cultures
A total of 23,319 patients with chemotherapy-induced FN were included, with 8,665 positive blood cultures. The mean age was 48.5 years ± 7.4 years, and the proportion of female patients ranged from 37.8% to 56% across studies.
In 17 studies,15,17–22,25–26,29–35 all patients (n = 17,960) had only hematologic malignancies. The remaining six studies reported a mixed population, with hematologic malignancy prevalence ranging from 19% to 89% and solid tumors (n = 456) ranging from 11% to 81%. Severe neutropenia (SNP) was reported in 100% of the patients in 10 studies (n = 3609).9,15,17,22,23,27,28,31,33,35 Three studies16,21,24 reported SNP in 30.2%–52.1% of cases, and one study 16 reported moderate neutropenia in 58.6% of patients.
Neutropenia lasted a median of 2 days (IQR 2–4), up to a maximum of 14 days (IQR 13–19). Notably, Caro et al. 29 reported a median duration of 23 days (IQR 15–38) with fluoroquinolone prophylaxis versus 30 days (IQR 21–51) without.
ICU admission was required in 4.2% to 54.3% of patients.20,26,32,33,35 Septic shock occurred in 10.2% to 17%,20,28,35 and vasopressor use ranged from 9.6% to 28.6% in two studies.23,35 Central venous catheter use was reported in seven studies9,23,24,27,31,33,35 with a prevalence of 22.9% to 77.9%. Mechanical ventilation was required in 3% to 36.1% across four studies.20,24,31,33 Reported mortality ranged from 2% to 70% 21 (Table 4).
Classification by groups of pathogens isolated in positive blood cultures.
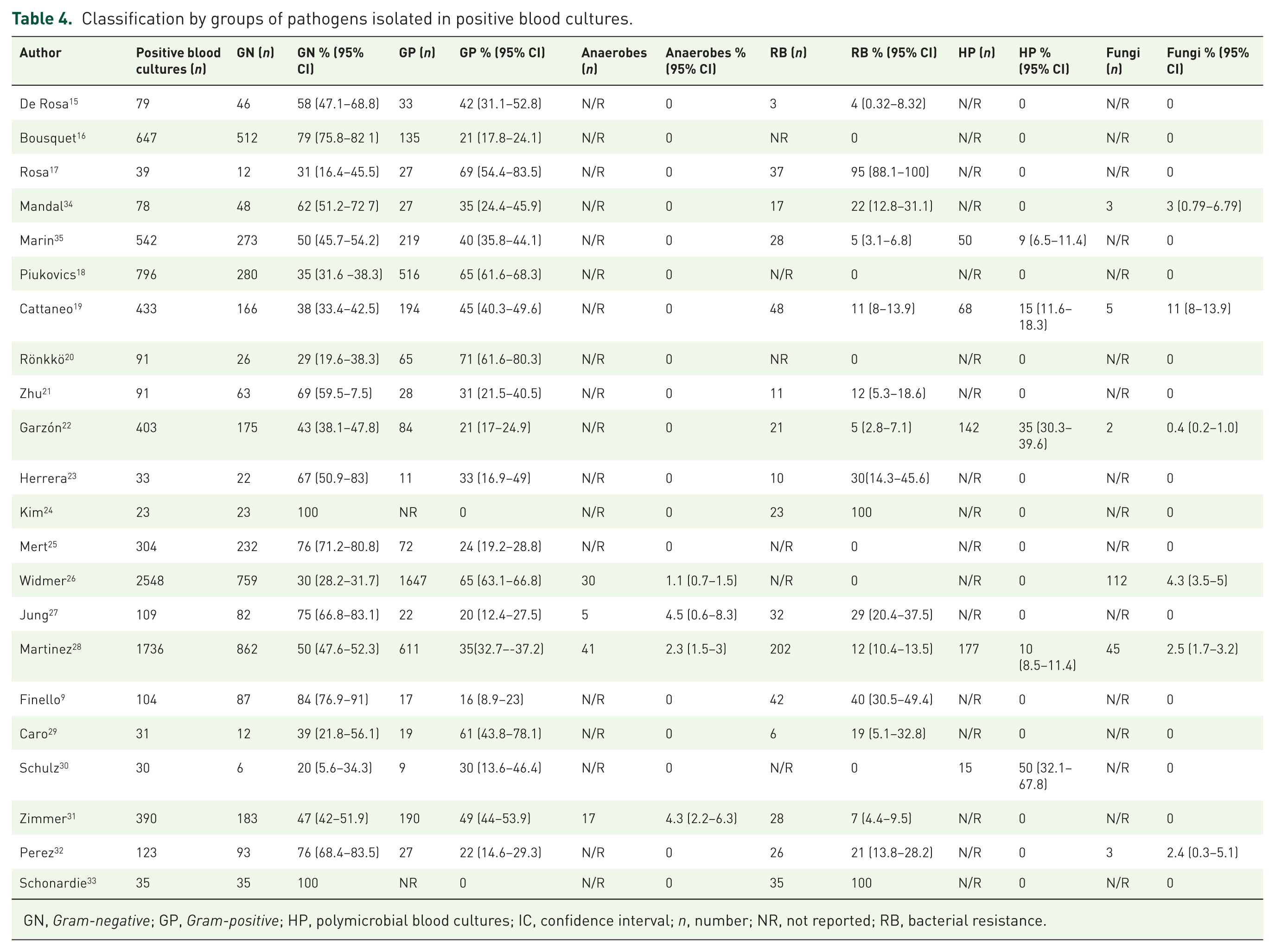
GN, Gram-negative; GP, Gram-positive; HP, polymicrobial blood cultures; IC, confidence interval; n, number; NR, not reported; RB, bacterial resistance.
ICU admission was required in 4.2% to 54.3% of patients.20,26,32,33,35 Septic shock occurred in 10.2% to mortality ranged from 2% to 70% 21 (Table 4).
Microorganisms identified in blood cultures in a patient with chemotherapy-induced febrile neutropenia
Table 4 summarizes the classification of microorganisms from 8,665 positive blood cultures, Gram-negative bacteria were the most prevalent (46.1%, 95% CI: 45.1–47.2), followed by Gram-positive (42.4%, 95% CI: 41.4–43.5), resistant bacteria (13.4%, 95% CI: 12.4–14.5), and fungi (3.2%, 95% CI: 2.8–3.7).
Among Gram-negatives, the most common were E. coli (46.2%, 95% CI: 44.6–47.8), P. aeruginosa (18.7%, 95% CI: 17.5–20.0), and K. pneumoniae (18.5%, 95% CI: 17.3–19.8) (See Supplemental Material). For Gram-positives, coagulase-negative Staphylococcus with (52.3%, 95% CI: 50.7–53.9), S. epidermidis (31.8%, 95% CI: 27.1–37.0), Enterococcus spp. (14.7%, 95% CI: 13.4–16.0), and S. aureus (9.2%, 95% CI: 8.3–10.1) (see Supplemental Material).
Bacterial resistance was reported in 572 cultures (13.4%), ranging from 3.8% to 100%. Of these, 74.1% (95% CI: 70.1–77.7) were Gram-negative bacteria, while 37.0% (95% CI: 32.5–41.7) were Gram-positive bacteria. The most prominent bacteria were: ESBL-producing E. coli (40.4%, 95% CI: 35.7–45.2), ESBL-producing Klebsiella spp. (15.2%, 95% CI: 11.9–19.3), MDR P. aeruginosa (22.2%, 95% CI: 18.3–26.7), methicillin-resistant coagulase-negative Staphylococcus (47.9%, 95% CI: 39.9–56.0), and MRSA (16.1%, 95% CI: 12.9–20.0) (See Supplemental Material).
Fungal isolations were found in six studies with a prevalence of 3.2% (95% CI: 2.8–3.7). The most frequent were Candida spp. (63.8%, 95% CI: 56.1–70.8), Candida Non-albicans (36.7%, 95% CI: 28.6–45.6) and C. Albicans (14.4%, 95% CI: 9.2–21.9) (see Supplemental Material). In a subgroup analysis, Gram-negative bacteria accounted for 59% (95% CI: 48–70) of bloodstream infections in developed countries (see Supplemental Material). Figures 2 and 3 present the analysis of the prevalence of Gram-positive and Gram-negative bacteria, including subgroup analyses stratified by risk of bias.

Forest plot of the prevalence of Gram-negative bacteria and subgroup analysis by risk of bias.
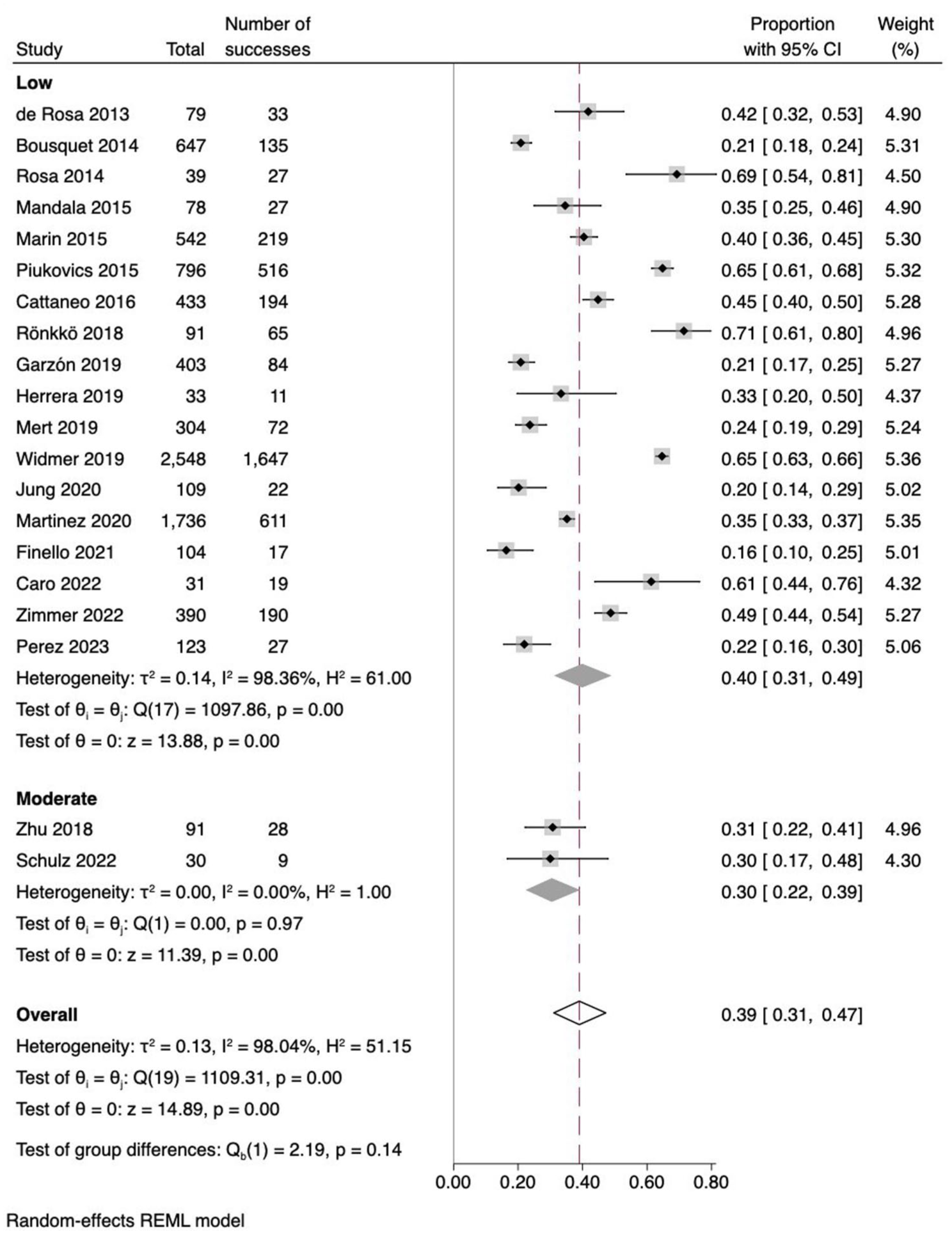
Forest plot of the prevalence of Gram-positive bacteria and subgroup analysis by risk of bias.
In developed countries, fungal pathogens accounted for 2% (95% CI: 1–4) of bloodstream infections (see Figure 4). Risk of bias stratification was not conducted, as all included studies were assessed as low risk.

Forest plot of fungal infection prevalence by World Bank Country Development status.
Source of heterogeneity assessment
In the univariate meta-regression, no significant results were obtained with the covariates: number of participants, risk of bias, and country development status.
Reporting bias
Reporting bias was assessed in studies reporting Gram-negative, Gram-positive, and fungal infections. Egger’s test showed p-values of 0.17, 0.80, and 0.94, respectively. Although funnel plots showed asymmetry (see Supplemental Materials), the Trim-and-Fill analysis did not impute any missing studies.
Discussion
Cancer patients—especially those with chemotherapy-induced acute leukemia—commonly experience profound and prolonged neutropenia, placing them at high risk for bacteremia and associated mortality. This is the first systematic review summarizing the distribution and characteristics of BSIs in FN patients post-chemotherapy. The primary finding is the high prevalence of both Gram-negative and Gram-positive bacteria and low prevalence of fungal infections. 33
Since 2000, infections by Gram-negative bacilli have increased, consistent with our finding that they account for 59% of BSIs. Contributing factors may include long-term catheter use, chemotherapy-induced mucositis, and widespread fluoroquinolone prophylaxis. 36 E. coli, K. pneumoniae, and other Enterobacteriaceae were the most common Gram-negative bacteria.
Gram-positive bacteria were the second most prevalent group. Literature attributes their infections largely to catheters and skin entry points. 37 Our results align with previous studies,36,38–40 identifying coagulase-negative Staphylococcus (52.3%), S. epidermidis (31.8%), and Enterococcus spp. (14.7%), and S. aureus (9.2%).
Gram-negative infections were more frequent in underdeveloped countries than in developed nations.37,41 This is consistent with findings reported in Latin America, where a BSI prevalence of 38.7% was documented during the 2019–2020 period, with Gram-negative bacteria accounting for 86% of cases. The most frequently identified bacteria were K. pneumoniae, E. coli, and P. aeruginosa. 39 Similarly, a hospital in China reported 62.5% Gram-negative and 33.1% Gram-positive infections from 2012 to 2019. 38 This trend continued in Latin America and Korea from 2020 to 2024, although Gram-positive prevalence is rising globally.
Fungal infections were less common (2%), potentially due to early empirical antifungal treatment in high-risk patients. 41 Diagnostic challenges may also delay detection.23,44
Pathogen distribution varies widely across regions, 40 emphasizing the importance of local epidemiology in guiding empirical antibiotic therapy. 41 While bacterial infections remain the primary concern, ongoing surveillance and improved diagnostics are essential for timely management in FN patients.
In infections produced by Gram-positive and Gram-negative organisms, universal quinolone prophylaxis is not recommended for low-risk patients and should be evaluated on a case-by-case basis in high-risk patients. Active monitoring is crucial to detect resistance emergence of resistance. 43
Several limitations of our study should be considered. First, most included studies were conducted in a single center, which introduces the possibility of selection and observation biases. Second, not all the studies included report bacterial resistance of the pathogens, which prevents a more complete representation of BSIs caused by resistant microorganisms in this group of patients. Third, patients with and without a central venous catheter as a risk factor for the development of BSI were not compared. Finally, information on predictors and mortality associated with BSI is limited. Despite this, our study provides relevant information on the distribution and characteristics of bloodstream infections in chemotherapy-induced febrile neutropenic patients. Also, there is an absence of studies in regions such as Africa and the Middle East.
Conclusion
Cancer patients with post-chemotherapy febrile neutropenia are at high risk for severe infections and mortality. Our analysis revealed a high predominance of Gram-negative (57%) and Gram-positive (39%) bacteria, where E. Coli and coagulase-negative Staphylococcus as the most common pathogens. Bacterial resistance was observed in 31% of cases, while fungal infections were rare (0.2%).
These findings highlight the importance of ongoing epidemiological surveillance and individualized clinical management to improve outcomes in this high-risk population.
Supplemental Material
sj-docx-2-tai-10.1177_20499361251376123 – Supplemental material for Microbiological characterization of bacteremia in patients with chemotherapy-induced febrile neutropenia: systematic review and meta-analysis
Supplemental material, sj-docx-2-tai-10.1177_20499361251376123 for Microbiological characterization of bacteremia in patients with chemotherapy-induced febrile neutropenia: systematic review and meta-analysis by Alvarez Franklin Correa, Paola Guasti, Luis Fuenmayor-González, Harold Alexander-León, Yunqi Yu Liu, Johana Elizabeth Salgado-Apunte, Jorge González Grijalva, Hernan Sánchez, Nayely García-Méndez and María Fernanda García-Aguilera in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-pdf-1-tai-10.1177_20499361251376123 – Supplemental material for Microbiological characterization of bacteremia in patients with chemotherapy-induced febrile neutropenia: systematic review and meta-analysis
Supplemental material, sj-pdf-1-tai-10.1177_20499361251376123 for Microbiological characterization of bacteremia in patients with chemotherapy-induced febrile neutropenia: systematic review and meta-analysis by Alvarez Franklin Correa, Paola Guasti, Luis Fuenmayor-González, Harold Alexander-León, Yunqi Yu Liu, Johana Elizabeth Salgado-Apunte, Jorge González Grijalva, Hernan Sánchez, Nayely García-Méndez and María Fernanda García-Aguilera in Therapeutic Advances in Infectious Disease
Footnotes
Appendix
Acknowledgements
We would like to express our profound gratitude to the Department of Research SOLCA, NUCLEO of Quito, for their invaluable support in conducting this study. In addition, we would like to express our profound gratitude to Dr. Fabricio Picoita, whose expert guidance throughout the research process was instrumental in the study’s success. In addition, we wish to acknowledge the assistance of Universidad Central del Ecuador and the Consejo de Postgrado Rodrigo Yépez for providing the essential resources and facilities that made this study possible.
Declarations
ORCID iDs
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
