Abstract
Chikungunya virus (CHIKV) has emerged as a serious candidate for “Disease X”—the name for an unknown agent that could cause a global pandemic. This paper searches for the unique traits of CHIKV that match that designation. Critical mutations like E1-A226V and E2 L210Q have driven CHIKV’s rapid adaptability and its transmission from Aedes mosquitoes to over 110 countries around the world. Public health impact is amped because the virus can cause debilitating diseases including chronic arthritis and severe neonatal complications. Populations remain vulnerable despite a recently approved vaccine, as there is little distribution of it and no treatments for the virus. By underscoring urbanization, climate change, and global travel as ecologic and genetic factors that enable the emergence and persistence of CHIKV, this paper emphasizes that these critical enablers of CHIKV need to be addressed both in the context of host range and transmission potential. However, phylogenetic studies and surveillance data for its capacity to sustain transmission cycles show how important it is to be included in improved global health strategies. Improved early detection, improved vector control, equitable vaccine distribution, and greater international collaboration are critical to reduce the pandemic potential of CHIKV. This paper aims to explore the genetic adaptations of CHIKV that have driven its increased transmission and expanded geographic spread. It examines how key mutations enable the virus to adapt to different mosquito vectors, contributing to its pandemic potential. The paper also assesses the epidemiological and environmental factors influencing CHIKV’s emergence and persistence, alongside the public health challenges posed by limited vaccine availability and treatment options. Finally, it highlights prevention strategies and the importance of global preparedness to reduce the risk of widespread outbreaks.
Plain language summary
Chikungunya virus (CHIKV) is a strong contender for ‘Disease X,’ a term used for an unknown disease that could lead to a global pandemic. Genetic changes, such as the E1-A226V and E2-L210Q mutations, have made the virus highly adaptable and capable of spreading through Aedes mosquitoes to over 110 countries. The virus poses a serious public health risk, causing severe illnesses like chronic arthritis and complications in newborns. Despite the recent approval of a vaccine, its limited availability and the absence of treatments leave many people unprotected. Urbanization, climate change, and international travel are major factors that help the virus spread and persist. Studies on its genetic adaptability and transmission cycles highlight the need for global attention. To reduce the risk of a pandemic, we must improve early detection, strengthen mosquito control, ensure fair vaccine access, and promote international cooperation. This research paper investigates the distinct characteristics of Chikungunya virus (CHIKV), focusing on its global epidemiology, genetic mutations, transmission dynamics, pathogenesis, clinical manifestations, and current preventive strategies and management approaches. Additionally, it evaluates CHIKV as a potential candidate for causing a future global pandemic.
Background
The World Health Organization coined a term to name an unknown pathogen that could cause a severe international epidemic or pandemic: “Disease X.” It is not indicative of the eventual specific threat, but rather a placeholder for an as yet identified future pandemic and underscores the need for good preparedness. In 2018, the idea came to be, when it was listed on the WHO’s Blueprint list of priority disease in urgent need of research and development.1,2 Deforestation and enhanced human–animal interactions are predicted to lead to increased opportunities for spillovers from animals to humans, also known as zoonotic spillovers, and these are a staple of the next pandemic, scientists say. It has been implicated before in past pandemics, including SARS-CoV2 and Ebola.2,3 The most likely causative agents of Disease X are members of families of known pandemic potential viruses, including coronaviruses, filoviruses (e.g., Ebola), and paramyxoviruses (e.g., Nipah). As these pathogens are additionally respiratory, have high mutation rates, and can rapidly spread from human to human, they are particularly concerning.2,4 Ahead of the curve, researchers and global health agencies are ramping up advanced surveillance and genetic characterization of high-risk pathogens while at the same time engaging in proactive development of adaptable medical countermeasures such as vaccines and diagnostics. The Coalition for Epidemic Preparedness Innovations’ 100 Days Mission was launched to develop pandemic vaccines within 100 days or three months after disease invasion is detected.1,4
Chikungunya virus (CHIKV) is an arbovirus responsible for primary transmission by Aedes aegypti and Aedes albopictus mosquitoes. The meaning of the word Chikungunya is “Bent Over In Pain”-taken from the Makonde language. It refers to the debilitating joint pain, which is a feature of the disease. Other symptoms can include fever and joint pain, rash, headache, muscle pain, fatigue, and joint pain that can last months to even years. While rarely fatal, chikungunya can give rise to profound complications, especially in high risk populations, including those aged 65 years and over, infants, and those with comorbidities.5–7 Previous to the current outbreak of the CHIKV, it has historically been confined to (and is continuously found on) a handful of locations in Africa and Asia, but has since expanded to over 110 countries across multiple continents. Its geographical spread, powered in part by intensifying global travel and urbanization as well as climate change, is a cause for concern for large scale outbreak. A vaccine has been recently made available but is only available for select at risk populations.5,7 Chikungunya demonstrates a number of “Disease X” characteristics. Because it can cause widespread outbreaks and lacks cross immunity to related viruses such as dengue or zika and depends on efficient mosquito vectors, it is a serious contender for a future pandemic. The elevated risk of increased transmissibility or severity further elevates its evolution and potential genetic adaptation, which, in turn, requires vigilant monitoring and proactive public health measures.5,7
Epidemiology
CHIKV, arbovirus primarily transmitted by Aedes aegypti and Aedes albopictus, exhibits unique epidemiologic patterns related to climatic factors, urbanization, and vector density. It has spread and resulted in large outbreaks worldwide. High temperatures (>25°C), which accelerate mosquito development and reduce their lifespan to around 14 days, are critical for sustaining transmission cycles. The extrinsic incubation period (EIC)—the time required for the virus to become transmissible inside the mosquito—ranges from 8 to 14 days, making temperature and mosquito survival tightly linked to outbreak potential. CHIKV has resulted in large-scale outbreaks across continents, particularly where environmental and social factors favor mosquito proliferation.
Brazil
In 2024, Brazil accounted for 84.8% of the Americas’ cumulative total, with 402,584 cases. However, since its 2014 introduction, Brazil has suffered annual outbreaks, with the northeast being hardest hit. Factors contributing to the difference are a high vector density, a tropical climate favorable to year-round transmission, and inadequate vector control measures. Nevertheless, spatial heterogeneity remains, determined by local immunity, differential socioeconomic conditions, and regional climate.8,9 A temporal trend of annual CHIKV cases in Brazil is illustrated in Figure 1. 10
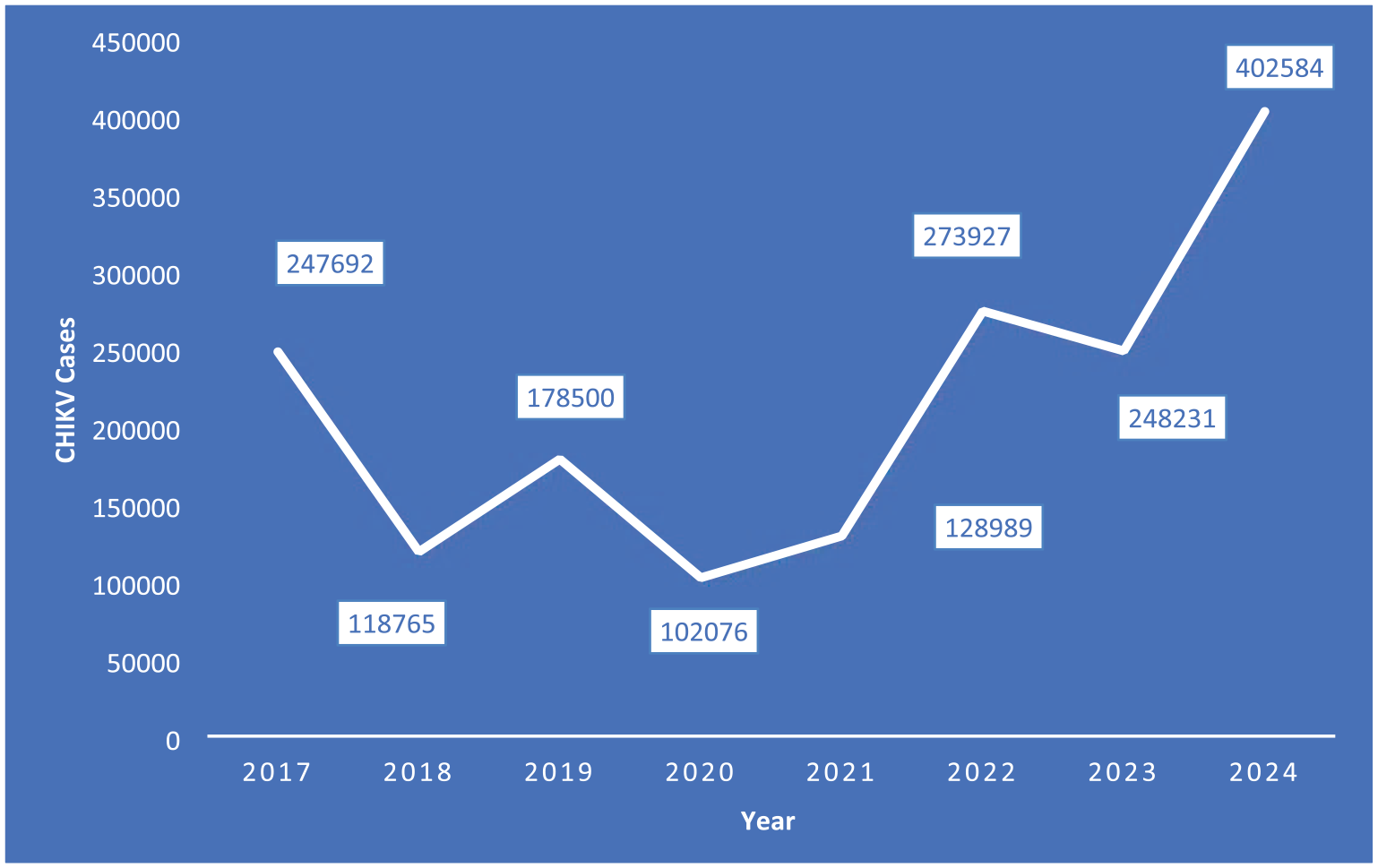
Temporal trend of CHIKV cases in Brazil which has experienced a significant outbreak in recent years.
India
In 2024, India reported 69,544 cases, of which a major part was from Karnataka and Maharashtra. A total of 31.6% of the cases were from Karnataka. Although these outbreaks season with the monsoon, urban waterlogging and poor waste management makes it worse. The broad geographic reach of chikungunya in India is illustrated by the fact that substantial case load was also seen in Maharashtra and Kerala.11,12 A Map of India showing the severity of cases confirmed in 2024 based on states is illustrated in Figure 2.12,13

Heatmap of India showing confirmed cases in 2024 of chikungunya (CHIKV) based on states.
A temportal trend of CHIKV cases in the two most affected states in India—Maharashtra and Karnataka—is illustrated in Figure 312,13

Temporal trend of confirmed chikungunya (CHIKV) cases in Maharashtra and Karnataka from 2018 to 2024.
Pakistan
In 2024, Pakistan reported 5706 cases, with the biggest outbreaks coinciding with monsoon seasons when vector breeding is at its highest. Sindh and Punjab provinces with high urban density and the insufficiency of mosquito control measures were hotspots. Although India has lower case numbers, surveillance gaps and underreporting very likely underestimate the true burden of disease. 14
Malaysia
In 2024, Malaysia had 80 cases and only localized outbreaks during rainy season. The larger epidemics have been prevented by effective vector control measures. But, post flood mosquito breeding is still a concern. 15
The Americas: beyond Brazil
Significant outbreaks had been reported in Paraguay (3104 cases), Argentina (768), and Bolivia (451) and other countries in the Americas. They together cover less area than Brazil but reveal circumscribed localization of chikungunya within these nations.8,16
Africa and Southeast Asia
While lower transmission levels than the rest of the regions, Senegal reported nine cases in Africa in 2024. 17 In Southeast Asia, the Maldives had 240 cases by May 2024 attributed to intermittent rains and subsequent vector proliferation. 18
Trends
Outbreaks occur in periods of inundating tropical rainy season and urban centers appear disproportionately affected because of resident vector density and environmental conditions. Vectors such as Aedes aegypti continue to expand their geographical range and thus threaten non endemically regions in response to climate change.9,19
Genetic mutation
As a single-stranded RNA virus with very high mutation rate, this virus can rapidly adapt to new vectors and hosts.20,21 Its genome consists of three major lineages: East/Central/South African (ECSA), Asian and the West African lineage. CHIKV’s epidemic potential has been mediated in part by adaptive mutations in the envelope proteins E1 and E2.9,22 Vectors and vertebrate hosts show enhanced infection and pathogenicity upon viral mutations like E1-M88L or E1-V156A. 20 CHIKV has a rapid rate of genetic change, with an estimation of about 5–10 mutations/year for its genome. 9 First discovered in Aedes albopictus mosquitoes during the 2005–2006 Indian Ocean outbreak, the E1-A226V mutation enhanced infectivity of CHIKV in these insects by a factor as large as 5. Previously considered a secondary vector, this mutation enhanced midgut infection and dissemination in this mosquito vector through its alteration of viral dependence on cholesterol.22,23 About 266,000 human cases of this mutation were linked to the large 2005–2006 epidemic on Reunion Island. This mutation increased the virus’s infectivity and dissemination in the laboratory settings in Aedes albopictus significantly compared with strains lacking the mutation. 23 Additional fitness advantages in Aedes albopictus were further obtained by subsequent CHIKV mutations, such as E2-L210Q, optimizing its adaptation to this vector.22,24 A 2022 study identified additional mutations in Brazilian ECSA-lineage strains, including changes in the E1 and nsP3 regions. 25 These mutations may contribute to viral persistence in populations and resistance to existing vector control strategies. Evolutionary flexibility of CHIKV in response to environmental and ecological pressures is illustrated by the mutations E1-A226V and E2-L210Q. Efficient transmission cycles were established in areas of high Aedes albopictus density, through these sequential adaptations. 22 Many of the Indian isolates from different locations had novel mutations like E1 patriarchal S295 rout, E1 pivotal P294 rout, and E1 virgin V322 rout, indicating the region adaptability and enhanced virulence potential of CHIKV. 26 This adaptive potential is of concern that the virus may be capable of generating more severe and widespread epidemics across regions in which Aedes albopictus is well established including temperate climates. Synergistic effects of double mutations, including E2-K252Q in combination with other mutations, were found for the virus adaptability and vector infection efficiency. 20 In Brazil, similar to what we had observed in Cuba, ECSA lineage strains devoid of E1-A226V dominated outbreaks in the face of widespread insecticide resistance caused by kdr mutations (e.g., V410L, V1016I, and F1534C), using Aedes aegypti and not Aedes albopictus, despite substitutions that confer insecticide resistance.9,27 This multimodal vector and geographic range in CHIKV raises concerns of its potential to become “Disease X.” Its genotypic plasticity permits adaptation to environmental and anthropogenic changes, including urbanization and vector expansion in response to climate change.9,24 Together with low immunity in naïve populations and the absence of widespread vaccination, CHIKV’s ability to adapt makes it a potential candidate for future global pandemics. Table 1 shows the location, effect, region, and advantages gained by the virus through the discussed mutations.
Mutation analysis table.

Transmission and spread of chikungunya
The transmission of CHIKV occurs usually via bite by the species of Aedes aegypti and Aedes albopictus mosquito.28,29 Transmission cycles include the enzootic (forest dwelling) sylvatic cycle in which the virus cycles between mosquitoes and nonhuman primates, and the urban cycle in which the virus cycles between mosquitoes and humans in densely populated areas.28,30–32 In the sylvatic cycle, CHIKV is primarily transmitted by Aedes species that inhabit forests, such as Aedes africanus and Aedes furcifer. These mosquitoes typically feed on non-human primates, with humans being an accidental host. Human infection occurs infrequently in this cycle. In contrast, the urban cycle occurs in densely populated areas, where Aedes aegypti and Aedes albopictus mosquitoes are the primary vectors. In this cycle, humans serve as the amplification host, and the virus spreads rapidly in urban environments with high mosquito densities and limited control measures. Densely populated areas are breeding grounds for mosquitoes, which increase risks to transmission because of stagnant water and urban heat islands environmental factors. 28 CHIKV enters host cells via clathrin-mediated endocytosis. The virus binds to host cell surface receptors, such as heparan sulfate proteoglycans and DC-SIGN, which facilitate virus adsorption. Once inside, the virus uncoats and releases its genome into the cytoplasm. The positive-sense RNA genome directly translates viral non-structural proteins (nsps) from the 5′ open reading frame. These nsps are essential for genome replication and viral RNA synthesis. The viral replication complex assembles on intracellular membranes, such as the rough endoplasmic reticulum, where replication of the RNA genome takes place. Subsequently, viral RNA is translated into structural proteins, including the envelope glycoproteins (E1 and E2), which are incorporated into newly formed virions. The newly synthesized virions are then packaged into vesicles and are released from cells via direct budding. 33 It is the adaptation of CHIKV to a variety of mosquito vectors that permits its rapid spread. Genetic mutations in the E1 glycoprotein of the virus that enhances its ability to be efficiently transmitted by Aedes albopictus have extended its range to temperate regions.28–30,32 Altogether, high human population density and the global travel has facilitated spread of the virus further.29–31 In the replication process, cell damage takes place and activates an immune response, which results in inflammation, fever, and joint pain, common to the disease. 28 Severe cases have high viremia levels (107 pfu/mL) facilitating efficient transmission to vectors, primarily to mosquito vectors. 32 Vertical transmission from mother to child is well documented; rates up to 50% are reported if the mother is viremic at the time of labor. However, severe neonatal outcome including encephalitis is one of the usual consequences of this route.30,32,34 Most vertical transmission cases occur when the mother is infected between 15 days before delivery and 4 days post-partum. 30 It has been demonstrated that the virus does cross the placental barrier from amniotic fluid and fetal tissues during stillbirth cases. 34 Furthermore, silent transmission from asymptomatic infections complicates the control of outbreaks.29,32 While asymptomatic infections do occur, most CHIKV infections are symptomatic, presenting with fever, rash, and severe joint pain. The symptomatic rate varies, but studies suggest that a significant proportion of infected individuals experience the characteristic symptoms of the disease, with only a small minority remaining asymptomatic. 19 Successful large-scale outbreaks are enabled by the virus’ dependence on both the efficiency of the mosquito vector and adaptation to human hosts combined with low immunity in population. 32 In Salvador, low seroprevalence (11.8%) indicated that there was not enough herd immunity to avert future outbreaks. In addition, viral lineage, mosquito exposure dose, and host genetic factors might influence the severity of infection as well as symptomatic rates. 31 Figure 4 shows the transmission cycle of chikungunya.26,29,30

Illustration of the two primary transmission cycles of chikungunya virus (CHIKV): the Sylvatic cycle and the Urban cycle.
Table 2 shows vertical transmission outcomes in chikungunya.26,28,29
Vertical transmission outcomes in chikungunya.

Disease pathogenesis of chikungunya
CHIKV pathogenesis involves a complex, distinct, acute-to-chronic progression, with systemic inflammation and severe symptoms, coupled with substantial long-term morbidity. CHIKV infection can be thus divided into three phases: acute, subacute and chronic phases.
Acute phase: Characterized by fever, severe arthralgia, myalgia, rash, and headache. These symptoms are present in the majority of cases. This phase may last up to 14 days.
In 75%–88% of cases the acute phase is usually 7–10 days long and characterized by fever, rash, polyarthralgia, myalgia, and fatigue.35–37 Because the elderly and neonates are particularly vulnerable, they can develop severe symptoms ranging from encephalitis to myocarditis, gastrointestinal complications and renal dysfunction.35–37
Subacute phase: During this phase, joint pain persists, and patients may experience ongoing fatigue. Chronic joint issues may begin to emerge, especially in those with pre-existing comorbidities. This phase may last from 15 days to 3 months.37,38
Chronic phase: Some patients develop persistent arthritis, fatigue, and neurological disorders, with symptoms lasting months to years. Chronic chikungunya is associated with significant long-term disability, particularly joint stiffness and neuropathic pain. This phase may last for greater than 3 months.
About 30%–50% of cases will develop chronic complications including persistent joint pain, fatigue and a form of inflammatory arthritis that can persist months to years.35,36 Neuropathic pain such as burning or stinging is common. These are known as atypical phases. The atypical phase of chikungunya infection often presents with symptoms that deviate from the classic fever-first pattern, notably joint pain appearing before fever, which is unusual and seen more in women. Patients may experience a broad range of manifestations involving multiple organ systems, including eye inflammation, oral ulcers, skin eruptions, neurological issues like memory loss, and occasional cardiovascular effects such as temporary blood pressure drops. These atypical symptoms occur across all ages, although eye problems are more common in children. 39 This compounds disability and limits the effectiveness of traditional treatments. 38 Neurological involvement was frequent; chikungunya RNA was detected in 52.9% fatal case specimens including CSF and was associated with encephalitis (21.4%). 37 Chronic chikungunya significantly impairs daily functionality, and 76% of infected individuals have walking disabilities one year post infection.36,38 Chronic chikungunya symptoms last from 3 months to 5 years affecting the quality of life significantly. 40 The psychological burden of long-term disease is underscored by increased risk of depression. 38
The rates of hospitalization range from 2.6% to 17%, and severe forms frequently require intensive care.36,40,41 People with comorbidities such as diabetes or other cardiovascular diseases are more vulnerable to hospitalization. 37 Mortality is primarily seen in the case of severe disease complicated with neurological or cardiovascular complications, with reported case fatality ratios between 0.1% and 0.7%. 36 However, a rare but serious outcome, Guillain-Barré syndrome, is reported in about 1.7 cases per 10,000 chikungunya infections. 41 During the subacute phase, patients with diabetes had a seven fold increased risk of death (odds ratio: 7.7). Most deaths occurred during the acute phase (18 days post symptom onset) although median time from symptom onset to death was 12 days. 37 In outbreaks, as occurred in the 2023 Brazilian epidemic, excess mortality was vastly underreported and the observed rates were 60 times higher than officially confirmed deaths. 42 Table 3 lists clinical manifestations of CHIKV by stage.36,38,41
Clinical manifestations by stage.

Therapeutic and preventive measures of chikungunya
Symptom alleviation is the focus of current management.43,44 Patients with acute pain are treated with paracetamol and opioids but non-steroidal anti-inflammatory drugs (NSAIDs) are avoided to control bleeding rates.43,45 NSAIDs, corticosteroids or for severe cases disease-modifying antirheumatic drug (DMARD) such as methotrexate, hydroxychloroquine are used for chronic arthritis.43,45,46 These DMARDs may be used for refractory conditions. 45 Critical to the mobility is physiotherapy and analgesics.44,45
There has been emerging research on antiviral treatments for chikungunya. One such candidate is sofosbuvir, a drug primarily used for hepatitis C. A study by Ferreria et al. 47 demonstrated that sofosbuvir may inhibit CHIKV replication in vitro, suggesting that it could be a promising therapeutic approach for managing CHIKV infections in the future. Aceclofenac is a non-steroidal anti-inflammatory drug that relieves pain and inflammation. During both acute and chronic phases, a short course of corticosteroids such as prednisolone will give relief from arthritis.45,48,49 Chronic arthritis is treated with methotrexate and hydroxychloroquine, though combined evidence shows improved results.49,50 Some experimental therapies include the combination of 2-fluoroadenine and metyrapone, which are also active antivirally in vitro. 51 Limited utility has been demonstrated in isolated studies of the use of ribavirin and interferon-α. 49 Treatment is supportive and follows a symptom based approach.
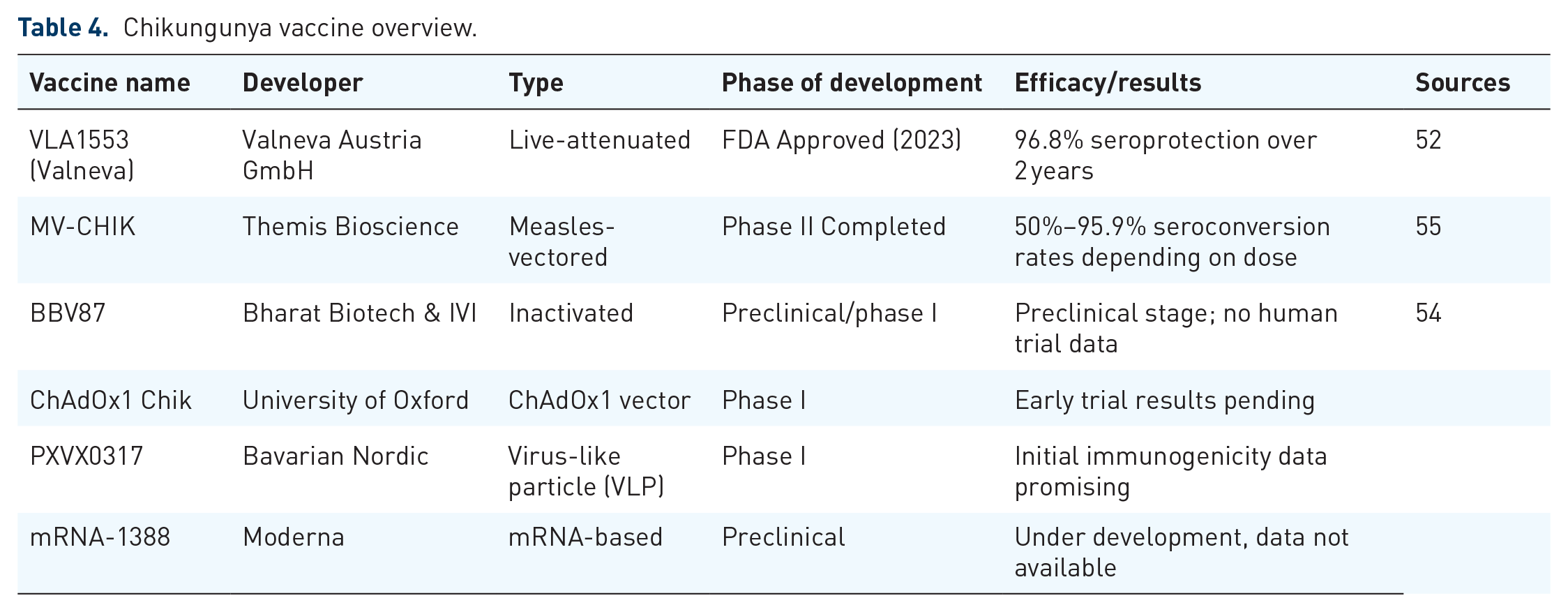
FDA approved chikungunya vaccine VLA1553 with seroprotection of 96.8% over two years in phase III trials.52,53 It uses a live attenuated virus approach that leads to long lasting neutralizing antibody, with high efficacy.52,54 Other vaccine candidates, MV-CHIK and mRNA-based vaccines, are still under development.55,56 Because of the rapid advancement of the regulatory pathways, the clinical trials were conducted with immunological markers rather than conventional efficacy trials.57,58 We still lag behind in widespread access of vaccines to endemic regions.52,57
Chikungunya can be prevented by common people by destroying Aedes mosquito breeding site, like stagnant water in drums, flowerpots, or drains28,59 Insect repellents containing DEET, picaridin, or oil of lemon eucalyptus, and wearing protective clothes, such as long sleeved shirts and pants, are advised.19,29 Window screens, mosquito nets, and cleaning the environment are known to greatly lower exposure. 19 As suggested by WHO, community engagement in integrated vector management reinforces the effect of single efforts.34,59 In endemic regions, awareness campaigns and behavioral interventions are critical.9,29 Table 4 shows various CHIKV vaccines that are available or are under development.52,54,55
Chikungunya vaccine overview.
Prediction of chikungunya to cause global pandemics
CHIKV is a credible candidate for future global pandemic because of its high adaptability, genetic evolution, and ability to spread broadly. The repeated emergence of CHIKV in highly populated and globally linked parts of the world is illustrated by large-scale outbreaks in the Indian Ocean islands, India, and the Americas where more than 3.7 million cases have been reported since 2013.9,28 The virus has mutated critically, including the E1-A226V mutation, which increased its fitness in Aedes albopictus allowing for its spread in areas where Aedes aegypti was less frequent.23,24 Moreover, epidemiologically advantageous additional mutations, such as E2-L210Q, have increased its epidemic potential, allowing the virus to establish sustained human–mosquito transmission cycles. 24 Its genomic plasticity and capacity to adapt to new ecological and vectorial niches emphasized the pandemic potential of CHIKV.9,21 In addition to the high attack rates, debilitating long-term symptoms and severe disease complications such as neurodevelopmental delay of neonates and chronic arthritis increase the public health burden.9,28 CHIKV geographical spread and local transmission are facilitated by a global distribution of Aedes vectors, along with climate change and urbanization.20,30 Rapid geographic expansion and persistence in many different environments are supported by surveillance data and phylogenetic studies.21,23 Hence, CHIKV meets the criteria of a potential candidate for “Disease X”: a disease that potentially can exploit an interconnected web of ecological, genetic, and social factors to cause global health crises.
Conclusion
CHIKV is a serious contender for its status as a future global health crisis as it has the remarkable capacity to adapt, spread, and survive in the different environments. Key mutations, most notably E1-A226V and E2-L210Q, have enabled it to be highly effective at infecting previously resistant mosquito species such as Aedes albopictus, which can now be found worldwide. Together with urbanization, climate change, and global travel, this adaptability has helped fuel huge outbreaks such as the more than 3.7 million cases in the Americas since 2013. Virus causes debilitating effects—chronic arthritis, severe complications in neonates, and prolonged illness—and with no widespread vaccines or effective treatments, it remains a major public health threat. Achieving control of CHIKV will need to take a new approach based on earlier detection and improved vector control strategies, along with equitable access to vaccines in the affected regions. The global “sync” that was developed to combat COVID is critical to avoid being overtaken by this adaptable virus that meets all the criteria for “Disease X,” which could utilize interconnected social, environmental, and biological factors to cause the next pandemic.
