Abstract
Background:
Immunosuppressive treatment can attenuate severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccine-induced immune responses. Moreover, SARS-CoV-2 has neuroinvasive potential and may induce a persistent pro-inflammatory milieu following infection.
Objectives:
To investigate if diminished post-vaccine humoral responses can be overcome with additional vaccine doses and/or breakthrough COVID-19 infections, and if COVID-19 infection can lead to a pro-inflammatory state with neuroaxonal/neuroglial injury in the intermediate-term in patients with central nervous system (CNS) neuroimmunological diseases.
Design:
A prospective observational study conducted at National Neuroscience Institute, Singapore.
Methods:
Serum levels of SARS-CoV-2 neutralising antibodies (NAbs) were measured in patients with CNS neuroimmunological diseases following their fourth SARS-CoV-2 mRNA vaccine (V4), or after breakthrough COVID-19 infection following three prior SARS-CoV-2 mRNA vaccinations, or both. Serum levels of pro-inflammatory cytokines interleukin-6 (IL-6) and tumour necrosis factor (TNF) were evaluated post-COVID-19 infection and post-V4, compared to baseline within individuals. Serum neurofilament-light chain (NfL) and glial fibrillary acidic protein (GFAP), biomarkers of neuroaxonal and astroglial injury, respectively, were measured at baseline and post-COVID-19 infection within patients with relapsing-remitting multiple sclerosis (RRMS) and neuromyelitis optica spectrum disorder (NMOSD).
Results:
Sixty-one patients with various CNS neuroimmunological diseases were recruited, including 34 with MS and 19 with NMOSD. All had received at least three doses of the SARS-CoV-2 mRNA vaccine. Patients on anti-CD20/sphingosine-1-phosphate-receptor modulators (S1PRM) showed significantly reduced NAbs levels in both post-V4 and post-COVID-19 infection scenarios, compared to patients on other immunotherapies. No significant differences between baseline and post-COVID-19 infection concentrations of IL-6 and TNF were observed. Within RRMS and NMOSD patients, NfL and GFAP levels remained similar between baseline and post-COVID-19 infection.
Conclusion:
Anti-CD20/S1PRM treatments are associated with persistently diminished humoral responses post-V4/infection. Patients with CNS neuroimmunological diseases do not show biomarker evidence of intermediate-term pro-inflammatory states and neural injury after COVID-19 infection.
Plain language summary
Patients with neuroimmunological diseases such as Multiple Sclerosis and Neuromyelitis Optica Spectrum Disorder are often put on immunosuppressive medications that aim to dampen the immune response targeting their own brain, spinal cord and optic nerve. Unfortunately, this may result in poor protective antibody responses to vaccines, including the COVID-19 vaccine. We sought to find out if additional doses can be used to bolster the poor response to the COVID-19 vaccines in these patients. Furthermore, previous research has shown that SARS-CoV-2, the virus responsible for COVID-19, can cause inflammation in the brain and spinal cord. Hence, we also looked at markers of neural damage and of inflammation in patients with neuroimmunological diseases after they contract COVID-19 despite previous vaccination. We recruited 61 patients on follow-up for their neuroimmunological disease at the National Neuroscience Institute, Singapore and collected data about their vaccination and COVID-19 history. Blood samples were also collected to test for the antibodies and markers. We found that patients on certain types of immunosuppressive medications (anti-CD20 and Sphingosine-1-Phosphate Receptor Modulators (S1PRM)) had poorer vaccine responses compared to those on other therapies or no therapy. Additional doses of COVID-19 vaccine did not improve vaccine responses for these patients. Markers of neural damage and inflammation after COVID-19 infection did not increase compared to baseline. This study reinforces that patients on certain types of immunosuppressive medications are likely to have an increased risk of COVID-19 although infections are generally mild. Reassuringly, no intermediate-term neural injury or a persistent inflammation resulted from COVID-19 infection in patients with neuroimmunological diseases who were previously vaccinated.
Introduction
Patients with neuroimmunological diseases such as multiple sclerosis (MS), neuromyelitis optica spectrum disorder (NMOSD) and myelin oligodendrocyte glycoprotein antibody-associated disease (MOGAD) are at an increased risk of COVID-19 infection as a result of certain immunosuppressive treatments, which may also dampen the protective immune response induced by severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) vaccinations and natural infection. Previous studies, including from our group, have shown that patients on anti-CD20 monoclonal antibodies and sphingosine-1-phosphate receptor modulators (S1PRM) have attenuated humoral responses to SARS-CoV-2 mRNA vaccinations,1,2 with an earlier time to COVID-19 infection after their third vaccine dose in comparison to patients on other immunotherapies or those without treatment. 3 Subsequent studies showed that an enhanced primary vaccination course moderately increased anti-Receptor Binding Domain (anti-RBD) seropositivity and antibody titres but neutralisation activity remained muted even after booster vaccine doses in MS patients on anti-CD20 treatment. 1 Even when primary vaccination was initiated before anti-CD20 treatment, immune responses after anti-CD20 treatment measured at 6 and 12 months post-vaccination were reduced and so was booster efficacy. 2 Nonetheless, given the paucity of studies, it remains uncertain if booster vaccine doses or symptomatic COVID-19 infection or hybrid responses after infection and vaccination improve protection given the diminished humoral responses seen in patients on anti-CD20 treatment.
Clinical and experimental reports have also demonstrated the neuroinvasive potential of SARS-CoV-2, sparking concerns of neuroaxonal and neuroglial injury following COVID-19 infection in patients with CNS neuroimmunological diseases who already have pre-existing neuroinflammation-driven neuronal/glial injury. Evidence suggests that dysregulation of innate immune responses can occur in COVID-19 infection, leading to cytokine release syndrome characterised by hyper-elevated pro-inflammatory cytokines such as interleukin-6, interleukin-1β, and tumour necrosis factor.4,5 This, together with muted or delayed type I interferon responses, has the potential to disrupt blood-brain-barrier integrity with resulting neurotoxicity.6,7 Emerging studies have also revealed elevated cytokine expression in the blood and CSF can persist in the post-acute phase, suggesting the continued neuroinflammatory processes may underlie the neurological symptoms in long COVID.8–10 However, it remains to be demonstrated if persistently elevated systemic inflammatory responses are present following COVID-19 infection in patients with CNS neuroimmunological diseases, especially those on immunomodulatory or immunosuppressive treatments, and if such elevations are associated with neuroaxonal or neuroglial injury in these at-risk individuals.
In this study, we sought to determine the serum levels of SARS-CoV-2 neutralising antibodies (NAbs) in patients with CNS neuroimmunological diseases following their fourth SARS-CoV-2 mRNA vaccine (V4), or after breakthrough COVID-19 infection following three prior SARS-CoV-2 mRNA vaccinations, or both. We postulated that protective humoral responses after both natural COVID-19 infection and after V4 would be muted in patients on anti-CD20 or S1PRM treatments compared to those on other immunotherapies or without treatment. We also aimed to measure the serum levels of pro-inflammatory cytokines, tumour necrosis factor (TNF) and interleukin-6 (IL-6), before and after COVID-19 infection or V4, to test the hypothesis that systemic pro-inflammatory responses are increased after these events. These two cytokines were selected given their roles in systemic and neuroinflammation in COVID-19 infection (both acute and chronic phases) and as key inflammatory drivers in the pathogenesis of CNS neuroimmunological diseases.11–14 Finally, we sought to determine the serum levels of neurofilament-light chain (NfL) and glial fibrillary acidic protein (GFAP), as markers of neuroaxonal and astrocytic damage, respectively, before and after COVID-19 infection, to identify potential evidence of neural injury following infection.
Materials and methods
Study and follow-up periods
This study was part of an ongoing prospective, pragmatic observational research project at the Neuroimmunology Clinic, National Neuroscience Institute, Singapore.3,15 Participants were recruited from April 2021 to March 2023 in tandem with their regular appointments for the management of their neuroimmunological conditions. The end of the recruitment period was defined as such given that beyond this time it was no longer mandated for symptomatic individuals to test for COVID-19 infection in Singapore, leading to uncertainty in confirming the presence or absence of COVID-19 infection.
Participants
The inclusion criteria for this study were: (1) patients with neuroimmunological diseases who had received three doses of either the Pfizer-BioNTech or Moderna SARS-CoV-2 mRNA vaccines and (2) had their first COVID-19 infection following the third vaccine dose (V3), or received a fourth vaccine dose (V4), or both. The patients were further subdivided into Same Treatment and Switch Treatment groups where the former maintained the same immunotherapy from the first vaccine dose (V1) till V4 and/or COVID-19 infection, while the latter did not. Demographic, clinical, immunotherapy, SARS-CoV-2 vaccination and COVID-19 infection information were obtained from the electronic medical records. COVID-19 infection was defined as the presence of symptoms consistent with COVID-19 infection together with a positive polymerase chain reaction or antigen rapid test.
All MS and aquaporin-4-antibody (AQP4+) NMOSD patients fulfilled the 2017 McDonald criteria for MS and 2015 International Panel for NMO Diagnosis (IPND) criteria for NMOSD, respectively.16,17 MOGAD patients had at least one core clinical demyelinating event with positive myelin oligodendrocyte glycoprotein (MOG) antibodies. 18 AQP4, MOG and autoimmune encephalitis-associated antibodies were detected on fixed or live cell-based assays.
Immunotherapy assignment and classification
Assignment of immunotherapy group for each patient was performed using strict criteria to ensure that any differences observed in outcomes, with immunotherapy as an independent variable, can be interpreted without confounding. For the Same Treatment group, the assigned immunotherapy had to have been started at least 3 months prior to receiving V1 and must be maintained till the point of V4 and/or COVID-19 infection. Patients who had received immune reconstitution therapies (IRTs) were ascribed to the IRT group regardless of the duration between treatment (before V1) to V4 and/or COVID-19 infection. Individuals on anti-CD20 treatments were assigned accordingly if they had received treatment within 8 months prior to V1 and maintained till either V4 and/or COVID-19 infection. Patients were assigned to nil treatment if they had not been on any immunotherapy for at least 3 months prior to V1 and up till V4 and/or COVID-19 infection. For the Switch Treatment group, assignment of immunotherapy group was based on the immunotherapy they were receiving at each vaccination or COVID-19 infection time point.
For the purpose of investigating the humoral response following V4 or COVID-19 infection, immunotherapies were classified into two broad groups: (1) anti-CD20 and S1PRM and (2) Others. Anti-CD20 therapies comprised Rituximab and Ocrelizumab while S1PRM consisted only of Fingolimod. The Others group included various immunotherapies; IRTs (i.e., cladribine, alemtuzumab, and autologous hemopoietic stem cell transplantation), disease-modifying anti-rheumatic drugs (DMARDs) (i.e., azathioprine and Mycophenolate Mofetil), other MS disease-modifying therapies (DMTs) (i.e., dimethyl Fumarate and Interferon beta-1α), and those not on treatment. Anti-CD20 and S1PRM were grouped together due to their known attenuation of humoral responses post-SARS-CoV-2 vaccination as shown in previous studies.1–3 Limited numbers precluded further stratification of the Others group.
Measurement of serum SARS-CoV-2 neutralising antibodies
Blood samples were collected at the patients’ clinical visit (BD Vacutainer® tube 367986), centrifuged within 4 h at 1300 g for 10 min, and the resulting sera stored at −80°C. NAbs against SARS-CoV-2 were measured using Genscript® cPass™ surrogate virus neutralisation test according to the manufacturer’s instructions. Variant RBDs were used as the viral strains in the assay. The antibody levels were expressed quantitatively as percentage inhibition (0%–100%); greater values indicate higher levels of NAbs. Negative values were rounded up to 0%. A threshold of ⩾30% represented a detectable NAbs response as per the manufacturer’s technical specifications.
Measurement of serum pro-inflammatory cytokines and neuroaxonal and astroglial injury markers
TNF and IL-6 concentrations were determined using a two-plex human cytokine magnetic bead panel (Milliplex®) on the multiplex bead-based Luminex (Intelliflex®) platform. Serum samples were analysed without dilution, and the concentrations of TNF and IL-6 were determined based on a standard curve generated using the Belysa® immunoassay curve-fitting software. NfL and GFAP levels were measured using the ultrasensitive Simoa® platform using methods as described previously. 19 All serum samples used for NAbs, cytokines, NfL and GFAP measurements were thawed only once.
Statistical analysis
For this study, sample size calculation was based on post-V3 NAbs data comparing Anti-CD20/S1PRM and Others treatments from our previous study. 3 This demonstrated an effect size (Cohen’s d) of 3.7, providing 90% power to detect differences in NAbs levels with an alpha of 5% using a sample size of 6 (in each comparative group). Given our biological hypothesis that NAbs responses would continue to be blunted post-V4 and/or post-COVID-19 infection, we performed statistical analysis only if the sample size (within each comparative group) was 6 or more (based on our sample size calculation). Comparative statistical analysis was not performed if any group had a sample size of 5 or less.
Statistical analysis and graphical representation were performed using GraphPad Prism (version 10, GraphPad Software, San Diego, CA, USA). NAbs levels between immunotherapy groups and among timepoints were compared using Mann–Whitney and Kruskal–Wallis tests as appropriate. The intervals between vaccine dose receipt or COVID-19 infection, and serum sampling for NAbs levels were analysed using two sample t-test with Welch’s correction or Welch ANOVA. Paired t-test was used to compare the pre- and post-event serum concentrations of TNF, IL-6, NfL, and GFAP within individuals, where the event-of-interest was either receipt of V4 or occurrence of COVID-19 infection. Statistical significance was set at p < 0.05.
Standard protocol approvals, registrations and patient consents
This research was conducted under local ethics approval (CIRB number 2021/2222), and all subjects provided written informed consent before participation in the study. The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement (Supplemental Material).
Results
Study population
Sixty-one patients who had received at least three doses of COVID-19 vaccines (all received either the BNT162b2 (Pfizer-BioNTech) or mRNA-1273 (Moderna) vaccines) met the inclusion criteria and were included in the analysis. There were 49 patients in the Same Treatment group and 12 patients in the Switch Treatment group.
Within the Same Treatment group, there were 25 (51.0%) MS (20 relapsing-remitting, 3 primary progressive and 2 secondary progressive), 17 (34.7%) AQP4+ NMOSD, 3 (6.1%) MOGAD and 4 with other CNS neuroimmunological diseases (including autoimmune encephalitis and overlap disorders) (see Table 1 for full list). Rituximab was the most frequently used treatment (19/49, 38.8%). A total of 25 patients were classified into the Anti-CD20 and S1PRM group while the remainder formed the Others group. As mentioned, all patients maintained the same treatment from V1 till V4 and/or COVID-19 infection.
Demographic and clinical characteristics of study population.

For the Switch Treatment Group, the Treatment is stated as that during either V4 or COVID-19 infection, whichever was earlier if both V4 and COVID-19 took place.
AHSCT, Autologous Hematopoietic Stem Cell Transplantation; AQP4+ NMOSD, aquaporin-4 antibody positive Neuromyelitis Optica Spectrum Disorder; BMI, Body-Mass Index; CLIPPERS, Chronic Lymphocytic Inflammation with Pontine Perivascular Enhancement Responsive to Steroids; MOGAD, Myelin Oligodendrocyte Glycoprotein antibody-associated Disease; PPMS, primary progressive Multiple Sclerosis; RRMS, relapsing-remitting Multiple Sclerosis; SPMS, secondary progressive Multiple Sclerosis.
Within the Same Treatment group, 36.7% (18/49) of patients had a COVID-19 infection and no V4. Twenty-three patients in the group had blood sampling after V4, whereas 30 patients had blood sampling after COVID-19 infection. The distribution of patients and blood sampling time points in relation to COVID-19 and V4 is summarised in Table 2 for both the Same Treatment and Switch Treatment groups.
Frequencies of patients and blood sampling time points grouped by treatment, V4 receipt and COVID-19 infection.

n refers to the number of patients; some patients contributed to more than one sampling time point, resulting in the total number of samples exceeding the number of patients.
NA, not applicable.
Same treatment group – neutralising antibodies measured after fourth vaccine dose
Thirteen patients in the Same Treatment group who received V4 without prior COVID-19 infection had post-V4 sampling for SARS-CoV-2 NAbs. Stratification by immunotherapy showed significantly reduced NAbs levels in the Anti-CD20 and S1PRM group (n = 7) compared to the Others group (n = 6) (median (IQR); Anti-CD20 and S1PRM 2.1% (0.0–42.6) versus Others 96.3% (95.1–97.4), p = 0.001), although there remained two patients in the former group that had detectable NAbs responses (Figure 1(a)). This difference was in the absence of a difference in the post-V4 sampling interval (given that NAbs levels are known to decrease with time from vaccination) between the two groups (mean (SD); Anti-CD20 and S1PRM 9.8 weeks (7.6) versus Others 15.1 weeks (8.4), p = 0.258) (Figure 1(b)). Ten patients in the Same Treatment group also received V4 and had post-V4 blood sampling but had a prior COVID-19 infection before receiving V4. The Anti-CD20 and S1PRM group (n = 8) and the Others group (n = 2) had median NAbs levels of 20.9% and 87.8% respectively (Figure 1(c)), with no difference in the post-V4 sampling interval (means of 9.8 vs 7.5 weeks respectively) between the two groups (Figure 1(d)). No statistical testing was performed in this subset of patients given the small sample size in the Others group.

SARS-CoV-2 NAbs and post-event sampling intervals stratified by immunotherapy groups. NAbs levels and sampling intervals post-V4 without (a, b) and with (c, d) prior COVID-19 infection. NAbs levels and sampling intervals post-COVID-19 without (e, f) and with (g, h) prior V4. NAbs levels are represented as medians with IQR whereas sampling intervals are represented as means with SD.
Same treatment group – neutralising antibodies measured after COVID-19 infection
Thirty patients in the Same Treatment group developed COVID-19 infection and had post-COVID-19 blood sampling. Three patients on Rituximab had COVID-19 infections of moderate severity, and the rest had mild infections (with severity classification according to local guidelines). 20 Of these 30 patients, 23 had not received V4 prior to the sampling. A significant reduction in NAbs levels was observed in the Anti-CD20 and S1PRM group (n = 12) compared to the Others group (n = 11) (median (IQR); Anti-CD20 and S1PRM 1.9% (0.0–36.5) vs Others 95.5% (95.4–97.3), p < 0.0001) (Figure 1(e)), with no significant difference in the post-COVID-19 sampling interval (mean (SD); Anti-CD20 and S1PRM 12.2 weeks (5.4) vs Others 11.9 weeks (7.2), p = 0.903; Figure 1(f)).
The remaining 7 of the 30 patients had COVID-19 infection after V4 with post-COVID-19 blood sampling. The Anti-CD20 and S1PRM group (n = 2) and the Others group (n = 5) had NAbs levels of 10.9% and 97.5%, respectively (Figure 1(g)). There was an observable difference in the means for the post-V4 sampling interval between the two groups (3.4 weeks for Anti-CD20 and S1PRM vs 9.8 weeks for Others) (Figure 1(h)) – this would have had, in any case, favoured higher NAbs levels in the Anti-CD20 and S1PRM group. No statistical testing was performed given the small sample sizes.
Same treatment group – comparing V4, COVID-19 infection, and their co-occurrence on NAbs levels within treatment groups
Within Same Treatment patients in the Anti-CD20 and S1PRM group, comparison of post-event NAbs levels in those who had V4 only (i.e., without prior COVID-19 infection) (n = 7) versus those with COVID-19 infection only (i.e., no additional V4) (n = 12) versus those who had V4 after a prior COVID-19 infection (with post-V4 sampling) (n = 8) revealed no significant differences (median (IQR); V4 only 2.1% (0.0–42.6) vs COVID-19 only 1.9% (0.0–36.5) vs V4 after prior COVID-19 20.9% (0.2–60.3), p = 0.514) (Figure 2(a)). There was no significant difference in their mean post-event sampling intervals (mean (SD); V4 only 9.8 weeks (7.6) vs COVID-19 only 12.2 weeks (5.4) vs V4 after prior COVID-19 9.8 weeks (5.0), p = 0.557) (Figure 2(b)). There were too few patients who had COVID-19 after a prior V4 (with post-COVID-19 sampling) to be included in the analysis. NAbs levels and corresponding post-event sampling intervals are also illustrated for V1–V3 for these patients for longitudinal comparison.
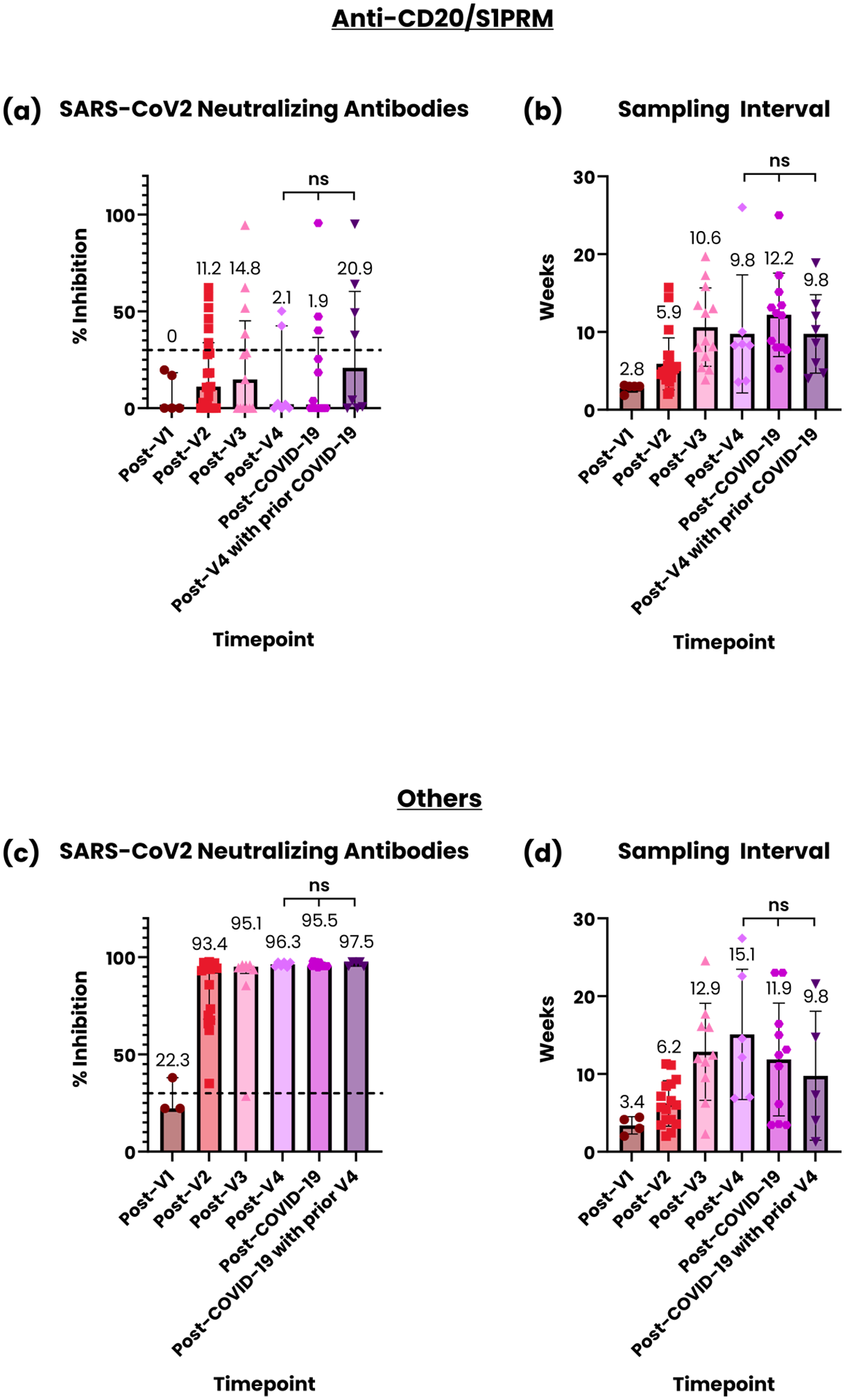
SARS-CoV-2 NAbs and post-event sampling interval across timepoints. NAbs levels (a) and sampling intervals (b) post-V1, post-V2, post-V3, post-V4, post-COVID-19, and post-V4 after prior COVID-19 for the anti-CD20/S1PRM group. NAbs levels (c) and sampling intervals (d) post-V1, post-V2, post-V3, post-V4, post-COVID-19, and post-V4 after prior COVID-19 for the Others group.
In the Others group of Same Treatment patients, there were no significant differences in post-event NAbs levels in those had V4 only (n = 6) versus those with COVID-19 infection only (n = 11) versus those who had COVID-19 infection after a prior V4 (n = 5) (median (IQR); V4 only 96.3% (95.1–97.4) versus COVID-19 only 95.5% (95.4–97.3) versus COVID-19 after prior V4 97.5% (95.4–97.6), p = 0.613) (Figure 2(c)). The mean post-event sampling intervals also did not differ significantly (mean (SD); V4 only 15.1 weeks (8.4) vs COVID-19 only 11.9 weeks (7.2) vs COVID-19 after prior V4 9.8 weeks (8.3), p = 0.544) (Figure 2(d)). There were too few patients who had V4 after a prior COVID-19 infection to be included in the analysis. NAbs levels and corresponding post-event sampling intervals are also illustrated for V1–V3 for these patients for longitudinal comparison.
Switch treatment group – longitudinal trend of neutralising antibodies
In the Switch Treatment group, patients who switched from another treatment to an anti-CD20/S1PRM therapy (n = 10) experienced a drop in NAbs levels following the switch at post-V3, post-V4, and post-COVID-19 time points (Figure 3(a) and (b)). Comparatively, a switch from a non-anti-CD20/S1PRM therapy to another non-anti-CD20/S1PRM therapy (n = 2) did not appear to affect NAbs levels (Figure 3(a)).
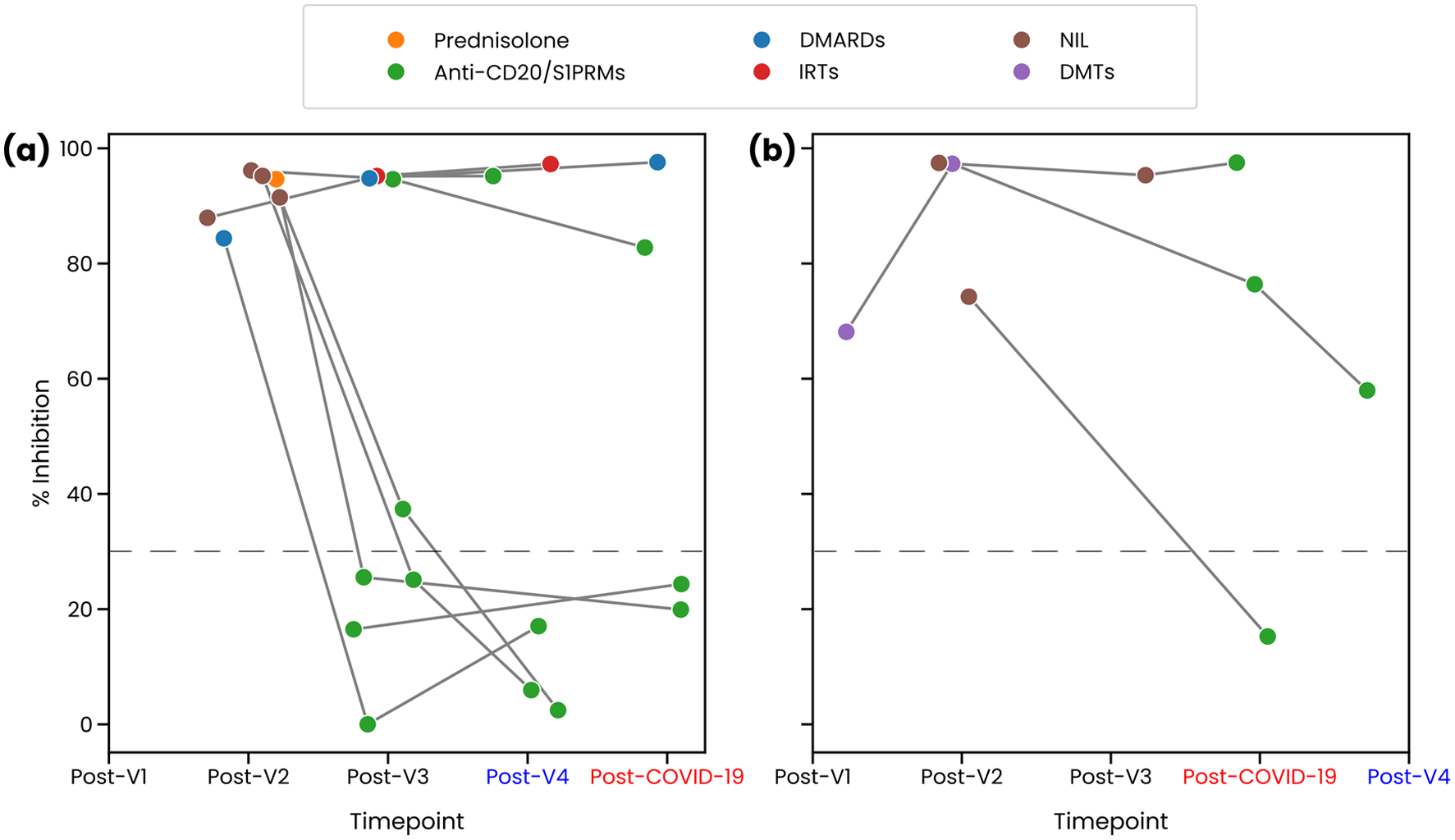
Longitudinal trend of SARS-CoV-2 NAbs in the Switch Treatment group. Subset of patients who had COVID-19 infection occurred after V4 (a). Subset of patients who had V4 following COVID-19 infection (b). Horizontal dashed lines represent percentage inhibition of 30%.
Same treatment group – pro-inflammatory cytokines before and after COVID-19 infection or V4
To determine if COVID-19 infection or V4 results in a persistent, systemic pro-inflammatory response, serum levels of TNF and IL-6 were measured at baseline (taken following V2) and at post-event in Same Treatment patients who had either a COVID-19 infection only (i.e., no prior V4) or receipt of V4 only (i.e., without prior COVID-19 infection). Sixteen patients with COVID-19 infection only and 13 patients with V4 only were included in this analysis. No significant differences were observed for both cytokines at baseline versus post-COVID-19 infection (mean of difference (95% CI); IL-6 −5.51 pg/mL (−19.42–8.40), p = 0.412; TNF −2.49 pg/mL (−9.65–4.67), p = 0.470) (mean (SD); baseline IL-6 10.55 pg/mL (21.2) vs post-COVID-19 IL-6 5.04 pg/mL (11.1); baseline TNF 22.29 pg/mL (13.36) vs post-COVID-19 TNF 19.80 pg/mL (16.26); Figure 4(a) and (b)). There were also no differences in the concentrations of both cytokines between baseline and post-V4 (mean of difference (95% CI); IL-6 4.94 pg/mL (−4.00–13.87), p = 0.252; TNF 2.70 pg/mL (−2.54–7.94), p = 0.284) (mean (SD); baseline IL-6 2.47 pg/mL (2.66) vs post-V4 IL-6 7.41 pg/mL (14.21); baseline TNF 24.77 pg/mL (15.27) vs post-V4 TNF 27.47 pg/mL (14.61); Figure 4(c) and (d)). The sampling intervals (mean, (SD)) for the baseline and post-COVID-19 sampling are 7.3 (5.3) and 12.1 (6.3) weeks, respectively. The sampling intervals (mean, (SD)) for the baseline and post-V4 sampling are 10.3 (6.1) and 12.2 (8.1) weeks, respectively. The baseline and COVID-19 sampling intervals were taken as the periods in weeks between receipt of V2 or the occurrence of COVID-19 infection, respectively, and blood sampling.

Paired serum concentrations of pro-inflammatory cytokines, and neuroglial and neuroaxonal biomarkers. Baseline and post-COVID-19 IL-6 (a) and TNF (b) concentrations. Baseline and post-V4 IL-6 (c) and TNF (d) concentrations. Baseline and post-COVID-19 NfL (e) and GFAP (f) within RRMS and AQP4+ NMOSD patients.
Same treatment group – neuroaxonal and astroglial injury biomarkers before and after COVID-19 infection
To determine if COVID-19 infection exacerbates neuroaxonal and astroglial injury in patients with relapsing-remitting MS or AQP4+ NMOSD, serum concentrations of NfL and GFAP were measured at baseline (taken following either V2 or V3) and post-COVID-19 infection. Only patients on immunotherapies in the Same Treatment group were included in this analysis, comprising 11 RRMS and 9 NMOSD patients. Baseline Z scores for NfL and GFAP in these 20 patients were obtained by computational referencing to published healthy cohorts,21,22 adjusted for age, sex and BMI. BMI was imputed with a value of 25 as these data were not available at sampling time point. Median (IQR) Z scores for NfL and GFAP were 0.025 (−0.46 to 1.16) and −0.64 (−0.78 to 1.28), respectively, suggesting low biomarker evidence of disease activity at baseline.
No significant differences were observed for both biomarkers at baseline versus post COVID-19 infection (mean of difference (95% CI); NfL −0.93 pg/mL (−2.55 to 0.70), p = 0.249; GFAP −7.36 pg/mL (−18.79 to 4.08), p = 0.194) (mean (SD); baseline NfL 8.62 pg/mL (5.17) vs post-COVID-19 NfL 7.69 pg/mL (2.65); baseline GFAP 94.54 pg/mL (67.15) vs post-COVID-19 GFAP 87.19 pg/mL (67.08); Figure 4(e) and (f)). The sampling intervals (mean, (SD)) for the baseline and post-COVID-19 samples were 8.0 (6.1) and 12.1 (6.5) weeks, respectively. The baseline sampling interval was taken as the period in weeks between receipt of either V2 or V3 and blood sampling. The COVID-19 sampling interval was taken as the period in weeks between the COVID-19 infection and blood sampling.
Discussion
In this study, we revealed that patients with CNS neuroimmunological diseases treated with anti-CD20 or S1PRM had significantly reduced levels of NAbs (compared to other immunotherapies or no treatment) after receipt of V4 or breakthrough COVID-19 infection (following V3). Our findings are consistent with previous literature that have shown decreased SARS-CoV-2 antibody responses following COVID-19 infection in patients with MS or NMOSD on anti-CD20 therapies compared to other immunotherapies 23 and following vaccination with V4 in MS patients on anti-CD20 therapies. 1 NAbs levels in patients that had sampling done after receipt of V4 (with prior COVID-19 infection), as well as after COVID-19 infection (with prior V4), showed similar trends although sample sizes were too small for statistical testing. We also demonstrated that patients who switched treatment to anti-CD20/S1PRM therapies had a reduction in NAbs levels measured after vaccination or infection, consistent with prior reports,1,2 despite initial robust humoral responses on other immunotherapies prior to switching.
We did not find a statistical difference in NAbs response induced by COVID-19 infection compared to V4 in patients receiving either anti-CD20/S1PRM therapies or Other immunotherapies. A previous study noted that prior COVID-19 infection markedly increases post-vaccine humoral, and to a smaller extent cellular responses, regardless of immunotherapy group. 24 This notion of hybrid immunity – immunity arising from both infections and vaccinations – has been suggested to be superior to vaccination-only immunity. 24 We observed a trend towards higher NAbs responses within patients on anti-CD20/S1PRM therapies who had post-V4 sampling with prior COVID-19 infection (median 20.9%), compared to those with V4 only (2.1%) and COVID-19 infection only (1.9%); this was not statistically significant, which may be attributable to the small sample sizes.
Our study found that COVID-19 infection did not increase serum levels of the pro-inflammatory cytokines, IL-6 and TNF, in CNS neuroimmunological diseases. Previous studies have noted elevated IL-6 and TNF levels in the acute phase of COVID-19 infection as well as in patients with Post-Acute Sequelae of COVID-19 (PASC) and long-COVID-19,8–10 postulating that persistent cytokine elevations underlie much of the pathophysiology for these longer-term complications. We also observed no increases of these two cytokines after V4, consistent with a previous study that reported that SARS-CoV-2 vaccination did not induce systemic inflammation through comparisons of vaccinated versus unvaccinated patients with various neurological diseases, 25 although systemic inflammation was only assessed by serum C-reactive protein, white blood cells and neutrophil-to-lymphocyte ratio in that study.
Notably, we found that serum NfL and GFAP were not increased after breakthrough COVID-19 infection in CNS neuroimmunological diseases. Markers of neuroaxonal and astroglial injury have been reported to be elevated in the acute phase of COVID-19 infection 26 and even after SARS-CoV-2 test negativization. 27 Plasma GFAP levels are higher in patients with severe COVID-19 infection compared to controls, and plasma NfL levels are increased in patients with severe infection compared to controls and patients with mild or moderate infection. 28 However, these differences dissipate by 6 months, and no correlation was found between these biomarkers in the acute phase and patients with persistent neurological symptoms. 28 Our study contextualises these findings to RRMS and AQP4+ NMOSD patients; reassuringly, we demonstrated no evidence of superimposed neural injury in these at-risk patients following COVID-19 infection in the intermediate-term (mean interval of 12 weeks from infection to blood sampling). This sampling interval is relevant especially for NfL, as it has been shown in clinical and experimental MS studies that serum levels peak only days to weeks after initial injury and remain elevated for prolonged periods thereafter.29,30 We did not measure serum NfL and GFAP following V4, as it has been reported that SARS-CoV-2 vaccination does not induce axonal loss (compared to unvaccinated patients) in various neurological diseases. 25
Our study’s limitations include small sample sizes due to the progressively decremental number of participants who had yet to have COVID-19 infection prior to V3 receipt in order to be included. This limited the power of the study and precluded the performance of comparative statistical analysis between certain groups. Longitudinal comparisons of the NAbs levels were also limited by sample size as the whole study cohort could not be comprehensively sampled following each vaccination or infection event, in part due to COVID-19 pandemic-induced restrictions on clinical and research visits during the early period of the study. Additionally, while there were no differences in sampling intervals between comparative groups for NAbs measurements after V4 only, after infection only, and after V4 with prior infection at the group level, inter-individual variability in sampling intervals may be present, which could have influenced our findings especially when it is known that NAbs concentrations wane over time. 31 T-cell immune responses were also not measured, limiting the assessment of the full repertoire of immune responses to vaccination and infection. Several studies have reported that cellular immunity contributes to the efficacy of SARS-CoV-2 vaccines and can protect against variants that evade NAbs as the targets of NAbs are restricted to proteins on the viral surface whereas T-cells can recognise a broader range surface and structural SARS-CoV-2 antigens.32,33 Apostolidis et al demonstrated that MS patients receiving anti-CD20 therapy produced strong, antigen-specific CD4+ and CD8+ T-cell responses following SARS-CoV-2 vaccination, however, CD4+ subsets were skewed while CD8+ responses were enhanced. 34 In contrast, SARS-CoV-2-specific T-cell responses appear to be attenuated in patients receiving S1PRM treatments.24,35 Despite these findings, it has been shown that amongst MS DMTs, anti-CD20 treatments confer the highest risk of severe COVID-1936,37; indeed all three patients who had moderately severe COVID-19 infection in the Same Treatment group were on Rituximab. This highlights that the complexity of B-cell and T-cell responses is yet to be fully understood, especially in individuals on immunosuppressive therapies. Our study was conducted at a single tertiary centre in Singapore on a cohort of predominantly Chinese ethnicity, factors which may affect the broader applicability of the findings to other populations or healthcare settings. The breakthrough COVID-19 infections identified in our study were mostly mild in severity and likely of the Omicron variant; these may limit the applicability of our findings to other and future variants, which may cause more severe disease and hence potentially result in differential cytokine or biomarker trends. Lastly, we also did not collect BMI data, which precludes the accurate determination of individual Z scores although this would not have affected the intra-individual trajectory (and paired analysis) as it is unlikely the BMI would differ significantly within individuals at baseline and at post-COVID-19 time points.
However, our study has several strengths including investigating both pro-inflammatory responses and neural injury after COVID-19 infection in patients with CNS neuroimmunological diseases. We also applied strict criteria for the assignment of patients to the immunotherapy groups to enable the clear delineation of the effect of treatments. Furthermore, the inclusion of multiple neuroimmunological diseases such as AQP4+ NMOSD and MOGAD allowed for a broader range of immunotherapies to be analysed. Finally, the specific use of a neutralising antibody assay as opposed to an anti-receptor binding domain antibody assay improved the ability to meaningfully translate the antibody levels to real-world humoral protection against infection.
Conclusion
With COVID-19 having become endemic globally and the continued emergence of new subvariants, which generally result in mild disease, most countries have done away with strict diagnostic testing for COVID-19. As a result, it is foreseeable that the study of immune responses following infections (and vaccinations) will be increasingly difficult. Our findings suggest that patients on anti-CD20/S1PRM therapies continue to have attenuated humoral responses post-V4, post-infection (after V3) and even after both events, underscoring the need for continued vigilance and infection prevention in these individuals although breakthrough infections appear to be mostly mild. Reassuringly, we demonstrated no intermediate-term neural injury or a persistent systemic pro-inflammatory state in patients with CNS neuroimmunological diseases after breakthrough COVID-19 infection. Further studies could examine the potential long-term effects of COVID-19 infection, particularly in patients with CNS neuroimmunological diseases who develop symptoms of long COVID-19, using longitudinal, sensitive clinical assessments and molecular markers to delineate COVID-19’s impact on the disease activity and progression.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251370471 – Supplemental material for Longitudinal quantification of serum SARS-CoV-2 neutralising antibodies, pro-inflammatory cytokines, NfL and GFAP before and after breakthrough COVID-19 infection in CNS neuroimmunological diseases: a prospective observational study
Supplemental material, sj-docx-1-tai-10.1177_20499361251370471 for Longitudinal quantification of serum SARS-CoV-2 neutralising antibodies, pro-inflammatory cytokines, NfL and GFAP before and after breakthrough COVID-19 infection in CNS neuroimmunological diseases: a prospective observational study by M. Hema Prashaad, Rachel Wan En Siew, Amelia Yun Yi Aw, Janice Hui Yi Tan, Muhammad Yaaseen Gulam Mohamed, Janis Tye Siew Noi, Kalpana Prasad, Kevin Tan, Jens Kuhle, Yinxia Chao, Ivy Ai-Wei Ho and Tianrong Yeo in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
