Abstract
Background/Aims: Data concerning differences in demographics/disease severity between the first and second waves of COVID-19 are limited. We aimed to examine prognosis in patients presenting to hospital with COVID-19 amongst different ethnic groups between the first and second waves in the UK.Methods: In this retrospective cohort study, we included 1763 patients presenting to a regional hospital centre in Leicester (UK) and compared those in the first (n = 956) and second (n = 807) waves. Admission National Early Warning Scores, mechanical ventilation and mortality rate were lower in the second wave compared with the first.Results: Thirty-day mortality risk in second wave patients was approximately half that of first wave patients [adjusted hazard ratio (aHR) 0.55, 95% confidence interval (CI) 0.40–0.75]. In the second wave, Black patients were at higher risk of 30-day mortality than White patients (4.73, 1.56–14.3). Conclusion: We found that disporportionately higher risks of death in patients from ethnic minority groups were not equivalent across consecutive waves of the pandemic. This suggests that risk factors for death in those from ethnic minority groups are malleable and potentially reversible. Our findings need urgent investigation in larger studies.
Introduction
The COVID-19 pandemic has caused significant global morbidity and mortality and has disproportionately affected ethnic minority groups. 1 In the absence of widespread immunity, many countries will continue to suffer surges and/or waves of infection, 2 in association with the implementation and release of lockdown measures. Ethnic minority groups have been disproportionately affected by COVID-19 – suffering increased infection, hospitalisation and death during the first wave of the pandemic. Multiple reasons exist for this, but it is thought that the disproportionate risk is mainly attributable to a higher risk of infection, from living in deprived areas, working in high-exposure or frontline occupations and living in larger, multigenerational households compared with White groups. 3 Limited evidence suggests that mortality in the second wave may be lower than that of the first, but it is not known whether this is true for all ethnic groups. 4
To address this, we compared the demographics and clinical outcomes of patients with COVID-19 admitted to a large UK centre serving a multiethnic population during the first and second waves of the current pandemic. We aimed to estimate the effect of ethnicity as an important determinant of mortality in both waves – and hypothesised that ethnicity was related to mortality in at least one wave of the pandemic.
Methods
This retrospective cohort study was undertaken at University Hospitals of Leicester (UHL) National Health Service (NHS) Trust. This consists of three hospitals: Leicester General Hospital, Glenfield Hospital and Leicester Royal Infirmary. The trust provides all acute care to patients with COVID-19 in the Leicestershire area [a population of just more than 1 million (2016 estimate)] within the United Kingdom, and provides secondary, tertiary and intensive care. Care for acute patients is provided by a government system (NHS) and free for all patients irrespective of socioeconomic status at the point of use. Private care for those hospitalised with COVID-19 does not exist in our region– therefore, our data provide complete coverage of the Leicestershire area. Disease severity scoring for patients in the UK is guided by the National Early Warning Score (NEWS) 2 alert system. Outpatient care is also provided by NHS hospitals.
We defined a case of COVID-19 as an adult patient with a positive molecular test for SARS-CoV-2 on nasopharyngeal swab. The first wave cohort included all cases between 1 March 2020 and data extraction on 28 April 2020. 5 The second wave cohort included all cases between 1 September 2020 and data extraction on 15 November 2020.
Testing for SARS-CoV-2 was based on the presence of symptoms during the first wave; however, a nationwide policy of universal testing for all emergency admissions was adopted from 27 April 2020. 6 Molecular testing was made available for hospitalised patients since the start of the first cases of COVID-19 in the United Kingdom, on 27 January 2020.
We extracted data from the electronic hospital record concerning age, sex, self-reported ethnicity, postcode, comorbidities (see Supplemental Table 1), NEWS 27 on admission, admission/discharge date, highest supplementary oxygen flow rate during admission, dexamethasone use, requirement for mechanical ventilation and date of death. Ethnicity was categorised as White, South Asian, Black and Other (see Supplemental Table 2). Postcode was used to derive Index of Multiple Deprivation (IMD).
Continuous variables were summarised as median [interquartile range (IQR)] and categorical variables as count (%). Comparison was by Wilcoxon rank-sum test for continuous variables and chi-square test for categorical variables.
We established survival time in days by calculating the difference between date of positive swab and date of data extraction or death and used univariable and multivariable Cox regression to establish factors associated with all-cause mortality within 30 days of COVID-19 diagnosis. Multiple imputation was used to replace missing data in all models fitted, and the multiple imputation model included all variables for those being imputed (for further details, see Supplementary information). To investigate the effects of multiple imputation on the results, we conducted a sensitivity analysis using only complete cases. All analyses were conducted using Stata (StataCorp. 2019). Values of p < 0.05 were considered statistically significant.
Results
Table 1 shows a description of the cohort stratified by pandemic wave. A total of 1763 patients were included in the final analysis, 956 (54.2%) from the first wave and 807 (45.8%) from the second wave. There were no significant demographic/comorbidity differences between the first and second wave cohorts.
Demographic and comorbidity characteristics of first and second wave patients.

IMD, Index of Multiple Deprivation; IQR, interquartile range; med, median
Markers of COVID-19 severity and duration of hospital stay stratified by ethnicity are shown in Table 2. Admission NEWS was significantly higher in the first wave compared with the second wave in those of White and South Asian ethnicity. The proportion of deaths after diagnosis with COVID-19 was higher in the first wave than in the second wave. This was observed in White and South Asian patients (26.1% versus 10.8%, p < 0.001 and 22.1% versus 10.9%, p = 0.001, respectively) but not in Black patients (16.3% versus 15.4%, p = 0.922). When those who died and those who were not discharged at the point of data extraction are excluded, median duration of hospital stay was longer in the second wave compared with the first (4 days, IQR 1–10 versus 5 days IQR 1–9.5, p = 0.016).
Markers of severity and outcome data stratified by pandemic wave and ethnicity.

IQR, interquartile range; NEWS, National Early Warning Score.
Wilcoxon rank-sum p < 0.05.
Chi-square p < 0.05.
Supplemental Table 3 shows Cox regression for survival to 30 days in the whole cohort. Follow-up time (time in days between diagnosis and death or data extraction) was significantly shorter in the second wave compared with the first [14 (6–26) versus 19 (9–27), p < 0.0001]. Factors associated with increased risk of 30-day mortality were increasing age, male sex, presence of diabetes and admission NEWS. Compared with those in the first wave, those in the second wave had around half the risk of 30-day mortality [adjusted hazard ratio (aHR) 0.55, 95% confidence interval (CI) 0.40–0.75].
Separate multivariable Cox regression models for survival to 30 days in the first and second waves are shown in Table 3. In both pandemic waves, age, male sex and admission NEWS score are significantly associated with a higher risk of 30-day mortality. In the first wave, ethnicity did not impact upon 30-day mortality; however, in the second wave, as compared with White patients, those of Black ethnicity were almost 5 times as likely to die within 30 days (aHR 4.72, 95% CI 1.56–14.43). When the waves were analysed together, the interaction between pandemic wave and Black ethnicity was not significant (aHR 3.25, 95% CI 0.87–12.07, p = 0.079). When only complete cases are analysed, significant findings do not change.
Multivariable Cox proportional hazards model for survival at 30 days from COVID-19 diagnosis in the first and second pandemic waves.
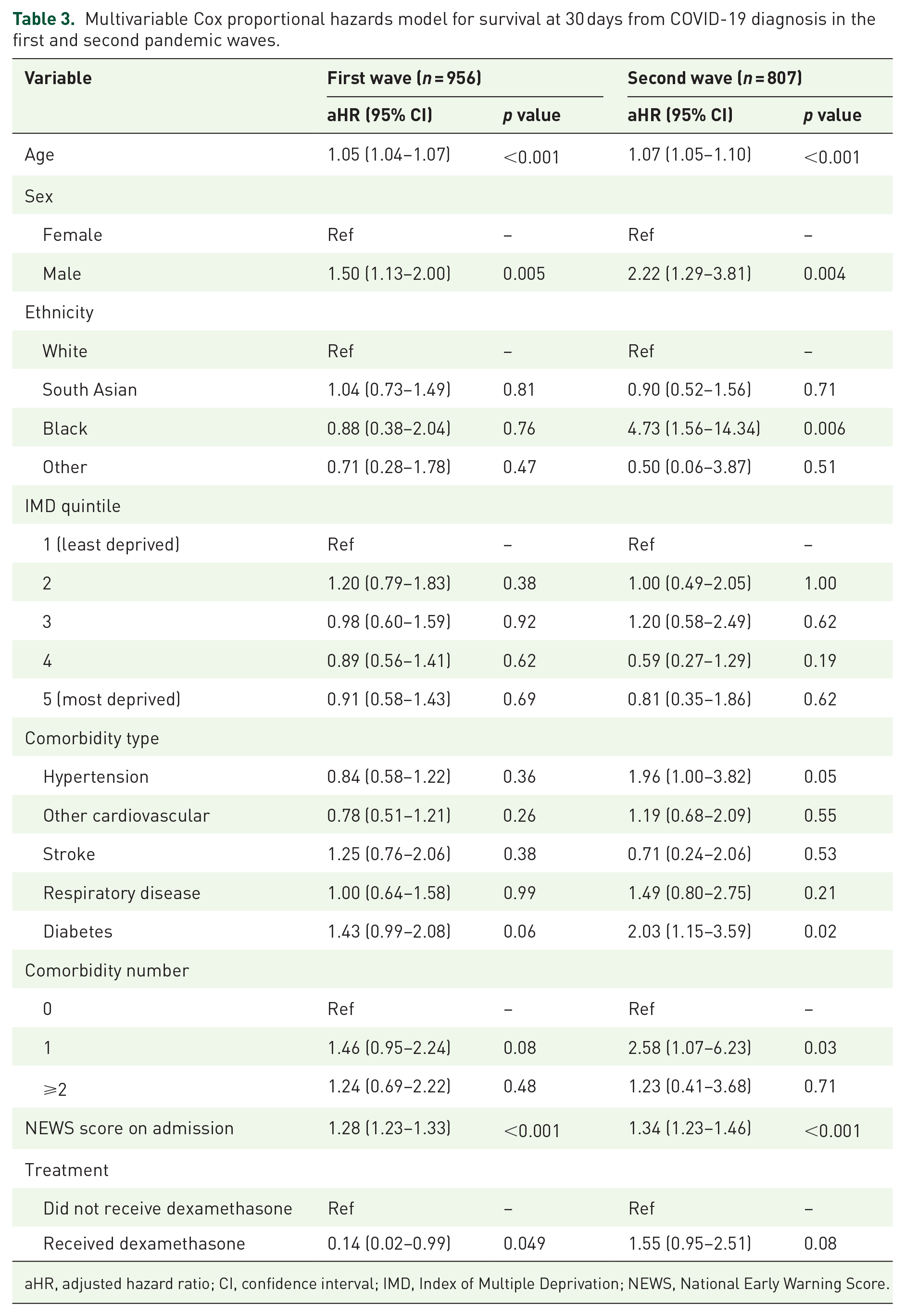
aHR, adjusted hazard ratio; CI, confidence interval; IMD, Index of Multiple Deprivation; NEWS, National Early Warning Score.
Discussion
In this study within an ethnically diverse cohort, we found a number of novel observations. First, there were no significant differences in demographic characteristics between patients in the first and second COVID-19 pandemic waves and that markers of severe COVID-19 (NEWS, requirement for mechanical ventilation and COVID-19-associated mortality) were lower in the second wave of the pandemic compared with the first. We found those of Black ethnicity were at higher risk of 30-day mortality than those of White ethnicity in the second wave.
COVID-19 case fatality rates have fallen in the second wave in 43 of the 53 countries with the highest total COVID-19-related deaths. 4 Foremost among the explanations for this observation is the increased availability of testing in the second wave which may increase the number of paucisymptomatic/asymptomatic individuals who are tested. In our study, the change in SARS-CoV-2 testing policy from symptom-driven testing to universal testing would be expected to pick up bystander infection in those attending hospital for reasons unrelated to COVID-19 and could generate bias towards a less symptomatic sample in the second wave. Other suggested mechanisms include the changing demographic profile of COVID-19 cases over time, with a smaller proportion of elderly patients (a group at high risk of adverse outcome from COVID-19) in the second pandemic wave with more people shielding,8,9 although we found no such differences in our dataset. In addition, in contrast to the early part of the first wave, there is an effective pharmacological treatment for severe COVID-19 (dexamethasone). 10 Clearly a reduction in COVID-19 mortality is desirable; however, increased survival from COVID-19 in the second wave may contribute to pressure on the healthcare system through increased hospital bed occupancy. Indeed our analysis shows that duration of hospital stay was longer in the second wave than in the first. Nonpharmaceutical interventions, such as mandating masks and lockdowns, may have also affected the number of patients infected and thus in turn, the number of deaths, but the city of Leicester remained in full lockdown (stay-at-home except for essential purchases, remote work, one exercise per day, cancellation of public gatherings and social events and no travel abroad) from 23 March 2020 to the end of the study.
We observed that mortality rates in the second pandemic wave may not be equal across ethnic groups. In our second wave cohort, those of Black ethnicity were at higher risk of 30-day mortality than White individuals (an effect not seen in the first wave cohort). These findings are opposite to a national UK study by Mathur et al., 11 which found that compared with the first wave, risks for death were attenuated for those from Black ethnic groups compared with White ethnic groups. Ethnicity is a social determinant of health. Therefore, it is likely that the differences in risks seen in our study reflect the number of infections that occurred in the local community and consequently, the number of people who present to hospital. A disproportionate number of infections in those of Black ethnicity may reflect socioeconomic factors, such as occupation, which prevent them from being able to stay at home on a second lockdown. Leicester is an especially ethnically diverse part of the United Kingdom, where approximately 30% of the population is born outside of Europe and North America, and individuals from the Indian subcontinent alone make up 15% of the population. 12 However, we recommend cautious interpretation of this finding, due to the small number of Black patients in our sample and the nonsignificant interaction between ethnicity and pandemic wave. Nonetheless, given the higher risk of SARS-CoV-2 infection and adverse outcome from COVID-19 in ethnic minority groups compared with those of White ethnicity,1,5,13,14 this observation requires urgent follow-up by larger studies with a greater proportion of Black participants and emphasises the importance of targeted interventions such as tailored public health messaging in these groups.
This study has limitations. Data are from a single centre and our data do not allow us to control for the change in testing policy between waves. We found a shorter median follow-up time in second wave patients. This is likely due to the fact that while the first wave data capture peak COVID-19 hospital admissions in early April with data extraction a month later, COVID-19 admissions continued to increase after data extraction for the second wave, meaning that a greater proportion of the second wave cohort were admitted close to the point of data extraction when compared with the first wave. However, we conducted an exploratory analysis excluding those admitted after the first wave ‘peak’ and still demonstrated a significant protective effect of being admitted during the second wave compared with the first. Mortality rates and severity of COVID-19 may change in subsequent waves of the pandemic, particularly with the emergence of the new variants of SARS-CoV-2. We only collected routinely available variables that were recorded within our clinical systems and as such, we do not have granular information – such as occupation – which may have affected their risk of infection. No patients in the United Kingdom were vaccinated at the time of study as part of national policy – these findings may be different now that a significant proportion of the UK population has been vaccinated. The emergence of novel variants, especially during the period of the second wave, may have had an impact on our study outcomes – however, genomic sequencing is not routinely performed in all our hospitals. Despite these limitations, our findings, particularly those related to ethnicity, must be investigated by larger studies to ensure that the inequalities of the first wave are not allowed to widen.
Supplemental Material
sj-docx-1-tai-10.1177_20499361221074569 – Supplemental material for Predictors of adverse outcome in the first and second waves of the COVID-19 pandemic: results from a UK centre
Supplemental material, sj-docx-1-tai-10.1177_20499361221074569 for Predictors of adverse outcome in the first and second waves of the COVID-19 pandemic: results from a UK centre by Christopher A. Martin, Daniel Pan, George Hills, Deborah Modha, Prashanth Patel, Laura J. Gray, David R. Jenkins, Linda Barton, William Jones, Nigel J. Brunskill, Pranab Haldar, Kamlesh Khunti and Manish Pareek in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
CAM and DP are National Institute for Health Research (NIHR) academic clinical fellows. LG and KK are supported by NIHR Applied Research Collaboration East Midlands (ARC EM). KK and MP are supported by the NIHR Leicester Biomedical Research Centre (BRC). The views expressed are those of the author(s) and not necessarily those of the NIHR, National Health Service (NHS) or the Department of Health and Social Care.
Author contributions
Conflict of interest statement
The authors declared the following potential conflicts of interest with respect to the research, authorship and/or publication of this article: KK is the Director of the University of Leicester Centre for Black Minority Ethnic Health, Trustee of the South Asian Health Foundation and Chair of the Ethnicity Subgroup of SAGE. MP reports grants and personal fees from Gilead Sciences and personal fees from QIAGEN, outside the submitted work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship and/or publication of this article: This work was supported by the National Institute for Health Research (NIHR). The funders had no role in design, data collection and analysis, decision to publish or preparation of the article. MP is supported by an NIHR Development and Skills Enhancement Award and funding from UKRI/MRC (MR/V027549/1).
Ethics statement
Ethical approval was not required after consultation with the National Health Service (NHS) Health Research Authority decision aid and the Caldicott Guardian and the study was approved as a service evaluation/audit (UHL10579).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
