Abstract
Bacterial infections are a major public health threat, with a substantial global burden of ∼5 million deaths in 2019, of which ∼1.27 million were attributed to antibiotic resistance. The formation of bacterial biofilms has significantly enhanced bacterial resistance to antibiotics. Worse still, it increases overall bacterial pathogenesis, contributing to inflammation and potentially to carcinogenesis in humans. Biofilm is implicated in approximately 65% of all bacterial infections and 78.2% chronic wound infections. Alarmingly, about 100–1000-fold increase in antibiotic concentration is required to eradicate bacteria within biofilms, further compromising the health of already ill-patients. Therefore, it is imperative to explore potential antibiofilm agents, especially ones with novel mechanisms of action, to clinically manage inpatient biofilms. Bacteriophage (phage) use is a promising evolutionary approach but is also challenged with potential resistance. Bacteria have developed several antiphage defense mechanisms, some of which exhibit synergistic antiphage activity. In this review, we provide several lines of evidence supporting the efficacy of phages against antibiotic-resistant clinical biofilm-forming bacteria. Observations reveal that phage enzymes disrupt biofilm structural components (e.g., EPS, pectate, and hyaluronic acid) and pave the way for phage infection of naked bacterial cells. We further provide insights into the recent advancements in phage use against biofilm-associated antibiotic-resistant bacteria in patients. Current knowledge shows that phages are rapidly evolving and counteracting antiphage bacterial mechanisms. Here, future perspectives to enhance phages efficacy against biofilm resistance are provided to establish their clinical antibiofilm application. Enhancing the clinical application of phages against biofilms requires addressing bacterial host biofilm resistance and optimizing strategies accordingly. Beyond phage cocktail and phage genetic engineering, conjugating phages with antimicrobial agents (eg., antimicrobial peptides) offers a compelling strategy to enhance phage antibiofilm efficacy.
Introduction
In previous centuries, sulfonamides, arsenicals, heavy metals, natural products, and drugs made by chemical tinkering with synthetic dyescrude were utilized in the treatment of a variety of bacterial infections, but these had significant side effects, limiting their systemic antibacterial application. 1 The discovery of penicillin by Fleming in 1928 and the subsequent development of various classes of antibiotics enhanced the golden age of the antibiotic era, where innumerable lives were saved from the devastating effects of bacterial infections. However, the widespread excessive dispensing, the irresponsible use of antibiotics, and the multifaceted bacterial capacity for “adversity-induced” evolution have resulted in the development of antibiotic-resistant bacterial strains.2–4 This has impacted the potency of antibiotics, and is rapidly driving bacterial infectious disease management into the “post-antibiotic era,” with many bacterial infections now requiring last-resort antibiotics in their treatment.5,6 Nevertheless, the introduction of lipopeptides in 1987 marked the last addition of a new class of antibiotics to the market, 7 leaving a gap of over three decades in antibiotic innovation. 3 The problem is further exacerbated by the continuous rise in infections caused by multidrug-resistant (MDR), extensively drug-resistant (XDR), and pan-drug-resistant (PDR) bacteria.8–11 The emergence and escalation of these drug resistance traits pose a major threat to public health on a global scale. A recent analysis indicates that antimicrobial resistant bacteria accounted for a substantial global burden in 2019. It was associated with an estimated ∼5 million deaths, of which ∼1.27 million were attributed to their resistance. 12 This menace is more concentrated in developing countries and may be higher than estimated due to the inadequate surveillance structures in these regions.12–16
The formation of bacterial biofilms increases overall bacterial pathogenesis, contributing to inflammation and potentially to cancer formation (carcinogenesis) in human systems. 17 Additionally, it significantly enhances bacterial resistance to antibiotics among patients, thereby facilitating bacterial dissemination to other potential sites for infection. This observation is supported by recent statistics that infections associated with biofilms account for approximately 65% of all bacterial infections and up to 78.2% of chronic wound infections.18–21 Worryingly, eradicating bacteria within biofilms requires a substantial increase in antibiotic concentration, often 100–1000 times higher than the usual dosage.22,23 This extremely high antibiotic concentration is not only toxic (e.g., nephrotoxicity) to consumers, but also erodes the protective physiological microbiota (dysbiosis). 24 Consequently, it is imperative to explore potential clinically applicable antibiofilm agents, especially ones with novel mechanisms of action 25 to overcome inpatient biofilms. The use of bacteriophages, also known as phages, has emerged as a plausible evolutionary approach.
Phages are natural predators of bacteria that target specific strains of bacteria and are the most abundant entity on earth, thereby making it possible to isolate phages specific to numerous clinically relevant bacterial strains. Phages can be grouped into two categories based on their life cycle: the lysogenic/temperate phage, which establishes a coexisting state with the bacteria by integrating its genetic material within the host bacterial genome, and the lytic phage (the type of phage under consideration in this review), which injects its genetic material into the host bacteria and utilizes the host ribosomes to synthesize its own proteins, thereby inducing bactericidal activity (Figure 1).19,26,27 Nonetheless, just like antibiotic resistance, phages are also challenged by bacterial resistance due to the multifaceted antibiotic resistance mechanisms of bacteria, especially biofilm-forming ones. This review, therefore, sought to provide insights into the basic science and recent advances of phage therapy for targeting antibiotic-resistant biofilm-forming bacteria. It further provides future perspectives on enhancing phage efficacy over biofilm-associated resistance to support its clinical application in antibiofilm therapy. Even though lytic phages are often combined with antibiotics in the treatment of biofilm-forming infections,28,29 this review focuses exclusively on the use of lytic phages.

The life cycle of lytic and temperate phages.
Phage interaction with bacterial biofilms
Bacterial biofilms are sessile communities of bacterial cells encased in a self-produced extracellular matrices. 30 They commonly adhere to biotic or abiotic surfaces, including lung tissues, medical implants, air–liquid interfaces, and inanimate surfaces as single-cell biofilms or mixed-species biofilms.31,32 The formation of biofilms involves a complex, yet finely regulated series of processes that commence with quorum sensing, and can be categorized into a few steps: (1) sensing, (2) initial contact with surface (reversible and irreversible attachments), (3) microcolony formation, (4) maturation and architecture, and (5) dispersion of biofilms (Figure 2).18,19,23,33 Biofilms of different bacterial strains may be made up of different matrices, but are generally comprised of core components such as a polysaccharide matrix, lipids (lipoprotein), fibrin, exopolysaccharides, proteins, nucleic acids; RNA, extracellular DNA (eDNA), ions like Ca2+, and other biomolecules to generate a hydrated, gelatinous, three-dimensional architecture biomaterial referred to as “extracellular polymeric substances” (EPSs), which is the core of biofilms.19,23,34–40 These mixed components provide mechanical stability, protect adhering bacteria from environmental assaults, enhance the default mode of growth, and impede the penetration of antibiotics by conferring rigidity and viscosity. The extracellular polymer matrix of the biofilm not only acts as a diffusion barrier but also possesses the capability of directly binding to antibacterial agents, effectively preventing their entry into bacterial cells.23,41–43 These culminate in the promotion of highly persistent bacterial infections, 44 due to the pathogens’ ability to withstand antimicrobials and evade the immune system.42,45 While biofilms prevent the entry of stressors and antibacterials, cells within the biofilm perform both active and passive diffusion for their movement and any associated molecules transfer, respectively. These movements prevent the bacteria from being trapped in a confined zone. 23

The biofilm formation cycle and bacteriophage invasion at each stage.
As with many other antibacterial agents, phages, which are highly specific in their actions toward bacterial species, first penetrate the biofilm structures that surround the bacterial colonies via intricate interactions with the components of the biofilm. Despite this prerequisite to therapeutic application on bacterial biofilms, phages are nonmotile and rely on Brownian motion, which is a random movement resulting from collisions with surrounding molecules, to navigate their environment and reach their targets.46,47 This, therefore, presents the perplexing question of whether phages can depend on Brownian motion to successfully traverse the biofilm and reach the target bacterial cells. This is, however, not entirely the case—the interaction of phages with these biofilm components involves not only the phage as a whole but proteins and enzymes produced by the phages. These enzymes and protein molecules enhance the favorable antibiofilm interaction between phages with biofilms. In this context, depolymerase enzymes, produced by some phages
48
degrade the polysaccharide layer of biofilms, resulting in potential damage to the biofilm structure by targeting the stages of microcolony formation and biofilm maturation.
48
In the case of naïve bacteria or bacteria with biofilm in the early development stages, the bacterial cells face internal lysis by lysine and putative T4-like lysozyme, effectively inhibiting their proliferation and preventing them from reaching the critical density required for mature biofilm generation.
48
Matured biofilms, on the other hand, can also be raptured with the aid of depolymerase enzymes of phages, degrading the bacterial polysaccharides,
23
alongside dispersing the chemical components of the biofilm matrix. A recent study by Park et al. identified a novel depolymerase, Dpo10, which exhibits the ability to specifically bind to and degrade the lipopolysaccharide of
Regardless of whether phages kill the bacterial cell, they penetrate and damage the biofilm during the biofilm control process.48,50 In addition to depolymerase and lysozymes on biofilm during the early and mature stages of biofilms, phage endolysins also digest the extracellular polymeric substance (matrix) of biofilms
51
and also weaken the bacterial cell wall by degrading the peptidoglycan, allowing for lysis and bacterial cell death (Figure 2).
52
The action of this enzyme potentially paves the way for phage entry into the bacterial colonies by destroying the protective layer of the biofilms. Additionally, the enzyme potentially enhances phage proliferation by weakening the bacterial cell walls to release phage progeny to infect other bacterial cells, consequently resulting in bactericidal activity. For instance, Schuch et al.,
53
in their assessment of phage-derived lysin CF-301 in clearing bacterial biofilms under different conditions, saw a complete eradication of
Moreover, phage itself can prevent the formation of mature biofilms from planktonic bacteria or biofilm in its early stages (Figure 2). An illustration of this is phage Bxb1, which when integrated into the genome of
Summary of phage encoded enzymes that degrade bacterial biofilms.


The interaction of phage enzymes with biofilm. (a) Enzymatic degradation of biofilm components by various phage enzymes and exposing naked bacterial cells. (b) Phages and lysins attach to and lyse naked bacterial cells.
Additionally, a selective combination of several lytic phages of different host range(s) into a single-phage cocktail formulation also provides a multifaceted interaction of phages with biofilms. While some phages in cocktails produce depolymerases, other phages that are lysis-producing or lysozyme-producing also generate their respective enzymes. This induces a simultaneous attack on biofilm components, and when bacterial cells are exposed, phage tails bind to specific receptors on bacterial cells to undergo the lytic life cycle. For example, Chhibber and his team reportedly reduced the mixed-species biofilm of
Bacteriophage against biofilms in patients and clinical isolates
Clinical isolates in vivo and animal models
Due to ethical limitations on the use of phage therapy in patients except as compassionate treatment in some countries,
65
its application remains largely limited to in vivo studies, where it yields encouraging results in many cases (Table 2). In the application of phages to control bacterial biofilms, there is an uneven distribution of studies across bacterial species. The strong biofilm formation of
Phage-mediated treatment of biofilms in preclinical studies; animal models and ex vivo or in vivo studies.
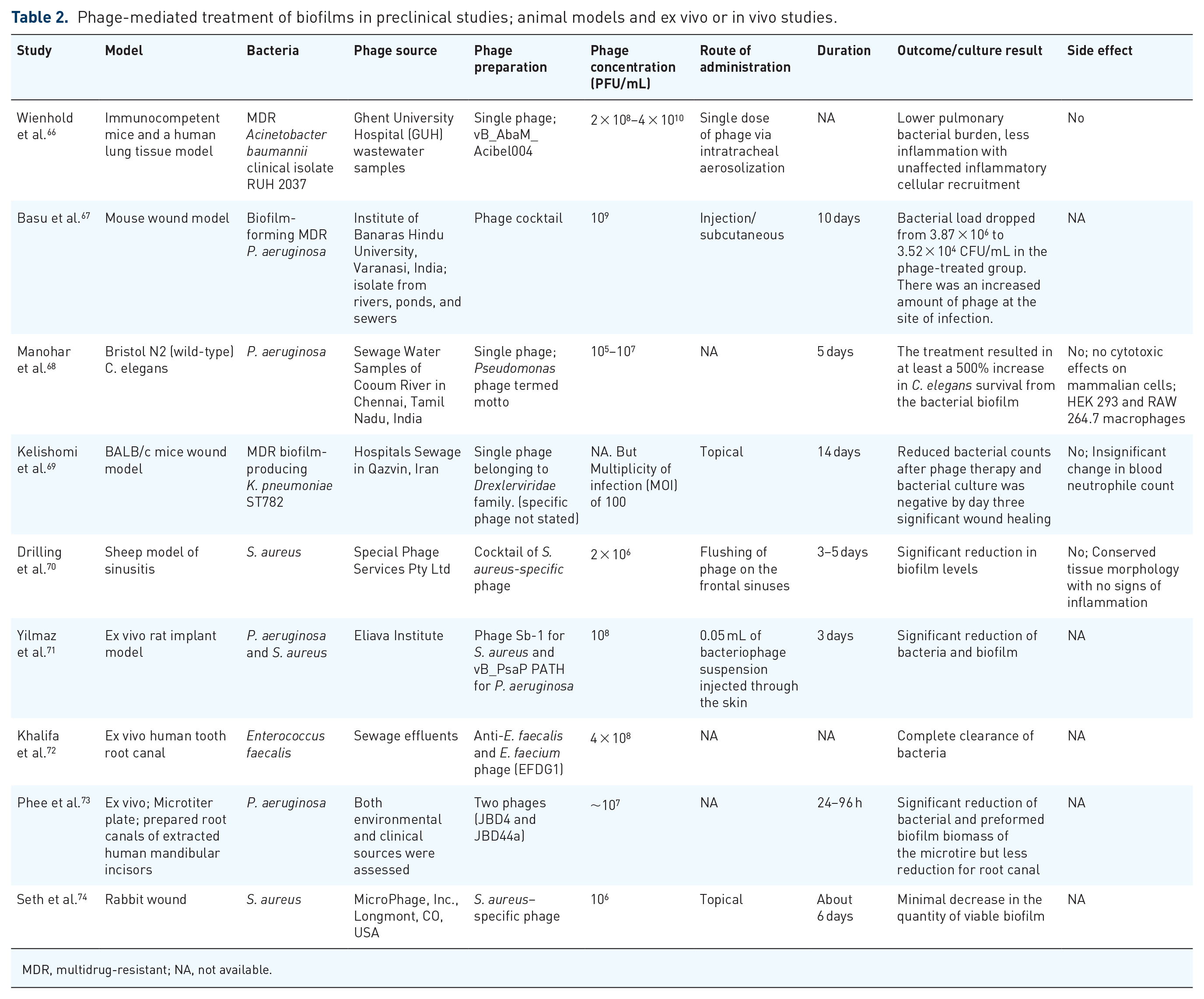
MDR, multidrug-resistant; NA, not available.
P. aeruginosa biofilm
Efficacy of monophage formulation against clinical isolates of P. aeruginosa biofilms
In a case report that attempted to determine the required minimum dosing of phage OMKO1 for the removal of
Jeon et al. conducted a comprehensive in vitro, in silico, and in vivo study, utilizing acute pneumonia mouse models to assess the efficacy of two novel phages (BΦ-R1836 and BΦ-R656) in fighting XDR
Efficiency of phage cocktail targeting clinical isolates of P. aeruginosa biofilms
In one of their studies, Debebe and his team investigated the efficacy of two phages (ΦNH-4 and ΦMR299-2) in combating nine clinical
Moreover, in their 2018 study, Forti et al. designed a six-phage cocktail, including two previously examined phages (PAK_P1 and PAK_P4) and four newly discovered phages (E217, PYO2, E215, and DEV), that effectively cleared MDR strains of
An in vitro study aimed at testing the activity of environmentally isolated phage against
Efficacy of phage formulation against E. faecalis, S. aureus , and A. baumannii biofilm
In addition to effectively lysing significant species of bacteria (Table 2), phages have demonstrated immense antimicrobial activity against biofilm-producing
Bacteriophage against biofilms within patients
Biofilms play a crucial role in enabling bacteria to endure in hostile environments, contributing to chronic infection, 88 as well as facilitating the dispersal of bacteria to different sites, leading to the initiation of new infections. 89 Phages are biocompatible and can be cleared via hepatic elimination by phagocytosing phages, making them a favorable applicable therapeutic agent. However, phages are challenged by with shortfalls such as short half-life and instability under varying pH.90–92 Concerning these shortfalls, the mode of phage administration to patients, which greatly influences phage–antibiofilm activity and adsorption, needs to be carefully determined based on the nature and location of the infection. 93 The findings from several preclinical studies suggest that to achieve a significant reduction in biofilms, it is necessary to administer phage directly to the precise location of the biofilm infection, 47 as observed in wound infection, where topical administration is the preferred mode of therapy.94,95 However, this idea becomes difficult to adopt when the site of infection is an internal part of the body and must therefore rely on indirect application or systemic delivery. Intravenous, intraoperative, and intrarectal phage administration are relatively unproblematic.93,95,96 These routes provide direct access to the bloodstream and relatively facilitate effective phage penetration into various tissues, particularly those affected by infection-induced inflammation, enhancing the permeability of the endothelial barriers as demonstrated in humans96,97 and animal models such as rats.93,98 Oral administration of phages is also quite effective, but inconsistent levels of different phages in the bloodstream after oral administration have been reported. Its adoption in clinical trials should therefore be preceded by preclinical experiments. 93 In the case of respiratory (lung and other pulmonary) biofilm-related infections, inhaled formulations (nebulization and other hydrogel formulations) have yielded promising results (Table 3), even though the loss of phage viability may be encountered. 99 Successful phage treatment via any of these routes is warranted by a variety of phage concentrations ranging from 104 to 1010 (Table 3).
Phage-mediated treatment of biofilms in human studies only.

Shown only are studies in which biofilm presence and treatment have been explicitly indicated.
MDR, multidrug-resistant.
Bacterial biofilms have been cleared in many parts of humans (summarized in Table 3), including, but not limited to the skin, femur, skeletal muscle, lungs, genitourinary tract, and biofilm-contaminated invasive devices. One such piece of evidence is a case report by Cano et al.
103
in which in vitro experimentation and subsequent therapeutic application on a 64-year-old patient was carried out at the Mayo Clinic Clinical Research and Trials Unit, a recovery of the knee function was observed. In the preclinical study, the treatment of
Another convincing outcome of lytic phage therapy was observed in another case study on a 72-year-old male with morbid obesity and hyperlipidemia with chronic MRSA prosthetic joint infections. 107 To successfully sterilize the patient’s chronic MRSA from the prosthetic joint infection, two doses of a selectively purified phage SaGR51Φ1 at 5.4 × 109 PFU were intra-articularly administered in 10 mL of normal saline at the end of surgery, after which antibiotic (IV daptomycin 1000 mg) administration was started and the subsequent daily intravenous phage (5.4 × 107 PFU/mL) administration. After 2 months, intraoperative cultures were again culture-negative.
There are documented instances of successful oral biofilm elimination, which is a key objective in preventing tooth decay. Phage SMHBZ8, obtained from samples of human saliva, demonstrated significant efficacy in preventing the formation of biofilm and reducing existing biofilm of
Recent advances by which bacterial cells embedded in biofilm can confer additional resistance against phages
Apart from the general inherent resilience of bacterial biofilms as shields against antibacterial agents (including antibiotics and phages), bacteria within biofilms employ a strategy of upregulating specific essential biofilm substances to capture viral particles, hindering phage invasion and safeguarding bacterial cells from potential damage.23,109 This is highly unfavorable to phages due to their movement being Brownian Motion. In addition to the shielding mechanism of biofilms, the bacterial cells within the biofilm also have recently been observed to protect themselves against their viral predator phages in a multifaceted manner: the release of phage-receptor decoys, release of outer membrane vesicles (OMVs), altering or concealing viral receptors, superinfection exclusion (Sie), retron production, expression of DNA-intercalating metabolites, use of toxin–antitoxin system, use of restriction-modification (R-M) systems, BREX system, abortive infection (Abi) systems, CRISPR-Cas and other mechanisms to counter the invasion of phage DNA (Figure 4).19,32,38,110–118 These novel defense systems possess a wide range antiphage defense mechanism, as noted by Ofir and his group in the case of a defense island system associated with restriction-modification (DISARM) in restricting incoming phage DNA. 113 Many of the defense systems exhibit synergistic antiphage activity. In this domain, Wu et al. 119 demonstrated that Zorya II synergizes with Druantia III and ietAS defense systems, while tmn exhibits synergy with co-occurring systems Gabija, Septu I, and PrrC.

Mechanisms of bacterial resistance to phage.
Release of phage-receptor decoys, OMV, and altering or concealing viral receptors are important separate mechanisms adopted by bacteria, enabling them to survive in the presence of phages. The outer membrane of Gram-negative bacteria has phage receptors that can be secreted and serve as decoys, causing phages to irreversibly bind to these decoys. This results in futile infection attempts by the phages. An example of this phenomenon is observed in
Expression of phage DNA-intercalating metabolites has been reported among some bacteria to confer antiphage activity. A study investigating molecules that inhibit phage φScoe2 and φScoe25, as well as
Sie is a bacterial defense phenomenon in which a preexisting viral infection prevents subsequent infections by closely related viral particles. 123 Bacteria that are capable of this mechanism can restrict the entry of additional phages into an already infected cell by producing proteins to hinder the secondary infection. For instance, protein Imm and protein Sp encoded by phage T4 prevent subsequent infection of the same bacteria by other similar T-even phages by ensuring the bacterial peptidoglycan is free from degradation, thereby hindering the phage DNA transfer across the membrane.124,125 When a Sie is bypassed and the bacteria are infected, Abi systems are triggered in some bacterial cells; a self-destruct mechanism that leads to the premature death of the infected cell (“suicide of the infected bacteria”).19,126 The precociously death of the infected cells prevent the concomitant replication and spread of phages, safeguarding the rest of the bacterial community from phage invasion.126,127 Additionally, retrons, which are bacterial genetic elements that produce small RNA molecules that can interfere with viral replication or other viral processes also activate their Abi system. This was evident when Millman et al. examined multiple retron systems and demonstrated their ability to confer defense against a wide array of phages through the mechanism of abortive infection. 112 Furthermore, some bacteria employ toxin–antitoxin systems as a defense mechanism to counter phage invasion 128 but the mechanism of action underlying this is currently elusive.
R-M, BREX, and CRISPR-Cas systems are separate and highly diverse mechanisms adopted by bacteria to enable them to survive phage predations via recognition and cleavage of phage DNA at specific sites while protecting their own genomes.19,129 R-M systems, comprising restriction endonucleases and methyltransferases as their two essential components, are vital defense mechanisms adopted by bacteria in preventing phage DNA invasion. The endonucleases recognize and cleave specific DNA sequences within the phage genome, rendering it nonfunctional, and further providing innate immunity against foreign genetic material. To prevent the bacteria’s DNA from being damaged by the cleaving ability of the endonuclease, methyltransferases serve a protective role in the R-M systems, methylating the host bacterial DNA at the same recognition sites, safeguarding it from the activity of the restriction endonucleases.130,131 Similar to the R-M system, BREX system also prevents phage genome replication, but its underlying mechanism is yet to be elucidated. It comprises methyltransferase (BrxX) and ATPase (BrxC). The BrxX is said to methylate adenine in the bacterial DNA, 118 while the latter may be providing energy to the system. This methylation is proposed to be the distinguishing feature of the bacterial genome and the phage genome. 132 CRISPR-Cas systems are adaptive immune defense systems in some bacteria, providing defense against phage and plasmids. These systems consist of CRISPR arrays, which are composed of short repetitive sequences interspersed with unique spacer sequences derived from previous phage encounters. When a phage invades, the Cas proteins associated with the CRISPR array use the spacers as a guide to recognize and cleave the phage DNA, effectively neutralizing the phage while maintaining the integrity of the bacterial genome.133,134
In addition to these underlying and novel resistance mechanisms, selecting the type of phage for biofilm eradication should be highly regarded, as it may enhance biofilm formation or tolerance. For instance, if selectively lytic phages are not carefully chosen, filamentous phages might be mistakenly used, resulting in the enhancement of bacterial stability and their biofilms. An example includes the interaction of filamentous phage with host and microbial polymers, resulting in the formation of a more complex liquid crystal structure within the biofilm matrix, thereby improving adhesion, survival in dry conditions, antibiotic resistance, tolerance, virulence, and dispersion capabilities of the biofilms.135,136
Mechanisms by which phages overcome bacterial resistance
To counter these resistance mechanisms, some phages rapidly evolve to carry genes encoding anti-R-M and anti-CRISPR proteins like AcrF1-5 (extensively reviewed in Refs. 137 and 138)), while other phages introduce mutations into the specific sequences targeted by CRISPR-Cas systems, including protospacer.137,139 Phages have also evolved to encode antitoxins produced by their host. For instance, ToxN toxin produced by
Moreover, in response to bacteria concealing phage-binding receptors and releasing phage-receptor decoys, some phages have evolved to recognize alternative receptors on bacterial surfaces. 143 In superinfection, where a first-time infection prevents subsequent infection by similar phages, the rapid phage mutation rate has enabled the bypassing of these bacterial defense mechanisms. For instance, the phage P22 encodes the SieA protein, which blocks DNA injections by other P22-like phages. However, mutant strains of phageP22 with amino acid changes at the C-terminal regions of the gene 16 and gene 20-encoded ejection proteins can overcome this block. 144
In cases where bacteria completely resist their predator phage, they often experience a fitness cost. This trade-off can render the bacteria more susceptible to other phages with different mechanisms or other antibacterial treatments due to the metabolic burden of maintaining energy and resources for the resistance.
145
Essentially, the metabolic burden of bacterial resistance mechanisms can slow down the bacterial growth rate, as observed in antibiotic-resistant bacteria.
146
While the bacteria may become resistant to one type of phage, they may become deficient in virulence traits,
147
including biofilm formation.
148
A recent study confirmed impaired biofilm formation of phage-resistant
Future perspectives on optimizing phage therapy against biofilms
The key to effectively treating biofilm-associated bacterial infections lies in preventing the biofilm maturation from the early stages of development, allowing conventional antibacterial agents to easily access the nascent bacterial cells. However, when dealing with preformed or mature biofilms, the successful eradication of biofilms depends on the capacity of antibacterial agents to rupture or penetrate the bacterial biofilm involved (Figures 2 and 3). Unfortunately, only a limited number of antibiotics possess such remarkable capabilities. Therefore, optimizing the antibiofilm activity of phages would be of utmost importance in advancing its therapeutic application in the near future (Figure 5).

Perspectives on enhancing the antibiofilm activity of phages.
Phage cocktail
The utilization of phage cocktails, harnessing the combined effect (sometimes synergistic) of individual phages, potentially enhances phage adsorption and spread within bacterial biofilms to lyse bacterial cells, as elaborated earlier. An example of this is when a phage enzymatically (e.g., depolymerase) breaks down the EPS structure of a multispecies biofilm, including those produced by its host and closely related species. This enzymatic action potentially improves the accessibility of other phages to their respective hosts within the biofilm.50,63,150
Furthermore, considering that, the isolation of phages that can lyse a variety of hosts (broad host range) is increasing, combining broad-host-ranged phages, such as the novel phages (e.g., ΦParuNE1 151 and HZ2201 152 known for their respective potent antibiofilm activity) into a phage cocktail can result in a highly effective treatment against a range of pathogens. This approach reduces the number of phages in the cocktail while maintaining their efficacy, simplifying the purification and safety assurance processes. Utilizing a smaller number of phages with a broad host range can also lead to cost savings in the formulation of the phage cocktail.
Genetic engineering as a beneficial tool for phage therapy
Harnessing the power of genetic engineering to address the limitations of phages holds tremendous potential for the effective eradication of biofilm infections. Incorporating genetic engineering techniques that augment phage shortfalls would empower phage therapy to overcome the inherent shortcomings in combating and eradicating bacterial biofilms. Phages can be engineered to incorporate anti-CRISPR proteins and anti-R-M system proteins to avoid phage DNA cleavage,153,154 as well as antitoxin proteins to neutralize bacterial secreted toxins. However, before these features come into play, phages must disrupt or penetrate the biofilm to reach the individual bacterial cells. In this regard, phages that lack genes encoding degradative enzymes can be genetically modified to express such enzymes. This enhances their ability to adsorb onto, penetrate, and diffuse through the exopolysaccharide matrix, ultimately facilitating biofilm destruction.
155
For instance, a study by Pei & Lamas-Samanamud engineered phage T7 to incorporate a lactonase enzyme with a wide-ranging capability to quench quorum sensing. This cell–cell communication mechanism (quorum sensing), mediated by acyl homoserine lactones (AHLs), is crucial for biofilm formation, particularly in the early stages. In this study, the authors demonstrated that an engineered T7 phage expressing the AiiA lactonase effectively degraded acyl homoserine lactones from various bacterial species. When introduced to mixed-species biofilms containing
In another study by Wu et al. that isolated and sequenced a phage SH-KP152226 (encoding depolymerase Dep42) that exhibits a specific affinity for infecting and lysing
Phage-based supramolecule development
A promising avenue for combating biofilms lies in the development and utilization of phage-based supramolecules (PBS). These supramolecules can be crafted by amalgamating phages with enhancing agents and other antibacterial components, like antimicrobial peptides, to cause direct damage to bacterial cells or as a delivery method of the phage. The resulting supramolecules exhibit a diverse range of forms, including broad-spectrum variants, while possessing inherent antibiofilm properties. This unique characteristic empowers these supramolecules to disrupt and ultimately eliminate both single and mixed bacterial species residing within biofilms.164–166 An important feature of developing phage-based supramolecules is the opportunity to alter the overall size of the developed supramolecule, as it offers larger multivalent surfaces that can be manipulated to provide selectivity against bacteria. Controlling the composition, size, and surface properties of these PBS offers a considerable tendency to generate an antibacterial agent with a diverse array of antibacterial activities. In a recent study, Rotello et al.
167
described that cationic nanomaterials are capable of penetrating biofilms. Synthesizing a supramolecule made of these nanomaterials coupled with appropriate phages may yield a promising antibiofilm effect. This is demonstrated using phage T4 with chitosan and polyethylene imine-coated alginate beads to treat
While the clinical implementation of PBS has been limited due to safety concerns, individual components of PBS have been shown to be independently biocompatible with human cell lines through in vivo and in vitro studies. This is demonstrated in a recent study where the combination of phage T7 and AgNPs was highly effective on bacterial biofilms and was safe for Jurkat E6-1 cells and adult human dermal fibroblasts, except that AgNP was observed to be toxic at a very high dose of 0.1 mg/mL. 170
Limitations and challenges of the prospects in developing phage therapies for clinical use
These perspectives on enhancing phage–antibiofilm activity hold promise, but they are not without limitations. Safety concerns surrounding phage therapy remain significant due to our limited understanding of its pharmacodynamics and pharmacokinetics. In addition to the controversies on strict ethical regulation of genetically modified therapies, 171 the genetic modification of phages to express degradative enzymes and the use of phage cocktails may face challenges such as the complexity of ensuring consistent and effective phage delivery to biofilms. Additionally, the clinical implementation of phage-based supramolecules may be hindered by safety concerns, including immunogenicity and potential toxicity, as well as the need for extensive research to validate their efficacy and biocompatibility.
Conclusion
This review has provided insight into the basic science and recent advances of phage therapy against biofilm-forming antibiotic resistance. It also highlighted some challenges with biofilm resistance to phages, as well as potential benefits of optimized phage administration systems. It is evident that phages are efficacious antibiofilm agents. However, the intricate interaction at the molecular level of how phages interact with biofilms is not fully exploited. Also, the biochemical interaction of phage enzymes with biofilms remains to be elucidated. Understanding these key concepts that underlie phage–biofilm interaction is imperative to the development of modified systems of phage-derived proteins and enzymes for combating clinically relevant antibiotic-resistant biofilms. In addition to phage cocktail and phage genetic engineering strategies, it would be promising to channel future studies into conjugating phages with other antimicrobial agents such as peptides to enhance phage antibiofilm efficacy.
