Abstract
Background:
Moderate-risk ampC beta-lactamase-producing Enterobacterales (HECK-Yes organisms) render many beta-lactams ineffective.
Objective:
This study evaluates selective reporting (SR) of antimicrobial susceptibility testing (AST) results to improve antibiotic prescribing for these infections.
Design:
A retrospective quasi-experimental study evaluating patients before and after the implementation of SR.
Methods:
SR of AST results for HECK-Yes organisms was implemented at a 1500-bed medical center. A retrospective study compared antibiotic prescribing before and after implementation in patients with positive blood or respiratory cultures.
Results:
Fifty patients were included in both pre- and post-implementation groups with similar baseline characteristics. Post-implementation, appropriate antibiotics within 24 h of AST report increased by 24% (62% pre vs 86% post, p = 0.01). A total of 30-day mortality, clinical success, and microbiological failure rates were similar between groups.
Conclusion:
SR improved appropriate antibiotic prescribing for moderate-risk ampC-producing Enterobacterales (e.g., HECK-Yes) infections.
Introduction
Antimicrobial stewardship programs (ASPs) in collaboration with microbiology departments have long utilized selective reporting (SR) as a strategic approach to influence antibiotic prescribing. In selective reporting, susceptibilities of certain antimicrobials are deliberately withheld, a strategy most often used to curb excessive use of broad-spectrum beta-lactams and fluoroquinolones.1–4
ASPs have traditionally leveraged SR to reduce the overuse of broad-spectrum antimicrobials and slow the development of resistance. We hypothesized that SR could further optimize antibiotic prescribing for infections caused by a distinct group of organisms collectively referred to as HECK-Yes (Hafnia alvei, Enterobacter cloacae, Citrobacter freundii, Klebsiella aerogenes, and Yersinia enterocolitica). This group of Enterobacterales poses a moderate risk for clinically significant production of ampC, an enzyme capable of hydrolyzing many beta-lactams.5,6 Emergence of beta-lactam resistance has been reported to be as high as 20% for HECK-Yes organisms, upon exposure to certain beta-lactam antibiotics.7,8 Consequently, the risk of clinical failure is higher with certain beta-lactams compared to others. 9
HECK-Yes organisms demonstrate intrinsic resistance to strong inducers/good substrates of ampC, including aminopenicillins, amoxicillin-clavulanate, first-generation cephalosporins, and cephamycins. 5 Resistance to weak inducers/good substrates may develop after exposure to these antibiotics, including piperacillin-tazobactam, aztreonam, and third-generation cephalosporins. In contrast, beta-lactams such as cefepime and carbapenems are more stable against ampC-mediated hydrolysis and have been associated with increased treatment success. 10
Methods
Study design
This retrospective, multicenter health system, quasi-experimental study aimed to compare antimicrobial prescribing practices before and after implementing SR for HECK-Yes organisms at Montefiore Medical Center (MMC). This study was conducted and reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement. 11
To steer ordering providers toward optimal antibiotic selection for infections with HECK-Yes organisms, we developed and implemented SR as shown in Figure 1. Beta-lactams categorized as strong inducers/good substrates of ampC are entirely suppressed from the susceptibility panel. Weak inducers/good substrates that risk the development of resistance after exposure, including piperacillin/tazobactam and ceftriaxone, are also excluded from the panel (of note, these antibiotics may still be considered for the treatment of simple cystitis, therefore, our group also developed a separate urine isolate cascade not discussed herein). Beta-lactams of choice, including cefepime and carbapenems, are released in the first panel. More extended-spectrum agents, such as meropenem/vaborbactam and ceftazidime/avibactam, are released only if resistance to agents in the first panel is demonstrated. Non-beta-lactam antibiotics such as ciprofloxacin and trimethoprim-sulfamethoxazole are also included within the panel. Such antibiotics are considered appropriate therapy if susceptibility is demonstrated, given that they are not impacted by ampC-mediated hydrolysis.
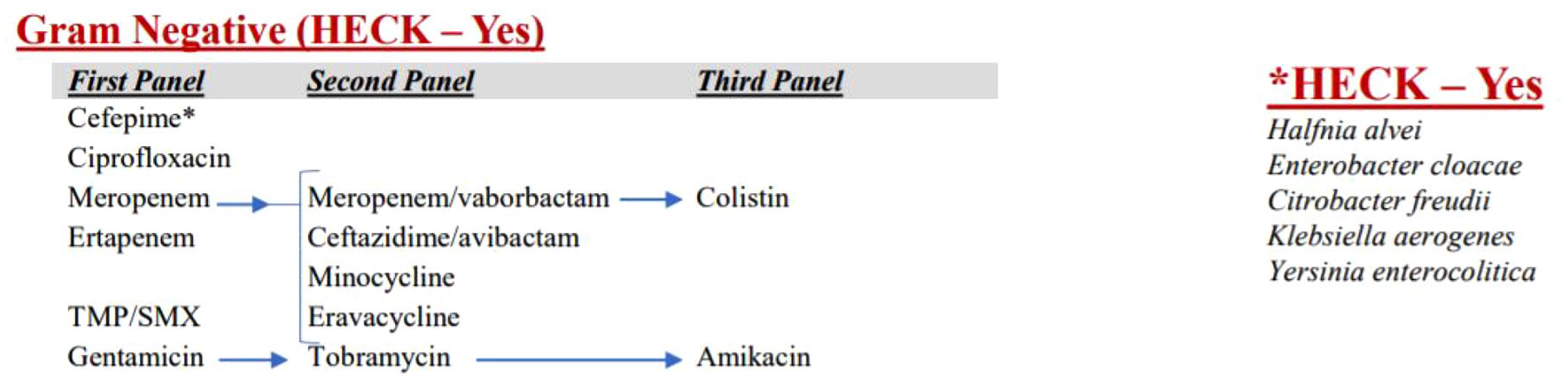
HECK-Yes selective reporting cascade algorithm.
Pre-implementation data were collected from July 1, 2021, to April 30, 2022, and post-implementation data were collected from July 1, 2022, to April 30, 2023. Patients were included if they were
Outcomes
The primary outcome was appropriate antibiotic therapy within 24 h of the antimicrobial susceptibility testing (AST) report, as defined by criteria outlined in the Infectious Diseases Society of America (IDSA) guidance on the treatment of antimicrobial-resistant gram-negative infections. 9 Secondary outcomes included time to appropriate antibiotic order and administration after AST report, 30-day mortality, clinical success, microbiological failure (for blood culture isolates), duration of antibiotics for HECK-Yes infection, and length of stay from index culture. Clinical success was defined as survival at 30 days with documented clinical improvement, including resolution of fever (<100.4°F) and/or leukocytosis (<11*103 cells/mm3) by the end of therapy. Microbiological failure, assessed only for bloodstream infections, was defined as failure to clear blood cultures within 72 h of antibiotic initiation.
Data collection
Baseline demographics, empiric antibiotic therapy, definitive antibiotic therapy, microbiologic data, 30-day mortality, and length of stay were collected from the electronic health record (EHR) and stored on an encrypted and secure file.
Statistical analysis
We determined a sample size of 79 based on an anticipated increase in appropriate antibiotic prescribing from 60% in the pre-implementation group to 80% in the post-implementation group. To accommodate margins of error, we enrolled 50 patients in both groups. Descriptive statistics, Student’s t-tests, Mann–Whitney U, or Fisher’s exact tests were performed to assess differences between pre- and post-implementation groups, as appropriate. A significance level of alpha <0.05 was applied for statistical significance.
Results
A total of 100 patients were included in the analysis. Baseline characteristics were similar between the two groups (Table 1). A similar proportion of patients received an infectious diseases consult in the pre- and post-implementation groups (92% vs 98%, respectively). No significant differences were observed between the groups regarding markers of illness severity, including intensive care unit stay (48% pre vs 40% post, p = 0.55), need for mechanical ventilation (40% pre vs 42% post, p = 1.00), the vasopressor requirement (40% pre vs 30% post, p = 0.40), and renal replacement therapy (14% pre vs 24% post, p = 0.31).
Baseline characteristics.
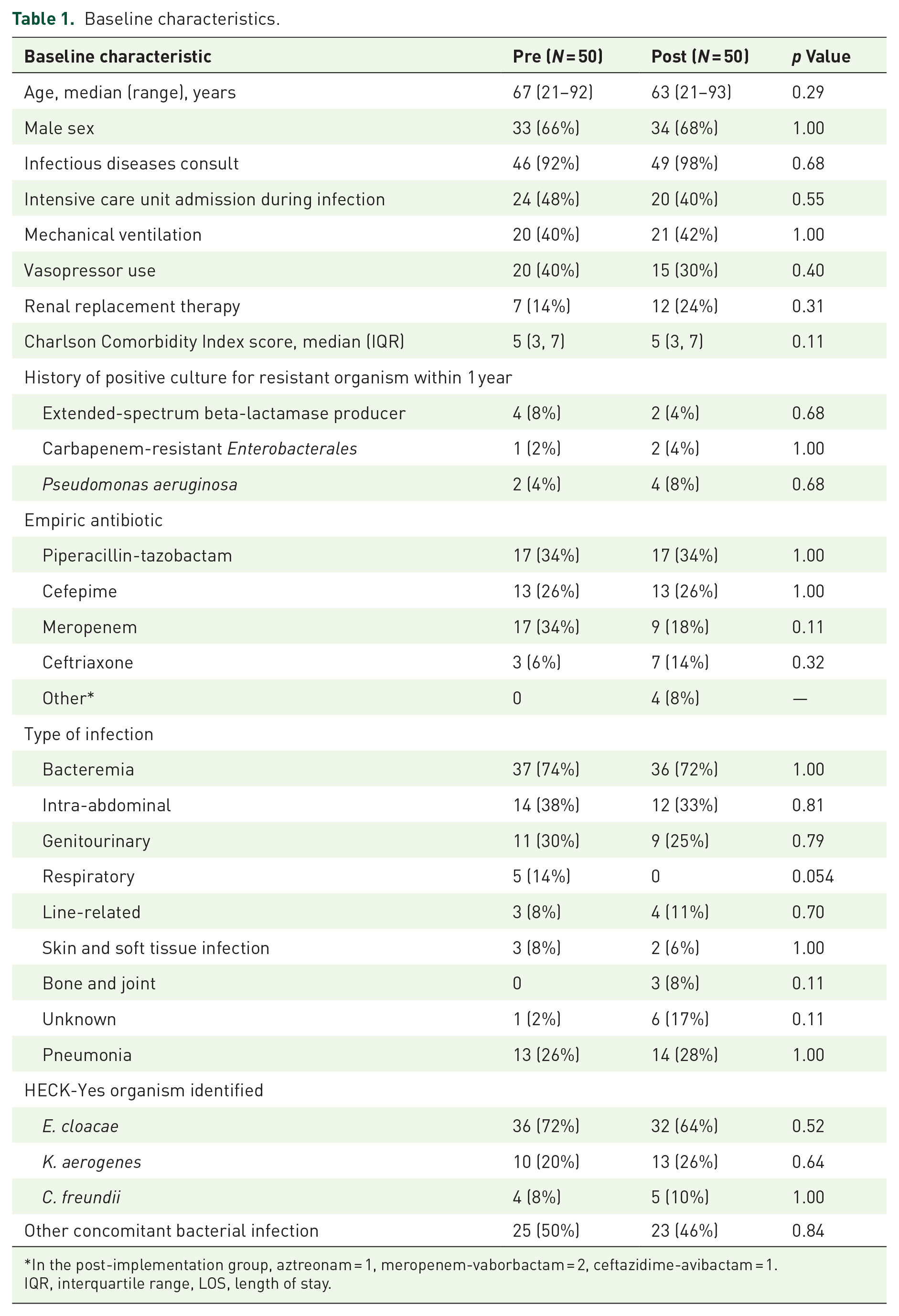
In the post-implementation group, aztreonam = 1, meropenem-vaborbactam = 2, ceftazidime-avibactam = 1.
IQR, interquartile range, LOS, length of stay.
Rates of positive cultures within 1 year for extended-spectrum beta-lactamase producers (8% pre vs 4% post, p = 0.68), Pseudomonas aeruginosa (4% pre vs 8% post, p = 0.48), and carbapenem-resistant Enterobacterales (2% vs 4%, p = 1.00) were similar between groups.
The majority of patients in both groups had bloodstream infections (74% pre vs 72% post, p = 1.00), with the most common sources being intra-abdominal (38% pre vs 33% post, p = 0.81), and genitourinary (30% pre vs 25% post, p = 0.79). Enterobacter cloacae was the most common organism isolated (72% pre vs 64% post, p = 0.52), followed by K. aerogenes (20% pre vs 26% post, p = 0.64), and C. freundii (8% pre vs 10% post, p = 1.00). No isolates of H. alvei or Y. enterocolitica were found in either group. Rates of concomitant bacterial infections were similar (50% pre vs 46% post, p = 0.84).
The majority of patients in each group received empiric piperacillin/tazobactam (34%) and cefepime (26%). The released AST report prompted antibiotic changes in nearly half of the patients in each group (44% pre vs 50% post, p = 0.69). During the pre-intervention period, significantly more patients were switched to an inappropriate antibiotic (41% pre vs 4% post, p < 0.01). Conversely, there was a significant increase in patients switched to appropriate antibiotic therapy in the post-implementation group (45% pre vs 80% post, p < 0.01).
A significantly higher rate of appropriate antibiotic therapy within 24 h of the AST report was observed in the post-implementation group (62% pre vs 86% post, p = 0.01; Table 2). No significant differences were observed between groups for any secondary outcomes, including time from release of AST report to appropriate antibiotic order or administration, total duration of antibiotics, 30-day mortality, clinical success, microbiologic failure for bloodstream infections, or length of stay. A subgroup analysis of bacteremic patients revealed similar outcomes to those of the whole cohort (Table 2).
Outcomes.
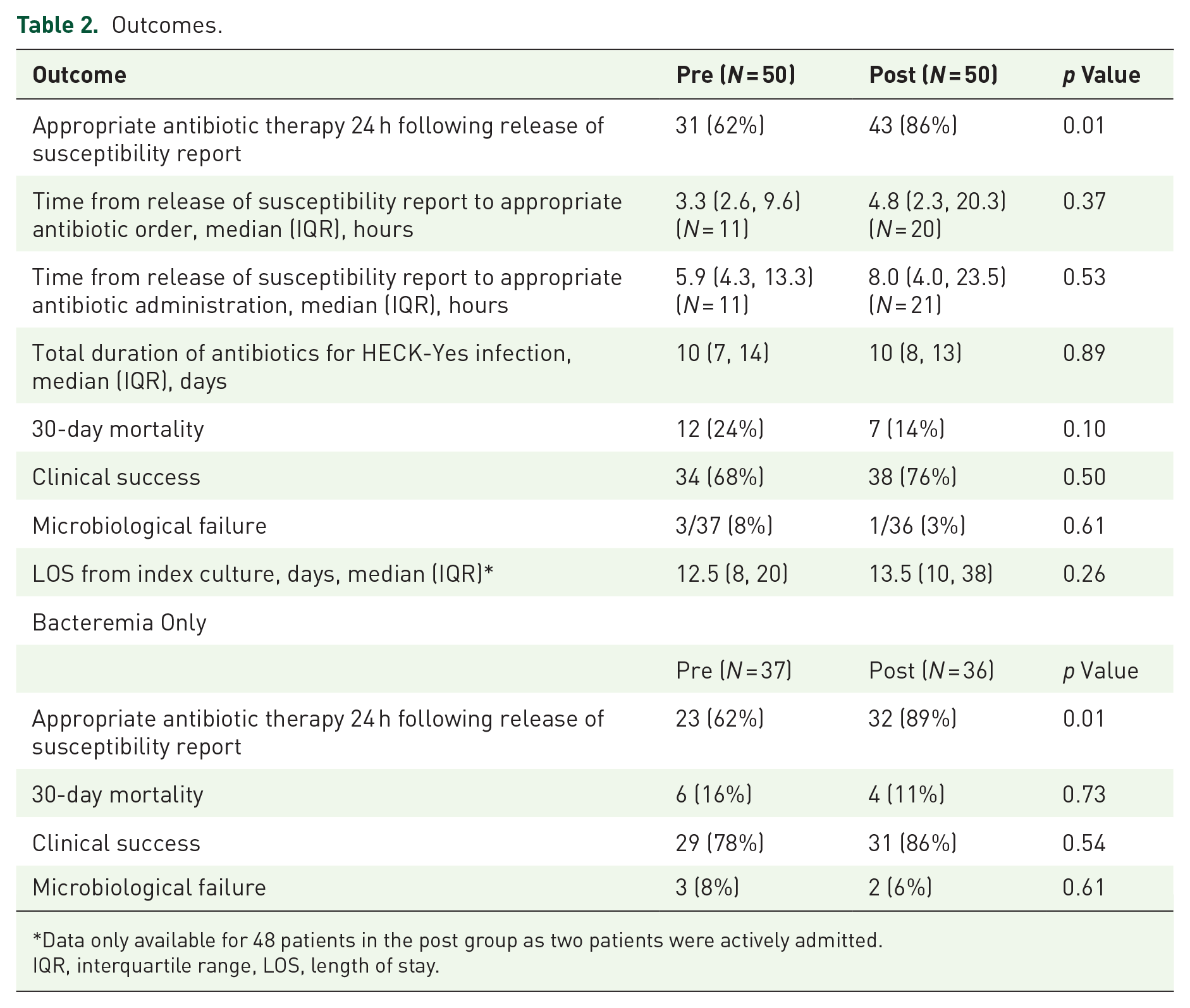
Data only available for 48 patients in the post group as two patients were actively admitted.
IQR, interquartile range, LOS, length of stay.
Discussion
Our investigation revealed SR was an effective nudging strategy that improved antibiotic selection for the treatment of respiratory and bloodstream infections with HECK-Yes organisms. To comply with IDSA guideline recommendations and in response to observed instances of guideline non-concordant treatment for infections with HECK-Yes organisms at our institution, our antimicrobial stewardship team engaged in strategic discussions with our microbiology department during routine bimonthly meetings. The objective was to explore strategies for influencing provider prescribing patterns through the manipulation of AST reporting. The development and implementation of SR proved to be a straightforward and easily applicable intervention across our health system.
Nonadherence to guideline-recommended therapies for infections with HECK-Yes organisms at our institution was likely multifactorial. Susceptibility to less preferred beta-lactams for treating invasive disease, such as ampicillin-sulbactam, cefazolin, ceftriaxone, and piperacillin-tazobactam often appear on AST reports prior to antibiotic exposure and upregulation of ampC production. Providers may have greater tendencies to select these agents if susceptibilities are reported. However, there is a known risk of development of resistance with repeat exposure to these agents. 5 IDSA guidance recommends cefepime or carbapenems as first-line treatment for certain HECK-Yes infections. 9 To steer providers toward these preferred agents, our stewardship and microbiology teams suppressed ampicillin-sulbactam, cefazolin, ceftriaxone, and piperacillin-tazobactam from the AST panel for non-urine isolates. Given high beta-lactam concentrations in urine and lesser concerns about therapeutic failure for simple cystitis with HECK-Yes organisms, IDSA guidance still allows consideration of these agents. 9 Accordingly, we implemented both a urine cascade (that reports ceftriaxone and piperacillin-tazobactam susceptibilities) as well as a non-urine cascade. Our group hypothesized that clinical outcomes may be more significantly influenced by antibiotic choice for non-cystitis cases. Therefore, our investigation focused solely on patients with positive blood and respiratory cultures and excluded urine-only isolates from the analysis.
Most of our patient cohort had bacteremia, most commonly of gastrointestinal or genitourinary sources, with E. cloacae as the predominant organism. Considering these patients were admitted to the hospital and at risk for infection with P. aeruginosa, our medical teams routinely initiated our institution’s antipseudomonal workhorses—piperacillin-tazobactam, cefepime, and meropenem—when gram-negative organisms were initially detected in blood cultures. Once organism susceptibilities were available, there were several instances in the pre-implementation period where patients were changed to inappropriate antibiotics; piperacillin-tazobactam and ceftriaxone were both chosen as definitive therapy in seven and two cases, respectively. Additionally, there were eight cases in the pre-implementation period where patients were started on empiric piperacillin-tazobactam, which was subsequently continued following susceptibilities. Following SR implementation, once piperacillin-tazobactam and ceftriaxone were suppressed from the panel, there was a significant increase in appropriate antibiotic prescribing, which was largely driven by cefepime as definitive therapy. Following SR implementation, there were no cases where piperacillin-tazobactam was selected as definitive therapy for HECK-Yes infection and only one case where antibiotics were de-escalated to ceftriaxone for definitive therapy. With respect to secondary outcomes such as time to optimal antibiotic order and administration, there were minimal differences between groups, likely reflective of the downstream nature of SR as an intervention. While SR improved appropriate antibiotic prescribing, clinical outcomes such as 30-day mortality and clinical success may not have shown significant differences due to the small sample size, the complexity of patient comorbidities, or the downstream nature of this intervention. Future research could explore more upstream interventions, such as incorporating electronic health record comments or notifications at the time of organism identification, to determine if there are quicker adjustments in therapy toward first-line recommended agents.
Our study had several limitations. First, this is a pilot study with a small sample size that limits the interpretability of findings for clinical outcomes such as 30-day mortality, clinical success, and microbiologic failure. Further, no adjustment for confounding was performed, and results should therefore be interpreted with caution. In addition, repeat blood cultures at 72 h were not systematically performed for all patients with bloodstream infections, which may subject the microbiological failure endpoint to selection bias due to differential surveillance practices. Despite these limitations, there were no observed harms to implementing SR. As implementation of this SR is trivial and without risk, the authors encourage other ASPs to adopt this intervention, which promotes the use of appropriate, guideline-concordant antimicrobials for HECK-Yes infections.
Future studies may warrant investigating the application of SR for other distinct groups of organisms posing therapeutic challenges, including but not limited to A. baumannii and S. maltophilia. Moreover, SR could be explored across a variety of settings, from resource-limited community hospitals to large academic medical centers, given its demonstrated ability to improve appropriate antibiotic therapy and the relative ease of implementing this intervention.
Conclusion
Implementing SR of AST results improved appropriate antibiotic prescribing for infections with moderate-risk ampC-producing Enterobacterales. Developing and implementing the cascade was a relatively simple intervention, making it potentially applicable to other institutions.
