Abstract
Background:
Understanding factors that predict progression to severe COVID-19 is critical. Antibodies targeting SARS-CoV-2 spike protein confer protection, while the N protein of SARS-CoV-2 plays roles in viral replication and immune dysfunction. This study explores the significance of N protein and anti-spike antibodies on disease severity, progression, and mortality.
Objectives:
To evaluate the relationship between SARS-CoV-2 N protein and anti-spike antibody levels with disease severity, clinical outcomes, and mortality in hospitalized patients with COVID-19.
Design:
A secondary analysis of serologic data from participants in the ACTIV-1 randomized clinical trial, which evaluated immunomodulators for the treatment of hospitalized patients with COVID-19.
Methods:
A subanalysis of the ACTIV-1 immune modulator trial was conducted. Samples collected at randomization were tested for N protein levels and anti-spike antibodies. Logistic regression and linear models were employed to examine the association between serological measures and clinical outcomes, including 28-day mortality as well as progression to high-flow nasal cannula (HFNC) and invasive mechanical ventilation (MV).
Results:
Among the 496 participants with detectable serum N protein, the median was 1143 ng/dL, and levels decreased from 2559 ng/dL in participants randomized at 6 days of symptom onset to 477.6 ng/dL at 11 days. Higher anti-spike antibody levels were seen as the days from symptom onset progressed or disease severity increased. Greater disease severity at randomization was associated with 28-day mortality, prolonged days of oxygenation, ventilation, hospitalization, and risk of new non-invasive ventilation, HFNC, MV, or extracorporeal membrane oxygenation use. N protein levels were associated with a higher risk of new non-invasive ventilation or HFNC use, longer oxygenation duration, and extended hospitalization. Anti-spike antibody serologies were not associated with clinical outcomes.
Conclusion:
N protein levels could provide insights into COVID-19 disease progression and prognosis. Further research is needed to explore the clinical implications of these findings to optimize patient care and enhance outcomes.
Plain language summary
COVID-19 severity can vary widely, and predicting who will develop severe disease is crucial for effective treatment. This study looks at two key components of the virus: the N protein, which helps the virus replicate, and anti-spike antibodies, which help the body fight off the virus. We studied blood samples from participants in a clinical trial to see how levels of these two components related to disease severity and outcomes, such as the need for advanced respiratory support or death. The study found that higher levels of the N protein were associated with more severe disease and worse outcomes, including a higher risk of requiring advanced respiratory support and longer hospital stays. On the other hand, anti-spike antibodies, which are typically seen as protective, did not show a clear relationship with the severity of illness or outcomes in this study. These findings suggest that the N protein might be a useful indicator for predicting how severe a person's COVID-19 illness might become, which could help doctors better manage and treat the disease. However, more research is needed to fully understand the role of these viral components in COVID-19 and how they can be used to improve patient care.
Introduction
The COVID-19 pandemic has caused significant morbidity and mortality and has challenged healthcare systems worldwide. 1 Identifying early biomarkers to predict severe disease or death is crucial for effective triage, timely management, and resource utilization. The severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) N protein is pivotal in viral replication, and recent studies have suggested its potential role in immune dysregulation and disease severity.2,3 Previous research has indicated that elevated N protein levels are associated with worse clinical outcomes, highlighting its potential as a prognostic marker. Furthermore, antibodies against the spike protein of SARS-CoV-2 are linked to protective immunity and reduced disease severity.2 –4 This study aims to build on existing knowledge by elucidating the relationship between these serologic markers—specifically serum N protein levels and anti-spike antibodies—and outcomes such as disease progression and mortality in COVID-19 patients. By understanding these associations, we hope to identify key markers that can help in the early identification of at-risk patients, thereby improving clinical management and outcomes.
Methods
ACTIV-1 study design
We performed a substudy analysis of the National Institutes of Health (NIH)-sponsored ACTIV-1 Immune Modulator (IM) study, aimed at exploring the significance of SARS-CoV-2 N protein and anti-spike serologies on COVID-19 disease severity, disease progression to higher supplemental oxygen needs, and mortality. The ACTIV-1 IM study was an international, multicenter, prospective study with a master protocol designed to evaluate multiple immune modulators plus standard of care in participants hospitalized with COVID-19 via three substudies comparing infliximab, abatacept, or cenicriviroc with placebo in a randomized, double-blinded design. 5 Participants ⩾18 years of age with laboratory confirmed SARS-CoV-2 infection within 14 days prior to enrollment, ongoing illness of any duration, and at least one of the following: radiographic infiltrates by imaging, blood oxygen saturation <94% on room air, requiring supplemental oxygen, or requiring invasive mechanical ventilation or extracorporeal membrane oxygenation (ECMO) were eligible and were randomly assigned to one of the three placebo-controlled arms. Exclusions included pregnancy, anticipated hospital stay <72 h, liver enzymes >10x normal, chronic liver disease, acute kidney injury with glomerular filtration rate <30 mL/min (stable chronic kidney disease permitted), severe heart failure, severe neutropenia or lymphopenia, known or suspected untreated infection including tuberculosis, or those who received cytotoxic or biologic-targeted immunomodulators within 4 weeks or five half-lives before screening (Supplemental Appendix). Upon enrollment, participants underwent collection of relevant clinical and laboratory data. One sample was collected from participants randomized in sites in the United States. Thus, our substudy did not include international ACTIV-1 sites and was not completely representative of the entire ACTIV-1 study population. Baseline serologies for N protein and anti-spike protein for a subset of patients were collected on randomization. This study was conducted between October 2020 and December 2021.
Substudy participants and data collection
Individuals who met the eligibility criteria of the ACTIV-1 IM study and had serology data at baseline were included in the substudy. Baseline serological assessments were conducted at randomization for a subset of participants, while those who did not have baseline serologies available were excluded. Quantitative N protein nucleocapsid levels were measured using the Quanterix Simoa SARS-CoV-2 N antigen assay (Quanterix, Billerica, MA, USA) according to the manufacturer’s instructions with a lower limit of quantification at 3 ng/L. Anti-spike antibody titers were determined using Quanterix Simoa anti-spike total IgG semi-quantitative antibody assay (Quanterix, Billerica, MA, USA), according to manufacturer’s instructions. Positivity for this assay has been defined as ⩾770 ng/mL, per manufacturer’s recommendation (hereafter defined as clinical cut-off).
Demographic, clinical, and laboratory data, including duration of oxygenation, ventilation, and hospitalization, were collected for each participant. Clinical status and safety data were captured daily during hospitalization through day 28 and post-discharge days 8, 11, 15, 29, and 60. Clinical status was evaluated using an 8-point ordinal scale (OS) developed for the ACTIV-1 study (Supplemental Table 1). Groupings of ordinal scale indices were selected to combine groups based on severity, OS2 with OS3 and OS4 with OS5, due to low sample sizes. Symptom duration was defined as time in days from symptom onset to ACTIV-1 study randomization. N protein levels were defined as high when above 1000 ng/dL.2,3 Days of symptoms at randomization and C-reactive protein (CRP) values (mg/dL) were tabulated by ordinal scale.
Statistical analysis
Descriptive statistics, such as means, medians, and standard deviations, were used to summarize the baseline characteristics of the participant population. The Spearman correlation coefficient was used to examine the correlation between N protein levels and anti-spike serologies. Descriptive statistics were used to assess any relationship between N protein levels, anti-spike serologies, and symptom duration. Descriptive statistics were also used to compare serology measures by baseline ordinal scale groups. For all descriptive analyses, N protein level was included as both a continuous and a binary measure of < or ⩾1000 ng/dL.
The association between serology measures, treatment group, and clinical outcomes, including 28-day mortality, progression to high-flow nasal cannula, and invasive mechanical ventilation, was assessed using logistic regression models for binary outcomes and linear models for continuous outcomes. Logistic regression models were specifically used for binary outcomes such as 28-day mortality, progression to high-flow nasal cannula (HFNC), and progression to invasive mechanical ventilation (MV). Linear models were used for continuous outcomes. Models were initially run with an interaction term between treatment group and the serology measure of interest. Treatment group was defined as assigned study drug of infliximab, abatacept, or a pooled treatment combination of either infliximab or abatacept. Participants receiving placebo study drug were included in the shared placebo group. The model was then rerun with the interaction term excluded if non-significant at the 0.05 level. All models were run separately for anti-spike serologies and serum N protein levels. All statistical analyses were conducted using SAS version 9.4 or higher software (SAS Institute, Inc., Cary, NC, USA).
Ethics
Institutional review board approval was obtained at WCG IRB (protocol number 20202874) and from all participating centers (Supplemental Appendix). Written informed consent was obtained from each participant prior to their inclusion in the study.
Results
Baseline characteristics
ACTIV-1 enrolled a total of 1971 participants between October 16, 2020 and December 31, 2021 at 95 hospitals (85 clinical research sites), of which 496 had baseline serologic specimens (Supplemental Figure 1). Sites outside of the United States were excluded from the analysis. Baseline characteristics are presented in Table 1. The median age of the participants was 57 years (interquartile range 48–66) and 39% were female. Minority representation included 24% of participants identifying as Black and 19% as Hispanic. At enrollment, most participants (58%) were receiving low-flow supplemental oxygen while 38% had high-flow oxygen requirements or higher and 4% had no supplemental oxygen requirements. The mean (±standard deviation) duration of symptoms was 9 (±4) days and most participants (70%) had obesity (defined as body mass index ⩾30 kg/m2).
Demographics and baseline characteristics of patients enrolled in the ACTIV-1 study.
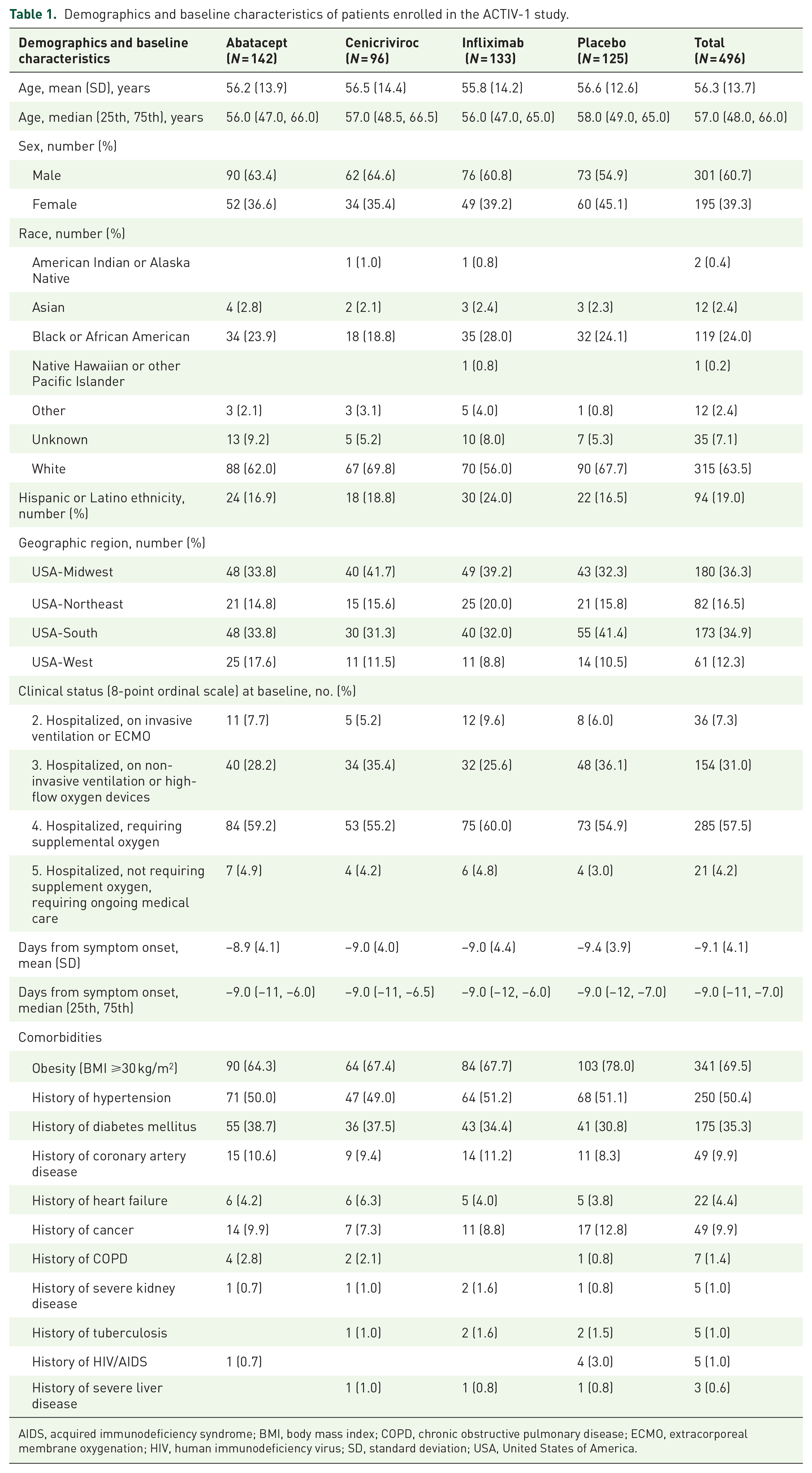
AIDS, acquired immunodeficiency syndrome; BMI, body mass index; COPD, chronic obstructive pulmonary disease; ECMO, extracorporeal membrane oxygenation; HIV, human immunodeficiency virus; SD, standard deviation; USA, United States of America.
Serum N protein, anti-spike antibody, and days of symptoms at ACTIV-1 study randomization
The baseline serum N protein levels were detectable in all patients with a median value of 1143 ng/dL (interquartile range 101–4771) and had an inverse relation with the days of symptoms at ACTIV-1 study randomization (Figure 1 and Supplemental Figure 2). As the days from symptom onset progressed, there was a notable decline in N protein levels from 2559 ng/dL within the first 6 days of symptom onset to 477.6 ng/dL at 11 days from symptom onset, representing an 81% reduction in values over time. Baseline values exceeded 1000 ng/dL for 64% of those who presented within 6 days of symptom onset but only for 37% of participants presenting >11 days from symptom onset (Figure 2 and Table 2). Participants randomized at later days from symptom onset, particularly those with higher disease severity (OS2 and OS3), exhibited elevated anti-spike antibody levels (Table 3, Figures 3 and 4, and Supplemental Figures 3 and 4). An inverse relationship between serum N protein levels and anti-spike serology values was noted, with higher baseline anti-spike antibody levels associated with lower serum N protein levels and vice versa (Figure 5). Participants with higher disease severity (OS2 and OS3) displayed a discernible increase in CRP levels after 11 days from symptom onset. In contrast, patients with lower disease severity (OS4 and OS5) maintained relatively stable CRP values, irrespective of the time elapsed since symptom onset (Table 3, Figure 6 and Supplemental Table 2, and Supplemental Figure 5).

Median N protein levels (ng/dL) by days since symptom onset at randomization.
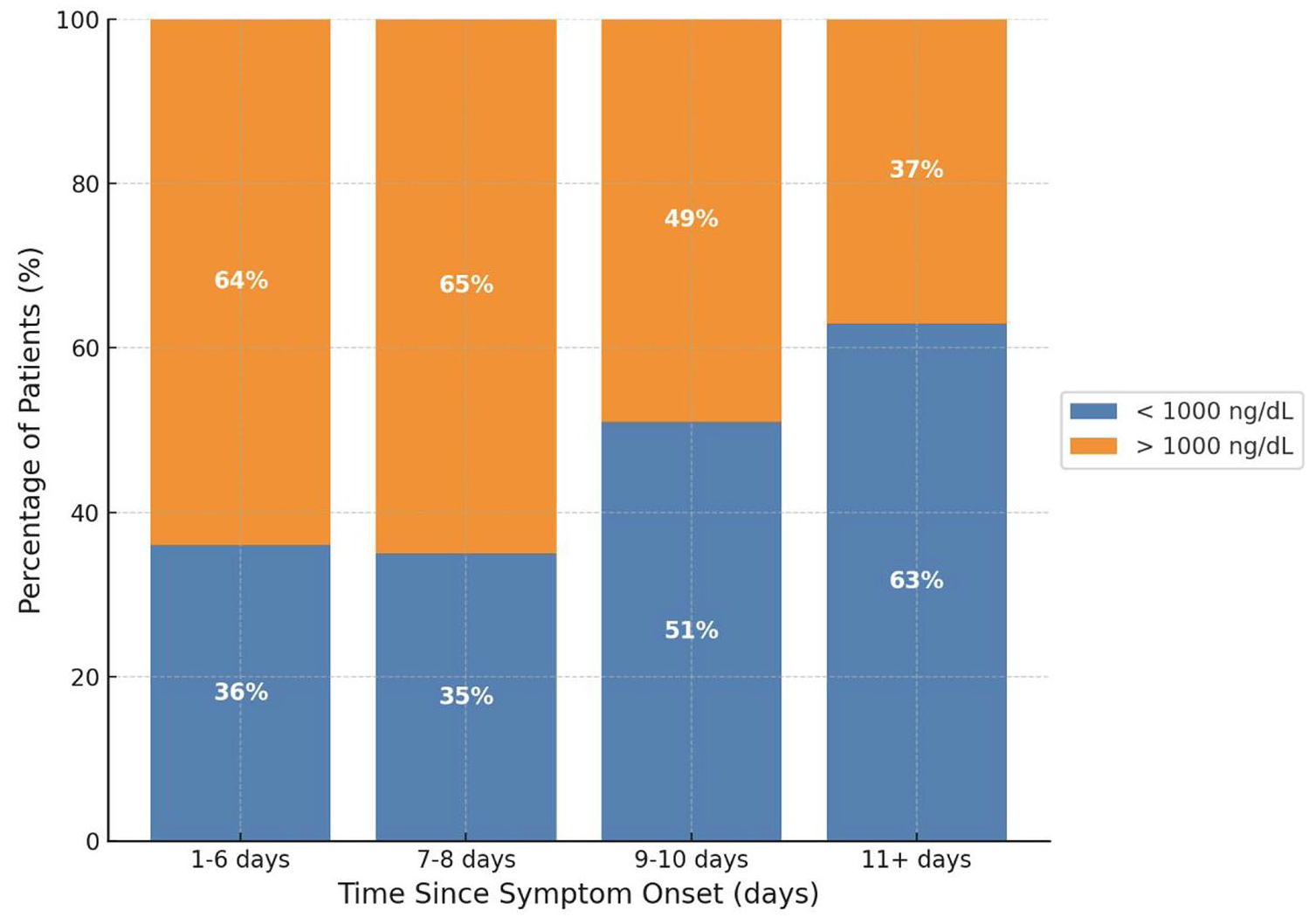
Proportion of patients with N protein above and below 1000 ng/dL, by days since symptom onset at randomization.
Days of symptoms at randomization and N protein percentile values stratified using a threshold of 1000 ng/dL.

Days of symptoms at randomization and percentile values by ordinal scale.


Median N protein levels (ng/dL) by disease severity (ordinal scale) and days since symptom onset at randomization.

Median anti-spike antibody levels (ng/mL) by days since symptom onset and disease severity (ordinal scale) at randomization.

Scatterplot of log-transformed serostatus relationship between N protein and anti-spike values.

CRP levels (mg/dL) by symptom duration and disease severity (ordinal scale) at randomization.
Associations between serology and COVID-19 severity, disease progression, and mortality
Higher disease severity at ACTIV-1 study randomization (OS2 and OS3) was strongly associated with multiple adverse outcomes, including 28-day mortality, duration of supplemental oxygen use, duration of invasive mechanical ventilation, duration of hospitalization, 28-day mortality, 28-day risk of new non-invasive ventilation or high-flow oxygen use, and 28-day risk of new invasive mechanical ventilation or ECMO use (p < 0.0001) (Table 4). Specifically, patients with more severe disease at randomization had a higher likelihood of mortality within 28 days, experienced prolonged durations of oxygenation and hospitalization, and were at increased risk for requiring advanced respiratory support. Serum N protein levels at ACTIV-1 study randomization were significantly associated with increased risk of requiring new non-invasive ventilation or high-flow oxygen, prolonged oxygen support, and extended hospitalization over 28 days (p < 0.0001), highlighting their potential as an early marker of severe disease. While N protein levels trended toward an association with prolonged invasive mechanical ventilation (p = 0.0787), this was not statistically significant. Conversely, anti-spike serologies did not show statistically significant associations with any of the measured outcomes. Additionally, no significant treatment effects were observed when comparing the three treatment groups to the pooled placebo group. These findings highlight the importance of disease severity and serum N protein levels as predictors of COVID-19 progression and mortality, underscoring the need for targeted interventions for high-risk patients. No treatment effect was noted in the three ACTIV-1 study arms compared with the pooled placebo group.
Associations of N protein (ng/dL), anti-spike (ng/mL), ordinal scale 2, and ordinal scale 3 at randomization with disease progression and clinical outcomes*.
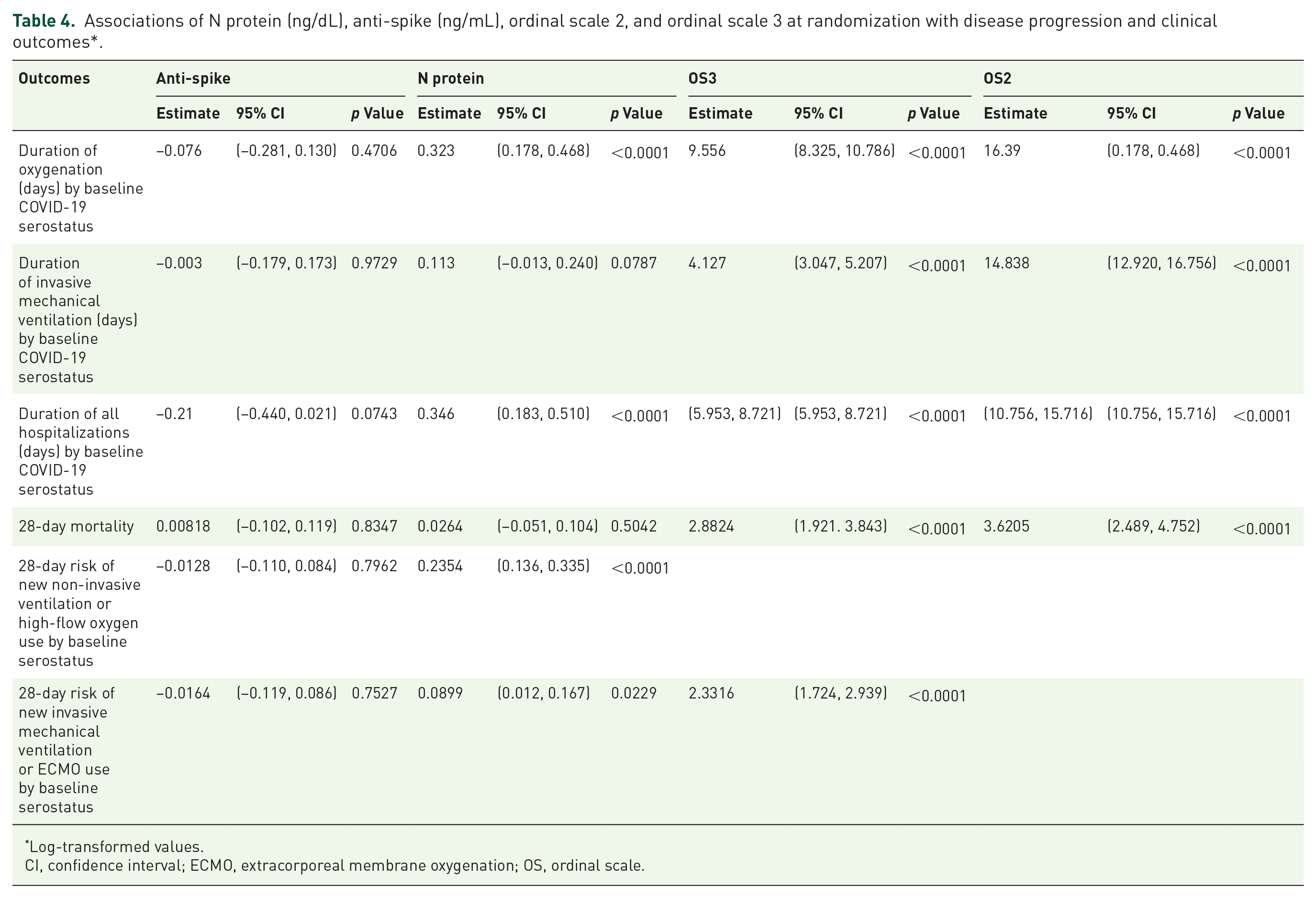
Log-transformed values.
CI, confidence interval; ECMO, extracorporeal membrane oxygenation; OS, ordinal scale.
Discussion
Our study highlights associations between serum N protein levels and clinical outcomes of importance. N protein levels at ACTIV-1 study randomization were associated with risk of progression of COVID-19 disease and duration of hospitalization and could offer valuable insights into patient prognostication and optimal resource allocation for the comprehensive management of COVID-19. Anti-spike antibody levels at ACTIV-1 study randomization were not associated with these outcomes. However, our study provides valuable insights into the dynamics of both serum N protein levels and anti-spike serology values and their relation to days of symptoms and severity at presentation in patients hospitalized with COVID-19.
Among the participants in the ACTIV-1 trial, the baseline mean serum N protein level was high at ACTIV-1 study randomization (3750 ng/dL), suggesting a substantial presence of SARS-CoV-2 viral activity in the study population. 6 Serum N protein values declined with time, particularly in patients with higher disease severity at ACTIV-1 study randomization (OS2 and OS3). The over 80% decline in N protein values after 11 days from symptom onset suggests a substantial decrease in viral activity over time.6,7 This trend aligns with the natural course of COVID-19, where viral load typically diminishes as the disease progresses to later stages of the disease, which are characterized by inflammatory responses and acute respiratory distress syndrome. The observed rise in CRP levels after 11 days in patients requiring higher oxygen (OS2 and OS3) supports this hypothesis. The pattern of N protein levels revealed a significant proportion of patients with high levels (>1000 ng/dL) in those randomized within 1–6 days from symptom onset. This pattern was inverted in patients randomized at >11 days from symptom onset, indicating a shift in viral activity and immune response dynamics. Anti-spike antibody levels were higher in patients presenting >9 days from symptom onset, particularly in those with higher disease severity (OS2/OS3). This finding suggests a potential interplay between viral activity and immune response, as indicated by the negative association observed between baseline anti-spike antibody levels and N protein levels. No treatment effect was noted in the three substudies. However, this may have been influenced by the small sample size and lack of power. The progression of COVID-19 from a viral phase to a pulmonary phase may take 5–10 days, aligning with the timing of patient presentation and enrollment in our study. Furthermore, in the ACTIV-3/TICO trial, early biomarker trajectories revealed heterogeneity, with some patients failing to clear nucleocapsid antigen despite antibody presence, which was associated with worse outcomes. 8
Higher disease severity at ACTIV-1 study randomization (OS2 and OS3) strongly correlated with 28-day mortality, duration of oxygenation, ventilation, and hospitalization, underscoring the importance of disease severity at presentation as a predictor of outcomes and healthcare resource utilization. N protein values exhibited no statistically significant association with mortality at 28 days. Nevertheless, N protein levels correlated with an elevated risk of progression to high-flow nasal cannula and invasive mechanical ventilation. This strong association with oxygen requirements and hospitalization suggests they may serve as an early marker for identifying patients at risk of severe disease progression. Baseline N protein levels also related to other important outcomes, such as duration of oxygenation and hospitalization. The link to ventilation days was not statistically significant. These findings emphasize disease severity as a key prognostic factor and suggest serum N protein levels as a potential marker for disease progression and resource utilization.
Our findings align with other studies showing the association between higher plasma antigen levels and severity of pulmonary illness and important patient outcomes. In a cross-sectional study of baseline plasma antigen level from 2540 participants, the initial severity of pulmonary illness correlated with serum N protein levels and the mean plasma antigen level was over three times higher in participants requiring invasive mechanical ventilation or non-invasive positive pressure ventilation than those requiring low-flow supplemental oxygen. 2 Male sex, reduced remdesivir use, and renal impairment were also linked to higher baseline N protein levels. In a prospective observational cohort of 256 hospitalized patients, a 500 pg/mL rise in plasma N-antigen was linked to a 1.05 adjusted odds ratio for worse clinical status. 9 N-antigen levels of ⩾1000 pg/mL at presentation predicted worsened status by day 7, intensive care unit admission, and ventilation. Notably, N protein was detectable in over 80% of participants in both studies. Serial measurements of N protein may enhance its prognostic utility by providing dynamic insights into disease progression. Future studies should explore this approach to refine its predictive value.
Our study has many limitations. Our laboratory data were collected at ACTIV-1 study randomization and thus represent only one point in time. Serial measurements before hospitalization and during illness could offer deeper insights into biomarker trends. Only one sample from sites in the United States was collected. Thus, our study was not completely representative of the entire ACTIV-1 cohort. Another key limitation is the time-related bias, as patients presenting later in the disease course may have different baseline profiles than those presenting earlier. This affects our findings on N protein and anti-spike antibody levels, indicating the need for stratifying participants by illness duration. The ACTIV-1 trial enrolled patients prior to the widespread introduction of vaccines and prior to the spread of the Omicron variant of SARS-CoV-2; thus the effect of vaccines and emerging SARS-CoV-2 variants on SARS-CoV2 serology is another important limitation. Other confounders that may influence biomarker levels and outcomes are detailed in Supplemental Table 3.
Conclusion
Our study highlights the potential of N protein as a clinical marker for progression of disease severity in COVID-19. Measuring N protein levels on admission could help provide valuable insights into possible disease progression, patient prognosis, and resource allocation in the management of COVID-19. Studies have consistently linked elevated plasma nucleocapsid antigen levels to worse COVID-19 outcomes, including higher hospitalization risk and delayed recovery.10,11 Our study further underscores the predictive value of serum N protein levels and their potential as an early biomarker to guide clinical risk stratification when integrated with disease severity scales. Further research is needed to explore the underlying mechanisms and clinical implications of these findings to optimize patient care and improve outcomes in individuals affected by COVID-19.
Supplemental Material
sj-docx-1-tai-10.1177_20499361251333617 – Supplemental material for SARS-CoV-2 N protein and anti-spike serologies: insights into COVID-19 disease severity and mortality—a secondary analysis of the ACTIV-1 trial
Supplemental material, sj-docx-1-tai-10.1177_20499361251333617 for SARS-CoV-2 N protein and anti-spike serologies: insights into COVID-19 disease severity and mortality—a secondary analysis of the ACTIV-1 trial by Alfredo J. Mena Lora, Kimi Enders, Huimin Wu, Luis Parra-Rodriguez, Christopher Palma, Katy Saliba, Sylvain Laverdurre, P Brian Smith, Kevin J. Anstrom, Samuel A. Bozzette and William G. Powderly in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-2-tai-10.1177_20499361251333617 – Supplemental material for SARS-CoV-2 N protein and anti-spike serologies: insights into COVID-19 disease severity and mortality—a secondary analysis of the ACTIV-1 trial
Supplemental material, sj-docx-2-tai-10.1177_20499361251333617 for SARS-CoV-2 N protein and anti-spike serologies: insights into COVID-19 disease severity and mortality—a secondary analysis of the ACTIV-1 trial by Alfredo J. Mena Lora, Kimi Enders, Huimin Wu, Luis Parra-Rodriguez, Christopher Palma, Katy Saliba, Sylvain Laverdurre, P Brian Smith, Kevin J. Anstrom, Samuel A. Bozzette and William G. Powderly in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
We thank the members of the ACTIV-1 Study Team (![]() ) for their many contributions in conducting the trial, and the participants and their families for their altruism in participating in this trial. We would also like to thank Cynthia Gonzalez, Jessica Springer, and Soju Chang from the National Center for Advancing Translational Sciences, Bethesda, MD, Megan Roebuck and Judi Willhide from Duke Clinical Research Institute, Durham, NC, and Sandy Butler from Technical Resources International (TRI), Bethesda, MD for their support in completing the manuscript.
) for their many contributions in conducting the trial, and the participants and their families for their altruism in participating in this trial. We would also like to thank Cynthia Gonzalez, Jessica Springer, and Soju Chang from the National Center for Advancing Translational Sciences, Bethesda, MD, Megan Roebuck and Judi Willhide from Duke Clinical Research Institute, Durham, NC, and Sandy Butler from Technical Resources International (TRI), Bethesda, MD for their support in completing the manuscript.
Declarations
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
