Abstract
Background:
Kentucky is one of seven states with high, sustained rural HIV transmission tied to injection drug use. Expanding access to pre-exposure prophylaxis (PrEP) has been endorsed as a key HIV prevention strategy; however, uptake among people who inject drugs (PWID) has been negligible in rural areas. Syringe services programs (SSPs) have been implemented throughout Kentucky’s Appalachian region, providing an important opportunity to integrate PrEP services.
Objectives:
The primary objective was to examine preliminary efficacy and effect sizes of the study interventions on PrEP initiation among HIV-negative PWID.
Design:
Parallel group randomized controlled trial.
Methods:
Eighty participants were enrolled from two rural SSP locations in southeastern Kentucky. Following informed consent, participants completed a baseline interview, and were randomized to the intervention comparators. The primary endpoint was PrEP initiation, measured by dispensed PrEP prescription, within the 6-month study period. Analyses employed intent-to-treat (ITT) and per protocol approaches.
Results:
In total, 77/80 enrollees (96.2%) completed at least one session of their assigned intervention, regardless of trial arm. Seventy (87.5%) were linked to the embedded PrEP provider for the initial clinical visit; 38 (47.5%) completed a follow-up clinical visit with the provider, 22 (27.5%) were issued a prescription, and 7 (8.8%) initiated PrEP during the study period. We observed a 12.1% difference (14.6% vs 2.5%; ITT) and 12.8% difference (15.4% vs 2.6%; per protocol) in the primary outcome (PrEP initiation), in favor of the experimental intervention.
Conclusion:
This pilot trial established proof of concept for integrated PrEP care within SSPs in rural areas, and demonstrated a clinically meaningful difference in PrEP initiation between interventions, which warrants examination in a larger trial. Rates of early care discontinuation indicate a need for ongoing patient engagement strategies and implementation support for community SSPs.
Trial registration:
Prospective registration with ClinicalTrials.gov, NCT05037513 (registered August 5, 2021).
Keywords
Introduction
Robust implementation of Ending the HIV Epidemic (EHE) strategies and evidence-based interventions has made notable progress in reducing HIV incidence in the United States, with recent surveillance data placing the number of new infections annually at the lowest level since the mid-1980s. 1 Yet despite this backdrop of declining incidence, people who inject drugs (PWID) are experiencing a countervailing increase in new HIV infections. 1 Driven in part by the ongoing opioid crisis, new HIV infections among PWID increased 4% between 2017 and 2021.1,2 The intersection of opioid use and rising rates of infectious disease, including HIV, has been widely observed in recent notable research.3,4 Importantly, recent HIV outbreaks among PWID also reflect growing HIV transmission in rural regions of the United States. 2 In fact, EHE identified seven US states with high sustained rural HIV transmission, including Kentucky, designating these as high priority jurisdictions for intervention.5,6 Kentucky’s most recent HIV surveillance data indicates that 14.8% of males and 54.2% of females newly diagnosed with HIV in 2021 had injection drug use-related transmission, which far exceeds national averages for this exposure category. 7
Increases in rural HIV transmission among PWID in the United States are propelled by limited resource availability for HIV prevention and treatment, provider shortages, and disparities in direct services availability, including testing, education, and HIV pre-exposure prophylaxis (PrEP). 2 In rural and remote areas, including in Kentucky, limited access to healthcare and HIV specialty care services, and provider scarcity limits access to PrEP along geographic boundaries.8–16 Given the current constraints on HIV prevention services in rural areas, expanding PrEP access through new, low-threshold approaches for PWID has potential to advance HIV prevention for this priority population. Among PWID, PrEP uptake remains severely limited,17–19 less than 1% by some estimates, 20 and no evidence-based or evidence-informed PrEP interventions specifically targeted for PWID are available. 21 A recent review evaluating published PrEP interventions noted a significant gap and lack of best practices for several populations, including PWID, 22 leading to a call for collaborative research with communities to develop interventions for increasing PrEP use and persistence.
Scientific consensus is growing with regard to differentiated service delivery models for PrEP care. 23 Differentiated PrEP service delivery is a client-centered approach that simplifies and adapts PrEP service provision through enhanced attention to service efficiency and integration, patient choice, and user preferences. 23 Within this context, interventions to increase PrEP services access by PWID in non-traditional, de-medicalized care settings may fill a need to improve the PrEP care cascade among rural PWID. For PWID in particular, there is broad scientific consensus supporting low threshold PrEP access24,25 through novel approaches that: shorten time to prescribing, increase point of care testing,24,26,27 utilize mobile PrEP or TelePrEP strategies,28,29 and integrate PrEP within harm reduction or syringe services programs (SSPs).19,30,31
Emerging research indicates that SSPs are acceptable and feasible locations for PrEP integration.19,30,31 PWID attending SSPs generally report greater awareness of PrEP and co-location of services could lower the threshold for PrEP access32–34; however, recent evidence on PrEP uptake in SSPs is mixed. Two recent studies found that although most PWID accessing SSPs expressed interest in using PrEP, none had initiated PrEP.35,36 Nevertheless, a recent study of women who inject drugs attending SSPs in Philadelphia demonstrated that a large proportion, more than two-thirds, initiated PrEP. 31
This article reports findings from a pilot randomized trial designed to integrate PrEP care into rural SSPs to expand the current service model with low threshold PrEP access. The pilot intervention integrates PrEP education, screening, prescribing, and monitoring within the SSP to reduce structural barriers to access through provision of co-located PrEP services. This structural change is paired with a newly adapted strengths-based case management (SBCM-PrEP) intervention designed to address individual and environmental barriers to PrEP initiation among rural PWID. The intervention was adapted from ARTAS HIV linkage case management, a weekly five-session SBCM intervention to link recently diagnosed persons with HIV care, which to our knowledge is the only published randomized controlled trial (RCT) of an HIV treatment linkage intervention with demonstrated efficacy.37,38 SBCM addresses environmental barriers impacting care—such as insurance or care costs and transportation—by identifying and resolving barriers in a detailed case management plan.39–41 Adapted SBCM approaches have shown efficacy in increasing HIV testing and linkage to medical care among people who use drugs in urban areas 42 as well as in linking HIV+ PWID to care. 43 Comparison of the newly adapted SBCM-PrEP intervention with standard PrEP counseling provides the opportunity to examine the value-added of a more robust, but brief, client-driven intervention that supports PrEP initiation through active advocacy to reduce multi-level barriers to uptake.
Methods
Trial design
Trial reporting follows CONSORT guidelines for pilot and feasibility trials. 44 Here, we implemented a pilot randomized parallel group trial to compare the preliminary efficacy of SBCM-PrEP versus brief standard PrEP counseling (CDC-PrEP) on the primary endpoint of PrEP initiation (measured by dispensed PrEP prescription) among rural PWID within the 6-month study period. Randomization, stratified by site and gender, was performed with a 1:1 allocation within strata (see Figure 1 study flow diagram).

Study flow diagram.
Sample
PWID participants were enrolled from Knox and Clay County Health Department SSP locations in rural southeastern Kentucky, which have been designated as high HIV vulnerability counties among PWID. 45 Study inclusion criteria were: (1) 18+ years of age; (2) at least one occasion of injection drug use in the past month; (3) an indication for PrEP in the past 6 months, consistent with CDC guidelines 46 ; (4) SSP participant at the time of study entry; and (5) willingness to participate in a multi-session intervention and follow-up. Exclusion criteria were: HIV+ serostatus and current PrEP care.
Trial enrollment
Enrollment included a brief structured eligibility screener conducted by trained study staff during regular SSP operating hours using a REDCap interface.47,48 Those eligible and interested participated in an informed consent process and provided locator information, followed by a structured face-to-face baseline interview examining patterns of substance use, health risk behaviors, health services utilization, mental health, HIV and substance use related stigma, PrEP awareness, knowledge, and interest. Trained study interviewers conducted face-to-face computer-assisted personal interviews with participants at baseline using REDCap.47,48 Participants were located for follow-up using detailed locator information collected at baseline. Project staff also had a routine presence in the SSPs to maintain ongoing contact with participants. Participants received a $30 incentive for data collection and transportation costs at baseline, 3-month, and 6-month follow-up. Immediately after baseline, participants were randomized to intervention.
Study interventions
Study intervention staff attempted to conduct the initial PrEP intervention session of the assigned arm immediately following baseline or within 1 week of study enrollment. Study intervention sessions were delivered by medical and public health graduate students, who were trained as HIV counselors and trained in SBCM protocols. In both arms, participants were offered linkage to embedded Advanced Practice Registered Nurse (APRN) PrEP providers within the SSPs for clinical PrEP screening, which involves an HIV risk assessment, PrEP counseling and point-of-care fingerstick and urine testing for PrEP eligibility using home test kits (HIV antigen and antibody, hepatitis B surface antigen, creatinine, and urine pregnancy testing, for women of child-bearing age with a uterus), prescribing and follow-up care. Test kits were collected at the SSPs and samples were mailed to a CLIA-certified lab; results were generally available within 7–10 days of kit receipt. Participants were asked to return for a second clinical visit in 2 weeks to review test results with the provider in order to receive a prescription for PrEP (emtricitabine/tenofovir disoproxil fumarate); up to 4 weeks was allowed for return in the event that the patient missed the 2-week appointment. At 4 weeks and beyond, repeat HIV testing was required prior to issuing a prescription. The study supported the cost for all laboratory testing. Participants received $10 transportation support gift cards for intervention visits and visits to the study PrEP providers. Study staff attempted to contact participants to remind them of intervention session appointments the day before each scheduled session, and participants were allowed to reschedule missed sessions. Participants who started PrEP obtained the medication through their insurance coverage and were offered onsite clinical follow-up visits at 3 and 6 months, with HIV testing performed at 3 months, and HIV and creatinine testing offered at 6 months.
Intervention comparators
The study intervention comparators are designed to provide brief exposure to structured educational content to increase basic awareness of HIV risk and PrEP care, which are considered critical initial steps or entry points to the PrEP care continuum. 49
The Comparison Condition is an in-use PrEP education intervention based on CDC guidelines, 46 and utilizing structured risk assessments and educational materials recommended by the CDC (CDC-PrEP). CDC-PrEP involves a single 20-min individual session that includes education on HIV and PrEP basics, costs of PrEP, the importance of medication adherence, and medication side effects. At the conclusion of the session, the study interventionist encouraged linkage to the embedded PrEP provider and participation in a clinical evaluation for PrEP. The use of an active comparator (CDC-PrEP) was driven by an ethical imperative to provide at-risk participants with needed prevention education and realistic potential to access PrEP if desired.
The Experimental Condition, SBCM-PrEP, is a multi-session SBCM intervention adapted for PrEP-related educational content. SBCM-PrEP provides up to five strengths-based sessions to deliver tailored PrEP education and engage in action planning to increase readiness for PrEP initiation. Formative work was conducted to adapt and refine the novel SBCM-PrEP intervention, following guidelines of the ADAPT-ITT framework. 50 Analysis of the in-depth interview data identified several important themes that led to intervention content and delivery strategy adaptations, which has been described in detail elsewhere. 30
Session 1 of the SBCM-PrEP intervention was a 1-h individual session emphasizing rapport building and engagement of the participant, whatever their initial attitudes regarding PrEP. The staff interventionist discussed the broad goals of SBCM, provided basic HIV education and tailored PrEP education, and engaged in an interactive dialogue to elicit participant information on their risk environment (e.g., concerns related to stigma, relationship dynamics/violence, housing instability, insurance coverage) that could have inhibited PrEP uptake. Strengths statements were used to reinforce participants’ abilities to make appropriate decisions about their health behaviors. The interventionist encouraged participants to link with the embedded PrEP provider and participate in a clinical evaluation for PrEP. The session closed with a summary of discussion and action planning to document goals, barriers, and strengths, and a warm hand-off if the participant was ready to link.
Following adaptation of the SBCM-PrEP intervention protocol, identification, hiring, and training of local study PrEP providers was undertaken. Importantly, these were local APRN providers who had not previously prescribed PrEP, but were interested to enter this area of practice and committed to harm reduction principles; one provider was newly licensed, the other had a prior clinical background in women’s health. Providers received study-related training, including human subjects training and PrEP content, and were subsequently embedded into offices within the two SSPs during operating hours, for 4–8 h per week. The study compensated providers or their employers at an hourly rate to offer these clinical services.
All study procedures were reviewed and approved by the Medical Institutional Review Board of the awardee institution. Trained and approved study staff who had completed required human subjects’ protection trainings obtained informed consent. The trial was prospectively registered (ClinicalTrials.gov, NCT05037513), and a Data and Safety Monitoring Board (DSMB) was convened to review the initial protocol elements and to provide ongoing monitoring of the progress, safety, and efficacy of the trial. Adverse events were collected and assessed at each study visit and reported to the DSMB and IRB as required.
Data collection and measures
Outcomes
The primary outcome of interest for this analysis was PrEP initiation (measured by dispensed prescription for emtricitabine/tenofovir disoproxil fumarate), examined by intervention condition, during 6 months post-baseline. Secondary outcomes of interest include: (1) completion of initial clinical visit with a study PrEP provider; (2) completion of second clinical visit with study PrEP provider to review initial lab results; and (3) issuance of initial electronic or paper PrEP prescription by study provider, also within 6 months post-baseline. Study providers maintained participant medical records, including history and physical information, PrEP testing information, laboratory results, prescription information, and follow-up visits. For each participant receiving a prescription, providers attempted to obtain pharmacy and patient verification that PrEP was dispensed (picked up by the patient), which was documented in the medical record.
Independent measures: Demographics, clinical, and environmental characteristics
The standardized baseline and follow-up assessments contained abbreviated segments of the Global Appraisal of Individual Needs (GAIN)51,52 to capture demographics, physical and mental health, healthcare and substance use treatment, housing situation, transportation, and health insurance, as well as items adapted from the Collaborative Injection Drug User Study III/Drug User Intervention Trial Assessment Instrument (CIDUS/DUIT) 53 to gather drug injection behaviors, SSP use, and HIV testing history. These core instruments were supplemented with validated measures of perceived HIV risk, 54 psychological resilience, 55 HIV-related community stigma, 56 and PrEP awareness, acceptability, and use. 57 Key measurement domains are indicated below.
Substance use
Drawn from the CIDUS/DUIT instrument, 53 current primary drug of injection was assessed with the question, “In the past month which drug did you inject most often?” Frequency of injection was gathered by asking participants, “How many times (number of injections) did you inject drugs in the last 30 days?”
Health status
We included a brief validated measure of perceived HIV risk 54 ; for this trial, we included the three-item subscale assessing cognitive judgments regarding likelihood of infection: “I am sure I will not get infected with HIV”; “There is a chance, no matter how small, that I could get HIV”; and “I think my chances of getting HIV are: zero to very large.” Participant responses were summed, with a possible range of 3–18, higher scores indicated higher perceived risk. There were no missing data on these items. Current health worry was assessed with one yes/no item drawn from the GAIN: “In the past 90 days, have you been worried about your health or your behaviors?” A brief 6-item mental health disorder screener from the GAIN 53 assessed recency of somatic, depressive, anxiety, trauma, psychosis, and suicidality symptoms. Endorsement of symptoms in the past 3 months were summed; scores of three and above were considered severe distress. Finally, the CD-RISC-10 55 assessed aspects of psychological resilience, with higher scores indicating higher resilience. Cronbach’s alpha in this sample was 0.75.
Health services use
Substance use treatment was assessed with the GAIN item querying, “When was the last time (if ever) you received any counseling, treatment, medication, case management or aftercare for your use of alcohol or any drug?” This variable was dichotomized to “within the past 90 days” or “not within the past 90 days.” Participants were asked a series of additional items to assess healthcare access and utilization, including “Have you received any care from a doctor in the past year?” For HIV testing, participants were asked to report the most recent occasion of testing: within the prior 6 months, more than 6 months ago but less than 1 year, or 1 year or more ago. Finally, to measure SSP utilization, we asked participants “How many times have you visited this SSP site in the past month?”
Social determinants of health
Participants were asked about their access to tangible resources. Current housing status was assessed with one item from the GAIN: “When was the last time, if ever, you considered yourself to be homeless?” This variable was dichotomized to “within the past 90 days” or “not within the past 90 days.” Access to transportation was assessed with one item: “What form of transportation do you usually use to get from place to place?.” Cellular access was assessed with one item: “Do you currently have access to a personal cell phone?” Health insurance status was assessed by asking: “Are your medical costs currently covered by any of the following insurance programs”: Medicaid, Medicare, private insurance, VA benefits, or other. Finally, we also included a validated 3-item scale to gather participants’ perceptions of public attitudes toward HIV as a measure of community HIV-related stigma, 56 where higher scores represent higher stigma.
PrEP knowledge, awareness, interest, and preferences
We adapted questions on PrEP awareness and interest, 57 using the following items: Have you ever heard of HIV-negative people taking a pill every day to reduce their chances of getting HIV infection (this is called PrEP, for Pre-Exposure Prophylaxis)? And, “Please rate your level of interest in learning more about PrEP?” measured on a Likert scale from “not interested” to “extremely interested.” An additional knowledge item was also included: “Please rate your level of knowledge about PrEP, what it is and how it is used?” where responses ranged from very low to very high.
Sample size
The pilot RCT was not designed to establish superiority of the SBCM-PrEP, but to generate preliminary evidence of effect sizes for application in a subsequent larger, fully powered trial. The sample size of 80 aligns with recommended sample sizes for two-arm pilot trials structured as planning for subsequent major trials.58–60
Randomization
Participants were randomly assigned to either the CDC-PrEP or SBCM-PrEP with a 1:1 allocation per a computer-generated randomization schedule stratified by site and gender. The study Co-Investigator biostatistician generated the allocation sequence and notified the Principal Investigator by secure email. The Study Coordinator assigned participants to intervention arms. The trial was not blinded.
Statistical methods
Descriptive statistics (e.g., frequencies, proportions, means, and standard deviations) were used to describe demographic, clinical, and environmental characteristics by trial arm using the {gtsummary} package in R (version 4.1.1). 61 Logistic regression models were utilized to assess intervention arm differences in PrEP initiation (primary; measured by dispensed prescription for emtricitabine/tenofovir disoproxil fumarate) during the 6-month intervention period and our secondary efficacy outcomes of interest: (1) completion of initial clinical visit with a study PrEP provider; (2) completion of second clinical visit with study PrEP provider to review initial lab results; and (3) issuance of initial electronic or paper PrEP prescription by study provider, also within 6 months post-baseline. Logistic regression models were adjusted for site and gender due to their use as stratification factors. Tests were two-sided at the 0.05 level. We used an intent-to-treat (ITT) approach, analyzing data from all participants based on the trial arms to which they were randomized regardless of whether they received or completed the randomized treatment to which they were assigned. We also examined outcomes per protocol, whereby those with no exposure to their assigned intervention were excluded from the analysis. Analysis of the primary outcome (PrEP initiation) and secondary outcomes were not affected by loss to follow up, as we used health department medical record data to examine clinical outcomes of interest.
Results
Eighty participants were enrolled into the pilot RCT from November 1, 2022 to October 31, 2023. Forty-one participants were randomized to the SBCM-PrEP intervention arm, and 39 to the CDC-PrEP arm (see Figure 1, study flow diagram). Table 1 presents demographic, clinical, and environmental characteristics of the sample at baseline, compared across study intervention conditions. Differences by intervention condition are not apparent, given our randomization procedures. As shown in Table 1, more than two-thirds of trial participants endorsed primary stimulant injection within the past month, which reflects overarching changes in the drug supply in the region. Of note, more than one-third of primary stimulant users also reported opioid injection within the past month, indicating substantial overlapping use (data not shown). On average, participants reported 3.2 times injecting per day, and 99 times in total, during the past month. Notably, the sample of PWID is impacted by social determinants of health, with over 40% indicating they were unstably housed within the past 3 months. Perceived risk for HIV was moderate overall, with a mean score of 12.1 (range 3–18) endorsed on the cognitive subscale of personal likelihood to acquire HIV. Additionally, a large majority (82.5%) reported very low or low knowledge about PrEP, yet half (50.0%) expressed very or extremely high interest in learning more about PrEP.
Baseline SSP sample characteristics by intervention group, N = 80.

Range 3–18.
Range 0–40.
Range 3–15.
PrEP, pre-exposure prophylaxis; SBCM, strengths-based case management; SSP, syringe services program.
As indicated in Figure 1, 96.2% (77/80) of study participants entered the PrEP care cascade by attending at least one PrEP intervention session, regardless of intervention assignment. Tables 2 and 3 display the results of logistic regression models for the primary and secondary outcomes measures by intervention arm, gender, and site, for ITT and per protocol analyses, respectively, within the 6-month study period. Overall, 88% (70/80) of participants were linked to the embedded PrEP provider and completed an initial PrEP clinical visit and point of care testing for PrEP eligibility (initial PrEP clinical visit); 48% (38/80) attended the second PrEP clinical visit (to review initial lab results); 28% (22/80) were issued an initial prescription for PrEP (issuance of initial electronic or paper PrEP prescription by study provider); and 9% (7/80) had PrEP dispensed (prescription picked up by the patient) during the 6-month study period. No participants who were linked to the PrEP provider (N = 70) tested positive for HIV during the course of the study. Of those who returned for PrEP test results but did not receive a prescription, reasons included: requiring re-testing due to untimely follow-up (9), losing PrEP indication (1), participant choosing to further consider prescription (1), actively declining prescription (3), and returning when provider not onsite (2). We observed a statistically significant effect of site on odds of completing the second clinical visit, both ITT and per protocol (adjusted odds ratio (aOR) 3.62, p = 0.007; aOR 3.62, p = 0.009) in favor of Site 2; additionally, all participants who initiated PrEP were enrolled from Site 2. No statistically significant differences were observed in primary or secondary outcomes by gender. By intervention condition, we observed a 13.6% difference (34.1% vs 20.5%; ITT) and 14.9% difference (35.9% vs 21.0%; per protocol) in prescription issuance and a 12.1% difference (14.6% vs 2.5%; ITT) and 12.8% difference (15.4% vs 2.6%; per protocol) in the primary outcome of dispensed PrEP prescription, in favor of the experimental SBCM-PrEP intervention, though neither of these comparisons reached statistical significance. One serious adverse event was documented during the course of the trial, which involved the death of a participant randomized to the SBCM-PrEP condition; this death was unrelated to trial participation.
Unadjusted and adjusted associations with PrEP outcomes among SSP clients (N = 80), intention-to-treat.
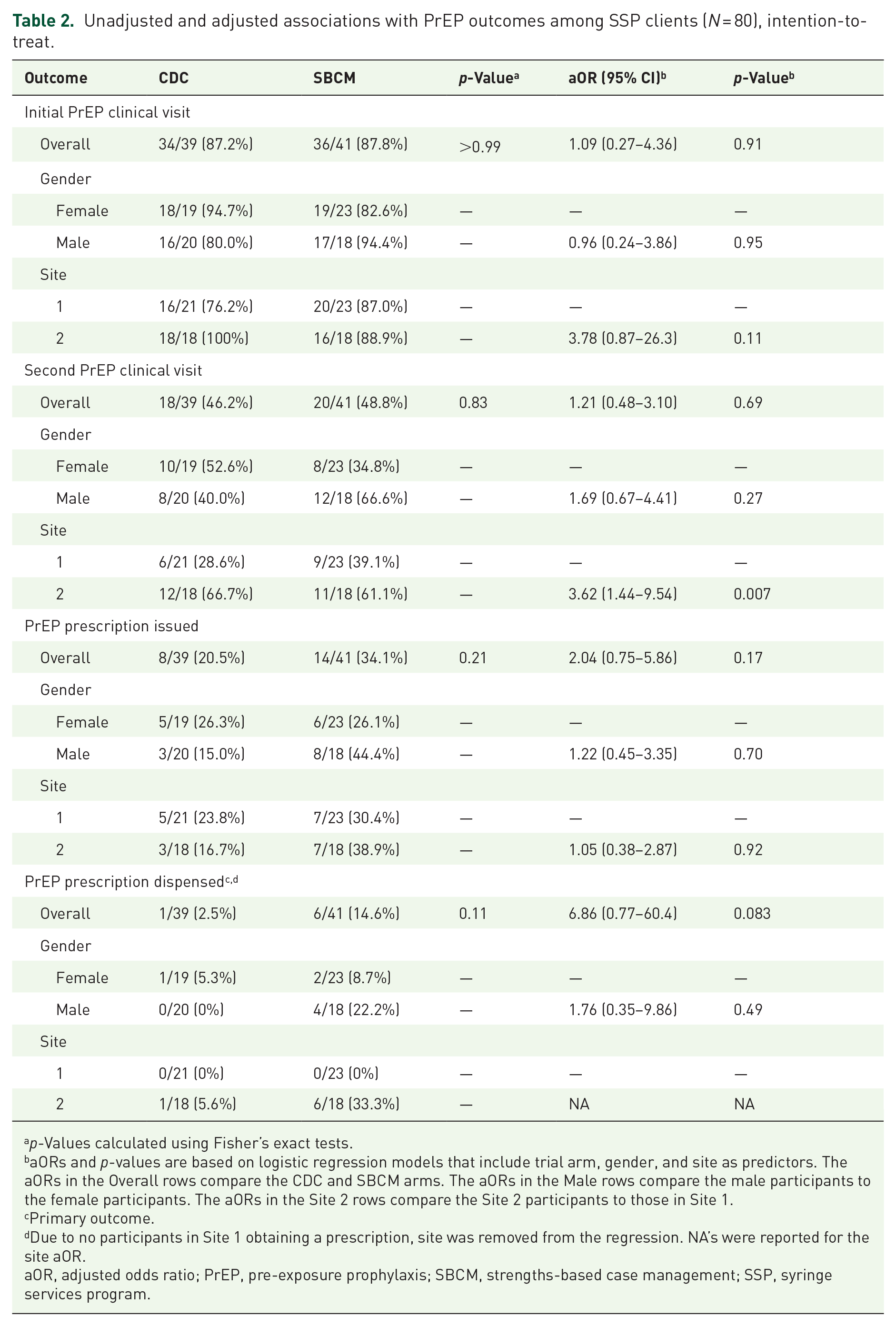
p-Values calculated using Fisher’s exact tests.
aORs and p-values are based on logistic regression models that include trial arm, gender, and site as predictors. The aORs in the Overall rows compare the CDC and SBCM arms. The aORs in the Male rows compare the male participants to the female participants. The aORs in the Site 2 rows compare the Site 2 participants to those in Site 1.
Primary outcome.
Due to no participants in Site 1 obtaining a prescription, site was removed from the regression. NA’s were reported for the site aOR.
aOR, adjusted odds ratio; PrEP, pre-exposure prophylaxis; SBCM, strengths-based case management; SSP, syringe services program.
Unadjusted and adjusted associations with PrEP outcomes among SSP clients (N = 77), per protocol.

p-Values calculated using Fisher’s exact tests.
aORs and p-values are based on logistic regression models that include trial arm, gender, and site as predictors. The aORs in the Overall rows compare the CDC and SBCM arms. The aORs in the Male rows compare the male participants to the female participants. The aORs in the Site 2 rows compare the Site 2 participants to those in Site 1.
Primary outcome.
Due to no participants in Site 1 obtaining a prescription, site was removed from the regression. NA’s were reported for the site aOR.
aOR, adjusted odds ratio; PrEP, pre-exposure prophylaxis; SBCM, strengths-based case management; SSP, syringe services program.
Discussion
The present study demonstrated proof of concept for integrated PrEP care within SSP settings in rural areas, indicated by high uptake of study intervention sessions and high initial linkage rates to embedded PrEP providers. Although PWID in rural KY report stigmatizing experiences from healthcare providers in traditional care sites, 62 data from this prospective pilot trial indicate high acceptability to participate in PrEP education, HIV risk assessment, and point of care PrEP testing within rural Appalachian SSPs, in communities that were otherwise without access to PrEP. Given that SSPs in rural communities tend to have high utilization 63 and are perceived to be non-stigmatizing environments by PWID,30,32 our findings suggest that enacting structural changes to support integrated PrEP care services at point of care in rural SSPs is a promising practice, with 96% in this study entering the PrEP care cascade by receiving a structured PrEP intervention session, and 88% completing an initial clinical visit for PrEP. Overwhelmingly participants linked to the PrEP provider after a single brief intervention session and the intervention assignment was unrelated to completing the initial PrEP clinical visit. This finding supports the importance of implementing structural changes to expand PrEP access, in this case, co-located PrEP services that were essentially available “on demand” at the time of the SSP visit.
Despite high initial linkage to PrEP care, we noted substantial levels of care discontinuation at the second PrEP clinical visit, at which time participants would receive and discuss their test results, engage in any additional testing if needed based on initial results, and receive a prescription for PrEP, if desired and appropriate. In the present analysis, we found that 38 (48%) participants returned for their second clinical visit and test results during the study period. Both study intervention staff and study providers made multiple attempts to contact participants for return using study locator information or directly onsite at the SSPs. Similar to uptake of the initial clinical visit, we found essentially no statistical differences by intervention arm related to attendance at the follow-up PrEP clinical visit. Nevertheless, significant site differences were apparent in return for the second clinical visit, with Site 2 participants having much higher odds of return than Site 1 participants. We speculate that this may be a function of SSP operational characteristics and implementation challenges that required a mid-study change in PrEP provider at Site 1, which may have potentially disrupted continuity of care for study participants. This finding also suggests that more deeply tailored implementation strategies may be required to better support unique site operational needs and internal challenges moving forward.
Importantly, we observed a 13.6% (ITT) and 14.9% (per protocol) difference in prescription issuance in favor of the experimental SBCM-PrEP arm, and a 12.1% (ITT) and 12.8% (per protocol) difference in the primary outcome of PrEP initiation (dispensed PrEP prescription picked up by the patient). Though neither of these comparisons reached statistical significance due to the small sample size in the pilot trial, the effects appear to represent clinically important differences that warrant further investigation. Notably, of the 99 individuals approached and screened for the trial, none had a prior history of PrEP prescription, which suggests that an approximate 15% PrEP initiation rate in the SBCM-PrEP arm is clinically impactful. These findings are of particular importance given that the study sample was largely comprised of primary stimulant users, among whom risk for HIV has been notably elevated in prior research.64,65
The pilot trial established a clinical workflow and protocol for integrated PrEP care in two rural SSP locations. Nevertheless, differences in the primary outcome of PrEP initiation indicate comparatively higher success in one site related to the implementation context. In particular, a close collaboration with local specialty pharmacy services in Site 2 was forged to facilitate seamless and timely access to PrEP medications, which was simply not feasible to accomplish in Site 1. As we consider the pilot results in the context of intervention and implementation challenges, it appears clear that augmented multi-level intervention and implementation support will be required to optimize PrEP uptake among rural PWID. Lazarus et al. 66 described an aspirational person-centered health system that prioritizes client and community needs, rights, and preferences as a model for achieving better HIV outcomes for clients, rather than choosing prevention and treatment regimens based on what is convenient for health systems. Building on the foundation of this pilot trial, strong local partnerships and key recommendations from stakeholder exit interviews, the deployment of stepped-care adaptive interventions67,68 would appear to be a reasonable next step in testing the optimal intervention pathways for PrEP uptake and persistence among rural PWID, by tailoring the interventions according to initial response and participant needs.
Additionally, consideration of novel implementation strategies to support same-day PrEP prescribing appear to be warranted 26 in the spirit of providing low threshold care for PWID, which aligns with our findings that attending a second PrEP clinical visit represented a substantial barrier for patients. Studies report that same-day PrEP prescribing is safe, feasible, and acceptable to men who have sex with men, 69 yet at this time data are largely unavailable for PWID. Our results demonstrated high uptake of initial point of care PrEP testing and no PrEP exclusions based on participant test results, which lends support to the safety of same-day prescribing. Current recommendations support lessening the time to PrEP prescribing under certain conditions, particularly if current HIV test results are available, to enable earlier PrEP initiation while awaiting full results if the provider and patient are amenable to this approach. 70 The recent publication of the US Preventive Services Task Force grade A recommendation statement on PrEP to prevent acquisition of HIV among people at increased risk, including PWID, may accelerate and empower implementation of additional PrEP programming for this population. 71
This study has limitations that are important to consider when interpreting the findings. The pilot trial we conducted involves a relatively small sample of participants collected from one specific geographic region of rural Kentucky, which may impact the generalizability of our findings to PWID in other areas and environments. Limited sample size also impacted our ability to engage in more extensive statistical modeling approaches. Additionally, we experienced significant loss to follow-up in the pilot trial as depicted in the study flow diagram. As noted earlier, 38 (48%) participants returned for their second clinical PrEP visit and test results, and 36 (45%) completed either a 3- or 6-month research follow-up interview visit. The primary and secondary outcomes we examined were derived from patient medical records, which is optimal due to high loss to follow-up over time. Follow-up was adversely impacted by high rates of arrest and incarceration and social determinant barriers that prevented return to the SSPs. In our pilot baseline data, 37% had been incarcerated in the past year, and several were incarcerated during the study period, limiting access for follow-up. The high level of attrition in the follow-up component of the study aligns with widely known challenges retaining people who use drugs in longitudinal HIV-related research,72,73 including in SSPs, 74 which indicates that more robust and person-centered retention approaches are needed moving forward.
Conclusion
This study adds to the growing evidence base to support expanding PrEP care opportunities for PWID outside of traditional medical settings in order to improve uptake of preventive care and treatment. 19 Utilizing SSPs as a venue to provide comprehensive PrEP services appears promising as a pathway to encourage more PWID to engage in HIV prevention services, and may be particularly important in communities that experience a lack of PrEP providers in other healthcare settings. Integration of PrEP services with existing public health programs provides a seamless pathway for entry to PrEP care, thus mitigating a major access challenge. 75 Innovative interventions to increase PrEP access, initiation and persistence by PWID in non-traditional care settings fill a critical need to improve the PrEP care cascade among PWID. Continued innovation to increase accessibility of novel PrEP formulations, most notably, long-acting injectables (LAIs), also represents an important area for future research in rural areas where HIV specialty care is limited, particularly considering that many PWID endorse a preference for LAI-PrEP.76,77 Developing academic–community partnerships and defining implementation strategies to support the successful rollout, delivery, and administration of LAI-PrEP in medically underserved areas represents a critical opportunity to expand care access among PWID.
Footnotes
Acknowledgements
The authors gratefully acknowledge the Knox County Health Department and Cumberland Valley District Health Department for their ongoing participation and partnership on the study, and the Christian County Health Department for their consultation and assistance with provider training. We would like to thank Appalachian Regional Healthcare Barbourville for their contributions to the study.
