Abstract
We describe a group of four travelers returning to the United States and Canada who acquired Leishmania braziliensis infection in the Peruvian Amazon. Pentavalent antimonials are the preferred treatment option for cutaneous leishmaniasis (CL) in most endemic countries in Central and South America. However, we initially treated our patients with liposomal amphotericin B (LAB) and miltefosine since these are the only two available Food and Drug Administration approved drugs in the United States. Refractory disease was common as three of the four travelers required repeated courses of miltefosine and two also received LAB. One patient required intravenous therapy with meglumine antimoniate (NMG), and one received intralesional NMG. Given the increasing number of cases of CL identified in the United States, there is an urgent need for expanded access to pentavalent antimonials for treating leishmaniasis acquired in Central and South America.
Introduction
An estimated 350 million people in 98 countries are at risk of acquiring cutaneous leishmaniasis (CL). 1 Transmission occurs predominantly through female phlebotomine vectors’ inoculation of the Leishmania parasite. International travel and growing immigration from countries endemic for leishmaniasis has led to an increasing recognition of this parasitic infection in travelers and newly arrived immigrants and asylum seekers in the United States. There are more than 20 species of Leishmania species, with each species having the potential to cause more than one clinical syndrome, including cutaneous, mucocutaneous, visceral, and viscerotropic forms. Optimal treatment of leishmaniasis requires identifying the Leishmania spp. to guide the choice of antiparasitic therapy.2,3
Leishmania (Viannia) braziliensis is associated with cutaneous disease and, in some cases, mucosal involvement of the oropharynx and nasopharynx. In most endemic countries in Latin America, systemic administration of the pentavalent meglumine antimoniate (NMG) remains the first line of treatment for CL. Other drugs used in the treatment of CL include miltefosine, intralesional pentavalent antimonials, liposomal amphotericin B (LAB), and pentamidine isethionate. The treatment of CL can be protracted and refractory, with recent studies showing that NMG may be more effective than LAB.4 However, the available evidence is only based on retrospective studies.3–6
In the United States and Canada, most forms of CL are identified in travelers returning or immigrating from endemic areas of Central and South America. Herein, we report a case series of four returning travelers who visited a field biological station in the Peruvian Amazon and acquired CL caused by L. braziliensis. The availability of pentavalent antimonial drugs in the United States is limited by the requirement to obtain investigational new drug (IND) approval, which is a complicated and time-consuming process that often delays antiparasitic therapy.7 Miltefosine is an orally administered antiparasitic drug that is Food and Drug Administration (FDA)-approved for the treatment of cutaneous leishmaniasis and is frequently prescribed for treating leishmaniasis in the United States.2,3 However, miltefosine is not consistently effective against all cases of CL, it is costly, sometimes may be associated with significant side effects, and it is teratogenic. Most of our patients had a protracted clinical course requiring LAB, extended courses of miltefosine, or the addition of NMG. Based on our experience caring for these four patients, we discuss some limitations of the drug armamentarium currently available to treat CL in the United States.
Case 1
A 40-year-old man with morbid obesity and pre-diabetes presented to an Infectious Disease clinic in New York City with progressive scaly skin lesions on his nose and dorsal left fifth proximal interphalangeal joint. One month earlier, he had returned from the Peruvian Amazon, where he spent 2 months at the “Los Amigos” biological field station working on a large-scale bio-surveillance project. He worked with reptiles, amphibians, birds, and primates. He highlights not using diethyltoluamide (DEET) since it is toxic to frogs. One week before returning to the United States, he noticed an insect bite over his nose and left fifth finger that became papular and increased in size within 2 weeks (Figure 1(a) and (b)).

Case 1: (a, b) Nose and finger lesion upon presentation and (c, d) nose and finger lesion during treatment with LAB.
We performed a biopsy of the finger lesion in his digit, revealing the presence of amastigotes within histocytes in the dermis (Figure 2(a) and (b)). The lesion over his nose progressed rapidly, concomitant with nasal congestion, edema, and induration around his bilateral orbits, suspicious for mucosal involvement (Figure 1(c) and (d)); thus, he was hospitalized for intravenous (IV) LAB and further workup. A rhinoscopy demonstrated no mucosal involvement. However, computed tomography of the sinuses showed marked soft tissue swelling along the nose extending into the columella concerning mucosal involvement; hence, he received an 8-day course of intravenous LAB at a dose of 4 mg/kg daily followed by a course of miltefosine 50 mg every 8 h. Upon discharge and completion of a 28-day course of miltefosine, there was minimal improvement, prompting a second course of miltefosine at the same dosage. Despite treatment with LAB and two courses of miltefosine, new satellite lesions developed around his left fifth finger with sporotrichoid lymphangitis 3 months later (Figure 3(a)). He underwent an emergent biopsy of the satellite lesion with tissue sent for path, culture, and polymerase chain reaction (PCR) testing; the latter of which was performed at the Centers for Disease Control and Prevention (CDC) and confirmed infection with L. braziliensis. Given the high suspicion of uncontrolled infection, he was admitted for a second course of IV LAB at a higher dose of 5 mg/kg every 24 h, along with oral miltefosine 50 mg every 6 h. In addition, an emergency investigational new drug (eIND) application was submitted to the FDA for IV NMG. On day 5 of the hospital stay, he started IV NMG at a dose of 1215 mg daily, and LAB along with miltefosine were discontinued. He completed a 28-day course of IV NMG with no significant side effects. Both lesions significantly improved within 1 week and eventually resolved (Figure 3(b) and (c))

Case 1: Histopathology showing amastigotes within histiocytes.

Case 1: (a) Satellite lesion after LAB and one course of miltefosine and (b and c) Lesions after completed treatment with IV NMG.
Case 2
A 24-year-old man was referred to the travel medicine clinic for evaluation of several skin lesions upon returning from the Amazonian region of Ecuador and Peru. He first developed a papular lesion in the abdomen wall that ulcerated over 4 weeks. He also noticed additional papular lesions in his left knee, left shoulder, and chest.
He recently spent many months in the Amazonian region of Ecuador and Peru, including the “Los Amigos” biological field station. He limited his use of DEET due to its toxicity to amphibians. He spent most nights working outdoors and reports multiple insect bites.
Upon examination, a 3 cm abdominal plaque with raised, violaceous borders with central ulceration had a honey-colored crust. He also had small papules on his left medial knee, left posterior shoulder, anterior chest, and upper abdomen. His oropharynx and nasopharynx were normal. We performed two punch biopsies from the abdominal ulcer edges and a shave biopsy of the lesion in his left knee lesion. The histopathology showed “dense perivascular and peri adnexal infiltrate of lymphocytes, plasma cells, and numerous histocytes forming a granulomatous inflammatory pattern. Numerous 2–4 µm organisms in histiocytes morphologically consistent with amastigotes of Leishmania.” The PCR test was positive for Leishmania (Viannia) braziliensis from his knee and abdominal biopsies. The patient was treated with cryotherapy and oral miltefosine 50 mg every 8 h for 28 days. After the biopsy, his left knee lesion ulcerated and became infected with Enterobacter spp., which was successfully treated with oral doxycycline. After completing a miltefosine course, the lesions were healing, and he returned to work in Ecuador, but after 2 weeks, the lesions worsened. He visited a Shell, Ecuador clinic where the abdominal and knee lesions were enlarging, with evidence of bacterial secondary infection (Figure 4(a)). This patient received intralesional NMG applied at a dose of 10.5 ml weekly.
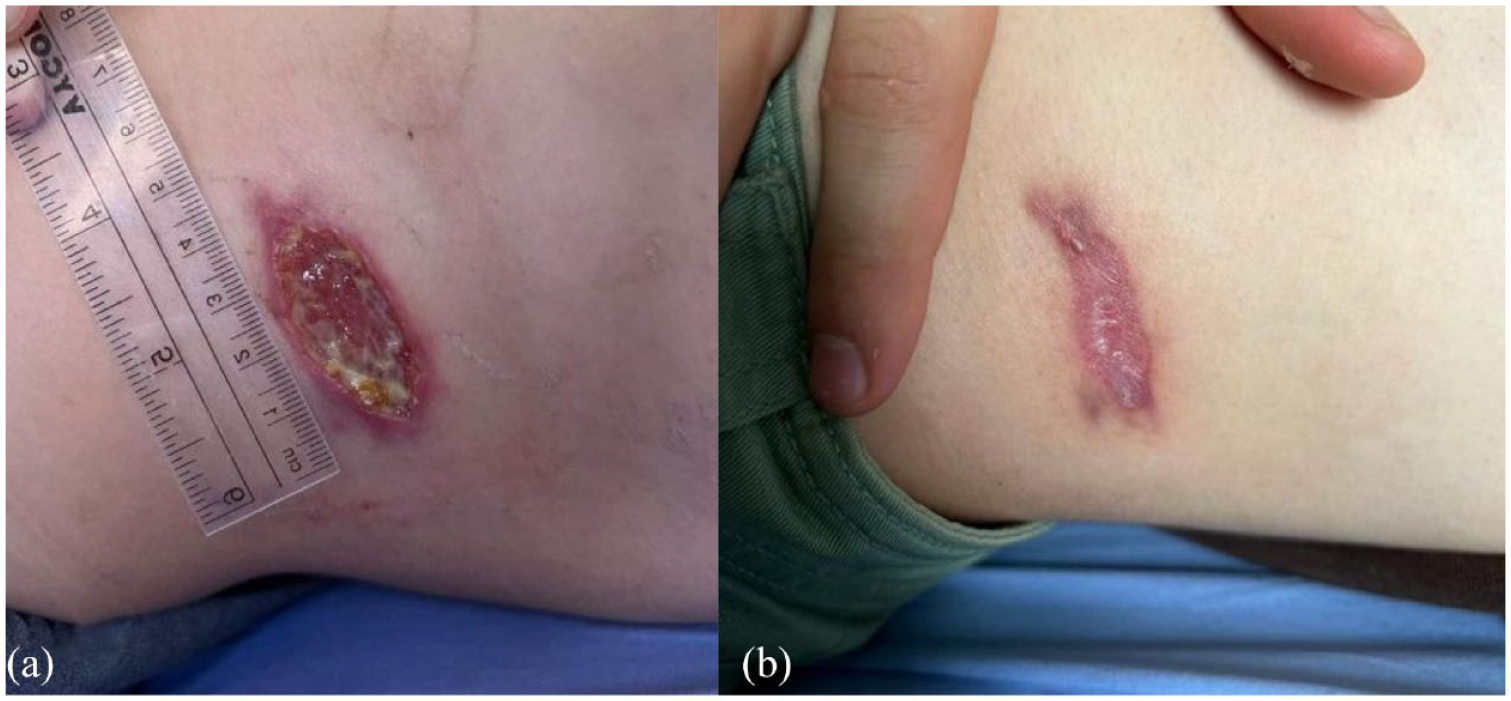
Case 2: (a) Abdominal lesion after one course of miltefosine and (b) abdominal lesion halfway through the second course of miltefosine along with intralesional NMG.
Five weeks into the intralesional NMG, he returned to the United States and a new papule was discovered in his left axilla, leading to a second 28-day course of oral miltefosine 50 mg every 8 h along with intralesional NMG (Figure 4(b)). In total, he received 12 doses of intralesional NMG with complete healing of lesions.
Case 3
A 24-year-old man was referred to the infectious disease clinic for facial lesions concerning leishmaniasis. Two months earlier, he noticed small bug bites along his jaw line bilaterally, which failed to heal and subsequently enlarged and became crusted.
He is a student working on a biological field station in the Peruvian Amazon where he spent 5 weeks. He worked mainly with reptile species and did not apply DEET but wore protective gear. He was seen at his university health service, where wound care was recommended. However, he was later informed that a fellow traveler had been diagnosed with leishmaniasis, resulting in his referral to an infectious disease clinic.
His examination revealed non-tender, non-pruritic, and symmetric lesions along his jawline, approximately 2 × 1.5 cm in size, with surrounding erythema. A biopsy showed tuberculoid granulomas with early caseation within the superficial and mid dermis with no evidence of amastigotes. Molecular PCR testing at Stanford University was reported positive for Leishmania braziliensis complex (L. panamensis, L. guyanensis, or L. braziliensis).
He had no evidence of mucosal involvement. He completed a 56-day course of oral miltefosine 50 mg every 8 h (two full courses). By the end of therapy, his lesions had healed, and 3 months later, there was no evidence of relapse. (Unfortunately, images are not available for this case). He developed severe testicular pain during the second course of miltefosine that resolved spontaneously.
Case 4
A 28-year-old healthy woman was evaluated at the local tropical medicine clinic for a non-healing ulcer on her forearm. She had spent 2 months in the Peruvian Amazon as part of a fieldwork expedition related to her research degree. Upon leaving Peru, she spent 1 week in Panama. By the end of her trip, she noted a small papule on her right forearm, which progressed into a chronic ulceration within 3 weeks. There were no other lesions and no apparent mucosal involvement. Prior to the presentation, she visited a walk-in clinic where she was prescribed 6 weeks of empiric fluconazole with no improvement after 3 weeks of treatment.
The shallow lesion measured approximately 2 × 3 cm with a superficial dry eschar. No sporotrichoid pattern, satellite lesions, lymphadenopathy, or surrounding cellulitis existed. Biopsy was not performed on this patient, given the high clinical suspicion of leishmaniasis. She received an initial 28-day course of oral miltefosine 50 mg every 8 h. Six weeks after completing the treatment, the lesions had notably improved with significant resolution of the eschar and a slowly healing keloid scar (Figure 5(a) and (b)).

Case 4: (a) Forearm lesion 2 months after completion of treatment with one course of miltefosine and (b) Forearm lesion 1 year after completion of treatment with one course of miltefosine.
Discussion
While there is autochthonous transmission of leishmaniasis in some areas in the United States, most cases of CL are diagnosed in international travelers and recently arrived immigrants from endemic countries of transmission.1,2,8 Direct parasitological visualization of the parasite is the gold standard of diagnosis. Molecular identification of Leishmania spp. offers the opportunity for tailoring antiparasitic therapy, and it is a crucial surveillance tool. 5
Pentavalent antimonials administered intramuscularly or intravenously have been the first-line treatment for L. braziliensis disease for decades in endemic countries in Central and South America. LAB and oral miltefosine constitute the only FDA-approved medications for treating CL in the United States. As such, three of our cases had cutaneous involvement without evidence of mucocutaneous disease and consequently were treated with miltefosine. However, in our first case, the location of the lesion and findings were concerning for mucocutaneous disease prompting treatment with LAB followed by a course of miltefosine.
Although treatment failure has been reported and associated with the host’s immunological response, little has been done to assess the effect of individual variation in pharmacokinetics or other drug-related responses in the host. 9 For instance, one patient had minimal clinical improvement after one LAB and two miltefosine courses. It is noteworthy that this patient was obese and thus did not achieve the recommended miltefosine dosing of 2.5 mg/kg/day with the institution of the standard dose, which likely contributed to treatment failure. Interestingly, he demonstrated a rapid clinical response to IV NMG, highlighting the efficacy of NMG in treating cutaneous and mucocutaneous cases caused by L. braziliensis.
Two of our patients were treated with NMG after failing to respond to FDA-approved therapy. One patient received a course of IV NMG obtained through an eIND request to the FDA, while a second patient received intralesional NMG in Ecuador, both achieving rapid clinical improvement. In an extensive series of French travelers, LAB demonstrated poor clinical responses in treating CL caused by species of the Viannia subgenus, including L. braziliensis. However, this drug provided excellent clinical outcomes in those infected with L. tropica and L. infantum. 10 Pentavalent antimonials have reduced efficacy when there is mucosal involvement; LAB plays a critical role in treating patients with proven mucosal or visceral involvement.
Conclusion
Our case series demonstrates the protracted clinical course of patients with L. braziliensis infection despite receiving FDA-approved treatment options in the United States. Increased availability of pentavalent antimonials is essential to improving the clinical outcomes of patients with CL. Obtaining NMG in the United States requires submission of an IND, which is a complicated and time-consuming process that delays patient care. Since most cases of leishmaniasis seen in the United States occur among travelers returning from countries endemic for Viannia subgenus, including L. braziliensis, we believe that the CDC should proactively improve access to pentavalent antimonial drugs. Likely, the number of cases of CL identified across the United States travel clinics will continue to increase in the upcoming years, linked to the increasing number of travelers bound to endemic countries and the augmenting flux of migrants seeking asylum.
Supplemental Material
sj-docx-1-tai-10.1177_20499361241274254 – Supplemental material for Challenges in the treatment of cutaneous leishmaniasis caused by L. braziliensis in four travelers: a case series
Supplemental material, sj-docx-1-tai-10.1177_20499361241274254 for Challenges in the treatment of cutaneous leishmaniasis caused by L. braziliensis in four travelers: a case series by Carolina Moreira Sarmiento, Guy Crowder, Bonnie Meatherall, Jacob Bezemer, Yenjean Hwang, Ariel Gordon, Aisha Mumtaz, Brittany Jackson, Juan David Ramírez, Alberto Paniz-Mondolfi, Carlos Franco-Paredes and Valida Bajrovic in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
