Abstract
Limited therapeutic options in visceral leishmaniasis (VL) make the treatment of this neglected disease very challenging. In addition to this, long treatment duration and toxic adverse effects make it even more difficult. With no effective vaccine available to date, treatment of VL is based only on chemotherapy. In the Indian subcontinent, a single dose of liposomal amphotericin B (L-AmB) and multidrug therapy (L-AmB + miltefosine, L-AmB + paromomycin [PM], or miltefosine + PM) are the treatments of choice for VL. In East Africa, however, combination therapy of pentavalent antimonials (Sbv) and PM remains the treatment of choice, and in the Mediterranean region and South America, L-AmB is the recommended drug. Fexinidazole and PA-824 are new promising drugs which have shown encouraging results in preclinical studies.
Introduction
In tropical countries protozoan parasitic diseases pose a major public health problem. After malaria, leishmaniasis stands second in having a high mortality and morbidity burden leading to economic loss. Leishmaniasis, is caused by an obligate intracellular protozoan of the genus Leishmania. It broadly manifests as visceral leishmaniasis (VL); also known as kala-azar, cutaneous leishmaniasis (CL) and mucocutaneous leishmaniasis. VL is caused by Leishmania donovani complex: L. donovani, is the causative organism of VL in the Indian subcontinent and Africa; Leishmania infantum (syn L. chagasi) causes VL in the Mediterranean basin, Central and South America [WHO, 2010].
VL transmission could be considered to be either anthroponotic or zoonotic depending on the mode of transmission. Anthroponotic VL is caused by L. donovani and is found in the Indian subcontinent and the Horn of Africa, with humans as the major reservoir of transmission [Magill, 1995; Pearson, 1999]. Sand fly species, Phlebotomus in the Old World and Lutzomyia in the New World are the only proven vectors inflicting humans [WHO, 2010]. Zoonotic VL is caused by L. infantum and found in the Mediterranean, the Middle East and Brazil, with the domestic dog being the most important reservoir host [Magill, 1995].
Worldwide, annual incidence of VL is estimated to be 0.2–0.4 million, and more than 90% of cases occur in just six countries: India, Bangladesh, Sudan, South Sudan, Ethiopia and Brazil. The burden of VL is further increased by rising prevalence of coinfection with HIV. The majority of HIV–VL coinfection cases were from southwestern Europe initially, but the number of cases is increasing in sub-Saharan Africa, especially Ethiopia, Brazil and South Asia [Alvar et al. 1997, 2008, 2012; Desjeux and Alvar, 2003].
VL-HIV coinfection is increasing in VL endemic regions of Bihar as evident by a recent study from Bihar, India, which showed that 2.4% of all VL patients ≥14 years of age presenting with VL were coinfected with HIV [Burza et al. 2014]. In north Brazil, the incidence of HIV-VL coinfection has increased from 0.32/100,000 in 2007 to 1.08/100,000 in 2010 [Albuquerque et al. 2014].
Clinically, VL manifests as prolonged fever, splenomegaly, hepatomegaly, pancytopenia, progressive anemia and weight loss, and is fatal unless treated. About 50% of patients with VL in Sudan and 1–3% in India develop post-kala-azar dermal leishmaniasis (PKDL) characterized by indurated nodules or depigmented macules [Thakur and Kumar, 1992; Zijlstra et al. 2003].
Recent development in the treatment of VL
Review of available antileishmanial drugs.

i.m., intramuscular; i.v., intravenous; VL, visceral leishmaniasis.
Pentavalent antimonials (SbV)
SbV is available in two forms: Sodium stibogluconate and meglumine antimoniate (MA). It is administered in doses of 20 mg/kg body weight for 28–30 days. In Bihar (India) and to some extent in adjoining Nepal there has been increasing resistance to Sbv and this has led to implementation of alternative treatment regimens for these regions [Sundar et al. 2000; Rijal et al. 2003]. However its efficacy remains high in other parts of world [Sundar and Chakravarty, 2015b].
Serious toxicities like prolonged QT interval is of electrocardiogram (QTc), cardiac arrhythmias, ventricular premature beats, ventricular tachycardia, ventricular fibrillation and torsades de pointes, can occasionally occur. Arthralgia, myalgia, elevated hepatic and pancreatic enzymes are other common associated toxicities. Response to SbV in patients with HIV–VL coinfection, however, has shown less efficacy and been associated with increased mortality as compared to HIV-negative VL cases [Ritmeijer et al. 2006]. Some adverse effects especially chemical pancreatitis is more common in HIV coinfected patients [Diro et al. 2014; Sundar and Chakravarty, 2015a].
Amphotericin B
Amphotericin B deoxycholate (AmB) is recommended for the treatment of refractory VL in India and used in doses of 0.75–1.0 mg/kg for 15–20 intravenous infusions with high cure rates (CR) ∼ 100% [Mishra et al. 1992; Thakur et al. 1999]. Nevertheless, its toxic side effect profile includes infusion reactions, nephrotoxicity, hypokalemia and myocarditis. It is also recommended for the treatment of PKDL in a dose of 1 mg/kg/day, up to 60–80 doses for 4 months in the Indian subcontinent [Thakur, 1997; WHO, 2010].
Adverse effects associated with AmB require close monitoring with increased hospital stays, which increase the cost of therapy. To combat this, various lipid formulations have been introduced. The rationale of using lipid formulations is that they enable targeted delivery of the drug with minimum adverse effects. The lipid formulations which are used in VL are, liposomal amphotericin B (AmBisome; Gilead Sciences; L-AmB), amphotericin B lipid complex (ABLC; Abelcet, Enzon pharmaceuticals, USA) and amphotericin B colloidal dispersion (ABCD; Amphotec, InterMune Corp.). L-AmB is the only approved drug by the US Food and Drug Administration (FDA). There is geographical variation in the total dose requirements of lipid formulations for the treatment of VL [Sundar and Chakravarty, 2015b]. In India, a total dose of 10 mg/kg results in a CR of >95% [Sundar et al. 2010]. A total dose of 18–21 mg/kg, administered in various regimens, has been recommended in the Mediterranean and South America [Davidson et al. 1996; Freire et al. 1997; Syriopoulou et al. 2003; Gradoni et al. 2004; Bern et al. 2006; Sundar and Chakravarty, 2015b]. From East Africa, although limited data are available, studies show that higher doses are required for treatment of VL. In a recent study from Sudan, L-AmB in a total dose of 30 mg/kg, over 10 days showed initial CRs of 92% with 1% treatment failures, 5% deaths and 7% relapses [Salih et al. 2014].
The major limiting factor in implementation of L-AmB is its high cost. However, a preferential pricing agreement with the World Health Organization (WHO) (agreement between Gilead and WHO of 14 March 2007) reduced the price of L-AmB for endemic regions of developing countries to $18 per 50 mg vial [Moon et al. 2011]. Encouraged by this, a study was conducted in India in which a single dose of 10 mg/kg of L-AmB was compared to the conventional AmB administered in 15 infusions of 1 mg/kg, given every other day during 29 days of treatment. CRs at 6 months were similar in the two groups: 95.7% (95% CI: 93.4–97.9) in the liposomal therapy group and 96.3% (95% CI: 92.6–99.9) in the conventional therapy group [Sundar et al. 2010]. Reduced cost and hospital stay made a single infusion of the liposomal preparation an excellent option for this region. In Bangladesh, this regimen was further tested at primary health centers, which showed a 97% CR at 6 month [Mondal et al. 2014]. A major hindrance is the requirement of a cold chain which demands infrastructure for it to be used at sub-district level [Maintz et al. 2014]. In a randomized controlled trial in East Africa, L-AmB was terminated prematurely because of low efficacy of all regimens [Khalil et al. 2014].
L-AmB at a dose of 4 mg/kg for 10 doses (days 1–5, 10, 17, 24, 31 and 38) up to a total dose of 40 mg/kg is recommended for treatment of HIV–VL coinfection [WHO, 2010]. The majority of the studies are from the Mediterranean region [Davidson et al. 1994; Russo et al. 1996; Montana et al. 2004]. A few studies of the effect of L-AmB on HIV–VL coinfected patients are shown in Table 2. In a recent retrospective study in Bihar, India, 102 patients with HIV–VL coinfection were treated by a combination of 30 mg/kg body weight L-AmB divided in 6 equal dose infusions given on alternate days, concurrently with 14 days of oral miltefosine. Cumulative incidence of all-cause mortality and VL relapse at 6, 12, and 18 months was 11.7%, 14.5%, 16.6% and 2.5%, 6.0%, 13.9%, respectively [Mahajan et al. 2015]. Like other opportunistic infections, secondary prophylaxis works well in HIV–VL coinfected patients. Secondary prophylaxis with ABLC (3–5 mg/kg per dose once) given every 3 weeks for 12 months has shown a relapse rate of 22% in comparison with 50% in patients without secondary prophylaxis at 1 year [Lopez-Velez et al. 2004; WHO, 2010]. L-AmB was again tested as a secondary prophylaxis at 5 mg/kg given every third week; the relapse free probability at 6 and 12 month was 89.7% and 79.1%, respectively and 55.9% both at 24 and 36 months [Molina et al. 2007].
L-AmB trials in HIV–VL coinfection.
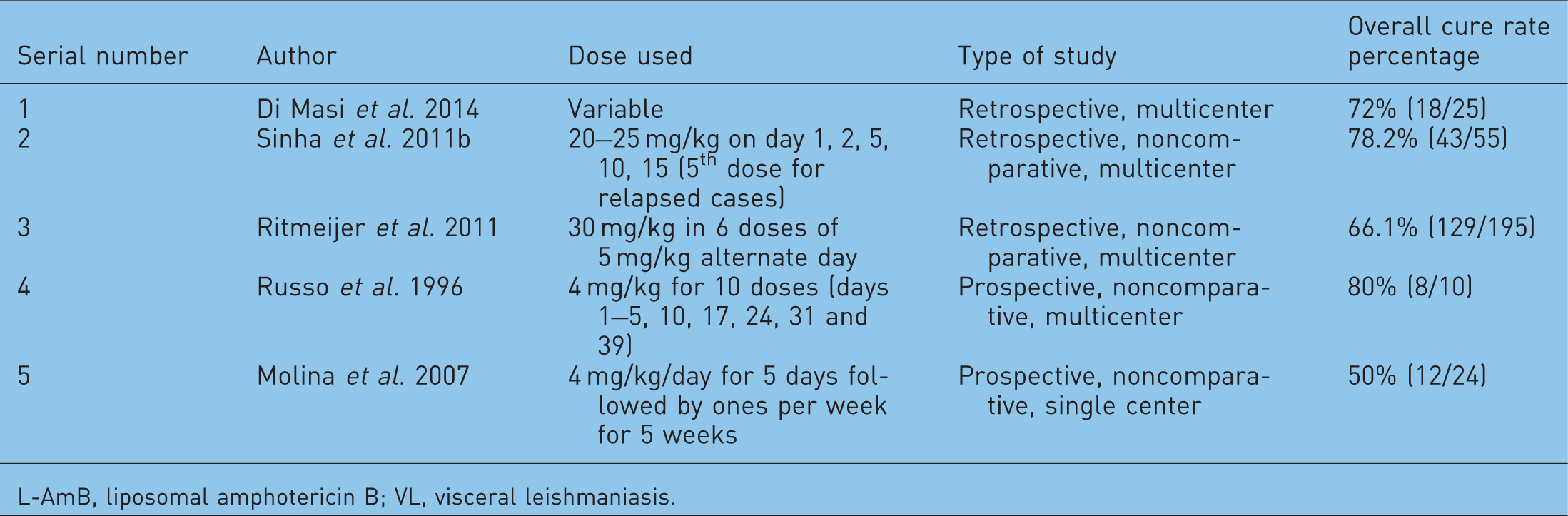
L-AmB, liposomal amphotericin B; VL, visceral leishmaniasis.
Miltefosine
Miltefosine was registered in India in 2002 as the first oral antileishmanial drug following a phase III trial which showed that 50–100 mg/ day of miltefosine for 28 days resulted in a long-term cure of 94% [Sundar et al. 2002]. The results were augmented by a large phase IV study which further showed CR of 95% [Bhattacharya et al. 2007]. Its efficacy, ease of use and applicability in the control program made this drug the backbone of the elimination program in India, Nepal and Bangladesh [Sundar et al. 2008a]. However, relapse rate doubled and efficacy dwindled after a decade of use of the drug in the Indian subcontinent [Sundar et al. 2012; Burza et al. 2013]. In Nepal, relapse rate of 10.8% at 6 and of 20.0% at 12 months was observed [Rijal et al. 2013], and in Bangladesh the CR was observed only in 85% patients [Rahman et al. 2011]. In Ethiopia, the CR among non-HIV-infected patients was 75.6% at 6 months [Ritmeijer et al. 2006]. In India, for PKDL, miltefosine was found to be effective in doses of 50 mg three times daily for 60 days or twice daily for 90 days [Ramesh et al. 2011]. A recent open-label, randomized multicenter trial of miltefosine, 100 mg/day for 12 weeks for patients with PKDL in India showed good CRs with very few adverse effects [Sundar et al. 2013]. It is recommended for the treatment of PKDL in the Indian subcontinent at the dose of 50–100 mg for 12 weeks [WHO, 2010]. In a recent study approximately 4% (3/73) patients of PKDL relapsed by the end of 12 months follow up, while a total of 15% (11/73) relapsed by the end of 18 months when treated with miltefosine [Ramesh et al. 2015].
The side effect profile of miltefosine includes gastrointestinal manifestations, hepatotoxicity and nephrotoxicity which requires monitoring thereby increasing the cost of therapy. Women of child-bearing age have to use contraception for the duration of treatment and for an additional 3 months as it is teratogenic. Its long half life (∼1 week) also makes it susceptible to the development of resistance in parasites.
Paromomycin (aminosidine)
A phase II study in India showed 93% CR at a dose of 16 mg/kg/day intramuscularly for 21 days [Jha et al. 1998]. Further a phase III trial showed a CR of 95% at a dose of 15 mg/kg paromomycin (PM) sulfate (11 mg base) for 21 days [Sundar et al. 2007]. Further trials confirmed its efficacy [Sinha et al. 2011a; Sundar et al. 2013]. In August 2006, PM was approved by the Indian government for the treatment of patients with VL. PM was compared to sodium stibogluconate (SSG) in Sudan, Ethiopia and Kenya in a phase III trail where PM was discontinued on account of its lower efficacy than SSG [Hailu et al. 2010]. Similarly, the efficacy of PM was found significantly lower than SSG (84.3 versus 94.1%) in a multicenter randomized-controlled trial (RCT) conducted in East Africa where efficacy and safety of PM (20 mg/kg/day for 21 days) and PM plus SSG in combination (PM, 15 mg/ kg/day and SSG, 20 mg/kg/day for 17 days) with SSG (20 mg/kg/day for 30 days) was compared in treating VL [Musa et al. 2012]. In a dose-finding phase II study in Sudan the efficacy of paromomycin was only 80% (95% CI: 56.3–94.3%) and 81% (95% CI: 58.1–94.6%) when used for a longer treatment duration (15 mg/ kg/day for 28 days) and at the higher dose (20 mg/kg/day for 21 days) respectively [Musa et al. 2010]. In a recent phase III, open-label, multicenter, single-arm trial that assessed the efficacy and safety of PM administered at 11 mg/kg (paromomycin base) intramuscularly once daily for 21 consecutive days to children and adults with VL in a rural outpatient setting in Bangladesh, the final CR at 6 months after end of treatment was 94.2% [Jamil et al. 2015]. There is no experience with this drug for VL in Mediterranean, Latin America where L. infantum is found.
Commonest side effects include pain at the injection site (55%), reversible ototoxicity (2%), and reversibly raised hepatic transaminases (6%). Despite being affordable (∼US $10 per case), its parenteral administration make its use in a control program of a developing country unfeasible at present. Also, monotherapy with PM might lead to development of resistance commonly seen with aminoglycosides [Sundar and Rai, 2005; Sundar et al. 2013].
Pentamidine
Pentamidine was used in the early 1980s for the treatment of refractory VL in India. However, it was discontinued due to associated serious toxicities such as insulin-dependent diabetes mellitus and declining efficacy [Jha et al. 1991]. The main adverse reactions related to pentamidine are pain, indurations and sterile abscess at the injection site, as well as nausea, vomiting, dizziness, myalgia, headache, hypotension, syncope, transient hyperglycemia and hypoglycemia [WHO, 2010; Sundar et al. 2013]. Recently it has been used in HIV–VL coinfection in Ethiopia and found to have a probability of relapse-free survival at six months and at 12 months of 79% and 71% respectively [Diro et al. 2015].
Combination or multidrug therapy
The rationale behind multidrug therapies are use of compounds with synergistic or additive activity acting at different sites, shorter duration of therapy and lower dose requirement, thereby lowering cost, chances of toxic side effects and emergence of drug resistance [Sundar and Chakravarty, 2013]. The emergence of resistance to anti-leishmanial drugs suggests that there is an urgent need to review the currently used monotherapy.
Combination therapy with L-AmB and miltefosine was studied in a multidrug therapy randomized, noncomparative, group-sequential, triangular design study where 181 subjects were assigned to treatment with 5 mg/kg of L-AmB alone, 5 mg/kg of L-AmB followed by miltefosine for 10 days, or 14 days of 3.75 mg/kg of L-AmB followed by miltefosine for 14 days. When it became apparent that all regimens were effective, 45 additional, nonrandomized patients were assigned to receive 5 mg/kg of L-AmB followed by miltefosine for 7 days. Final CRs was similar in all groups (>95%) [Sundar et al. 2008b].
In a subsequent large phase III study in the Indian subcontinent, three drug combinations (single injection of 5 mg/kg L-AmB and 7-day 50 mg oral miltefosine or 10-day 11 mg/kg intramuscular PM; or 10 days each of miltefosine and PM) showed an excellent CR (>97%) in treatment of VL [Sundar et al. 2011a]. Another combination trial in India where single dose of L-AmB 5 mg/kg and miltefosine 2.5 mg/kg/day for 14 days, showed a CR of 91.9% by intention to treat (ITT) and 97.6% by per protocol analysis [Sundar et al. 2011b].
A large retrospective field evaluation by Medecins Sans Frontieres (MSF) in Southern Sudan where combination therapy with PM plus Sbv for 17 days was compared with 30 days of Sbv monotherapy showed an initial CR of patients to be 97% and 92.4% respectively [Melaku et al. 2007]. It was further strengthened by a recent, large multicenter trial which showed comparable efficacy of combination therapy for 17 days with SSG treatment making it the preferred regimen in this region [Musa et al. 2012].
In East Africa, an exploratory study with miltefosine alone and combinations of single dose of L-AmB (10 mg/kg) with SSG (20 mg/kg) for 10 days and L-AmB (10 mg/kg) with miltefosine for 10 days is underway [Omollo et al. 2011]. Combination therapy of PM and SSG was a well-tolerated regimen and just as effective (CR 91.4%) as antimonials on their own (CR 93.9%), thus being a good option for treatment in Eastern Africa [Musa et al. 2012].
Thus, these combination therapies are not only well tolerated and effective options but associated with shorter duration of hospitalization and an excellent option for treating VL.
Future prospects
The limited therapeutic options and emerging resistance to the available drugs highlight the importance of searching for new antileishmanial agents. Among the long list of drugs under investigation, fexinidazole and PA-824 have reached phase II trials and have shown encouraging results [Sundar et al. 2015].
Fexinidazole
The Drugs for Neglected Disease initiative (DNDi) has rediscovered fexinidazole, a nitroimidazole used to treat human African trypanosomiasis (HAT) [Torreele et al. 2010]. Fexinidazole causes DNA, lipid and protein damage by acting as a prodrug releasing cytotoxic metabolites by enzyme-mediated reduction by nitro-reductases [Raether and Hanel, 2003]. Fexinidazole has shown potent activity against L. donovani in vitro and in vivo in a VL mouse model, assessed in studies in healthy volunteers and shown to be well tolerated when given as a single dose or as repeated dosing over 14 days. Various phase I studies of fexinidazole have been completed [Hovsepian, 2010, 2011, 2012. Currently, a phase II proof-of-concept trial to determine efficacy of fexinidazole at doses of 1800 mg/day (3 tablets) for 4 days followed by 1200 mg/day (2 tablets) for 6 days in VL patients in Sudan is underway [Musa and Khalil, 2015].
PA-824
PA-824, is a 4-nitroimidazo-oxazine. It has bactericidal properties against both replicating and nonreplicating Mycobacterium tuberculosis and is in a phase II trial for tuberculosis [Barry et al. 2004; Nuermberger et al. 2008; Ginsberg et al. 2009].
R-enantiomer of PA-284, has minimal activity against M. tuberculosis but has around a five-fold greater inhibitory effect on L. donovani growth compared with the S-enantiomer in in vitro studies [Patterson et al. 2013].
(S)-PA-824 is believed to function as a prodrug which requires bioreductive activation catalyzed by an unusual deazaflavin (F420)-dependent nitroreductase in M. Tuberculosis [Stover et al. 2000; Manjunatha et al. 2006; Singh et al. 2008]. This nitroreductase did not play a role in the activation of PA-824 in L. donovani as overexpression of nitroreductase in promastigotes fail to significantly change the sensitivity to either (S)-PA-824 or (R)-PA-824. In vitro drug combination studies suggest that fexinidazole and (R)-PA-824 are additive, whereas (S)-PA-824 and (R)-PA-824 showed mild antagonistic action [Nuermberger et al. 2008]. Although the in vitro and in vivo pharmacological profiles of the two enantiomers are similar, (R)-PA-824 is more efficacious in the murine model of VL, with >99% suppression of parasite burden when administered orally at 100 mg /kg of body weight, twice daily for 5 days [Patterson et al. 2013].
A phase I study was done in 58 healthy male volunteers using single oral doses (50, 250, 500, 750, 1000, 1250 or 1500 mg) or multiple doses of 200, 600 and 1000 mg of (S)-PA-824 each day for 7 days [Winter et al. 2013]. (S)-PA-824 was well tolerated at all doses in the phase I study, with no serious adverse events. Headache was the most common adverse event, followed by elevated serum creatinine levels, stomach discomfort (nausea, vomiting, flatulence or diarrhea) and back pain. Currently, (S)-PA-824 has completed a phase II trial in which the combination of (S)-PA-824-moxifloxacin-pyrazinamide was found suitable for treating drug-sensitive and multidrug-resistant tuberculosis [Diacon et al. 2012].
New AmB formulations
AmB is one of the most potent antileishmanial agents; however, its major drawbacks are need for prolonged infusions, toxicity and cost. Recently, novel drug delivery systems including liposomes, niosomes, microspheres, nanoparticles, and carbon nanotubes (CNTs) have been used to provide targeted delivery of the drug to macrophages [Pham et al. 2013]. The lipid formulations have excellent efficacy and are less toxic but cost is a limiting factor. Thus, efforts are ongoing to make cheaper and less-toxic formulations of this drug and to make it orally bioavailable.
Nano-AmB
The rationale behind testing nanoparticles of AmB deoxycholate in leishmaniasis is that nanoparticles are recognized as foreign bodies and phagocytosed by the macrophages, which also harbor the parasite, leading to target specific delivery of the drug. Nanoparticle delivery has shown promising results in in vitro and in vivo studies [Muller et al. 2001; Manandhar et al. 2008; Asthana et al. 2013].
Carbon nanotubes (CNTs)
Functionalized CNTs (f-CNTs) are prepared by carboxylation and amidation with AmB. This covalent modification by the organic functionalization of end groups and side walls of f-CNTs increases the solubility of f-CNTs in a range of solvents, including water [Georgakilas et al. 2002]. It has shown in in vitro and in vivo experiments in mice to be more effective and safer than AmB. The leishmanicidal efficacy of oral f-CNT-AmB was tested in hamsters which showed 99% inhibition of parasite growth at 15 mg/kg body weight for 5 days [Prajapati et al. 2012].
Others: An oral lipid-based formulation of AmB (iCo-009) composed of monoglycerides, diglycerides and distearoylphosphatidyle-thanolamine PEG 2000 significantly reduced liver parasite load in a murine model of VL at 10 and 20 mg/kg twice daily for 5 days [Wasan et al. 2009a, 2009b, 2010; Sivak et al. 2011; Ibrahim et al. 2013]. There are other new preparations which are in experimental phase [Nicoletti et al. 2009; Gupta et al. 2014; Les Ka, 2014].
Other investigational drugs
There are many natural and synthetic agents which have shown antileishmanial activity in in vitro and in vivo studies as described elsewhere [Sundar et al. 2015]. Their safety profile and efficacy need further studies.
Vaccine development
To date no vaccine is recommended for treatment of VL. However, deciphering the genomic sequence of Leishmania has helped in understanding the pathogenesis and host immune response caused by different Leishmania species. Several recombinant antigens that have protective properties against Leishmania infection have been studied in experimental models [Connell et al. 1993; Gurunathan et al. 1997; Stober et al. 2005; Rafati et al. 2006; Carrillo et al. 2008; Noazin et al. 2009]. Despite most of these recombinant antigens showing immunogenicity and protective efficacy in animal models, only a few have progressed to clinical trials [Kumar et al. 2010; Chakravarty et al. 2011; Singh et al. 2012].
Conclusion
The risk of increasing drug resistance and paucity of new antileishmanial drugs highlights the need to search for molecules with antileishmanial properties. In South Asia, single dose of L-AmB and short-course combination therapy is recommended in the treatment of VL [Carrillo et al. 2008]. Studies are underway to assess the feasibility, safety, and efficacy of these regimens at primary healthcare services and district hospitals in the endemic regions of Bihar, India by the Drugs for Neglected Diseases initiative (DNDi) and WHO Special Programme for Research and Training in Tropical Diseases.
In Africa, combination therapy of Sbv and PM is recommended as L-AmB, miltefosine and PM are less effective against L. donovani of this region. Whether combination therapy with single and multiple dose of L-AmB is effective for this region is still to be explored. In the Mediterranean region, L-AmB is the preferred drug, with a paucity of studies done with other drugs. For VL in the New World, L-AmB, SbV or conventional AmB can be used.
For HIV–VL coinfection, L-AmB is the recommended drug based on studies done in the Mediterranean region. But in Ethiopia which has a huge burden of HIV–VL coinfection, L-AmB has limited efficacy and therapeutic options need to be explored.
Recently not-for-profit organizations such as the DNDi have searched for newer drug targets for various neglected tropical diseases. Such entities could play a vital role in identifying the novel agents and hasten the process of drug development.
Footnotes
Funding
The work was funded by National Institute of Allergy and Infectious Disease, USA (Division of Microbiology and Infectious Diseases - DMID funding mechanism: Tropical Medicine Research Center grant number P50AI074321).
Conflict of interest statement
The authors declare no conflicts of interest in preparing this article.
