Abstract
We report a case of a 65-year-old male seen in a North Texas dermatology clinic with three erythematous nodules possessing central ulceration and scaling on the left lateral shoulder, present for months. Head, ears, lips, oral mucosa, and other body surfaces did not reveal similar lesions, and review of systems was negative. Shave biopsy was performed and histopathological findings demonstrated granulomatous inflammation in the dermis and parasitized histiocytes containing peripherally located amastigotes. Leishmaniasis was diagnosed and patient was educated on the disease while communication with the Centers for Disease Control and Prevention was initiated. The patient declined systemic medications from infectious disease specialists and, 3 weeks later, returned for follow-up treatment with cryotherapy.
Introduction
Leishmaniasis is an infectious disease caused by different species of parasitic intracellular protozoan. The protozoans are transmitted by two genera of sand fly vectors, Phlebotomus for Old World, and Lutzomyia for New World. The protozoans can be found in a variety of mammalian reservoirs.1,2 Leishmaniasis can be classified geographically in areas where they predominantly occur. The two regions are Old World (eastern hemisphere) and New World (western hemisphere).1,2 The three major clinical types are cutaneous leishmaniasis (CL), mucosal leishmaniasis (ML), and visceral leishmaniasis (VL).1,2 A recent study by McIlwee et al. 2 identified human CL in the United States as being endemic, with a significant number of cases occurring in Texas. There is not one generalized treatment regimen for CL as therapy should be individualized based on the patient, presentation, and parasite.3,4 We present a North Texas man who presented with CL that was treated with cryotherapy. CL and cryotherapy are discussed and reviewed.
Case report
We evaluated a 65-year-old male who presented to a North Texas dermatology clinic with chief complaint of upper body skin lesions. Lesions have been present on the left shoulder for months and have never been treated. The patient described lesions as non-healing, red, and mild. Past medical history included arthritis, hearing loss, and hernia repair, but reports no history of skin cancers. Dermatologic history included dry skin, seasonal allergies, and previous poison ivy exposure. Family history was negative for melanomas. The patient did not report any recent travel outside of Texas. The patient took one pantoprazole delayed release 50 mg tablet by mouth daily. Review of systems was negative, except for new skin lesions. Physical examination showed a well-appearing man with skin type Fitzpatrick type 2. Three erythematous nodules with central ulceration, crust, and scaling were distributed on the left lateral shoulder (Figure 1). The head, ears, lips, oral mucosa, and other body surfaces were spared of similar lesions. Differential diagnosis included basal cell carcinoma, squamous cell carcinoma, and picker’s nodule. Shave biopsy was performed for all three lesions. Follow-up appointment 5 days later revealed histopathological findings demonstrating mononuclear infiltration and granulomatous inflammation in the dermis (Figure 2). The dermal macrophages contained 2–4 µm peripherally located oval-shaped organisms demonstrating a marquee sign. The organisms also contained bar-shaped kinetoplast. This was consistent with a diagnosis of leishmaniasis. Leishmaniasis species determination was not completed. The patient was educated on the nature of the protozoal infection and potential pentavalent antimony treatment from the Centers for Disease Control and Prevention (CDC). The patient declined oral medication from infectious disease specialists. At a 3-week follow-up appointment, the patient received a single session of cryotherapy to all three lesions with subsequent resolution of symptoms. One year after treatment, the patient’s lesions have remained resolved.
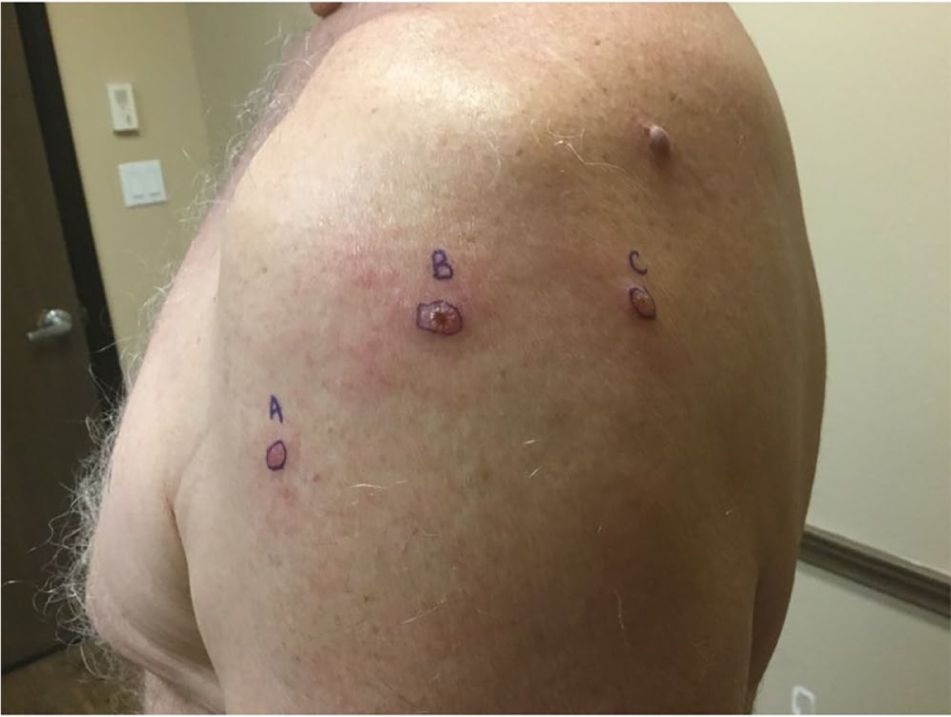
Three erythematous nodules with central ulceration, crust, and scaling of cutaneous leishmaniasis distributed on the left lateral shoulder.

Leishmaniasis histopathological findings demonstrating dermal granulomatous inflammation, mononuclear infiltrates, and parasitized macrophages with peripheralization of amastigotes: (a) Hematoxylin and eosin stain, × 200. (b) Hematoxylin and eosin stain, × 1000.
Discussion
Leishmaniasis is a parasitic infection designated by the World Health Organization as a Neglected Tropical Disease.4,5 Epidemiology data are uncertain with CL annual incidence estimates ranging from 0.7 to 1.2 million cases worldwide. 5 Computer-generated models have calculated potential autochthonous leishmaniasis exposure of 27 million in 2050 for North America.2,6
Pathogenesis of Leishmania infection begins with inoculation from the bite of the female sand fly as seen in the CDC life cycle image presented in Figure 3.1,5 Flagellated promastigotes are injected into the skin and phagocytized. Subsequently, promastigotes transform into aflagellated amastigotes, multiply, and continue to infect other cells.1,5 Upon future sand fly blood meal, amastigotes are consumed, ingested, and transform back into promastigotes in the gut of the sand fly.1,5 Promastigotes multiply and migrate to the sand fly proboscis, completing the cycle.1,5 Human infection is caused by at least 20 species of Leishmania, including L. aethiopica, L. amazonensis/L. garnhami, L. braziliensis, L. colombiensis, L. donovani/L. archibaldi, L. guyanensis, L. infantum/L. chagasi, L. lainsoni, L. lindenbergi, L. major, L. martiniquensis, L. mexicana/L. pifanoi, L. naiffi, L. panamensis, L. peruviana, L. shawi, L. siamensis, L. tropica/L. killicki, L. utingensis, and L. venezuelensis.1,5,7 McIlwee et al.’s2 cross-sectional observational study highlighted endemicity of CL in 41 autochthonous Texas cases and speciation identified L. mexicana 100% of the time on 22 cases tested.

Leishmaniasis life cycle (Courtesy of CDC. Available at https://www.cdc.gov/parasites/leishmaniasis/biology.html).
Clinical presentation of CL is commonly described as a self-limited course with solitary, well-circumscribed papules often distributed on exposed areas.1,4,5,8 Lesions can enlarge to form nodules or plaques with scaling and ulceration.1,4,5,8 The lesion may spontaneously heal over a period of months.1,4,5,8 Signs of healing include decrease in lesion size, re-epithelialization, and no new lesions. 3 The patient may have associated regional lymphadenopathy.1,4,5,8 L. mexicana classically affects the ear as the chiclero ulcer which is notable in chicle gum tree harvesters. 8 Upon resolution, atrophic scarring and pigmentation changes may leave the patient with significant cosmetic concerns.1,4,5,8 Complications include scarring, superinfection, persistent wounds, and dissemination in immunocompromised individuals. 3 CL can progress to ML and needs to be monitored, with the exception of the L. mexicana which rarely evolves.1,2,8
Histopathological findings of CL depend on timing, with early stage acute changes showing dermal infiltrates consisting of lymphocytes, plasma cells, neutrophils, and parasitized histiocytes that contain peripherally located basophilic oval-shaped amastigotes (Leishman-Donovan bodies) with rod-shaped kinetoplasts.1,2,5,8 Later-stage chronic findings reveal tuberculoid granulomatous changes with decreased Leishman-Donovan bodies.1,2,5,8
Various diagnostic modalities can be utilized and include smears, cultures, histopathology, polymerase chain reaction, and Montenegro skin test.1,3,5,8 The CDC is available to guide diagnosis and treatment. 5 Guidelines from the Infectious Diseases Society of America (IDSA) and the American Society of Tropical Medicine and Hygiene (ASTMH) strongly recommend full-thickness skin biopsy from lesions cleansed of cellular debris and eschar/exudates, using multiple diagnostic methods while simultaneously testing other differential diagnosis. 3 Strong recommendations are also made for parasite isolation with reference laboratories (CDC, Walter Reed Army Institute of Research (WRAIR)) and molecular amplification and DNA-based assays, while skin test or serologic testing should not be done. 3 A weak recommendation for speciation determination in CL is made as it may guide management. 3 Patients with travel history and compatible skin lesion have a weak recommendation for empiric treatment after considering risk and benefits. 3
The IDSA/ASTMH guideline states there is no universally applied therapy for CL and strongly recommends individualized treatment regimens be guided by a number of factors like agent, dose, and duration. 3 Clinically simple spontaneously healing lesions can be observed periodically without speciation or treatment, provided the patient is agreeable, immunocompetent, and at low risk of ML progression. 3 Periodic monitoring should include nasal and oropharyngeal examinations for 1 year, and if increased ML risk, 2 years. 3 Evaluation for ML, with fiberoptic examination if applicable, should be performed in any patient with current or history of CL that present with nasal or oropharyngeal signs or symptoms. 3 Systemic treatment is recommended for those with complex, diffuse, or disseminated CL, increased ML risk, specifically Leishmaniasis recidivans, impractical local therapy, or if rapid healing preferred. 3 Parenteral therapies available are conventional amphotericin B deoxycholate, lipid-based amphotericin B, pentavalent antimonials, and pentamidine. 3 Oral options include miltefosine and antifungals (e.g. ketoconazole and fluconazole). 3 Local therapies (e.g. heat, cryotherapy, topical paromomycin, intralesional pentavalent antimonial, photodynamic, and laser) are preferred for clinically simple Old World CL, and useful for New World CL without increased ML risk, but should be debrided of eschars and have secondary infections managed. 3 Treatment factors to consider include ML risk, susceptibilities, adverse events, age, pregnancy, obesity, hepatic, pancreatic, renal, and cardiac comorbidity, administration routes, response time, daily activities, psychosocial, logistics, availability, cost, insurance. 3 IDSA and ASTMH provide an organized, quick reference, list of medications, dosing, and additional information in their treatment approach for leishmaniasis in Table 3 of their guidelines. 3 Treatment response should be assessed clinically for 6–12 months by physical appearance, with additional treatment for new, worsening, or incompletely healed lesion 3 months after therapy. 3 An alternate treatment approach should be made for inadequate responses. 3 Ulcerations generally heal and reepithelialize 3 months after treatment. 3 Paradoxical inflammatory response may occur 2–3 weeks after treatment, appearing similar to therapeutic failure. 3 If lesions are healing, no repeat parasitologic testing need be done. 3 Management should include wound care, documentation of lesion evolution, and patient education of treatment failure/relapse and ML. 3
Our patient was managed with local cryotherapy, an efficacious and safe treatment alternative for CL. 9 A recent meta-analysis by Lopez-Carvajal et al. 9 looked at eight total studies, where three studies reported on per-lesion efficacies for cryotherapy and pentavalent antimonials, 67.3% and 67.7%, respectively, finding no statistical difference. The same meta-analysis looked at five studies for per-patient efficacies of cryotherapy in 271 patients and pentavalent antimonial in 199 patients, finding no statistical difference with respective intent-to-treat efficacies of 54.2% and 68.3% (relative risk comparison of efficacies 0.73 (0.42–1.29)), and per–protocol efficacies of 63.6% and 74.7% (relative risk comparison of efficacies 0.78 (0.45–1.34)). 9 Safety analysis was limited, but notable cryotherapy adverse effects included hypopigmentation, hyperpigmentation, erythema and edema, pain, and ardor. 9 Cost per patient for cryotherapy was approximately $4, while alternative options ranged from $30 to $1500. 9 The authors concluded favorable use of cryotherapy as it has similar efficacy to pentavalent antimonials with the advantage of shorter treatment duration and better adherence, particularly in situations where medications are contraindicated, areas are low risk for ML, or patients have a history of therapeutic failure. 9
Conclusion
This case report describes New World Cutaneous Leishmaniasis in a North Texas male with a review of epidemiology, pathogenesis, clinical presentation, histopathology, diagnosis, and management, and a particular focus on cryotherapy treatment. A detailed evaluation and thorough workup are important as therapeutic regimens are individualized using multiple elements that consider Leishmania species, patient-related factors, presentation, and severity. Cryotherapy may be a practical, cost-effective treatment option with similar efficacy to pentavalent antimonials.
Footnotes
Acknowledgements
The authors acknowledge Dr. Stephen E. Weis, Medical City Weatherford Dermatology Program, Weatherford, Texas, USA.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics approval
Our institution does not require ethical approval for reporting individual cases or case series.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Informed consent
Written informed consent was obtained from the patient for their anonymized information to be published in this article.
Availability of data
Data sharing is not applicable to this article as no data sets were generated or analyzed during the current study.
