Abstract
Chronic pulmonary aspergillosis (CPA) is a challenging respiratory infection caused by the environmental fungus Aspergillus. CPA has a poor prognosis, with reported 1-year mortality rates ranging from 7% to 32% and 5-year mortality rates ranging from 38% to 52%. A comprehensive understanding of the pathogen, pathophysiology, risk factors, diagnosis, surgery, hemoptysis treatment, pharmacological therapy, and prognosis is essential to manage CPA effectively. In particular, Aspergillus drug resistance and cryptic species pose significant challenges. CPA lacks tissue invasion and has specific features such as aspergilloma. The most critical risk factor for the development of CPA is pulmonary cavitation. Diagnostic approaches vary by CPA subtype, with computed tomography (CT) imaging and Aspergillus IgG antibodies being key. Treatment strategies include surgery, hemoptysis management, and antifungal therapy. Surgery is the curative option. However, reported postoperative mortality rates range from 0% to 5% and complications range from 11% to 63%. Simple aspergilloma generally has a low postoperative mortality rate, making surgery the first choice. Hemoptysis, observed in 50% of CPA patients, is a significant symptom and can be life-threatening. Bronchial artery embolization achieves hemostasis in 64% to 100% of cases, but 50% experience recurrent hemoptysis. The efficacy of antifungal therapy for CPA varies, with itraconazole reported to be 43–76%, voriconazole 32–80%, posaconazole 44–61%, isavuconazole 82.7%, echinocandins 42–77%, and liposomal amphotericin B 52–73%. Combinatorial treatments such as bronchoscopic triazole administration, inhalation, or direct injection of amphotericin B at the site of infection also show efficacy. A treatment duration of more than 6 months is recommended, with better efficacy reported for periods of more than 1 year. In anticipation of improvements in CPA management, ongoing advances in basic and clinical research are expected to contribute to the future of CPA management.
Introduction
Chronic pulmonary aspergillosis (CPA), a chronic lung infection caused by Aspergillus, is a significant health concern. 1 In 2007, it was estimated that there were approximately 372,000 cases of CPA developed worldwide due to pulmonary tuberculosis cases. 2 The recent CPA model using WHO’s global tuberculosis figures in 2020 estimated 1,837,272 cases of CPA arising annually from previous pulmonary tuberculosis cases. 3 Moreover, it has been alarming that approximately 340,000 patients with CPA die within the first year of disease onset. 3 Despite its status as a challenging infectious disease with low survival, CPA remains underappreciated compared to invasive pulmonary aspergillosis (IPA). 4 It suffers from a lack of awareness, resulting in delayed diagnosis and frequent misdiagnosis due to under-recognition and association with comorbidities. As a result, the true prevalence of CPA is likely underestimated. 5
The management of CPA is complicated by multiple comorbidities, complex clinical presentations, drug interactions, toxicities, and treatment resistance.6,7 A better understanding of CPA, including its etiology, clinical manifestations, appropriate treatment strategies, and various attempts to treat refractory cases, is needed to identify this devastating disease early and direct it to more appropriate treatment. 8 However, much remains to be learned about this patient population, the disease itself, and the optimal use of available therapeutic modalities.9,10
Historically, limited research on CPA has resulted in limited treatment options. In recent years, however, there has been a surge in public interest, resulting in an increased number of reports. 11 This evolving landscape suggests a growing recognition of the importance of studying and understanding CPA, potentially paving the way for expanded therapeutic options in the future. This review aims to provide a thorough exploration of the pathogen, pathophysiology, risk factors, diagnosis, treatment, and prognosis associated with CPA. As we delve into these aspects, we recognize the ongoing efforts of the mycological community to advance the care of CPA patients and emphasize the need for continued research to improve our understanding of this complex condition.
Aspergillosis-causing fungi
How Aspergillus causes infections
The genus Aspergillus, which causes aspergillosis, is classified as a filamentous fungus. 12 It reproduces by extending filamentous structures called hyphae. 13 When certain conditions are met (e.g. in the most virulent Aspergillus fumigatus, exposure to sufficient oxygen concentration and gas phase), Aspergillus forms specialized structures called conidiophores on exposed surfaces, giving rise to conidia [Figure 1(a) and (b)].13–15 Conidia are a type of asexually produced spore. The color of many large colonies is attributed to the pigmentation of these conidia [Figure 1(c)]. 13 The surface of conidia is highly hydrophobic, allowing it to float in the air for long periods without being surrounded by water molecules. 16 With a particle diameter of approximately 3 μm, 13 these conidia continue to float without settling, making them susceptible to inhalation. 17 Aspergillus primarily resides in soil and plant matter, with its conidia being dispersed into the air through various mechanisms, such as mechanical disturbances or environmental factors. Consequently, these airborne conidia can infiltrate our living environments. 18

Micromorphological characteristics and macroscopic colony morphology of Aspergillus fumigatus. (a) Microscopic view of A. fumigatus slide culture stained with lactophenol cotton blue. Bar = 50 μm. (b) A. fumigatus conidial heads have a columnar shape with uniseriate conidiogenous cells. Smooth-walled conidiophore (red arrow), subclavate vesicle (orange arrow, 20–30 μm wide), and (sub)spherical conidia (yellow arrow, 2.5–3.0 μm diameter) are highlighted. Bar = 20 μm. (c) Colonies exhibit a dark blue-green color characterized by a dense felt of conidiophores intermingled with aerial hyphae. 13 The image of this slide culture (A) is from Tashiro et al. 14
Pathogenic Aspergillus species
Aspergillus spp. are common fungi, and many are not pathogenic to humans. 12 However, certain species, including A. fumigatus, A. niger, A. terreus, A. flavus, A. nidulans, and A. versicolor, etc. are pathogenic to humans, with A. fumigatus being the most pathogenic and commonly implicated in the cause of aspergillosis.19–23 Some Aspergillus spp., referred to as related or cryptic species, are difficult to identify using conventional identification methods based on characteristics such as colony morphology and conidiophore observation.23,24 Cryptic species are less common and rarely associated with human infections; however, clinical concerns arise from the low susceptibility of these cryptic species to certain antifungals.25–33 Cryptic species that are pathogenic to humans and have shown low drug susceptibility include the following strains: A. fumigatus complex (A. lentulus, A. udagawae, A. viridinutans, A. felis, A. fischeri, and A. thermomutatus); A. niger complex (A. welwitschiae, A. tubingensis).34–43 Reports from Indonesia indicate that cryptic species were identified in 24% of patients with CPA, emphasizing the need for accurate identification. 44 Matrix-assisted laser desorption ionization coupled to time-of-flight mass spectrometry (MALDI-TOF/MS) is increasingly being used for identification, and some closely related species can now be identified.45,46 However, we have experienced a case in which A. udagawae was misidentified as A. thermomutatus because it was not registered in the MALDI-TOF/MS database, 47 and we believe that genetic identification is still necessary to determine the exact species. 48
Types of pulmonary aspergillosis
In the lungs of healthy individuals, conidia adhering to the airways are expelled by ciliary movement and cleared from the lungs.49,50 Conidia that reach below the bronchioles, where cilia are absent, are phagocytosed, killed, and cleared by alveolar macrophages and neutrophils.51–53 When these defense mechanisms are compromised, as in various immunocompromised patients or those with lung disease with structural destruction of the lungs, pulmonary aspergillosis can manifest. 51 If the immune response, primarily characterized by neutrophil depletion or dysfunction, fails to clear the conidia, IPA with tissue invasion may result. CPA develops when the lungs have structural damage that fails to physically eliminate Aspergillus, such as pulmonary tuberculosis, chronic obstructive pulmonary disease (COPD), or other lung diseases (Figure 2). CPA differs from IPA in that most cases of CPA are characterized by minimal or no tissue invasion. 54 Aspergillus-related lung diseases also include allergic diseases such as asthma, allergic bronchopulmonary aspergillosis (ABPA), and hypersensitivity pneumonitis.55–57 Reports indicate that 5% of patients with CPA have concomitant ABPA, highlighting the need for vigilance in diagnosing both conditions when managing patients with CPA.58,59 Thus, pulmonary aspergillosis is broadly classified into three types: acute (invasive), chronic (non-invasive), and allergic (Figure 2). These classifications represent a spectrum that allows for the existence of intermediate, transitional, and mixed forms.
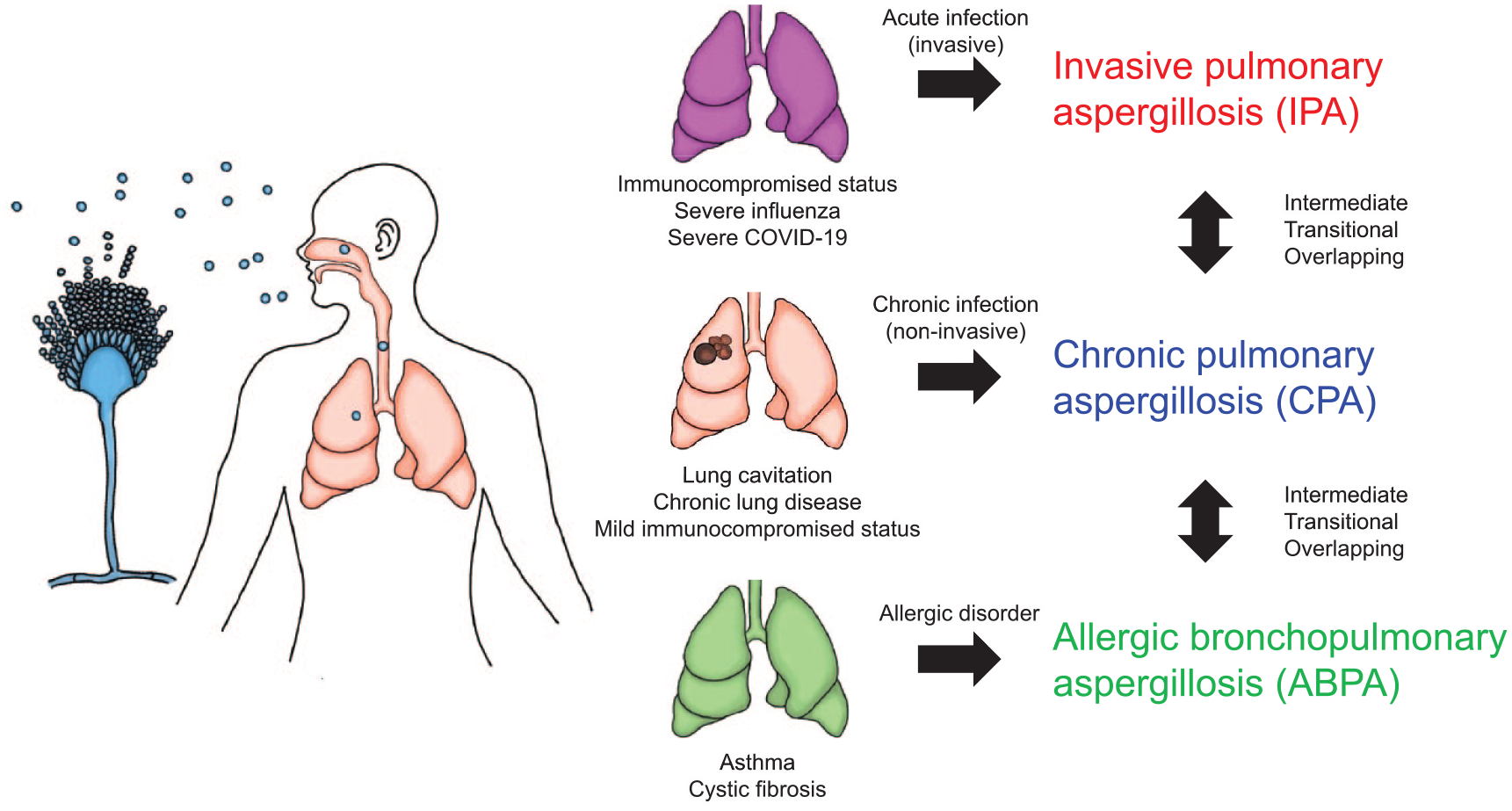
Illustration demonstrating the relationship between the three types of pulmonary aspergillosis and underlying comorbidities, as well as the continuum between each type. Pulmonary aspergillosis encompasses three distinct types: IPA, CPA, and ABPA. Each type is closely associated with the patient’s underlying comorbidities. Transitioning between types over time, overlapping manifestations of each type, and the presence of intermediate states resembling a blend of the different types represent the diverse patterns observed.
Subtypes of CPA
Classification of CPA
CPA encompasses a spectrum of complex pathologies, leading to the use of multiple subtypes to characterize different disease manifestations (Figure 3). In practical clinical settings, distinguishing between these subtypes can be challenging and overlap can occur, making it difficult to use these terms as a single disease entity. 54 Nevertheless, these terms are valuable in conceptualizing and discussing patients’ conditions. In addition, the use of these subtype names is essential when considering treatment strategies, including surgical interventions. The guidelines formulated by the European Society for Clinical Microbiology and Infectious Diseases (ESCMID), the European Confederation of Medical Mycology (ECMM), and the European Respiratory Society (ERS) define five subtypes of CPA. 54 This review also discusses additional subtype terms such as ‘chronic necrotizing pulmonary aspergillosis’, ‘complex aspergilloma’, and ‘chronic progressive pulmonary aspergillosis’ (Figure 4). In addition to these introduced subtypes, CPA has been reported to exhibit different clusters based on the distribution of serum IgE and peripheral blood eosinophil counts. 60

Illustration of Aspergillus infection progression in the human respiratory system. Inhaled conidia can lead to rapid invasive pulmonary aspergillosis in immunocompromised patients, while immunocompetent individuals may develop chronic pulmonary aspergillosis. The figure outlines specific manifestations, including SA, CCPA, CFPA, SAIA, and CNPA, as well as AN.

Characteristics of subtypes of chronic pulmonary aspergillosis are delineated by five parameters. Changes within each parameter are continuous and distinctions may not be clear. The eight subtypes identified often have overlapping features. CA overlaps with CCPA, while SAIA overlaps with part of CNPA. CPPA is an umbrella term that includes both CCPA and CNPA.
Simple aspergilloma (SA)
When Aspergillus spp. forms a fungus ball in the cavity of a host, it is referred to as an aspergilloma. 54 Aspergilloma is created as fungal masses grow along the cavity wall and detach. 61 SA is the term used for cases in immunocompetent patients with a single existing cavity where an aspergilloma forms, accompanied by minimal systemic symptoms and inflammation, and with radiographic stability for more than 3 months. 54 The cavity wall is typically thin (Figure 5). 14 Diagnosis is based on characteristic imaging findings of aspergilloma, along with serologic or microbiologic evidence suggesting the presence of Aspergillus spp. 54 SA is an important subtype of CPA, among other clinical manifestations.62,63 Despite the lack of symptoms, SA can be a cause of hemoptysis, making it a target for treatment. Observation is a management option for patients with a simple, uncomplicated aspergilloma with stable cavity size over 6 to 24 months.54,64 In the treatment of SA, surgical resection of the lesion is recommended primarily for patients presenting with hemoptysis, as outlined by published recommendations.54,64 However, it is essential to recognize regional variations in treatment approaches. While surgery may be a primary option in certain regions, antifungal drug therapy plays a crucial role in the management of SA, particularly in areas where surgery is technically challenging or not readily available. Therefore, both surgical resection and antifungal drug therapy are important components of the treatment strategy for SA, with their prioritization dependent on individual patient factors and regional considerations.

CT image showing an aspergilloma (*) with a thin-walled cavity and no evidence of infiltrative shadows (yellow arrow), suggestive of a SA occurring within an enlarged bronchus in a 43-year-old woman. The image of the case is from Tashiro et al. 14
Chronic cavitary pulmonary aspergillosis (CCPA)
CCPA refers to a condition in which Aspergillus spp. grow in lung cavities and progress slowly over 3 months or more. The number of cavities or the presence of aspergilloma is not a restrictive criterion, although approximately half of CCPA patients have aspergilloma.65,66 CCPA is a major subtype of CPA.54,63,67 The diagnosis of CCPA requires the convergence of three conditions over 3 months: the presence of respiratory symptoms (such as cough and dyspnea) or systemic symptoms (such as fatigue), progressive changes in the lung cavities (including the presence of aspergilloma or thickening of the cavity walls) or changes in the surrounding lung tissue (expansion of cavities or appearance of infiltrative shadows) for 3 months or more, and serological or microbiological evidence suggesting the presence of Aspergillus spp (Figure 6).14,54 Histopathologic analysis shows erosion of the cavity walls and granulation tissue, with granulomas in some cases. Surrounding tissues show chronic inflammation, occasionally characterized by organizing pneumonia, bronchiectasis, enlarged vessels, and/or fibrosis around the cavities. 68 In addition, concomitant organizing pneumonia around infected cavities is believed to be a significant contributor to respiratory failure.66,69 Treatment for CCPA is primarily antifungal drug therapy.

CT image showing CCPA with a thickened cavity wall (yellow arrow) and an aspergilloma (*) in a 69-year-old man after recovery from tuberculosis. The image of the case is from Tashiro et al. 14
Complex aspergilloma (CA)
CA refers to CCPA accompanied by aspergilloma. The ESCMID/ECMM/ERS guidelines include CA under CCPA. 54
Chronic fibrosing pulmonary aspergillosis (CFPA)
CFPA is a condition in which the fibrotic destruction of the lungs in CCPA patients extends to multiple lobes.54,67 It is a rare complication of CPA and may manifest several years after disease onset, particularly in patients not receiving antifungal therapy. 70 The fibrosis observed in CFPA is similar to the ‘destroyed lung’ syndrome seen after tuberculosis treatment. 70 Patients with CFPA may have significantly elevated Aspergillus IgG titers. 70 As a progressive form of CCPA, individuals with CFPA have typically been treated with antifungal agents, potentially leading to toxicity or resistance. 65
Subacute invasive aspergillosis (SAIA)
SAIA refers to a subacute course of CPA over 1 to 3 months (Figure 7).14,54 It is characterized by tissue invasion, and its pathophysiology is similar to that of IPA.54,71 The majority of patients have mild to moderate immunodeficiency and in some cases meet the criteria for the diagnosis of IPA.72–77 The prognosis of SAIA is poor compared to other subtypes of CPA. 78 Unlike other CPA subtypes, SAIA is often positive for serum Aspergillus galactomannan antigen, reflecting tissue invasion. 79 It encompasses part of chronic necrotizing pulmonary aspergillosis.54,71 The treatment for SAIA generally follows the principles of IPA and involves antifungal drug therapy.

SAIA in a 64-year-old man with underlying undiagnosed diabetes and pulmonary emphysema. (a) CT image showing the formation of a cavity within the consolidation of the left upper lobe, with the appearance of aspergilloma inside (yellow arrow). (b) Macroscopic examination of the dissected lung revealed black fungal masses within the cavity, with Aspergillus niger confirmed by culture. (c) Pathological images of the left upper lobe, with GMS staining on the left and H&E staining on the right, at 4× magnification. A. niger invasion resulted in necrosis of lung tissue, leading to the formation of cavities containing fungus balls. (d) Polarized light microscope image at 200× magnification, showing deposition of calcium oxalate crystals (oxalosis) in the patient’s tissues due to A. niger infection. The CT image of the case is from Tashiro et al. 14
Chronic necrotizing pulmonary aspergillosis (CNPA)
CNPA is characterized by Aspergillus invasion of lung tissue and the formation of cavities over a chronic course.71,80 While cases progressing over 1 to 3 months overlap with SAIA, leading to their classification under SAIA in the ESCMID/ECMM/ERS guidelines, 54 it is important to recognize the nuances and variations in presentation and progression within CNPA. Our experience includes cases demonstrating tissue invasion into the lung over several years, highlighting the heterogeneity of CNPA (Figure 8). Previous definitions of CNPA may differ from the current SAIA definition, indicating the need for a nuanced understanding of these conditions. 71 Therefore, some latitude should be given in defining CNPA, recognizing its potential overlap with other CPA subtypes and its spectrum of clinical manifestations.

CT images showing CNPA in a 56-year-old man, depicting the condition’s progression over 3 years. The patient sought medical attention due to worsening abnormal chest shadows identified during a routine health check-up. Despite multiple bronchoscopic examinations, a definitive diagnosis could not be made and the patient was kept under observation. Over a 3-year follow-up period, the appearance and enlargement of the cavity were observed, accompanied by a fungus ball-like shadow within the cavity and seroconversion of Aspergillus precipitin antibody. Video-assisted thoracoscopic surgery with resection of the right upper lobe was performed for both diagnostic confirmation and therapeutic intervention. Pathologic examination of excised lung tissue stained with EVG stain confirmed lung tissue destruction and hyphae invasion into alveolar and vascular walls. The images of the case are from Tashiro et al. 14
Chronic progressive pulmonary aspergillosis (CPPA)
In situations where the progression of previous imaging findings cannot be traced, it is difficult to clinically differentiate between CNPA with cavitation and CCPA based on a single imaging observation.62,81 Given the lack of therapeutic differences between CCPA and CNPA, the term CPPA has been proposed to include both.82,83
Aspergillus nodule (AN)
AN refers to a condition in which nodular lesions form in areas without preexisting cavities in the lungs (Figure 9).14,54 The average size of AN is approximately 20 mm (ranging from 5 to 50 mm), and in some cases, multiple nodules may be present.84,85 Among AN cases, 20% show spiculation, 39% show calcification, and 61% show cavitation. 86 Pathologically, Aspergillus can be identified within necrotic tissue with granulomatous inflammation involving multinucleated giant cells.84,85 Tissue invasion is not observed. Distinguishing AN from other nodular lesions, such as lung cancer, by imaging can be challenging.84,87,88 In patients with suspected lung cancer, AN is often missed.87,88 AN also accumulates on fluorodeoxyglucose (FDG) positron emission tomography (PET), creating the potential for false-positive results. 89 Most AN show stability or improvement after biopsy, regardless of antifungal treatment. 90 However, CCPA can occasionally develop, suggesting ongoing radiologic follow-up for unresected nodules. 90

CT images showing AN in a 60-year-old man with a history of pulmonary tuberculosis treatment 30 years ago. The lung nodule (yellow arrow), discovered during evaluation for hemoptysis, showed enlargement and consolidation over 2 years. Suspecting lung cancer, video-assisted thoracoscopic surgery with a right upper lobe biopsy was performed. Pathologic examination of the excised lesion revealed the presence of Aspergillus clumps within an enlarged bronchus and caseous necrotic nodules (tuberculous nodules). The image of the case is from Tashiro et al. 14
Risk factors for CPA
Underlying lung conditions in CPA patients
Many CPA patients have underlying lung disease associated with the destruction of existing lung structures. In particular, lung cavities are a significant risk factor for CPA. In a follow-up study of pulmonary tuberculosis patients, the incidence of CPA was significantly higher in cases with cavities (6.5% per year) compared to those without cavities (0.2% per year) (p < 0.001). 91 The presence of lung cavities is also considered a risk factor for CPA in patients with non-tuberculous mycobacterial infections.92–94 In addition, a strong association has been reported between the presence of lung cavities and levels of anti-Aspergillus IgG antibodies. 95 These facts emphasize the importance of lung cavities as a risk factor for CPA.
Underlying diseases in CPA patients
The most common underlying diseases in CPA patients include pulmonary tuberculosis, non-tuberculous mycobacterial infections, ABPA, COPD, bronchiectasis, pulmonary fibrosis with lung cysts, pneumoconiosis, pulmonary cystic disease, sarcoidosis, history of pneumothorax, and post-lung cancer surgery, among others.96–103 Pulmonary tuberculosis stands out as the most important underlying disease,63,97,104–106 while in countries with low tuberculosis prevalence, non-tuberculous mycobacterial infections and COPD become crucial.96,97,107,108 The prevalence of each underlying disease varies significantly between countries. For example, in Japan from 2006 to 2011, 273 CPA patients prospectively enrolled in two randomized controlled trials had the following underlying diseases: pulmonary tuberculosis 40.7%, COPD 23.1%, non-tuberculous mycobacterial infections 14.7%, idiopathic pulmonary fibrosis 8.4%, history of pulmonary surgery 7.7%, bronchiectasis 5.9%, history of pneumothorax 5.1%, pneumoconiosis 3.7%, and pulmonary cysts 1.8%. 97 By contrast, a cohort of 126 CPA patients in the United Kingdom from 2002 to 2009 had the following underlying conditions: classical tuberculosis 15.9%, non-tuberculous mycobacterial infection 14.3%, ABPA ± asthma 11.9%, COPD and/or emphysema ± bullae 9.5%, pneumothorax ± bullae 9.5%, lung cancer survivors 9.5%, community-acquired pneumonia 7.9%, sarcoidosis 7.1%, thoracic surgery 4.8%, rheumatoid arthritis without immunosuppression 3.2%, asthma without ABPA or severe asthma with fungal sensitization 2.4%, severe asthma with fungal sensitization 1.6%, and bullae without COPD and pneumothorax 0.8%. 96 The risk of CPA complications is highest in pulmonary tuberculosis. It is estimated that CPA develops in 5–20% of patients after pulmonary tuberculosis is cured.74,91 In patients with non-tuberculous mycobacterial infection, the reported rate is 7.2%, 109 and in individuals after lung resection surgery, the cumulative incidence rate over 10 years is 3.5%.110,111
Characteristics and immunological abnormalities in CPA patients
Studies comparing CPA patients with individuals with Aspergillus colonization show distinct characteristics in CPA patients, including a lower body mass index (BMI) (18.45 versus 21.09 kg/m2). 21 ‘Aspergillus colonization’ in this context was clinically defined as the lack of radiological or clinical findings suggestive of IPA or CPA or ABPA in patients with Aspergillus species isolates from respiratory specimens in the absence of hematologic malignancy, 21 while the definition of clinical criteria to evaluate fungal colonization of the respiratory airways differs between experts. 112 Various degrees of immunological abnormalities have been reported in CPA patients. Lymphocyte depletion is observed in 58% of CPA patients. 113 In some CPA cases, impaired peripheral blood interferon (IFN)-γ production in response to stimuli has been reported. 114 Macrophages from CPA patients show lower expression of Toll-like receptor 3 (TLR3) and TLR10 and higher expression of Triggering Receptor Expressed on Myeloid Cells 1 (TREM1) compared to healthy individuals. 115 Genetic analysis of CPA patients has revealed polymorphisms in TLR3, TLR4, TLR10, interleukin-1 (IL-1) pathway, IL-15, and other immune regulation disorders. 116 Abnormalities in the Janus kinase-signal transducer and activator of transcription 3 (JAK-STAT3) pathway associated with high IgE syndrome have been reported in patients with CPA. 117 Corticosteroid use has been identified as a risk factor for CPA in patients with non-tuberculous mycobacterial infections,92,94,109 suggesting that mild immunosuppression may increase the risk of developing CPA. These findings highlight the diverse immunological landscape in CPA patients and underscore the importance of understanding individual immune responses in the context of CPA.
Pathogen-related factors in CPA
Research on pathogenic factors of the Aspergillus primarily focuses on A. fumigatus, which is considered the predominant fungus in all forms of aspergillosis. 118 Although A. fumigatus typically inhabits soil, many of its characteristics for survival in this environment are repurposed to adapt to the human host environment. 119 The pathogenicity of A. fumigatus is considered to arise not from specific virulence factors but rather from its ability to adapt to the human host environment.49,120 Key factors implicated in the establishment of A. fumigatus infection can be categorized into three aspects: multiple compensatory reactions of the stress response, nutritional flexibility, and resistance to the immune system.49,121 The human host environment poses significant stress for many microorganisms. 122 However, A. fumigatus demonstrates remarkable adaptability to various stressors, including high temperatures within deep tissues of the human body,48,123,124 low pH conditions like those found in phagolysosomes,49,125 low oxygen levels in tissues,126–128 changes in osmotic pressure,129–131 reactive oxygen species (ROS),132,133 and microbiota.134,135 Furthermore, A. fumigatus produces various hydrolytic enzymes, facilitating the acquisition of essential nutrients such as carbon, nitrogen, and ions (iron, copper, zinc, calcium, magnesium, and manganese) required for survival within the human body.136–139 In addition, A. fumigatus produces galactosaminogalactan (GAG) and secondary metabolites that act as toxic immunosuppressors and evade the immune system by masking pathogen-associated molecular patterns.140–145 Although these factors are based on experimental results primarily focused on IPA, it is reasonable to assume similar factors contribute to the pathogenicity of A. fumigatus in CPA. However, specific pathogenic factors of A. fumigatus in CPA patients have not been elucidated. Whole-genome sequence (WGS) analysis of 17 strains of A. fumigatus isolated from CPA patients revealed distinct subclades based on the analysis of 42,345 consensus sites or single nucleotide polymorphisms. 146 However, the relationship between these subclades and clinical features remains unclear. Nonetheless, such analyses utilizing WGS or next-generation sequencing hold promise for revealing the characteristics of A. fumigatus in CPA patients in the future. 147
Diagnosis of CPA
Characteristics of CPA diagnosis
Of note, CPA diagnosis typically involves the use of multiple diagnostic tests rather than relying solely on the results of a single test. Consensus among various microbiological, serological, and molecular tests is often required for accurate diagnosis and effective patient management. This multifaceted approach to diagnosis ensures a comprehensive assessment of the patient’s condition and aids in determining the most appropriate treatment strategy. In addition, the diagnostic features of CPA vary depending on the subtype (Figure 4). 54 SA, CA, CCPA, and CFPA can be diagnosed based on imaging features (presence or formation of lung cavities, thickening of the cavity walls, or presence of aspergilloma) along with evidence suggestive of Aspergillus spp. (serological or microbiological testing).54,148,149 Guidelines emphasize the importance of combining chest CT with specific Aspergillus IgG antibody measurements in the diagnostic process.150,151 AN lacks distinct imaging features and its diagnosis often requires biopsy or surgical resection to directly demonstrate Aspergillus infection within the lesion. 86 The significance of serologic testing in the diagnosis of AN remains unclear, 86 but diagnosis based solely on serologic results is considered inadequate. Some SAIA can be evaluated using the diagnostic criteria for IPA.76,77
Diagnosis of CPA in resource-limited settings
Diagnostic capabilities may be limited in low- to middle-income countries where CT scans and Aspergillus IgG testing may not be available. These regions often have a high prevalence of pulmonary tuberculosis, a primary underlying cause of CPA, which may lead to the underdiagnosis of many cases of CPA. 152 Consequently, an international panel of experts has proposed a case definition for CPA in resource-limited settings that focuses primarily on CCPA. 152 Specifically, CPA is defined as a disease that persists for more than 3 months and meets all of the following criteria: (1) weight loss, persistent cough, and/or hemoptysis; (2) progressive cavitary infiltration on chest imaging and/or fungal balls and/or fibrosis, infiltration or pleural thickening; and (3) positive Aspergillus IgG measurement or other evidence of Aspergillus infection. In such settings, confirmation of Aspergillus IgG by point-of-care testing is considered valuable.153,154
Aspergillus antibody testing
Detection of Aspergillus IgG is a crucial aspect of the diagnosis of CPA.54,64,155–158 The performance of IgG antibody testing in the diagnosis of CPA is reported to have a sensitivity of 90% and a specificity of 90%. 159 The diagnostic ability is maintained even in immunocompromised patients. 160 However, in patients with AN, 42% were reported to test negative for IgG, questioning the significance of antibody testing in the diagnosis of AN. 86 One-third of 79 healthy controls were reported to have false-positive results, highlighting the importance of selecting patients with a high pre-test probability for testing.161,162 In regions where non-fumigatus Aspergillus is prevalent, additional testing for IgG antibodies to other Aspergillus spp. may be considered in cases where testing is negative in patients suspected of having CPA.163,164 Concerns have been raised about the appropriateness of the manufacturer’s IgG positive cutoff values for diagnosing CPA, with several reports suggesting inadequacy.165,166 Various cutoff values have been proposed, but no definitive conclusion has been reached.167–170 Due to geographic variations in the serum prevalence of Aspergillus IgG and CPA, a universal cutoff for Aspergillus IgG may not exist. 171 Enzyme-linked immunosorbent assay is considered superior for Aspergillus IgG testing, 172 with several commercially available assays, although no conclusive comparison of diagnostic performance has been established. 54 The practicality of lateral flow devices utilizing immunochromatography for the detection of Aspergillus IgG has been reported, making them suitable for the diagnosis of CPA in several studies.153,154,173–176 There are conflicting reports regarding the ability of Aspergillus IgG to reflect treatment response.64,177–179 While Aspergillus IgM and IgA antibody assays have been reported, their utility in the diagnosis of CPA appears to be limited.180–183 Total serum IgE has been suggested as a potential biomarker of disease activity in CPA. 184
Aspergillus galactomannan antigen testing
Serum galactomannan (GM) antigen testing in CPA is generally not recommended for diagnosis due to its low sensitivity (23–66%) and prevalence of false-positive results.54,154,155,185–188 In cases of SAIA, which closely mimics the pathophysiology of IPA, serum GM tends to be more frequently positive.
54
The use of bronchoalveolar lavage fluid (BALF) GM antigen testing in CPA patients has been reported to have higher sensitivity and specificity compared to serum GM.79,170,185,187–191 However, there is concern due to significant variability in reported sensitivity and specificity, possibly due to differences in BALF collection methods between institutions. In addition, the cutoff values for BALF GM antigen for the diagnosis of CPA are inconsistent. Sputum GM antigen has been studied, but its recommendation is hampered by the constant risk of environmental Aspergillus contamination and the inherent variability in sputum sample quality.
192
While attempts to simultaneously measure BALF GM antigen and BALF β-
Mycological culture testing
Detection of Aspergillus in respiratory specimens alone is not sufficient to confirm a diagnosis of CPA. 54 In particular, a positive culture from sputum is not considered diagnostic due to the ubiquitous presence of Aspergillus as an environmental fungus.54,193 However, in CPA patients, the issue of drug resistance of A. fumigatus during treatment emphasizes the crucial importance of culture for drug susceptibility testing.194–196 Detection using BALF provides more specific results, with a reported positive culture rate of approximately 70%.197–199 The use of fungus-specific culture plates has been identified as a strategy to increase the positivity rate. 200 In addition to the uneven distribution of Aspergillus in liquid samples such as BALF due to its filamentous nature, the mucous nature of respiratory specimens further complicates the task of obtaining homogeneous specimens even after agitation. Unlike bacterial or yeast cell forms that disperse uniformly upon agitation, Aspergillus tends to remain unevenly distributed in liquid samples, making the addition of a portion of the sample to the culture plate potentially insufficient for detection. Therefore, increasing the number of samples cultured or increasing the volume of the sample on the culture plate has been reported to improve the sensitivity of the culture.54,201,202
Nucleic acid testing
Polymerase chain reaction (PCR) testing is considered to be more sensitive than culture; however, similar to culture, there are concerns about false-positive results due to environmental contamination with Aspergillus.54,203 In CPA patients, BALF is the primary specimen considered, with consistent sensitivity reported189,204 but challenges with standardization of testing methods are noted.203,205 Additional nucleic acid tests, such as real-time PCR and loop-mediated isothermal amplification (LAMP), are under consideration.204,206,207 A novel genetic test for the five major Aspergillus spp. using LAMP has been developed, focusing on respiratory specimens. 208 The diagnostic performance for CPA was reported to be 71.4% sensitivity and 87.0% specificity. 208 While sputum PCR testing is generally not recommended due to concerns about false-positive results, Aspergillus PCR positivity in sputum has been associated with azole resistance in CPA patients undergoing treatment. 209
Markers of inflammatory response
Blood tests commonly used in clinical settings, such as procalcitonin, C-reactive protein (CRP), and erythrocyte sedimentation rate, are not considered useful in the diagnosis or follow-up of CPA. 210 However, in more extensive forms of lung involvement, such as SAIA or CFPA, there is a tendency for elevated levels of inflammatory markers and counter-immunoelectrophoresis (CIE) titers. 211 Increased levels of circulating inflammatory mediators, including, IL-8, regulated upon activation, normal T cell expressed and secreted (RANTES), tumor necrosis factor-alpha (TNF-α), intercellular adhesion molecule 1 (ICAM-1), and mediators involved in endothelial activation and thrombosis (von Willebrand factor, tissue factor, plasminogen activator inhibitor-1) have been reported in CNPA patients. 212 As a potential candidate for assessing the characteristics of CPA patients, we have also reported a specific increase in the IL-10/IL-5 ratio in CPA patients. 213
Characteristics of exhaled components and respiratory function tests in CPA patients
An attempt was made to analyze volatile organic compounds (VOCs) in the exhaled breath of CPA patients for diagnostic purposes. 214 In comparison to healthy individuals and those with community-acquired pneumonia, characteristic VOCs identified in the exhaled breath of CPA patients include phenol, neopentyl alcohol, toluene, limonene, and ethylbenzene. Limonene concentration was found to be significantly correlated with Aspergillus IgG antibody titers. Analysis of respiratory function tests in CPA patients was also reported. Of 112 CPA patients, 46% had restrictive impairment and 54% had obstructive impairment. 215 This impairment of respiratory function probably reduces the quality of life of CPA patients.216–218 However, these results are strongly influenced by the underlying lung diseases associated with CPA, and the clinical significance of these findings in CPA remains unclear.
Surgery for CPA
Benefits and risks of surgery for CPA
Surgery may be a curative treatment. A retrospective analysis of 240 CPA patients with aspergilloma showed a significant improvement in 10-year survival in 135 cases who underwent surgical resection compared to 105 cases who were treated conservatively (84.8% versus 56.7%, p < 0.001). 219 In the analysis of 24 CPA patients who underwent surgery for hemoptysis, 83% experienced cessation of hemoptysis. 220 However, there is variability in reported postoperative mortality rates ranging from 0% to 5.7%,221–239 suggesting the need for cautious consideration of surgical indications in CPA patients. 54 Postoperative complications have been reported at rates ranging from 11% to 63%,221,223–237,239,240 with a higher incidence in cases where CPA developed in lungs with tuberculosis. 231 Recent improvements in surgical techniques and a decrease in the incidence of pulmonary tuberculosis may contribute to a reduction in postoperative complications.232,233 Common postoperative complications include recurrent pneumothorax (2.3–26.3%), prolonged air leak (2.3–23.0%), abscess formation (1.2–20.0%), bleeding (1.6–17.4%), respiratory failure (1.9–14.3%), incomplete lung re-expansion (9.3–12.1%), wound infection (2.0–4.5%), and bronchopleural fistula (1.6–2.5%).221,223–227,230,234–237,239 The postoperative recurrence rate of CPA varies from 0% to 41%,86,220,224,227,234,235,239,240 and of 25 recurrent cases studied, 80% had a recurrence within 3 years. 220
Selection of CPA patients for surgery
Surgical resection of aspergilloma is an important therapeutic option for CPA patients with sufficient pulmonary function.54,236,241,242 Surgery should be considered for all CPA patients with severe hemoptysis.54,243 However, many patients may be physically frail, which increases the risk of mortality and perioperative complications. 235 Therefore, careful patient selection is essential before deciding to proceed with surgery.235,238 Postoperative mortality rates for each subtype are reported as SA: 0%, CCPA: 1.9–8.1%.222,225,230,244 In terms of postoperative complications, SA: 0–9.3%, CCPA: 0–30.0%, indicating increased risks with CCPA.225,230,244,245 While surgery is an appropriate choice for SA, careful management of CCPA is warranted due to the potential for higher risks.235,237,238,246,247 Recurrence rates after surgery vary, SA: 0–43%, CCPA: 0–75%, AN: 0–40%, CFPA: 7.7%.86,220,222,244,245,248 An analysis of 85 surgically treated CPA cases, including SA, CCPA, CFPA, and AN, showed equivalent recurrence rates. 248 However, postoperative complications were significantly associated with CFPA, highlighting its role as a significant risk factor. 248
Surgical approaches for CPA
The choice of surgical procedure for CPA patients is critical to achieving complete aspergilloma excision without fungal spillage into the pleural cavity, ensuring fungal eradication and control. 54 Basic surgical options include bullectomy, segmentectomy, sublobar resection, wedge resection, lobectomy, pleurectomy, and pneumonectomy. 54 Choosing selective surgical treatment with fewer lung resections may reduce the risk of postoperative complications. 249 In recent years, robot-assisted surgery has been attempted. 250 Both single-port and multi-port video-assisted thoracic surgery have been proposed as alternative techniques to open surgery, potentially reducing the number of complications and hospital stays.249,251–253 These methods are suggested for resection of SA and multifocal disease without hilar infiltration.54,249,251 In patients deemed unsuitable for lung resection due to age, severe respiratory failure, or general poor health, cavenostomy is considered when the aspergilloma is peripherally located.238,254,255 Cavenostomy is followed by thoracoplasty with simultaneous muscle flap transplantation.256–261 Although video-cavernoscopy for aspergilloma removal has been attempted, recurrence has occurred in approximately half of the cases. 262 In patients ineligible for surgery, bronchoscopic mechanical aspergilloma removal has been reported in cases where the cavity is accessible by bronchoscopy. 263
Perioperative antifungal therapy
There is no established protocol for perioperative antifungal therapy, and several reports suggest no benefit from antifungal administration during this period.264–266 SA that can be resected without a spread of Aspergillus probably does not require adjuvant antifungal therapy. 54 However, some reports suggest that preoperative or perioperative administration of antifungals may prevent recurrence.220,235,267 In cases of CCPA or when surgical techniques are complex and fungal spread is anticipated, preoperative antifungal therapy weeks before surgery may be preferable. 54 Postoperative antifungal therapy is considered necessary if Aspergillus is expected to remain in the body (e.g. if specimens obtained during or after surgery are culture positive). 54 In cases of intraoperative or postoperative fungal spread into the pleural cavity, the method of injecting 2% taurolidine into the pleural cavity has been reported. 235
Management of hemoptysis in CPA
Characteristics of hemoptysis in CPA
Among 143 CPA patients with aspergilloma, 50.3% had clinically significant hemoptysis. 268 Patients with a mean cavity diameter of 22 mm or more and an aspergilloma diameter of 18 mm or more had an increased risk of hemoptysis. 268 However, no significant correlation was found between the severity of hemoptysis and imaging features. 269 In a study investigating the causes of hemoptysis in 25 patients with CPA, 68 bleeding arteries were identified. Of these, 36 were bronchial arteries (52.9%), 15 were intercostal arteries (22.1%), 9 were internal thoracic arteries (16.2%), 5 were inferior phrenic arteries (7.4%), and 3 were pulmonary artery branches (4.4%). 270 CPA patients with hemoptysis had significantly elevated blood levels of VEGF. 271
Management of hemoptysis in CPA
Bronchial artery embolization (BAE) is a safe and minimally invasive procedure for the treatment of hemoptysis that allows for repeat interventions. 272 BAE is indicated in cases where surgery is delayed or deemed inappropriate. 54 Because it directly targets the source of the bleeding vessels, 64–100% of CPA patients achieve hemostasis within 24 h of BAE.273–275 However, among the various causes of hemoptysis, the presence of aspergilloma significantly correlates with a high recurrence rate. 276 The recurrence rate of hemoptysis in CPA patients treated with BAE is approximately 50%.275,277 Although there appears to be no difference in success rates between SA and CCPA, CCPA patients tend to have a higher recurrence rate than SA patients (55% versus 33%). 275 Preoperative arterial embolization is expected to reduce intraoperative bleeding; however, at least two retrospective studies have not confirmed a significant reduction in bleeding volume.278,279 Some attempts have been made to use plumbage surgery to control hemoptysis in CPA patients. 280 Tranexamic acid (1 g orally or intravenously every 8 h), a fibrinolysis inhibitor proven to reduce bleeding in other patient populations, is prescribed for its potential to suppress hemoptysis in CPA patients, although data on its efficacy in this setting are limited.54,281–283
Pharmacological treatment of CPA
Oral triazole therapy for CPA
Triazoles represent the only class of drugs for the oral treatment of CPA and serve as the first choice for the treatment of CCPA, SAIA, and CNPA.54,64,83 Surgical resection remains the preferred approach for SA, with antifungal therapy considered for cases ineligible for surgery. 54 There is limited research on the efficacy of triazole therapy for asymptomatic SA. Triazole treatment for CFPA is considered beneficial for overall patient stability, although its impact on dyspnea is considered limited. 67 SAIA follows the treatment strategy for IPA. 54 Most AN remained stable regardless of the effect of the antifungal drugs. 90 Four triazoles – itraconazole (ITCZ), voriconazole (VRCZ), posaconazole (PSCZ), and isavuconazole (ISCZ) – are currently available for the treatment of CPA.54,284 Adverse effects of triazoles are common and include peripheral neuropathy, heart failure, elevated liver enzymes, QTc prolongation, and sunlight sensitivity. 67 ITCZ, VRCZ, and PSCZ may have significant interindividual variability in drug concentrations, making therapeutic drug monitoring (TDM) essential during azole therapy.67,285 In addition, the potential for drug–drug interactions is a critical consideration when using triazoles for the treatment of CPA. 67
Oral itraconazole therapy for CPA
In a prospective randomized controlled trial comparing patients with CCPA, the group receiving oral ITCZ at 400 mg/day for 6 months had a significantly higher rate of improvement than the group receiving supportive therapy alone (76.5% versus 35.7%). 286 Combining results from other clinical trials, the efficacy of oral ITCZ for CCPA ranges from 43.5% to 76.5%.67,97,287–290 The recurrence rates at 2 years after initiation of ITCZ therapy were 38% with 6 months of treatment and 10% with 12 months of treatment. 291 Meta-analysis of observational data showed no significant differences in outcomes between ITCZ, VRCZ, and PSCZ. 292 However, based on its proven efficacy in a randomized controlled trial and substantial support from other clinical research, oral ITCZ is recommended as first-line therapy for CCPA.54,64 ITCZ treatment is associated with cardiotoxicity in 29% of patients. 293 Dose adjustment based on body weight (200 mg/day for 30–39 kg, 300 mg/day for 40–49 kg, and 400 mg/day for 50 kg and above) has been reported to reduce adverse effects and allow for long-term administration. 294 Although several formulations of ITCZ exist, capsule formulations tend to have lower blood concentrations. 295 Quality concerns have been raised regarding generic versions that may affect blood concentration levels. 296
Oral voriconazole therapy for CPA
The reported efficacy of oral VRCZ for CCPA ranges from 32% to 80%.97,198,297–301 Compared to CCPA, CNPA has a higher reported efficacy (58% versus 32%). 297 In a retrospective analysis of 160 CPA patients, the percentage of patients showing improvement was higher in the VRCZ group compared to the ITCZ group (40.0% versus 18.2%). However, when stable patients were included, the percentages in the VRCZ and ITCZ groups were 52.6% and 50.9%, respectively, with no significant difference. 97 VRCZ is considered the first-line treatment for CCPA and can be used after failure or intolerance of ITCZ therapy.54,64 VRCZ treatment is associated with skin adverse events in 28% of patients. 293 Long-term use of VRCZ has been associated with an increased risk of skin cancer, so caution should be taken when using it for prolonged periods in CPA patients. 302 TDM is recommended for dose adjustment of VRCZ. 54 TDM is required when initiating VRCZ, changing its dosage or formulation, or initiating medications that interact with VRCZ. In addition, blood concentrations of VRCZ in CPA patients have been reported to correlate with CRP, highlighting the importance of monitoring VRCZ levels in patients with changing CRP. 303
Oral posaconazole therapy for CPA
The reported efficacy of oral PSCZ for CPA ranges from 44% to 61%.304,305 PSCZ is available in multiple formulations, but PSCZ delayed-release tablets exhibit superior biological availability compared to liquid suspension. 306 Even at a lower dosage (200 mg once daily), which is below the usual dose (300 mg once daily), sufficient drug concentrations have been reported in CPA patients. 306 This lower dose has the potential to achieve therapeutic levels while reducing the risk of long-term side effects.
Oral isavuconazole therapy for CPA
In a randomized, open-label study comparing ISCZ with VRCZ as a control, the overall response rate at the end of treatment for CPA patients in the ISCZ group (n = 52) and the VRCZ group (n = 27) was 82.7% and 77.8%, respectively. 307 Adverse drug reactions were 61.5% in the ISCZ group and 85.2% in the VRCZ group. Another retrospective study suggested a lower incidence of adverse events with ISCZ compared to VRCZ (60% versus 86%, p = 0.02). 308 As ISCZ has stable pharmacokinetics, TDM is usually not required. 309 The typical dose of ISCZ is 200 mg orally once daily. However, in cases where continued administration is challenging due to adverse events, a reduced daily dose of 100 mg has been reported to achieve adequate blood concentrations, improve tolerability, and allow for prolonged treatment. 309
Intravenous echinocandin therapy for CPA
Echinocandins are characterized by lower toxicity compared to other antifungal agents. 310 In addition, they are less likely to be substrates for cytochrome P450 enzymes, resulting in fewer drug interactions compared to azole antifungals. 310 The reported efficacy of echinocandins for CPA ranges from 42.4% to 77.8%.311–316 In a multicenter, open-label, randomized study comparing micafungin (MCFG) with VRCZ (n = 97), there was no significant difference in efficacy between the MCFG group (60.0%) and the VRCZ group (53.2%) (p = 0.499). 311 Safety evaluation showed fewer adverse events in the MCFG group compared to the VRCZ group (26.4% versus 61.1%, p = 0.0004). 311 In a randomized, double-blind study directly comparing caspofungin (CPFG) and MCFG (n = 120), efficacy against CPA was equivalent (CPFG 46.7% versus MCFG 42.4%) and there was no statistically significant difference in safety. 313 In a multicenter prospective study of combination therapy with ITCZ and MCFG, the efficacy was 58.8%. 317 Echinocandins are administered exclusively by intravenous infusion, making long-term administration challenging. As an alternative, outpatient treatment with MCFG via outpatient parenteral antimicrobial therapy or intermittent administration of echinocandins has been attempted with reported efficacy.318,319
Intravenous amphotericin B therapy for CPA
In intravenous amphotericin B therapy for CPA, liposomal amphotericin B (L-AMB) is used as a single agent or in combination with other drugs for safety reasons.320,321 Intravenous infusion of amphotericin B deoxycholate is considered less beneficial and is not recommended for CPA patients.74,67,199 The reported efficacy of L-AMB for the treatment of CPA ranges from 52.9% to 73.8%.321,322 A multicenter, open-label, randomized study comparing the effects of L-AMB (2.5–5.0 mg/kg once daily intravenously, n = 51) and VRCZ intravenous therapy (n = 59) in CPA patients showed no statistically significant difference in efficacy at the end of treatment (L-AMB 52.9%, VRCZ 67.8%, p = 0.111). 321 The median duration of treatment was 21 days for L-AMB and 28 days for VRCZ. 321 Adverse events in the L-AMB group included hypokalemia (34.9%) and renal dysfunction (16.9%). 321 In a retrospective analysis of 71 CPA patients treated with L-AMB, 73.8% responded within the first 6 weeks, but 75% experienced a greater than 25% decline in estimated glomerular filtration rate (eGFR) during initial treatment, with 25% developing acute kidney injury. 322
Intrabronchial triazole therapy for CPA
In a randomized controlled trial (n = 60) evaluating the adjunctive effect of intrabronchial VRCZ therapy for CPA patients with mild to moderate hemoptysis, the combination group demonstrated a significant reduction in hemoptysis severity compared to the control group (86.7% versus 36.7%, p < 0.0001). 323 However, the adjunctive use of intrabronchial VRCZ therapy did not show a preventive benefit for hospitalization or BAE. 323 In a retrospective study that served as the basis for the randomized trial, cases (n = 82) receiving 400 mg VRCZ dissolved in 20 mL 0.9% saline and administered intrabronchial at weekly intervals for 4 weeks showed a 68.3% reduction in hemoptysis and a 54% reduction in aspergilloma size. 324 Other case reports have also highlighted instances where intrabronchial ITCZ therapy resulted in the disappearance of aspergillomas. 325
Inhaled amphotericin B therapy for CPA
A prospective, non-inferiority, open-label, randomized controlled trial (n = 33) compared the efficacy of inhaled amphotericin B therapy (50 mg/day for 7 days) with oral ITCZ therapy (400 mg/day for 6 months) in patients with CPA. 326 Patients inhaled levosalbutamol 1.25 mg over 10 min, followed by inhaled amphotericin B (50 mg/10 mL) over 30 min. 326 The results at the 6-month follow-up showed equivalent treatment effects between the two regimens (67% versus 65%). 326 Inhaled amphotericin B therapy resulted in cough in 40% of patients, but did not cause renal impairment, a serious adverse effect of amphotericin B. 326 In another case, successful treatment with inhaled L-AMB (25 mg, twice daily) in combination with VRCZ has been reported. 327
Intracavitary administration of amphotericin B for CPA
In cases of intractable hemoptysis in patients with CPA where surgical resection is not feasible and there is no associated bleeding disorder, infusion of amphotericin B into the infected cavity may be considered.54,328 However, it has been reported that the efficacy of this treatment tends to decrease in patients with a longer duration of disease. 329 Antifungal agents can be administered either percutaneously or bronchoscopically into the lung cavity. Many percutaneous approaches involve the placement of a percutaneous catheter, which reduces the patient burden of repeated antifungal administration.329–334 A retrospective analysis of 20 cases in which percutaneous intracavitary amphotericin B was administered reported that bleeding was stopped in 85% of cases and pneumothorax occurred in 26% of catheter insertions. 330 The standard dose is 50 mg of amphotericin B dissolved in 20 mL of 5% dextrose solution and injected in an amount based on the size of the cavity.54,330,331 In cases without a percutaneous catheter, an alternative method involves combining amphotericin B with a paste or gelatin for prolonged retention in the cavity, with percutaneous injection guided by CT.335,336 During injection, patient positioning is adjusted to prevent leakage into bronchi other than the target bronchus. 54 Complications may include cough, chest pain, pneumothorax, and bronchial reflux. 54 One case has been reported in which repeated bronchoscopic administration of L-AMB resulted in the disappearance of aspergilloma. 337
IFN-γ immunotherapy for CPA
IFN-γ deficiency has been observed in some cases of CPA, prompting the exploration of adjunctive immunotherapy with IFN-γ.54,338 There are reports of clinical efficacy confirmed in CPA patients with IFN-γ administration.67,339,340 A retrospective analysis of patients (n = 20) who received IFN-γ immunotherapy for more than 12 months showed a significant reduction in acute exacerbations of CPA and hospitalizations during the 12 months following initiation of treatment compared to the previous 12 months. 340 However, due to the limited number of documented cases and the lack of controlled trials, IFN-γ immunotherapy is not currently recommended for CPA. 54
Corticosteroid therapy and CPA
The use of corticosteroids poses a significant risk for the progression of CPA. 54 If the continuous administration of corticosteroids or immunosuppressive agents is necessary to treat the underlying disease, appropriate antifungal therapy is essential. 54 On the other hand, in specific pathologies within CPA where an excessive inflammatory response is observed, cases have been reported where the adjunctive use of corticosteroids was beneficial.69,341 Certain fungal species, such as A. niger and A. tubingensis, produce oxalic acid during infection.342,343 Oxalic acid reacts with calcium in the host to form oxalate crystals. 344 Deposition of these crystals in tissues can cause severe inflammation with potentially fatal outcomes, a condition known as oxalosis [Figure 7(d)].345,346 In a case where treatment with MCFG and VRCZ was ineffective in a CPA patient with confirmed oxalosis, the addition of corticosteroids successfully controlled the intense inflammatory response associated with oxalosis, resulting in improvement. 341 In addition, some patients with CPA develop concomitant organizing pneumonia.68,69,342 One case has been reported in which the adjunctive use of corticosteroids effectively treated organizing pneumonia complicating CPA. 69
Follow-up of antifungal therapy for CPA
Evaluation of response to antifungal therapy for CPA
Evaluation of the efficacy of antifungal therapy for CPA is performed after a minimum of 3 months and ideally continues for more than 6 months of treatment. 347 Clinical or radiologic deterioration after 6 months of treatment is considered treatment failure. 347 It is essential to exclude other potential causes of clinical or radiologic deterioration. Including weight loss of 5% or more as a criterion for deterioration has been reported to increase the sensitivity of treatment failure detection by 36%. 348 Serum tests should not be relied upon to determine treatment response. 347 While changes in imaging findings are variable, the disappearance of aspergillomas, reduction in cavity wall thickening, and reduction in pleural wall thickening correlate strongly with clinical improvement. 65 By contrast, changes in cavity size are not associated with the progression of CPA. 65
Duration of antifungal therapy and risk of recurrence in CPA
The recommended duration of antifungal therapy for CPA is at least 6 months.54,64 In addition, the efficacy of continued treatment for more than 1 year has been reported.291,349,350 In a single-center, open-label, randomized, controlled trial comparing the 6-month treatment group (n = 81) with the 12-month treatment group (n = 83) using ITCZ, the proportion of patients who experienced a recurrence 2 years after treatment initiation was significantly lower in the 12-month group (10%) compared with the 6-month group (38%), with an absolute risk reduction of 0.29 [95% confidence interval (CI) 0.16–0.40]. 291 The mean time to first recurrence was significantly longer in the 12-month group (23 months) than in the 6-month group (18 months) (p < 0.0001). 291 In a retrospective analysis of 196 cases, treatment duration of 12 months or more was independently associated with a reduced risk of recurrence (adjusted hazard ratio: 0.48, 95% CI: 0.28–0.80). 349 CPA patients who continued antifungal treatment for 1 year also showed a sustained decrease in Aspergillus IgG levels throughout the year. 351 Since the goal of antifungal therapy for CPA is not only to improve symptoms and prevent recurrence but also to prevent hemoptysis and further fibrosis, continuing antifungal therapy in stable patients may be beneficial by suppressing the worsening of CPA. 54 After discontinuation of CPA treatment, 20–30% of patients experience recurrence within 1 year.60,352,353 Risk factors for recurrence of CPA include involvement of more than one lung lobe, a large initial maximum cavity diameter, and concurrent infection with nontuberculous mycobacteria.352–354
Prognosis of CPA
Mortality and prognostic factors in patients with CPA
The 1-year mortality rate in CPA patients ranges from 7% to 32% and increases to a substantial 38–52.6% at 5 years, indicating a poor prognosis.105,355–359 Factors contributing to mortality in CPA can be broadly categorized into patient-specific characteristics, comorbidities, and various laboratory parameters. Patient-specific characteristics associated with CPA-related mortality include age 65 years or older, male sex, underweight (BMI < 18.5 kg/m2), worsening St George’s Respiratory Questionnaire activity domain scores, and malnutrition.97,355–358,360,361 Comorbidities associated with mortality in CPA include COPD, non-tuberculous mycobacterial lung disease, diabetes, interstitial lung disease, history of lung cancer, pulmonary hypertension, high Charlson Comorbidity Index, and baseline corticosteroid use.92,93,97,357,358,360,362–364 Laboratory parameters associated with CPA-related mortality include low serum albumin, elevated CRP, high TNF-α, high IL-1β, elevated Aspergillus IgG, and the presence of an aspergilloma.356,357,359,362,363,365 Of note among specific patient backgrounds is the impact of impaired IFN-γ production, which has been reported to negatively influence CPA prognosis. 114 Conversely, factors predictive of favorable outcomes include higher BMI, lower Charlson Comorbidity Index, and antifungal therapy for more than 6 months.361,366 In our study of 273 CPA patients, exacerbation of CPA accounted for only 31% of fatal cases, bacterial pneumonia complications accounted for 25%, and 23% succumbed to comorbidities. 97 This underscores the significant impact of comorbidities associated with CPA on overall prognosis. 367
Assessment of CPA activity
When assessing the activity of CPA, understanding the impact of the disease on patients’ quality of life becomes a critical outcome measure. The St. George’s Respiratory Questionnaire is used as an objective tool to assess factors such as fatigue and general health, which are thought to reflect the activity of CPA.217,368 This questionnaire has demonstrated significant reliability and validity in measuring the health status of individuals with CPA. 369 Symptom worsening on the St. George’s Respiratory Questionnaire and patient self-reported worsening have been reported to be associated with clinical relapse. 370 In addition, serum IL-1β and total IgE have been reported as useful markers for monitoring the activity of CPA.184,371
Limitations
The evolving nature of research in this area means that newer studies may have been published after the review, potentially affecting the comprehensiveness of the synthesized information. In addition, there is a potential for selection bias, as citations may have been chosen subjectively.
Conclusion
In conclusion, the pathophysiology of CPA is complex, emphasizing the need for personalized medical care for each patient.372,373 A thorough understanding of the characteristics of the causative fungi, epidemiology, pathogenesis, risk factors, diagnosis, treatment, and prognosis, as outlined in this review, is essential for optimal patient-tailored management. Although treatment options for CPA remain limited, the ongoing momentum in clinical trials, led by global organizations such as the Chronic Pulmonary Aspergillosis Network (CPAnet), is promising. This gradual accumulation of evidence from clinical research raises expectations for future improvements in the management of CPA.374–376
Footnotes
Acknowledgements
We are deeply grateful to Cana Fukumaki for her invaluable contributions in creating the figures for this review article. In addition, our sincere thanks go to Takayoshi Tashiro for providing valuable insights and guidance during the development of this article. We would like to thank Yuichiro Nakano for his valuable advice on the morphology of Aspergillus. Their dedication and expertise have greatly enhanced the quality and impact of our work, and we express our sincere appreciation for their significant contributions to this project.
