Abstract
Background:
Despite adoption of the ‘test-and-treat’ strategy, a high proportion of antiretroviral therapy (ART) naïve people living with HIV (PLHIV) enrol in care with, and die of advanced HIV disease (AHD) in Uganda. In this study, we aimed to determine the prevalence of AHD among ART naïve adults enrolling in care and associated factors at selected public health facilities in Kampala, Uganda.
Methods:
From April to July 2022, we conducted a mixed-methods study at Kiswa Health Centre III, Kitebi Health Centre III, and Kawaala Health Centre IV. The study involved cross-sectional enrolment and evaluation of 581 participants, utilizing an interviewer-administered questionnaire and chart reviews. Modified Poisson regression was employed to identify factors associated with AHD, complemented by a qualitative component comprising fifteen in-depth interviews, with data analysed through thematic analysis.
Results:
Overall, 35.1% (204/581) of the study participants had AHD. Being male [adjusted prevalence ratio (aPR): 1.4, 95% CI: 1.04–1.88] and aged 35–50 years (aPR: 1.81, 95% CI: 1.14–2.88) were associated with AHD. Participants with no personal health perception barriers had 37% lower odds of presenting to care with AHD (aPR: 0.63, 95% CI: 0.46–0.85). Qualitative findings indicated that individual factors, such as waiting until physical health deteriorated and initially opting for alternative therapies, took precedence in contributing to enrolment in care with AHD.
Conclusion:
Over one in every three ART naïve adults presents to public health facilities in Uganda with AHD. Male gender, age 35–50 years, and personal health perception barriers emerged as significant factors associated with AHD; emphasizing the need for targeted interventions to address these disparities and enhance early detection and engagement in care. Routine HIV testing should be emphasized and incentivized especially for men and persons aged 35–50 years.
Introduction
Globally, 39 million people were living with HIV in 2022. Of these, more than half (20.8 million) were in Eastern and Southern Africa. 1 Uganda contributed approximately 4% of the global HIV burden. 2 In late 2015, the World Health Organization (WHO) made the ‘test and treat’ recommendation where all criteria (WHO clinical stage and CD4 cell count) on eligibility for anti-retroviral therapy (ART) following an HIV positive test were removed. 3 This followed evidence to support immediate ART initiation.4,5 The test and treat policy was adopted by the Ministry of Health in Uganda in December 2016, and in 2022, coverage of adults and children receiving ART was 84%. 2 Following the 2015 consolidated guidelines on the use of ART drugs for treating and preventing HIV infection, the WHO published guidelines on the management of advanced HIV disease (AHD). ‘For adults and adolescents, and children older than 5 years, AHD was defined as CD4 cell count less than 200 cells/µL or WHO stage 3 or 4 events. All children younger than 5 years old with HIV were considered to have AHD’. 6
Globally, AIDS-related deaths have been reduced by 51% since 2010. 1 Despite this success, in 2022, 630,000 people died from AIDS-related illnesses worldwide. 7 This was higher than the UNAIDS target of reducing AIDS-related deaths to fewer than 500,000 as part of the commitments to end AIDS by 2030. 8 In Eastern and Southern Africa; 260,000 people died from AIDS-related illnesses, contributing 41% to the global burden of mortality related to HIV. 1 In the same year 2022, 17,800 people died due to AIDS-related illnesses in Uganda. 2 Individuals who start ART later in the course of the infection are at a higher risk of death compared to those who start earlier, with disease progression to death and AIDS clustered among patients starting ART with a CD4 cell count less than 200/µL.9,10
Prior studies conducted in Uganda to determine prevalence of AHD used inconsistent and different criteria to define AHD. Additionally, they were all conducted prior to adoption of the ‘test and treat’ recommendation. The current prevalence of AHD among ART naïve PLHIV enrolling in care and the associated factors during the ‘test and treat’ era in Uganda are unknown. In order to establish the burden of the problem, determine and understand associated factors, and subsequently inform prevention strategies, a mixed methods study design was conducted.
Methods
Study design
We conducted a mixed methods study employing both qualitative and quantitative data collection methods. Qualitative data collection using in-depth interviews allowed probing and a deeper understanding of reasons for enrolling in care with AHD. Responses were fully explored, explanations and clarifications sought where the response seemed ambiguous.
Study area
The study was conducted at three primary healthcare centres in Kampala. Kampala is the capital and largest city in Uganda, and is divided into five political divisions of Nakawa, Central, Makindye, Rubaga and Kawempe. It is administered by the Kampala Capital City Authority (KCCA). There are six KCCA public health facilities providing comprehensive HIV prevention, care and treatment services; these include Kiswa HC III in Nakawa, Kawaala HC IV in Rubaga, Kitebi HC III in Rubaga, Komamboga HC III in Kawempe, Kisugu HC III in Makindye and Kisenyi HC IV in Central division. Participants for this study were recruited from Kiswa HC III, Kitebi HC III and Kawaala HC IV. The health centres were randomly chosen using a random number generator in Microsoft Excel.
Approximately 22,600 PLHIV are in care at the three facilities with Kawaala HC IV having the largest number of clients at approximately 9000, 7400 at Kitebi HC III and 6200 clients at Kiswa HC III. All health centres offer a wide range of other services including maternal and child health services, emergency and outpatient services.
Study population
ART naïve PLHIV aged 18 years and above enrolling in care at the study sites were considered for the study.
Inclusion criteria
ART naïve PLHIV enrolling in care between January 2021 and July 2022 at Kiswa HC III, Kawaala HC IV and Kitebi HC III.
Exclusion criteria
Clients who were mentally ill and those unable to give verbal responses to the interview questions.
Sample size and sampling procedure
With the primary aim to determine the prevalence of AHD among ART naïve PLHIV enrolling in care, sample size was determined using Kish Leslie formula. The total sample size required was calculated to be 547 participants, and a minimum number of 164 participants per site was determined using a formula by Bennet and colleagues. 11 Fifteen in-depth interviews to explore reasons for enrolling in care with AHD were conducted.
Participants were systematically sampled from a line list of clients who had newly enrolled in care between January 2021 and March 2022. This line list was obtained from the Uganda Electronic Medical Records. Sampled individuals were then invited back to the health facility to participate in the study.
For the qualitative component, individuals presenting with AHD were purposively selected for in-depth interviews.
Study variables
AHD was defined as WHO stage 3 and 4 or CD4 cell count of less than 200 cells/µL at enrolment in care.
Demographic and individual factors; these included age, sex, marital status, education level, employment status, number of recent sexual partners, competing needs to health care, that is, failure to access health care in favour of basic needs like food, shelter, and clothing and failure to access the basic needs in favour of health care and perceived personal barriers to health care. Responses were also recorded to questions related to perceived barriers to seeking HIV testing and medical care. Service delivery barriers included ‘have to wait too long to see the health care worker’, and ‘not treated with respect by the health care worker’. Financial barriers included ‘high costs associated HIV medications’ and ‘could not afford the cost of transport to and from the HIV clinic’. Personal health perception barriers included ‘didn’t think it was necessary because didn’t feel sick’ and ‘felt too sick to go to HIV clinic’ Logistical barriers included ‘could not get time off work to attend the clinic’ and ‘had to take care of someone else’. Structural barriers included ‘could not get to the clinic during the hours it was open’, and ‘did not know where to seek HIV care from’. Interpersonal factors included living in a household with others and failure to disclose HIV status to one’s spouse.
HIV programme and health system-related factors included sources of HIV care, distance from the health facility, time taken to travel to the HIV clinic, HTS approach; Provider Initiated Testing and Counselling (PITC) and Voluntary Counselling and Testing (VCT), HIV testing model and time from HIV diagnosis.
Data collection and study procedure
Quantitative data were collected using an interviewer-administered questionnaire and chart reviews by trained research assistants from April to July 2022. The questionnaire was pretested on 10 ART naïve clients who were not part of the study population. This was to ensure that proper, adequate and correct information is obtained during the study using these tools. Data collected from client charts included date of birth, CD4 level at ART initiation and presence of WHO stage 3 or 4 disease, while the rest was collected using an interviewer administered questionnaire. Qualitative data were collected from participants enrolling in care with AHD through in-depth interviews (IDIs) that were audio recorded. These were conducted at the study site by experienced qualitative interviewers. Participants were classified as having AHD by the research assistants through chart reviews and invited back to the facility for the IDIs. These were purposively selected to include females, males, those aged 25 and above, and those aged 24 and below. Qualitative data were collected on facilitators and barriers to HIV testing, knowledge on HIV testing and treatment services, disclosure, challenges in enrolling in HIV care, HIV care services and the health care system in general. IDIs were conducted until saturation. Authors had no access to information that could identify individual participants during or after data collection.
Data analysis
Participant characteristics were presented in a table as frequencies and percentages. Continuous variables were additionally analysed using mean, median, and standard deviation. Bivariate analysis was conducted to determine association of AHD with the individual risk factors using modified Poisson regression model. To allow incorporation of the perceived barriers into multivariable models, while also minimizing collinearity, they were categorized into five groups including service delivery, financial, personal health perception, logistical, and structural barriers. A modified Poisson regression model with robust standard errors was used to determine the factors associated with AHD at multivariable analysis. Prevalence ratios (PRs) were used as the measure of association. This was because the prevalence of the outcome was high at 35.1%, and the risk ratio would have been overestimated if odds ratios were used.12–14
The reporting of this study conforms to the Strengthening of the Reporting of Observational Studies in Epidemiology. 16
Results
Characteristics of study participants for the quantitative component
Quantitative data were collected from 581 participants and included in the analysis. Age ranged from 18 to 67 years (mean 32.3 years, SD 8.5, IQR 26–37). Nearly two-thirds of the participants (65.9%; 383/581) were females and 45.8% (266/581) were aged 25–34 years. Only 11.7% (68/581) had an underlying or chronic disease or disability and most participants (82.3%; 478/581) had never been diagnosed with HIV/AIDS before (Table 1).
Characteristics of participants.

AHD among the study participants
Among 581 participants, 12.7% (74/581) had WHO stage 3 or 4 disease, while 65.7% (382/581) had a CD4 ⩾ 200 cells/µL at ART initiation. Overall, 35.1% (204/581) of patients were considered to have AHD according to the WHO definition.
Bivariate analysis for the association between AHD and participant characteristics
At bivariate analysis, factors that were significantly associated with AHD included sex, age, perceived personal health barriers, and HIV testing point. The prevalence of AHD was 1.59 times higher among males compared to their female counterparts. It was also 2.22 times higher among participants aged 35–50 compared to those aged 18–24; and 2.89 times higher among those tested at the TB clinic compared to VCT (Table 2).
Bivariate analysis for the association between AHD and participant characteristics.
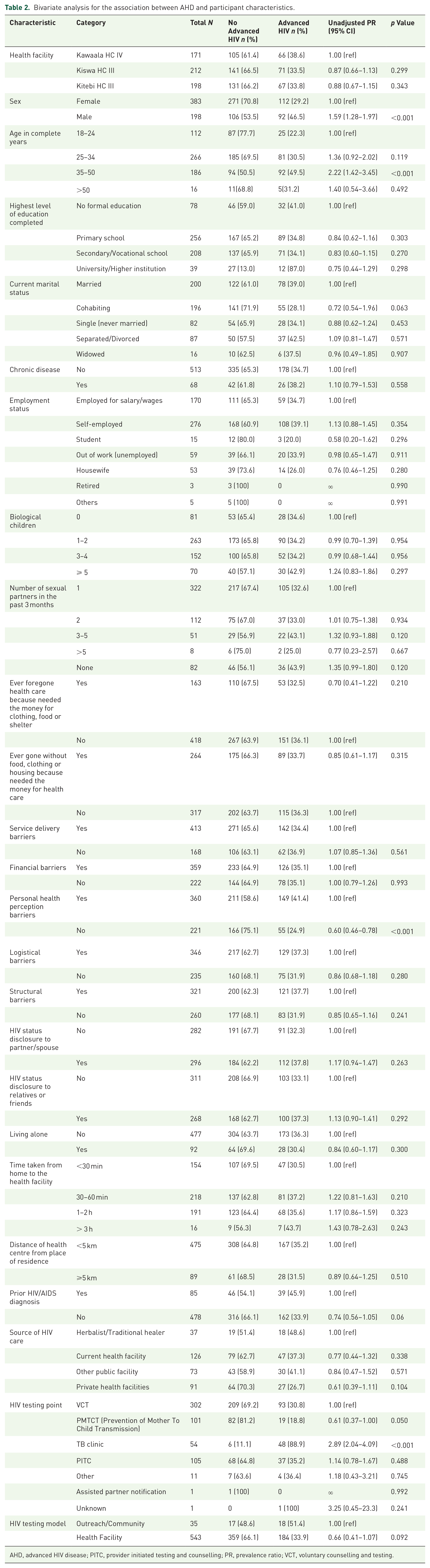
AHD, advanced HIV disease; PITC, provider initiated testing and counselling; PR, prevalence ratio; VCT, voluntary counselling and testing.
Multivariable analysis for the association between AHD and participant characteristics
At multivariable analysis, after adjusting for covariates, the prevalence of AHD was 1.4 times higher [adjusted PR (aPR): 1.40, 95% CI: 1.04–1.88] among males compared to their female counterparts. It was also 1.81 times higher (aPR: 1.81, 95% CI: 1.14–2.88) among participants aged 35–50 years when compared with those aged 18–24 years. When compared with participants who reported personal health perception barriers, the prevalence of AHD was 37% lower (aPR: 0.63, 95% CI: 0.46–0.85) among those who did not report any personal health perception barriers (Table 3).
Multivariable analysis for the association between AHD and patient characteristics.

Adjusted variables: HIV testing model, HIV testing point, Prior HIV/AIDS diagnosis, Current marital status, Care seeking, Disclosure to partner/spouse, Number of sexual partners in the past 3 months, district of residence.
AHD, advanced HIV disease; PITC, provider initiated testing and counselling; PR, prevalence ratio; VCT, voluntary counselling and testing.
Qualitative analysis
Exploring reasons for presentation with AHD at enrolment in care
Eight women and seven men enrolling in care with AHD were interviewed at the three health centres. 7/15 (46.7%) were aged 35 and above. The study found eight emergent subthemes that were grouped under three pre-determined themes based on literature review and previous studies conducted.
Theme 1: Intrapersonal factors
Exploration of intra personal factors revealed four emergent subthemes, which included feeling healthy, use of alternative therapies, fear of HIV diagnosis, and attitude towards HIV-related health education messages and testing. From the findings, it was evident that feeling healthy took precedence as the intrapersonal factor that contributed to enrolling in care with AHD. Alternative therapies including herbal medicine, self-medication, and seeking care from clinics first took second place.
Feeling healthy
Majority of the participants mentioned that what caused their late enrolment in care with AHD was the perception that they were still healthy. They mentioned they did not know that they were sick and some perceived their HIV risk to be low. A newly enrolled client mentioned that; ‘
Use of alternative therapies
About half of the participants reported that when they got frequent illnesses; they resorted to taking herbal medication, which gave them relief for some time, until when the herbal medicine became ineffective. Some confessed to having self-medicated; buying drugs from clinics for some time to cure their sickness before going to the hospital. ‘
Fear of HIV diagnosis
A few female participants mentioned that fear of being told that they were HIV positive contributed to their delay in seeking HIV care. ‘
Attitude towards HIV-related health education messages and testing
Almost all participants reported having paid minimal attention to HIV-related health education messages despite having heard them on several platforms including radios, community outreaches, and from peers. They were not motivated to heed the HIV-related messages. Below are remarks from two female participants; ‘
Theme 2: Interpersonal factors
Advice from friends and relatives
From an interpersonal perspective, majority of the respondents mentioned that they had received advice and support from friends and family members to go for HIV testing. However, from the responses, it was evident that peer and family support was of no effect in contributing to seeking HIV care timely. This was possibly because the social support came in late when the disease had already progressed. ‘
Theme 3: Health system-related factors
Role of health workers
Some of the participants mentioned having been referred for an HIV test by the health worker but this was at an advanced stage of the disease. Others mentioned that no healthcare worker had referred them for an HIV test. ‘
Discussion
In this study, over a third of ART naïve adults enrolling in care had AHD, 32.4% had a CD4 less than 200 cells/µL, while only 12.7% had a WHO stage 3 or 4 disease. Our findings show that the burden of AHD among ART naïve adults was high despite introduction and implementation of the Test and Treat strategy. These findings are similar to findings from Botswana, where the prevalence of AHD increased from 15.5% before implementation of the Test and Treat policy to 24.7% during Test and Treat implementation. 17 Likewise, in the African Cohort Study (AFRICOS), there was no significant decrease reported in the prevalence of CD4 < 200 before and after the Treat and Treat era. 18 In Senegal and Ethiopia, a prevalence of 70% and 60% of AHD were reported among clients newly diagnosed with HIV during the Test and Treat era respectively.19–20 The aforementioned studies were conducted at tertiary healthcare facilities including national referral hospitals and district hospitals respectively, and this could explain the difference in the prevalence reported by this study that was conducted at out-patient primary care facilities. Reasons that could explain this high prevalence of AHD include low perception risk among patients, low HIV literacy levels, lack of access to HIV-related messages and poor health seeking behaviours. Consequences of this include continued HIV-related morbidity and mortality at individual level; onward transmission of HIV infections, with subsequent failure to achieve HIV epidemic control.
The prevalence of AHD was higher among males and those aged between 35 and 50 years. Additionally, the prevalence of AHD was lower among those who did not report any personal health perception barriers. The former has previously been attributed to women having more frequent health care visits including family planning and antenatal care visits, and wider access to HIV testing during antenatal care.21,22 Men are also less likely to test or link to care due to stigma, the psychological implications of a positive HIV result, poor health seeking behaviour and due to issues related to occupation and culture.23–26 However, with the scale-up of HIV services, one would have hoped that the gender differences would have been attenuated due to the fact the advantage of women having more frequent healthcare visits would no longer stand. Interventions that address men’s health in a targeted way are needed to ensure that they are not left out.
Age 35–50 years was also found to be associated with AHD. Other studies have also reported older people being more likely to present with AHD.22,27 This is assumed to be due to a low perception of HIV risk among older people, and health workers being less likely to consider HIV infection in older people, however, this may require more research. Health workers offering HIV prevention, care and treatment services therefore need to design HIV prevention services targeting the older people. Health facility staff also need to ensure that older individuals newly enrolling in care are screened for AHD.
The prevalence of AHD was significantly lower among those who did not report any personal health perception barriers. Qualitative data further revealed that the majority of the clients who enrolled in care with AHD perceived themselves to be still healthy, and this was true for both men and women. They reported to have always recovered from the frequent sickness they suffered before enrolling in care and therefore saw no need for HIV care until the illness persisted or when they became symptomatic. This is especially important because HIV infection is a chronic illness and one may take a long time without being symptomatic. However, during this time, the body’s immune system is getting destructed and most times to irreversible levels even after ART is initiated. This implies a gap regarding the provision of HIV testing services and health education in Uganda. There are currently minimal incentives for early HIV testing, a lack of adequate knowledge and awareness on the frequency of routine HIV testing in the general population, and lack of timely access to HIV testing services. In a qualitative study conducted in Uganda, Lofgren
There was no association between AHD and where the client first sought care including herbalists/traditional healers. This is in contrast to a recent study from Senegal
19
and one study conducted in Uganda before Treat and Treat.
29
However, almost half of the interviewed IDI participants reported resorting to herbal medication first. Others reported first seeking care from clinics and self-medicating from pharmacies and drug shops. In a study by Namuddu
Limitations of this study include the fact that PLHIV who may have previously been on ART and dropped out of care could have showed up as ART naïve. Approximately 18% of the study participants reported to have tested HIV positive prior to enrolling in care, however data on how far back this diagnosis was made was not available. Additionally, this study was conducted at outpatient ART clinics in selected public facilities in Kampala city, which is the country’s capital. Therefore, results from this study may not be generalizable to the whole country. However, we adopted the standard WHO AHD definition incorporating both CD4 cell count and the WHO stage. The CD4 cell count cut off used was 200 cells/µL as recommended by WHO unlike several studies conducted prior that had differing cut-offs.
Conclusion
The study findings show that the burden of AHD among ART naïve PLHIV enrolling in care was high in the Test and Treat era in Uganda. The prevalence was higher among males and individuals aged 35–50 years compared to their female counterparts and those aged 18–24 years. Findings further reveal that perceived personal health barriers contribute to enrolling in care with AHD, with most individuals perceiving themselves to still be healthy and waiting till physical health deterioration before enrolling in care. There is therefore an urgent need for implementing partners, HIV care and treatment clinical teams in facilities to innovate HIV testing strategies engaging men and aged individuals aged 35–50 years. HIV testing during flexi hours (extended working hours) targeting men should continue to be implemented, however, these services should also be extended to workplaces especially where casual jobs are carried out because this subpopulation may not benefit from flexi hours at health facilities.
The Uganda Ministry of Health and its partners need to consider revising the national HIV Testing Services Policy and implementation guidelines to include incentives for early and routine HIV testing apart from the provision of free HIV testing services. Additionally, MoH together with partners need to enhance investments in HIV literacy specifically for men and people aged between 35 and 50 years, with emphasis on routine testing regardless of one’s perception of their health status. Lastly, provision of free HIV testing services in private clinics, pharmacies, and drug shops by the Ministry of Health may improve uptake of HIV testing services.
Supplemental Material
sj-docx-1-tai-10.1177_20499361241251936 – Supplemental material for Prevalence of advanced HIV disease and associated factors among antiretroviral therapy naïve adults enrolling in care at public health facilities in Kampala, Uganda
Supplemental material, sj-docx-1-tai-10.1177_20499361241251936 for Prevalence of advanced HIV disease and associated factors among antiretroviral therapy naïve adults enrolling in care at public health facilities in Kampala, Uganda by Bridget Ainembabazi, Elizabeth Katana, Felix Bongomin, Phillip Wanduru, Roy William Mayega and Aggrey David Mukose in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-pdf-2-tai-10.1177_20499361241251936 – Supplemental material for Prevalence of advanced HIV disease and associated factors among antiretroviral therapy naïve adults enrolling in care at public health facilities in Kampala, Uganda
Supplemental material, sj-pdf-2-tai-10.1177_20499361241251936 for Prevalence of advanced HIV disease and associated factors among antiretroviral therapy naïve adults enrolling in care at public health facilities in Kampala, Uganda by Bridget Ainembabazi, Elizabeth Katana, Felix Bongomin, Phillip Wanduru, Roy William Mayega and Aggrey David Mukose in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-pdf-3-tai-10.1177_20499361241251936 – Supplemental material for Prevalence of advanced HIV disease and associated factors among antiretroviral therapy naïve adults enrolling in care at public health facilities in Kampala, Uganda
Supplemental material, sj-pdf-3-tai-10.1177_20499361241251936 for Prevalence of advanced HIV disease and associated factors among antiretroviral therapy naïve adults enrolling in care at public health facilities in Kampala, Uganda by Bridget Ainembabazi, Elizabeth Katana, Felix Bongomin, Phillip Wanduru, Roy William Mayega and Aggrey David Mukose in Therapeutic Advances in Infectious Disease
Footnotes
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
