Abstract
Background:
Persistent low-level viraemia (PLLV) is a risk factor for virologic failure among people receiving antiretroviral therapy (ART).
Objectives:
We assessed the prevalence and predictors of PLLV among individuals receiving Dolutegravir-based ART in southern Nigeria.
Design:
This retrospective cohort study used routine program data from electronic medical records of persons receiving Dolutegravir-based first-line ART in 154 PEPFAR/USAID-supported health facilities in Akwa Ibom and Cross Rivers states, Nigeria.
Methods:
Clients on first-line Dolutegravir-based ART ⩾6 months, who had a viral load result in the 12 months preceding October 2021 (baseline), and a second viral load result by September 2022 were included. Persons with low-level viraemia (LLV) (viral load 51–999 copies/ml) received additional adherence support. The outcome analysed was PLLV (two consecutive LLV results). Indices were summarized using descriptive statistics, and predictors of PLLV were determined using multivariate logistic regression.
Results:
In total, 141,208 persons on ART were included, of which 63.3% (n = 89,944) were females. The median age was 36 [29–44] years, median ART duration was 19 [11–42] months. At the end of the study, 10.5% (14,759/141,208) had initial LLV, 90.1% (13,304/14,759) of which attained undetectable viral load (⩽50 copies/ml), and 1.1% (163/14,759) transitioned to virologic failure (⩾1000 copies/ml) by the end of the study. PLLV prevalence was 0.9% (1292/141,208). Increasing ART duration [adjusted odds ratio (aOR) = 1.9; 95% confidence interval (CI): 1.54–2.44; p < 0.001] and viral suppression (<1000 copies/ml) before initial LLV (aOR = 1.7; 95% CI: 1.50–2.00; p < 0.001) were positively associated with PLLV, while receipt of tuberculosis preventive therapy reduced the likelihood of PLLV (aOR = 0.3; 95% CI: 0.10–0.94; p = 0.039).
Conclusion:
PLLV was uncommon among individuals receiving dolutegravir-based ART and was associated with longer ART duration, prior viral suppression, and non-receipt of tuberculosis preventive therapy. This strengthens recommendations for continuous adherence support and comprehensive health services with ART, to prevent treatment failure.
Introduction
Antiretroviral therapy (ART) is a life-saving intervention that reduces morbidity and mortality among persons living with HIV. It prevents viral transmission by halting HIV replication, resulting in the rapid reduction of plasma viral load to undetectable levels (<50 copies/mL).1,2 HIV viraemia is a marker of viral replication and a predictor of disease progression and transmission. 3
ART coverage has increased globally from 2% in 2000 to 76% in 2022, with 29.8 million of the estimated 39.0 million people living with HIV (PLHIV) receiving ART.4,5 Viral suppression rates have also improved globally from 81% in 2015 to 93% in 2022. 5 PLHIV who have viraemia levels considered ‘undetectable’ level will not transmit the virus sexually, 6 making achieving and maintaining sustained virologic suppression a primary goal in the management of PLHIV.7–9
Despite significant advancements in ART, studies have shown that PLHIV receiving ART experience low-level viraemia defined as detectable plasma HIV RNA between 50 and 999 copies/ml. 10 Some authors have described low-level viraemia as either transient or persistent 11 based on the frequency of detectable viraemia, while other authors have classified low-level viraemia based on the level of viraemia (viral load copies).2,12 Persistent low-level viraemia (PLLV) has been subject to varying definitions but generally refers to the sustained presence of plasma HIV RNA above the lower limit of detection of a specific assay but below the defined threshold for virologic failure, among individuals receiving ART.13,14
The clinical significance of low-level viraemia and its management, while unclear7,13–15 is of particular interest because of the increased risk of virologic failure and drug resistance associated with both low-level viraemia and PLLV.16–23 The origin of PLLV is subject to debate, but its occurrence has been associated with several risk factors, such as higher baseline viral load, inconsistent adherence, and use of non-nucleoside reverse transcriptase inhibitor and protease inhibitor-based regimens.2,13 However, this has not been thoroughly investigated in the context of the widespread use of integrase strand transfer inhibitors such as dolutegravir, especially in resource-constrained settings, like Nigeria where dolutegravir-based regimens are the recommended options for first-line ART. 24
Nigeria is home to a substantial burden of HIV, with an estimated 1.9 million PLHIV as of 2021. 4 The introduction of dolutegravir-based ART as the preferred first-line regimen by the World Health Organization, 25 and subsequent adoption by Nigeria in 2018, 26 has raised expectations of improved virologic suppression and treatment outcomes.27,28 While dolutegravir has demonstrated high potency and rapid virologic suppression,29–31 the prevalence and predictors of PLLV among individuals on dolutegravir-based ART, particularly in the context of real-world clinical outcomes, warrants investigation, as this can be crucial for optimizing treatment strategies, improving patient care, and guiding public health policies.
This study aimed to determine the prevalence and identify predictors of PLLV among clients receiving dolutegravir-based first-line ART regimens in Nigeria.
Methods
Study design
This was a retrospective cohort analysis of secondary data routinely collected from electronic medical records for program implementation.
Setting
The study was conducted in two states in Southern Nigeria (Akwa Ibom and Cross River States), where Excellence Community Education Welfare Scheme receives funding from the United States President’s Emergency Plan for AIDS Relief through the United States Agency for International Development to support the government of Nigeria in providing comprehensive HIV care and treatment. Over 180,000 recipients of care receive ART across 154 supported health facilities (105 primary, 47 secondary, and 2 tertiary). Mobile teams also provide decentralized services in community settings. 32
Participants
The study extracted data from PLHIV receiving first-line dolutegravir-based ART regimens across 154 health facilities. Eligibility for inclusion in the study cohort included PLHIV on ART for ⩾6 months who had one viral load result in the 12 months preceding October 2021, and who had a second viral load result between October 2021 and September 2022. This aligns with Nigeria’s treatment guidelines, which recommend a first viral load test after 6 months on ART, and annual viral load tests subsequently if virally suppressed. 24
Ethics approval and consent to participate
Permission to analyse these program data was obtained from the Health Research Ethics Committee in Akwa Ibom state on 22 December 2022 (HREC No. AKHREC/25/11/22/120). Approval was also obtained from the Office of International Research Ethics on 24 February 2023 (PROJECT #: 2026278-1).
Patient informed consent was not required because only routine, anonymous and operational monitoring data were collected and analysed.
Description of low-level viraemia management in the study setting
Adherence support is provided to recipients of care with low-level viraemia by trained healthcare workers, in line with the National HIV treatment guidelines for managing low-level viraemia. 24 Once low-level viraemia is detected, clients are invited for a clinical review. The review involves identifying the risk factors for poor adherence and counselling on how to mitigate these factors. Follow-up counselling support and medication reminders are provided on a monthly basis and viraemia levels are re-assessed after 3 months of this intensive adherence monitoring.
Data sources/variables/measurement
Data for this study were obtained from the electronic medical records – Lafiya Management Information System in each health facility. 33 The outcome variable abstracted and analysed was PLLV defined as two consecutive low-level viraemia results. The prevalence of PLLV was calculated as the proportion of total study participants who experienced PLLV.19,34,35 Explanatory variables extracted from the electronic medical records for the study include age, sex, date of ART initiation, viral load categories, most recent World Health Organization clinical stage during the last clinic encounter prior to initial viral load, history of tuberculosis preventive therapy, and differentiated service delivery model (Community ART Clubs, Fast Track, Adolescent Refill Clubs, Community Pharmacy ART Refill Program, and Community ART Groups). The Differentiated Service Delivery models are described by Sanwo et al. 36 ART duration was derived by comparing the period between the date of ART initiation and the date of low-level viraemia result and categorized as ⩽12 or >12 months. World Health Organization clinical stage was determined during the most recent clinic visit prior to the initial viral load result and classified as ‘Asymptomatic’ (if stage 1) or ‘Symptomatic’ (if stages 2, 3 or 4). Viral load categories were defined based on the client’s previous viral load history (i.e. last viral load before the baseline used in the study) into the first viral load, prior viral suppression (if viral load <1000 copies/ml), and prior viral unsuppression (viral load ⩾1000 copies/ml). History of tuberculosis preventive therapy completion and devolvement to differentiated models of ART prior to initial low-level viraemia was collected.
Data analysis
Descriptive statistics were used to analyse categorical variables using frequencies. Binary logistic regression was used to determine predictors of PLLV among the cohort with initial low-level viraemia. Significant variables in the univariate analysis were included in the multivariate model, adjusting for all variables. All analyses were done using STATA version.14 (produced by StataCorp LLC, Texas, USA) with a significance set at 0.05.
The reporting of this study conforms to the Strengthening the Reporting of Observational Studies in Epidemiology statement. 37
Results
Data were extracted for 168,783 PLHIV, of which 141,208 (83.7%) had a documented viral load result as of October 2021 and a subsequent viral load result within the 12-month study period, and were eligible for inclusion in the study (Figure 1). The median interval between viral load tests was 11 (9–11) months. Of the 4678 individuals excluded because of not having two viral loads, 3998 (85.4%) had an undetectable viral load, 588 (12.6%) had low-level viraemia, and 92 (2.0%) had an unsuppressed viral load at baseline.
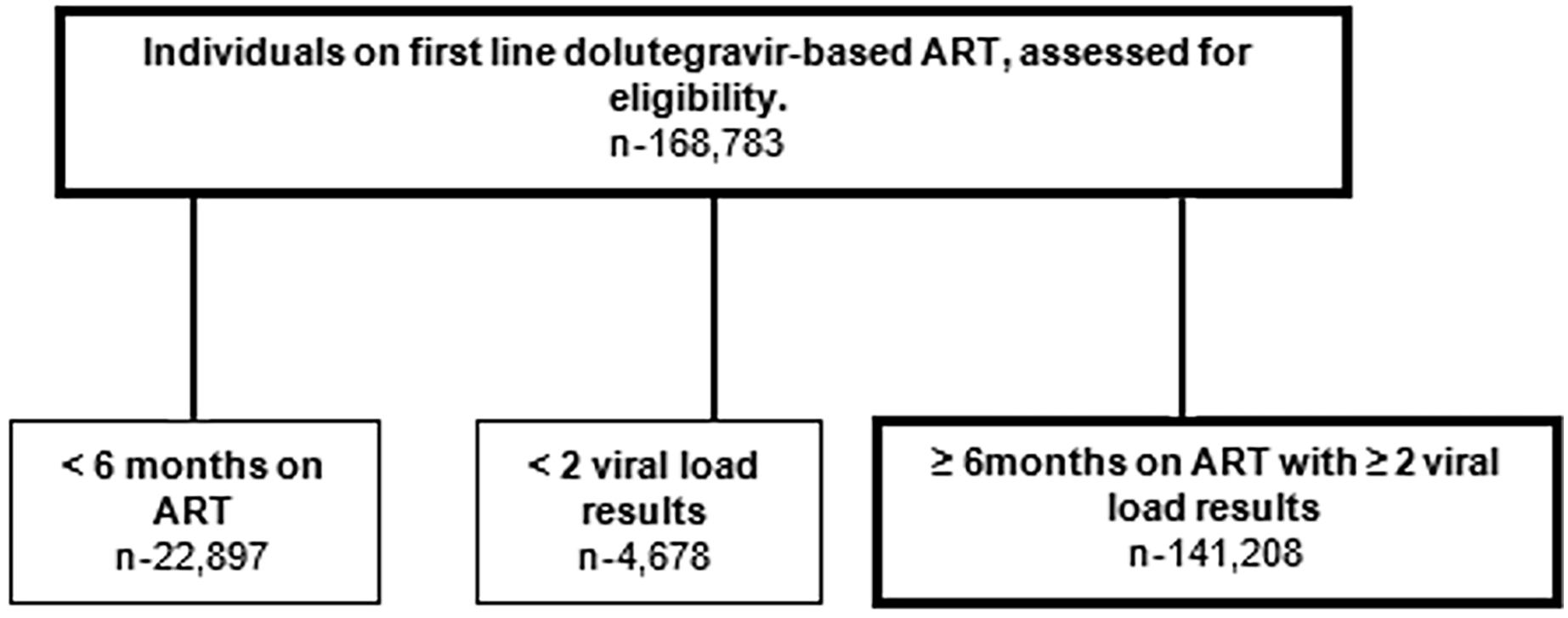
Flow chart of selection criteria for PLHIV receiving dolutegravir-based regimen as of October 2021 in southern Nigeria.
The majority [63.7% (n = 89,944)] of clients included were females, with a median age of 36 years [29–44 years], 99.1% were receiving tenofovir-based dolutegravir regimen and median ART duration was 19 (11–42) months. Over 99.9% had completed tuberculosis preventive therapy, with 89.8% enrolled in Differentiated Service Delivery, while 99.0% had asymptomatic clinical stage prior to initial low-level viraemia and 67.5% were ART naïve (Table 1). In total, 99.1% (n = 139,877) of participants were receiving tenofovir disoproxil fumarate with dolutegravir, while the other 0.9% of participants received Abacavir with dolutegravir.
Characteristics of PLHIV cohort on dolutegravir-based first-line ART for at least 6 months, with at least two viral load results between October 2021 and September 2022 in Southern Nigeria.

ART, antiretroviral therapy; PLHIV, people living with HIV.
As shown in Figure 2, 10.5% of individuals had low-level viraemia at the initial assessment, and 0.91% (n = 1292/141,208) had two consecutive low-level viraemia results (PLLV). The majority of PLHIV with initial low-level viraemia (90.1%; n = 13,304/14,759) attained undetectable viral load level (⩽50 copies/ml), and only 1.1% (n = 163/14,759) transitioned to Virologic Failure (viral load ⩾1000 copies/ml) during the study period.

Flow chart of viral load outcomes at follow-up, for persons living with HIV receiving a dolutegravir-based regimen, who had baseline viral load results.
The prevalence of PLLV was comparable by sex (0.9% among males and 0.9% among females). By history of tuberculosis preventive therapy, PLLV was lower among those with a history of completing tuberculosis preventive therapy (0.9%) compared to those without a history of tuberculosis preventive therapy completion (23.8%). When analysed by viral load categories, PLLV was lowest among those with first viral load (0.4%) compared to those who had prior viral suppression (1.1%) or those with prior viral unsuppressed status (0.8%) (Table 2).
Prevalence of PLLV by client characteristics for the cohort of PLHIV with initial low-level viraemia.

ART, antiretroviral therapy; PLHIV, persons living with HIV; PLLV, persistent low-level viraemia.
In multivariate analysis, increasing duration of ART [adjusted odds ratio (aOR) = 1.9; 95% CI: 1.54–2.44; p < 0.001] and viral suppression prior to initial low-level viraemia (aOR = 1.7; 95% CI: 1.50–2.00; p < 0.001) were positively associated with the presence of PLLV. In addition, those who had a history of tuberculosis preventive therapy completion were less likely to have PLLV compared to those with no history of tuberculosis preventive therapy completion (aOR = 0.3; 95% CI: 0.10–0.94; p = 0.039) (Table 3).
Predictors of persistent low-level viraemia for PLHIV cohort with initial low-level viraemia.

Logistic regression analysis, STATA 14.0.
aOR, adjusted odds ratio; ART, antiretroviral therapy; CI, confidence interval; PLHIV, persons living with HIV.
Discussion
This study aimed to investigate the prevalence of PLLV among PLHIV receiving dolutegravir-based first-line ART. Our findings indicate that at a prevalence of 0.9%, PLLV was a rare occurrence in this population and that increasing ART duration, prior viral suppression, and absence of tuberculosis preventive therapy were associated with PLLV.
Other studies have reported a substantially higher prevalence of PLLV, including the US Military HIV Natural History Study (NHS) which documented a 7% PLLV prevalence. 38 Similarly, the AIDS Clinical Trials Group (ACTG) Studies A5142 and A5095 reported prevalence rates of 7% and 4%, respectively, 34 an observational cohort study in Canada reported prevalence rates of 12%, 8%, and 5% after 6, 9, and 12 months of follow-up, respectively, 19 and a study focused on pregnant women in South Africa 35 recorded a 9.8% prevalence of PLLV. Although two of these studies evaluated PLLV differently due to the absence of a standard definition for PLLV, this may not sufficiently account for the differences in prevalence. The two ACTG studies, 34 the Canada study 19 and ours defined PLLV based on repeat low-level viraemia measurements between 50 and 1000 copies/ml yet reported varying PLLV prevalence rates, whereas the NHS 38 which defined PLLV based on >25% low-level viraemia measurements over a 27-year period reported similar prevalence as the ACTG study A5142 despite the difference in PLLV evaluation method. The South Africa study 35 defined PLLV based on repeat detectable viraemia over a 4-week period.
One critical difference, however, is that our study included only individuals on dolutegravir-based regimens, while the NHS, 38 ACTG 34 and Canada studies 19 included individuals receiving non-nucleoside reverse transcriptase inhibitors and protease inhibitors for ART, which could account for the differences in PLLV prevalence especially as several studies have associated dolutegravir with sustained viral load suppression29–31 while others have linked non-nucleoside reverse transcriptase inhibitors and protease inhibitors with low-level viraemia and PLLV.13,23,38,39 The South Africa study 35 only included clients on first-line ART but did not specify the regimen class for clients in their cohort. The progression of 98.9% of individuals with low-level viraemia to undetectable viral load levels without a change in regimen further strengthens reports on the high efficacy of dolutegravir in suppressing HIV replication and preventing resistance development. 40 More so in our study, adherence counselling was provided to patients with low-level viraemia, while only the South African study reported adherence support for clients with low-level viraemia. However, the time interval between the two viral load tests was 4 weeks in the South African study, compared to our study where adherence counselling was provided for at least 3 months before a repeat viral load test. The longer duration allowed for sufficient time to adequately assess, identify and address barriers to adherence, which could account for the lower PLLV prevalence in our study. Suboptimal ART adherence has been linked with higher risks of low-level viraemia40,41,42 across various ART regimens, 43 and our findings further suggest that adherence counselling for an adequate duration may be an effective intervention for patients on dolutegravir-based ART with low-level viraemia, especially in resource-constrained settings with limited access to drug resistance testing.
In multivariate analysis, our study identified factors positively associated with PLLV, which are longer ART duration and viral suppression prior to initial low-level viraemia, while a history of tuberculosis preventive therapy completion was associated with a lower risk of PLLV. This is in contrast to previous research linking longer ART duration with lower risk of PLLV, although previous studies have focused on clients receiving nucleoside reverse transcriptase inhibitors, non-nucleoside reverse transcriptase inhibitors, and protease inhibitors, and none included clients on dolutegravir-based ART.44–46 However, a large Nigerian study 47 reported associations between longer duration of ART and risk of low-level viraemia, which is similar to our findings. This may be a result of declining ART adherence over time, which has been linked to longer ART duration,48,49 even among members of adherence clubs, 50 and points to the need for continuous person-centred adherence support with increasing ART duration. Previous research has also associated longer ART duration with mental health issues, 51 which may consequently impact ART adherence adversely,52,53 and this underscores the need to integrate mental health services with adherence counselling 54 for individuals with low-level viraemia. Our findings may also reflect the accumulation of minor resistance mutations over time in individuals on ART for longer durations as has been reported in other studies in Africa and the United States.55,56 Although our study did not assess drug resistance, the progression of only a minority (0.1%) of individuals with low-level viraemia to virologic failure reflects the need to prioritize adherence counselling for low-level viraemia57–59 in the era of dolutegravir-based ART especially in resource-limited settings, with the high costs of drug resistance testing.60,61
Our study found associations between viral suppression prior to initial low-level viraemia and an increased likelihood of PLLV, and while this relationship has not been properly understood 62 other researchers have associated persistent viraemia with ongoing cycles of viral replication even in patients who have achieved viral suppression <50 copies/ml.63,64 Replication can occur due to suboptimal drug penetration in anatomical reservoirs or the production of virions that do not infect new cells by clonally expanded infected T cells. 62 The association could also be related to similar factors in long ART duration, including treatment fatigue and declining adherence, which can be addressed with continued adherence monitoring and support for clients who have attained viral suppression. 65
Tuberculosis preventive therapy completion is a reflection of good medication adherence and may explain the relationship between tuberculosis preventive therapy completion and reduced likelihood of PLLV. Clients who have completed tuberculosis preventive therapy may have received continued psychosocial counselling during clinic visits, which can improve both tuberculosis preventive therapy and ART medication adherence. Subtherapeutic drug levels and suboptimal adherence to ART medication have been implicated in episodes of low-level viraemia.38,66,67 Some authors have also suggested an interplay between immune response and viral dynamics. Individuals who complete tuberculosis preventive therapy may have better immune function than those who do not, 68 which might contribute to more effective viral suppression. 69 This raises broader questions about the potential impact of other co-morbidities that affect immune status on the dynamics of PLLV, which warrants further research. Our results also suggest that comprehensive health interventions can have a broader impact on viral suppression.70,71
Our study found no associations between PLLV and age or sex, which is consistent with previous research that reported no links between client demographics and PLLV.11,13,72–74 The absence of correlation between PLLV and individuals’ most recent World Health Organization clinical stage suggests that dolutegravir-based ART is effective in achieving and maintaining viral suppression in most clients, regardless of their demographic or clinical characteristics.75,76 Also, the consistency of PLLV outcomes irrespective of the Differentiated Service Delivery model lends credibility to reports that Differentiated Service Delivery does not compromise the virological efficacy of dolutegravir-based ART.77,78
It is plausible that integrase inhibitor-based therapies, such as dolutegravir, could contribute to reduced PLLV over time and enhanced virological control. The mechanism behind this effect warrants further investigation and could offer new insights into optimizing ART regimens to achieve better treatment outcomes. Resistance genotyping should be considered in patients with PLLV when feasible, and treatment should be modified if resistance is detected. There is a dearth of clinical evidence to guide management when genotyping is not feasible. 13
Our study had some limitations. First, the use of retrospective data limited the type of analysis that was done as data on pre-ART HIV RNA, pre-ART CD4 count, previous ART regimen, adherence levels and drug resistance testing, which have been associated with low-level viraemia in other studies were not available. Secondly, the paucity of studies on PLLV in sub-Saharan Africa also limited comparisons in similar settings. However, the improved viral load suppression following adherence support is a fair proxy for adherence in the study cohort. Other potential predictors of PLLV, such as viral subtype, drug interactions, and comorbidities, which were not assessed in this study, as well as the clinical impact of PLLV on long-term outcomes including comorbidities, which remains unclear, require more research.
To the best of our knowledge, this study is the first to report on the prevalence and predictors of PLLV among a large cohort of PLHIV on dolutegravir-based ART in sub-Saharan Africa and is strengthened by its large sample size and the use of real-world data from routine clinical practice. Findings from the study have important implications for the clinical management and public health response to HIV, especially in resource-limited settings where dolutegravir is part of the recommended first-line ART regimen. The lack of consistency between definitions of PLLV makes comparisons between studies challenging. Our definition of PLLV which evaluates the consecutive incidence of low-level viraemia reflects the consistency and stability of viral suppression which are important for clinical management, and the use of two consecutive low-level viraemia results allows for timely intervention, whereas non-consecutive definitions may capture transient episodes of viral blips that do not indicate virologic failure or drug resistance.
Conclusion
Our study found that PLLV is uncommon among PLHIV on dolutegravir-based ART in southern Nigeria, and most cases of low-level viraemia progress to undetectable viraemia when adherence support is provided. Continuous adherence support especially for individuals who have attained viral suppression or with longer treatment durations and comprehensive healthcare service delivery for PLHIV are effective interventions to reduce the risk of PLLV. These findings contribute to knowledge on the interplay of factors influencing virological outcomes in the dolutegravir era, and support the design of personalized interventions to enhance viral suppression and prevent virologic failure. The long-term clinical consequences of PLLV remain largely unknown and merit further investigation.
Supplemental Material
sj-doc-2-tai-10.1177_20499361241242240 – Supplemental material for Prevalence and predictors of persistent low-level HIV viraemia: a retrospective cohort study among people receiving dolutegravir-based antiretroviral therapy in Southern Nigeria
Supplemental material, sj-doc-2-tai-10.1177_20499361241242240 for Prevalence and predictors of persistent low-level HIV viraemia: a retrospective cohort study among people receiving dolutegravir-based antiretroviral therapy in Southern Nigeria by Ogheneuzuazo Onwah, Esther Nwanja, Uduak Akpan, Otoyo Toyo, Chiagozie Nwangeneh, Babatunde Oyawola, Augustine Idemudia, Kolawole Olatunbosun, Onyeka Igboelina, Dolapo Ogundehin, Ezekiel James, Okezie Onyedinachi, Adeoye Adegboye and Andy Eyo in Therapeutic Advances in Infectious Disease
Supplemental Material
sj-docx-1-tai-10.1177_20499361241242240 – Supplemental material for Prevalence and predictors of persistent low-level HIV viraemia: a retrospective cohort study among people receiving dolutegravir-based antiretroviral therapy in Southern Nigeria
Supplemental material, sj-docx-1-tai-10.1177_20499361241242240 for Prevalence and predictors of persistent low-level HIV viraemia: a retrospective cohort study among people receiving dolutegravir-based antiretroviral therapy in Southern Nigeria by Ogheneuzuazo Onwah, Esther Nwanja, Uduak Akpan, Otoyo Toyo, Chiagozie Nwangeneh, Babatunde Oyawola, Augustine Idemudia, Kolawole Olatunbosun, Onyeka Igboelina, Dolapo Ogundehin, Ezekiel James, Okezie Onyedinachi, Adeoye Adegboye and Andy Eyo in Therapeutic Advances in Infectious Disease
Footnotes
Acknowledgements
We would like to thank all those who were involved in the implementation of the Excellence Community Education Welfare Scheme, ACE 5 project in Nigeria, the Government of Nigeria, and especially the frontline service providers at health facilities and community service delivery points for their life-saving work.
Declarations
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
