Abstract
Outpatient parenteral antimicrobial therapy (OPAT) in Australia has evolved from modest beginnings to a well-established health service with proven benefits in patient outcomes. This is a comprehensive review of the current state of art Australian OPAT with vignettes of the types of OPAT models of care, antimicrobial prescribing and antimicrobial use. In addition, we highlight the similarities and differences between OPAT to other countries and describe Australian OPAT experiences with COVID-19 and paediatrics. Australian OPAT continues to advance with OPAT antifungals, novel treatment options and upcoming high-impact research.
Keywords
Background
Outpatient parenteral antimicrobial therapy (OPAT), which is most commonly administered in Hospital in the Home (HITH) programmes in Australia, originated in the United States in the mid-1970s primarily for cost reasons when delivering intravenous (IV) antibiotics outside of the hospital setting for uninsured patients. Since then, this concept has been adopted in numerous countries with varying models of care.1,2
The Australian healthcare system, Medicare™, provides universal access to essential medical services for all citizens and permanent residents of Australia. This means that regardless of the income or socioeconomic status, the entire Australian population has access to healthcare through Medicare™.
OPAT service is a specialized program that allows patients to receive IV antibiotics outside of a hospital setting, usually in their homes. While OPAT services are available nationwide, capacity can vary from region to region.
Access to OPAT services can depend on various factors, including the patient’s medical condition, the availability of healthcare facilities in their area and the discretion of healthcare providers. Generally, OPAT services are offered to patients who meet the specific criteria and can benefit from receiving ongoing IV antibiotics outside the hospital.
OPAT services originated in Australia from Victoria in the mid-1990s as a cost-saving, acute bed substitution initiative.3,4 Pharmacokinetic and efficacy studies for OPAT were initially limited to 12-hourly cefazolin 5 and then to once-daily cefazolin plus oral probenecid 6 to demonstrate the efficacy and safety of managing uncomplicated cellulitis in the OPAT/HITH setting.
The advent of peripherally inserted central catheters (PICC) and favourable antimicrobial stability data have allowed continuous infusions of additional antimicrobials (especially other beta-lactams) to be delivered in the OPAT/HITH setting through portable infusion devices. This development greatly expanded the number of indications that can be successfully managed as an outpatient.3,6
A substantial proportion of HITH patients receive parenteral antimicrobials, or ‘OPAT’. A variety of models of care exist for OPAT/HITH in Australia. OPAT is commonly provided by HITH services, where patients receive treatment at home as a substitute for a hospital bed.
7
Early programs featured governance by nursing staff; however, medical governance, either
The features and benefits of a successful OPAT/HITH program are well-described, including4,8,9:
- Efficacy: must be equivalent or better compared with hospital-based care with antimicrobials prescribed according to Antimicrobial Stewardship (AMS) principles (e.g. avoiding the use of broad-spectrum agents for convenience).
- Cost: management at the patient’s residence rather than as a hospital inpatient is usually less costly.
- Patient and carer satisfaction: increased.
AMS principles govern the Australian OPAT model, similar to the United Kingdom and Infectious Diseases Society of America OPAT Guidelines.9,12–14 OPAT/HITH continues to evolve worldwide, including the use of complex oral antibiotic therapy early in the management of indications such as infective endocarditis and bone and joint infections (BJIs) 13 as well as playing a pivotal role during the COVID-19 pandemic in delivering antiviral and antibody therapy to patients outside of the hospital inpatient setting. 15
This narrative review aims to describe the evolution and current state of OPAT/HITH in Australia, including the various models of care, medical governance, common indications for treatment and the use of OPAT/HITH care for COVID-19 and paediatrics.
Models of OPAT care in Australia
Current models of OPAT care in Australia were developed by health organizations to avoid or shorten hospital admission for patients under their care and to reduce resource use and the cost of treatment. Australia has a strong and comprehensive public sector health system administered by the Australian states and territories. Several state Ministries of Health have published guidelines governing OPAT services under the broader banner of HITH.16–18 The HITH Society of Australasia is the peak HITH body in the Asia-Pacific region that actively lobbies the government at local and federal levels to influence health policy on HITH-related issues. 19
In many jurisdictions, patients receiving OPAT under HITH are classed as hospital-admitted inpatients rather than outpatients, as the term OPAT implies. Examples of organizational structures are listed in Table 1. The Therapeutic guidelines: Antibiotic is considered the Australian consensus guidelines of infection diseases and includes a chapter on Community-based parenteral antimicrobial therapy, which sets out guiding principles. 9
Examples of organizational structures of OPAT services in Australia.
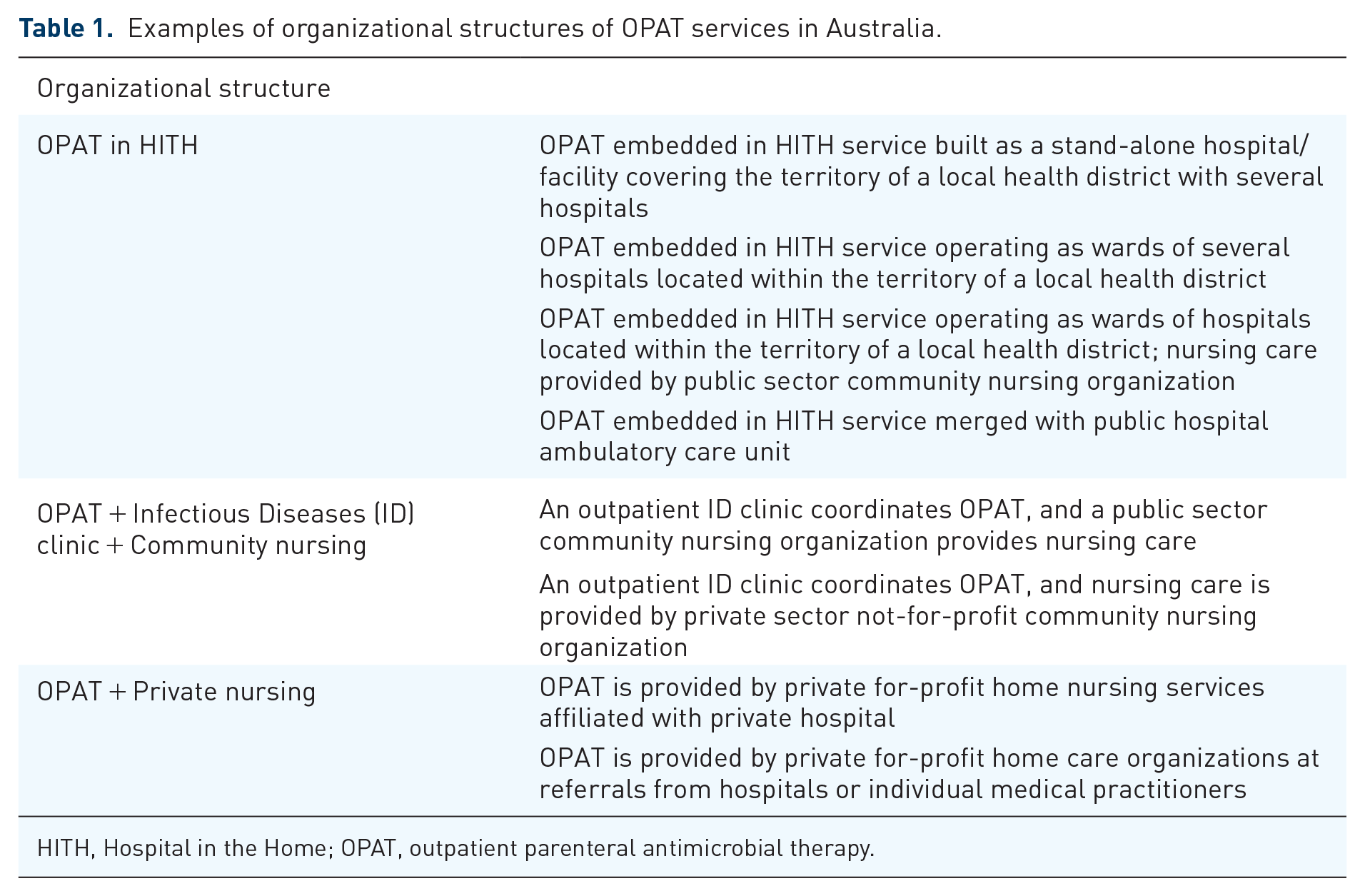
HITH, Hospital in the Home; OPAT, outpatient parenteral antimicrobial therapy.
In addition to patients receiving OPAT, HITH supports patients with various care needs to facilitate early discharge from the hospital or to avoid/substitute for hospital admission. HITH services are typically staffed by a multidisciplinary team (MDT) and have access to clinicians from various disciplines. While OPAT services coordinated by outpatient ID clinics are usually medically led by an ID physician, HITH services can be nursing led or medically led by a more generalist doctor on staff. Pharmacists are embedded in either the OPAT/HITH service, ID/AMS service, or both.
20
OPAT/HITH care by doctors in Australia can range from daily to weekly consultations. Models of nursing care, with service delivery by nursing staff from within the service or external nursing providers. Public OPAT services in Australia are either block funded by the state Ministry of Health or
Many Australian private hospitals provide OPAT through either a nursing service affiliated with the hospital or parent company or contractual arrangements with a private home nursing care organization. In contrast to publicly funded programs, medical governance and management usually remains with the specialist(s) who cared for the patient during the private hospital admission. The patient’s private health insurer usually funds some of the costs of private OPAT.
OPAT/HITH services receive referrals from hospital teams, ED departments (to avoid hospital admission) and outpatient clinics and general practitioners (GP) (to prevent hospital presentation). The HITH team assesses referrals, and a treatment and medical review plan is agreed upon with the referring team. Medical governance for the OPAT episode either remains with the referring consultant or is transferred to the HITH team.
OPAT services typically offer once-daily home visits. However, some services can visit patients’ multiple times a day or arrange for patients to attend clinics. Suitable patients may administer their treatment at home with or without telehealth supervision by a nurse. Services with MDTs can support patients with home visits by allied health clinicians alongside their OPATs as needed. Medical progress reviews generally occur at least weekly, most commonly by the HITH or referring team in a clinic setting. However, Telehealth Virtual Care is becoming more common for suitable patients, and some centres offer home visits by doctors.
The OPAT service or the referring hospital usually provides medication. Elastomeric infusion devices are commonly used in Australia to administer OPAT for drugs requiring dosing more frequently than once or twice daily. The OPAT service may load doses at the aseptic compounding facility of the referring hospital or at an external compounding service. Some OPAT services use battery-operated Computer Ambulatory Delivery Device (CADD) pumps or smart pumps as an alternative to elastomeric devices. CADD pumps consist of a cassette pump placed in a bag that can be worn and is programmed to deliver an exact dose per hour at exact times. Smart pump technology uses dose error reduction software with a programmed drug library and provides user alerts with override functions. 21 In general, OPAT services use the standard Australian reference publication Australian Injectable Drug Handbook, 22 databases (e.g. eTG 9 Stabilis 4.0, and in-house data from compounding companies to reference drug stability for continuous infusions). As individually compounded products, these routinely manufactured infusion devices are exempt from the standard medicines’ approval process by the government regulator, the Therapeutic Goods Administration. Published evidence23–25 guides individual agents, which in some cases has not been adopted by compounding companies. Unlike the United Kingdom, there is currently no standard Australian benchmark document for stability requirements for antimicrobial continuous infusion devices used routinely in OPAT. 26 The absence of defined standards has led to challenges, including conflicting stability data from in-house and published data resulting in discrepancies between commercially available compounded products for some drugs.
OPAT prescribing in Australia
Antimicrobial selection in OPAT in Australia is commonly operated under an AMS program; however, some Australian OPAT services may not involve AMS or ID consultation.9,13,27 Factors including the spectrum of coverage, stability data, dosing interval, monitoring requirements, storage conditions and indication/individual patient factors are carefully considered in the prescribing process and often imbedded into policies, procedures and guidelines.9,28 This setting is, however, an area where more focused AMS interventions could improve practice. The Australian Commission on Safety and Quality in Healthcare has a defined chapter in the AMS Book highlighting opportunities for improved stewardship in the OPAT setting. For example, regularly reviewing the patient to ensure timely de-escalation of antimicrobial therapy is essential to avoid prolonged, unnecessary IV treatment.
The most extensive Australian study on OPAT prescribing is the OPAT National Antimicrobial Prescribing Survey 2021 (OPAT NAPS). 7 OPAT NAPS evaluated the appropriateness of antibiotic use in children and adults by surveying 23 Australian HITH services representing regional and urban areas throughout Victoria, New South Wales, Queensland and Tasmania. Western Australia and South Australia did not participate in the OPAT NAPS. The study audited 1154 OPAT prescriptions across 715 patients and reported cefazolin (22%) and flucloxacillin (12%) as the most frequently prescribed antibiotics, followed by ceftriaxone and piperacillin-tazobactam.
Of the 1154 OPAT prescriptions audited, 87% were prescribed appropriately. Despite this, one in three ceftriaxone OPAT prescriptions was inappropriate for various reasons (e.g. unnecessarily broad-spectrum relative to the indication or inappropriate duration relative to the indication). Piperacillin/tazobactam OPAT prescriptions were often inappropriately prescribed (10%) for incorrect indications. 7
Cellulitis, osteomyelitis and pneumonia were the three most common indications in the NAPS OPAT. Other infections safely and effectively treated with OPAT in Australia include endocarditis, deep abscesses, pyelonephritis, vascular graft infections, malignant otitis externa, bacteraemia and empirical sepsis. Respiratory tract infections (RTIs) and cellulitis were the indications with the most inappropriate prescriptions, which may be attributed to limited ID consultation for these indications at some centres. BJI, deep abscesses and endocarditis, which typically warrant ID consultation, were the indications with the most appropriate prescribing. 7 This is consistent with previous reports where ID consultation during OPAT significantly reduces hospital readmission rates and improves healthcare outcomes. 29
On the international stage, Australian OPAT prescribing differs from UK OPAT prescribing, where parenteral once-daily antibiotics accounted for 91% (UK)
When comparing OPAT indications of other countries to Australia, studies from Singapore and the United States report that the most common OPAT indications were osteomyelitis (15%) and BJI (22%), respectively.31,36 The indications are similar to those stated in the Economic analysis commissioned by the HITH Society of Australasia.
4
A unique indication for OPAT care in Australia is an infection caused by
OPAT utilization in Australia
Antimicrobial utilization surveillance in Australian hospitals is conducted by the National Antimicrobial Utilisation Surveillance Program (NAUSP) using World Health Organization Daily Defined Doses (DDD) as the metric and pharmacy dispensing data. NAUSP was initially started in 2004, monitoring inpatient antimicrobials that excluded OPAT data; however, since 2021, Australian hospitals have contributed OPAT usage to NAUSP to enable them to monitor their usage over time and identify any unexpected trends. 39 A total of 61 hospitals (57 public, 4 private) are registered to contribute OPAT data to NAUSP, with 58 sites submitting data between January 2021 and December 2022; Figure 1 shows the geographic distribution of the hospitals. In addition, 14 (45%) of the 31 principal referral hospitals in Australia contribute OPAT data to NAUSP.

Hospitals contributing OPAT data to NAUSP.
From the OPAT usage data analysis contributed to NAUSP, flucloxacillin is the most commonly used antibiotic comprising 30.9% of all OPAT usage. Cefazolin is the second most common, comprising 21.0%. This is consistent with the Australian OPAT NAPS, where cefazolin and flucloxacillin were most prescribed. 40 The 12 antimicrobials used in the OPAT setting (by DDDs) are provided in Table 2.
Antibacterials used in OPAT in Australia (data contributed to NAUSP for 2021 and 2022).

Includes tobramycin, amikacin, tigecycline, cefoxitin, gentamicin, clindamycin, aztreonam, imipenem-cilastatin.
DDD, daily defined dose; NAUSP, National Antimicrobial Utilisation Surveillance Program; OPAT, outpatient parenteral antimicrobial therapy.
Negligible quantities (less than 0.1% of total OPAT dispensing) of other antibacterials were included in data submitted to NAUSP, including ceftazidime-avibactam, amoxicillin-clavulanic acid, lincomycin, colistin, azithromycin, ceftaroline, ceftolozane-tazobactam, metronidazole, ampicillin, moxifloxacin and ciprofloxacin.
Australian Health Services’ high rate of OPAT utilization has also been demonstrated for key infective diagnoses. At one large Victorian Health Service, 34% of all adult episodes of care for extended beta lactamase (ESBL) harbouring
COVID-19 and OPAT in Australia
The COVID-19 pandemic propelled OPAT services into unique circumstances posing challenges and opportunities. The mature nature of Australian OPAT/HITH services placed them at the forefront of Australia’s ambulatory response to COVID-19.11,42
The overall impact of COVID on Australia’s health service’s activity over the pandemic was varied. For example, in the first year of data encompassing the pandemic (July 19–June 2020), national hospitalizations experienced a 2.8% decrease compared with the previous year. This was primarily due to a federal decision to suspend elective surgeries at the emergence of the pandemic. However, the subsequent year (July 2020–June, 2021) demonstrated increased activity (+6.3%) related to catch up on elective surgeries and increasing demand for COVID-19-related care. 43 OPAT/HITH program’s role within this changing landscape comprised three key components deployed at various times during the pandemic.
OPAT services have been extensively utilized as a substitute for ward-based care in patients with COVID-19. Patients typically selected for admission diversion to HITH programs were those who had moderate symptoms, without oxygen therapy, or with medical comorbidities or social vulnerability, placing them at high risk of disease progression.44,45 Due to the varied nature of HITH/OPAT programs in Australia, care was delivered by different models, including physical reviews at home, telehealth/virtual care and
As a second role in the COVID-19 response, Australian OPAT programs have delivered COVID-19 therapeutics for ambulatory patients, including synthetic antibodies such as sotrovimab and the antiviral remdesivir.47,48 The model of delivery has varied by service and jurisdiction, including in-centre infusions and home administration.
A third key role for OPAT/HITH services during the pandemic was in caring for patients with diagnoses other than COVID-19. In addition to antimicrobial therapy, many Australian programs provide care, including acute anticoagulation management, IV diuretics, complex wound management, early postoperative care and cancer therapies. 49 This broad ‘Hospital in the Home’ role has supported Health Services to utilize ward-based beds more efficiently for acute patients (COVID & non-COVID) and increased volumes of elective surgeries to catch up for delayed activity during COVID-19. At-home care also protects vulnerable patients from COVID-19 exposure during attendance at healthcare facilities.
Paediatric OPAT in Australia
Australian OPAT is well established in paediatrics and presents unique challenges for OPAT care. These include different central venous access issues, dedicated paediatric nursing skill requirements and complications of infections.40,50–52 Children’s central lines are more susceptible to blockage and require small gage central venous catheters, increasing the risk of readmissions and catheter-related adverse effects.40,53 The benefits of OPAT are described to be greater in paediatrics than in adults due to the significant developmental and psychosocial impacts of hospitalization due to the child’s absence from school and family.54,55
The most extensive Australian paediatric OPAT study (
The choice of OPAT options may be different in children compared to adults. For example, in children, ertapenem is given 12 hourly, rather than 24 hourly in adults, due to the higher clearance <12 years of age. 57 Paediatric OPAT clinicians have a more extensive range of antibiotics to prescribe as continuous 24-h infusors because more dilute concentrations allow for increased stability. There are more allowances to use antibiotics in paediatric OPAT with controversial 24-h stability at paediatric dose concentrations (e.g. meropenem and ceftazidime).24,58,59
Future direction and challenges of OPAT in Australia
Newer antimicrobial agents, antifungal OPAT, novel routes of administration and treatment options are currently being explored in Australian OPAT practice. These newer OPAT agents include the use of tigecycline, 60 dalbavancin, ceftazidime-avibactam, 61 ceftaroline 62 and ceftolozane-tazobactam. 25 Australia is not alone in the increasing concerns of antimicrobial resistance, where there are limited OPAT options for multidrug-resistant organisms such as carbapenem-resistant Enterobacterales (CRE). 63 It is promising to see Australian-led research investigating antimicrobial stability in the OPAT setting by the Centre of Research Excellence – Personalising antimicrobial dosing to reduce resistance (CRE RESPOND). A once-a-day dose of tigecycline has been studied in Australian OPAT to treat multidrug-resistant infections. 60 This study showed that a selection of patients for tigecycline OPAT is vital due to the high rates of treatment failure, readmissions and adverse effects. Dalbavancin has been an attractive option in Australia for bone and skin infections as its long half-life allows for once-weekly dosing. It has increasingly been used in OPAT for people who inject drugs without leaving vascular access in situ between weekly doses.64,65
Western Australia (WA) is leading the treatment of invasive fungal infections in the OPAT setting, where published experience is scant.
66
A recent retrospective study in WA (
At the 2023 Australian Society of Infectious Diseases (ASID) Annual Scientific Meeting, data in the OPAT setting was presented on the use of thrice-a-week micafungin (3 mg/kg/dose) for IFI prophylaxis as a safer alternative to L-amb. It was associated with significantly less potassium supplementation and infusion reactions. 68 We look forward to the possible utility of the recently approved long-acting echinocandin, rezafungin, in the Australian antifungal OPAT setting.69,70 A recent analysis demonstrated the feasibility of delivering once-weekly antifungal in an Australian OPAT program based on a historical cohort of patients who received daily echinocandin therapy on OPAT. 71
Subcutaneously administered antibiotics are a novel approach to OPAT in Australia. Ertapenem displays favourable pharmacokinetics when given subcutaneously compared to the IV route of administration.
72
A large OPAT service in WA has reported that ertapenem is safe and effective in various infections, including diabetic foot, urinary, intra-abdominal and respiratory foci.
73
The Australian authors of this study found that the most frequently used patient group to use subcutaneous ertapenem were patients with chronic kidney disease who might progress to renal replacement therapy to preserve venous access. The main benefit of the subcutaneous (SC) route is the quick turnaround of patients in outpatient clinics without needing a PICC line insertion – a much more invasive and costly procedure. They report that nursing staff can easily perform SC needle insertion without specific training in peripheral cannulation or PICC insertion. Other antibiotics administered subcutaneously that can be future options in the OPAT setting are ceftriaxone and teicoplanin. In addition to this, there is growing literature to support the use of other antibiotics given
Bacteriophages (phage) are a novel treatment approach in the OPAT setting and have a resurgence in managing complicated multi-drug resistance infections. Successful Australian case reports of phage therapies given intravenously through OPAT for treating complicated mycobacterial infections exist. 75 Treatment protocols and clinical trial evidence are continually developing. Phage Australia is leading this work internationally with the STAMP study. 76
There are potential practice-changing Australian-led clinical trials that may change OPAT care. The Staphylococcus aureus bacteraemia Network Adaptive Platform (SNAP) trial is an international multicentre randomized adaptive platform trial looking at
Seeing how Australian OPAT has evolved since its humble beginnings of treating cellulitis with cefazolin is promising and exciting. Australia has a well-established OPAT service nationwide with a range of governance structures, national surveillance and AMS programs, policies, procedures, clinical resources and expertise. We continue to be at the cutting edge by exploring novel treatment options and addressing the gaps in research in OPAT clinical practice.
